Abstract
Introduction
Chronic subdural hematoma (CSDH) is associated with high recurrence rates. Middle meningeal artery embolization (MMAE) has emerged as a promising treatment option. In this systematic review and meta-analysis, we aimed to assess the safety and efficacy of MMAE for CSDH using liquid embolic agents and compare them with particles.
Methods
We systematically reviewed all studies describing MMAE for CSDH with liquid embolic agents, according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. Additionally, we included a cohort of patients from our institution using liquid and particle embolic agents. Data were analyzed using random-effects proportions and comparisons meta-analysis, and statistical heterogeneity was assessed.
Results
A total of 18 studies with 507 cases of MMAE with liquid embolic agents (including our institutional experience) were included in the analysis. The success rate was 99% (95% confidence interval [CI]: 98–100%), all complications rate was 1% (95% CI: 0–5%), major complications rate was 0% (95% CI: 0–0%), and mortality rate was 1% (95% CI: 0–6%). The rate of hematoma size reduction was 97% (95% CI: 73–100%), complete resolution 64% (95% CI: 33–87%), radiographic recurrence 3% (95% CI: 1–7%), and reoperation 3% (95% CI: 1–7%). No significant differences in outcomes were found between liquid and particle embolic agents. Sensitivity analyses revealed that liquid embolic agents were associated with lower reoperation rates in upfront MMAE (risk ratio 0.13, 95% CI: 0.02–0.95).
Conclusion
MMAE with liquid embolic agents is safe and effective for the treatment of CSDH. Outcomes are comparable to particles, but liquids were associated with a decreased risk of reoperation in upfront MMAE. However, further studies are needed to support our findings.
Keywords
Introduction
The incidence of chronic subdural hematoma (CSDH) has been increasing with time, with a shift toward aging populations. 1 CSDH is a collection of blood in the subdural space with hypodense or isodense, and sometimes with superimposed acute components on computed tomography scan. 2 The pathophysiology of CSDH formation consists of multiple steps involving trauma, dural border cell damage, and proliferation as a healing response that sustains inflammation, angiogenesis of fragile blood vessels resulting in new leaky vascular membranes, and fibrinolysis.3,4 Craniotomy and burr hole irrigation are the standard of care for symptomatic CSDH, but recurrence reaches up to 30%, often requiring reoperation.5,6
Middle meningeal artery embolization (MMAE) is an increasingly popular alternative or adjunctive treatment option for CSDH. 5 MMAE might act on the underlying pathophysiology; devascularization of the membranes reduces blood leakage and enhances hematoma resolution.7,8 Stand-alone MMAE has been suggested for asymptomatic or high surgical risk patients, pre-evacuation MMAE for severely symptomatic patients, and post-evacuation MMAE if emergent evacuation is needed. However, ongoing trials will answer when and for whom to perform MMAE. 9 Several studies have shown the efficacy of MMAE for CSDH using embolic materials, including particles and liquid embolic agents, with or without coils.4,8,10,11 A recent systematic review and meta-analysis on MMAE with particles for CSDH showed that it is an effective and safe technique, whether primary or concurrent with conventional treatment, 12 while another one reported results for all different embolic agents. 32 In this systematic review and meta-analysis, including our institutional experience, we aimed to assess the safety and efficacy of MMAE using liquid embolic agents in CSDH and compare them with particles.
Methods
Protocol and guidance
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 13 The protocol of this systematic review was registered on the International prospective register of systematic reviews (PROSPERO) with the registration code CRD42022374649.
Eligibility criteria
Original studies with at least three patients undergoing MMAE with liquid embolic agents for the treatment of CSDH (reporting post-embolization outcome data, in comparison with conventional treatment, other embolic agents, or as sole intervention) were deemed eligible for inclusion. The exclusion criteria were: (1) irrelevant articles, (2) articles not in English, Spanish, Arabic, Greek, Russian, or Romanian, (3) narrative or systematic reviews and meta-analyses, (4) letters to the editor, editorials, and comments without primary patient data, (5) conference abstracts and abstracts with no published full text, (6) in-vitro, animal, and cadaveric studies, (7) and articles with overlapping published data in a more recent series (checked based on the authors, center, and country). No search filters were used.
Literature search strategy
Two independent reviewers (GSS and IMM) searched through the MEDLINE (via PubMed), Embase, Scopus, and Web of Science databases (end-of-search date: January 30, 2022) to identify eligible studies. We used the Covidence software platform for systematic reviews for all search and study selection stages. 14 The following search algorithm was used: (CSDH OR SDH OR “chronic subdural hematoma” OR “subdural hematoma” OR “refractory subdural hematoma”) AND (“middle meningeal artery” OR MMA OR “embolization”). The complete search strategy for every database can be found in Supplemental Table S1. A third reviewer (OS) resolved any disagreements. The reference lists of the included studies were then searched for missed studies based on the “snowball” methodology. 15
Data collection
Two reviewers (GSS and OS) independently extracted the data from the included studies using a standardized, prepiloted form. Any disagreements were resolved through discussion with a third reviewer (JVS) when necessary. The following data were extracted from each study: (1) study characteristics: first author, publication year, design, country, and number of patients/cases, (2) patient characteristics: age and sex, (3) procedure characteristics: type of embolization, (4) efficacy outcomes: radiographic recurrence (an increase in hematoma thickness on follow-up imaging), technical success (no procedure abortion), hematoma size change, reoperation (required surgical evacuation for recurrent or persistent CSDH after MMAE), (5) and safety outcomes: mortality (all cause), major complications (any adverse event related to the endovascular procedure that caused permanent morbidity, including new neurologic deficit, blindness, or acute ischemic or hemorrhagic changes on imaging), and any procedure-related complications.
To group outcomes, we defined three indications: (1) Upfront or primary or stand-alone MMAE: patients with a previously untreated CSDH; (2) Prophylactic or adjunct or concurrent MMAE: before or after surgical evacuation without evidence of interval postoperative CSDH recurrence, usually during the same admission; and (3) MMAE for recurrent or reaccumulated CSDH after failure of previous surgical evacuation.
For each outcome, we collected data (1) for all cases combined using any liquid embolic agent (with or without coils, without being combined with particles), (2) separately for each liquid embolic agent (without being combined with coils or particles), and (3) for particles (with or without coils, without being combined with liquid embolic agents) only if reported in studies comparing liquid embolic agents with particles.
Quality of evidence assessment
The tool developed by the National Heart, Lung, and Blood Institute (NHLBI) was used to assess the quality of included studies. 16 The NHLBI scale ranges from 0–9; a score from 0–3 denotes poor study quality, 4–6 fair quality, and 7–9 high quality. The follow-up period was arbitrarily set to 90 days. Two independent reviewers (OS and IMM) rated each eligible study, and final decision was reached by discussion with another author (MMS). The mean and standard deviation (SD) were calculated.
Data pooling
Categorical variables were presented as frequencies and percentages, while continuous variables were presented as means and SDs. When continuous data were described as medians and ranges or interquartile ranges, we estimated the means and SDs by applying the methods by Hozo et al. 17 and Wan et al. 18 All relative rates were estimated based on the available data for the variables of interest. We performed all data handling according to the principles in the Cochrane Handbook. 19
Meta-analysis
We calculated proportions for all outcomes for all liquid embolic agents combined using a random-effects meta-analysis of proportions using a generalized linear mixed model to calculate pooled rates and 95% confidence intervals (CIs). Additionally, we compared all outcomes between particles and all liquid embolic agents combined using the Mantel–Haenszel method to calculate risk ratio (RR) and 95% CIs for each outcome. Statistical heterogeneity across studies was assessed with the I2 test (>50% suggests significant heterogeneity). In addition, to investigate sources of heterogeneity, we performed sensitivity analyses for each specific liquid embolic agent (proportions for nBCA [TruFill, Cerenovus, Irvine, CA, USA], Onyx [Medtronic Neurovascular, Irvine, USA], and Squid [Balt, Montmorency, France]), and each of the three indications (proportions for all liquid embolic agents combined and comparisons between particles and all liquid embolic agents). A p-value less than 0.05 was considered statistically significant. All analyses and plots were generated using R statistical software (version 4, 1.3) and R Studio.
Institutional experience
We performed a retrospective review of all MMAEs performed in our tertiary medical center between May 2019 and September 2022. Institutional review approval was obtained, and informed consent was waived due to the retrospective nature of the study. One hundred seventeen consecutive cases (90 patients) that underwent MMAE using liquid embolic agents or particles were included in this study. Data collection included all the aforementioned variables. The MMAE techniques used were similar to those described in the literature.
Results
Study selection
A total of 2196 documents were identified, and 1176 duplicates were removed. After screening 1020 documents by title and abstract, 883 were excluded. From the 136 documents in the full-text evaluation, 119 documents were excluded (Supplemental Table S2). After screening the references of the 17 initially selected studies, one more article was identified. Finally, 18 studies were included (Figure 1).
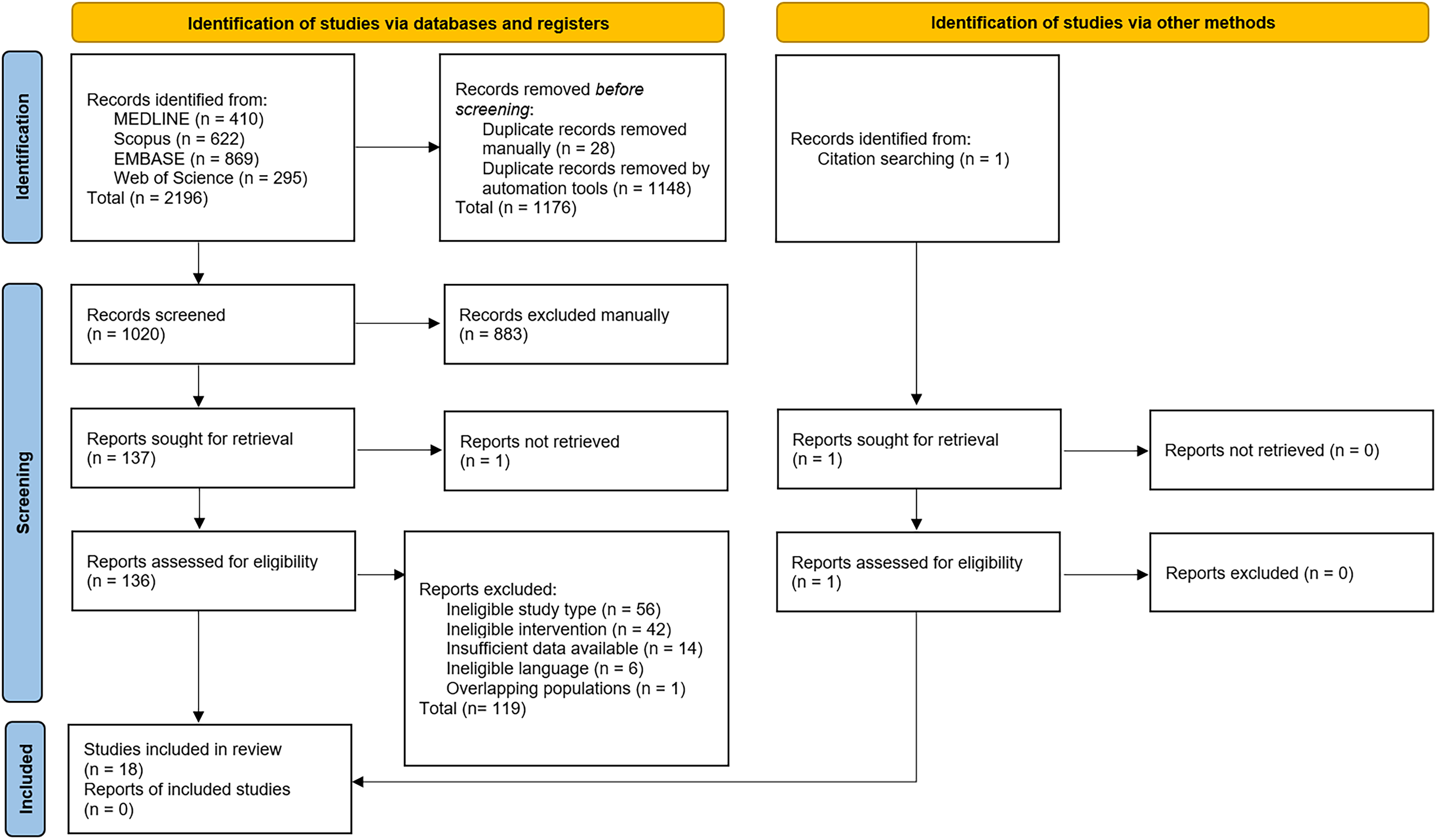
Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 2020 flow diagram for new systematic reviews.
Study characteristics
Eight studies were conducted in the United States,4,6–8,11,20–22 six in Japan,10,23–27 two in China,28,29 one in Russia, United Kingdom, and Germany, 30 and one in the United Kingdom. 31 A total of 507 cases of MMAE with liquid embolic agents were included in our meta-analysis, including our institutional experience. The mean age was 71.3 ± 12.7 years, and 106/415 (25.5%) were females after summarizing all cases with liquid embolic agents with available demographic data. Additional characteristics of all included studies are shown in Table 1, and all outcomes described are shown in Table 2.
Characteristics of the included studies.
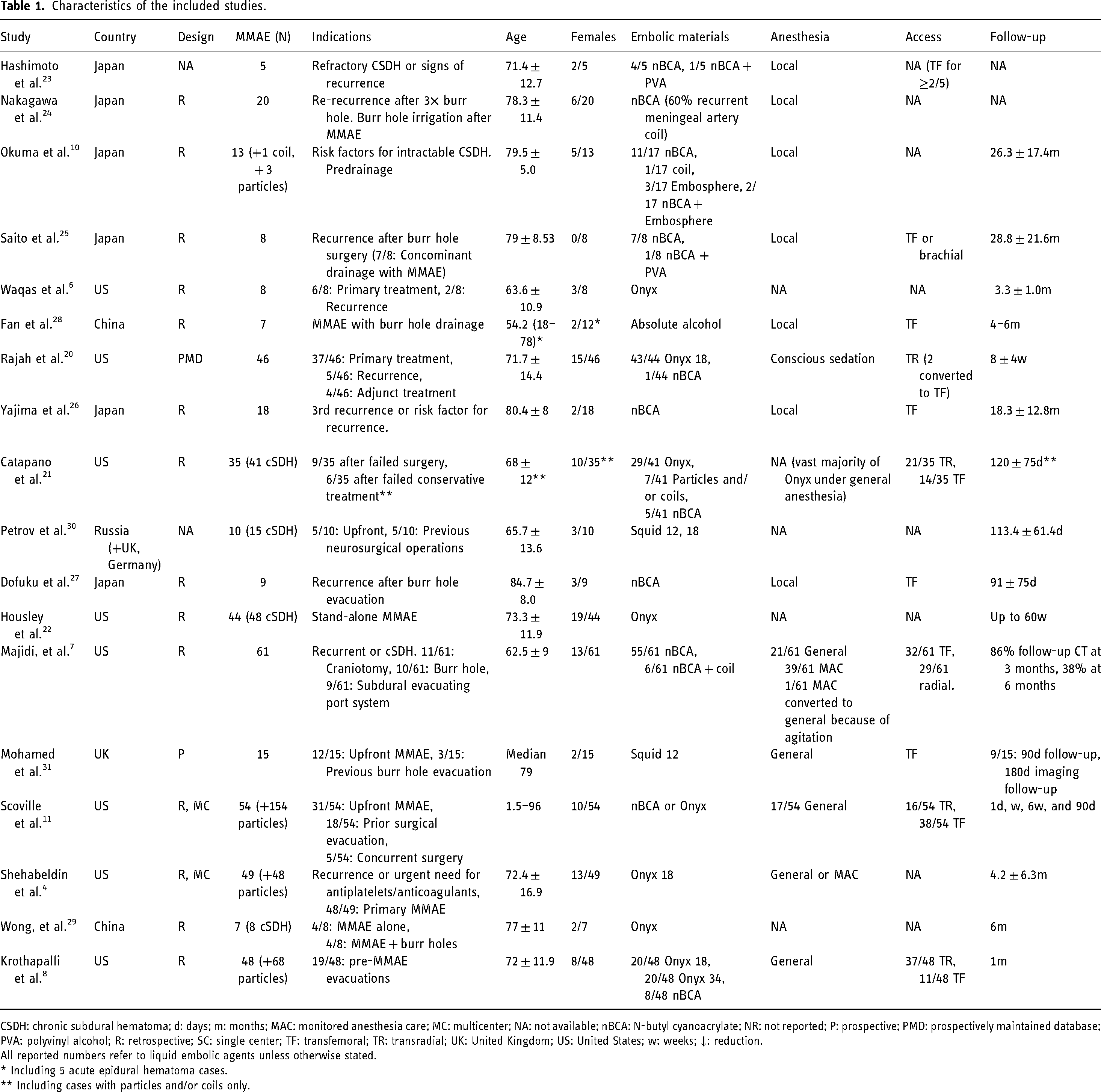
CSDH: chronic subdural hematoma; d: days; m: months; MAC: monitored anesthesia care; MC: multicenter; NA: not available; nBCA: N-butyl cyanoacrylate; NR: not reported; P: prospective; PMD: prospectively maintained database; PVA: polyvinyl alcohol; R: retrospective; SC: single center; TF: transfemoral; TR: transradial; UK: United Kingdom; US: United States; w: weeks; ↓: reduction.
All reported numbers refer to liquid embolic agents unless otherwise stated.
* Including 5 acute epidural hematoma cases.
** Including cases with particles and/or coils only.
Outcomes in the included studies.
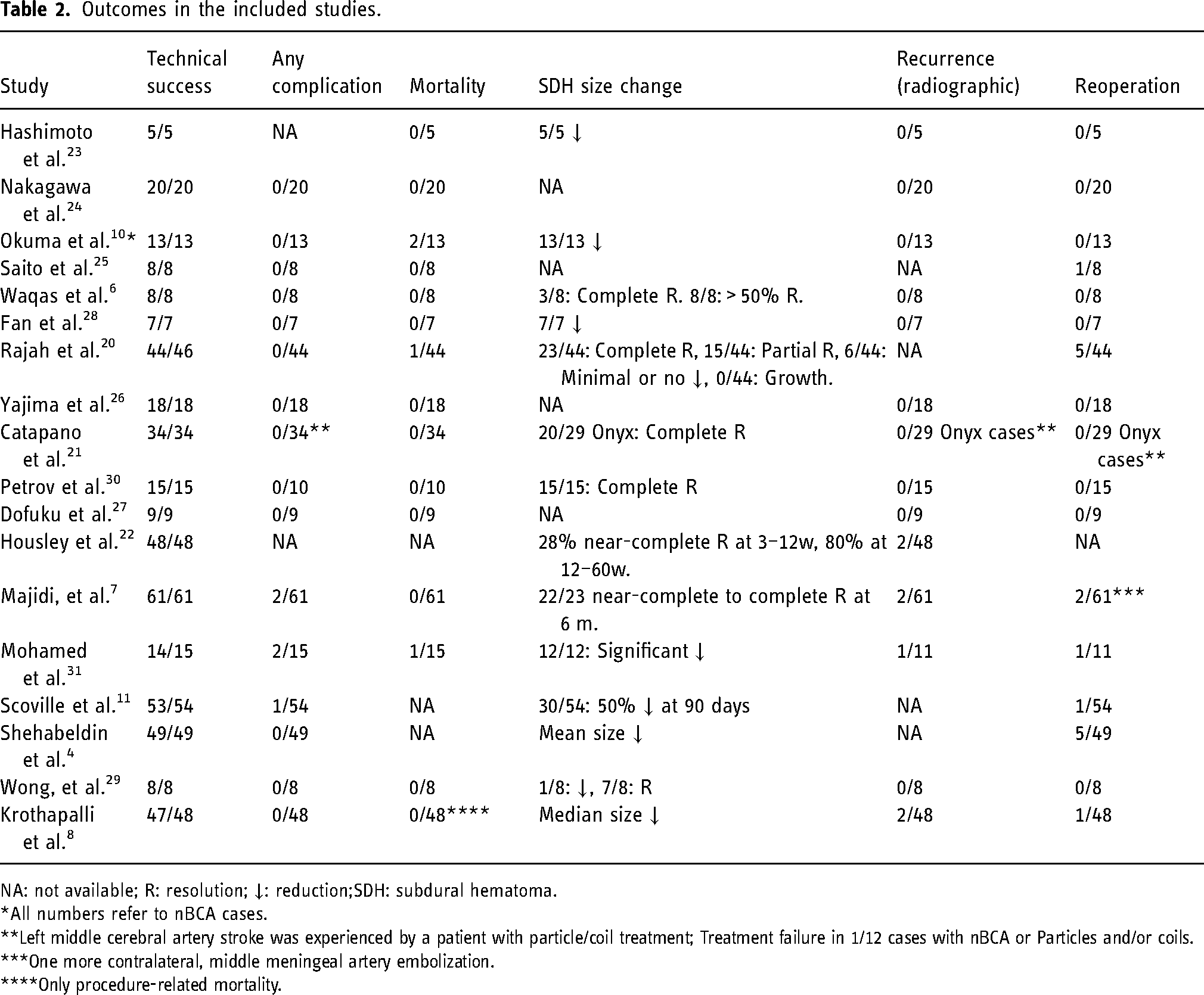
NA: not available; R: resolution; ↓: reduction;SDH: subdural hematoma.
*All numbers refer to nBCA cases.
**Left middle cerebral artery stroke was experienced by a patient with particle/coil treatment; Treatment failure in 1/12 cases with nBCA or Particles and/or coils.
***One more contralateral, middle meningeal artery embolization.
****Only procedure-related mortality.
Institutional experience
In our institution, 90 patients (117 cases) underwent MMAE. The mean age was 71.6 ± 10.4 years; 21.1% were females, 62.4% were stand-alone MMAEs, and liquid embolic agents were used in 32.5% of cases (Supplemental Table S3).
Quality of evidence assessment
The mean score of the NHLBI scale was 7.6 ± 1.1 (Supplemental Table S4). On average, the included studies were of good quality. Egger's tests showed no publication bias for any outcome in the proportion analysis for liquid embolic agents (Supplemental Table S5), but data were insufficient for Egger's tests in the comparison analyses. Graphical assessment of the Funnel plots identified publication bias in all outcomes for the proportion analysis for liquid embolic agents (Supplemental Figure S1), whereas no publication bias was identified in the comparison analyses for liquid embolic agents (Supplemental Figure S2).
Liquids embolic agents
MMAE with liquid embolic agents was technically successful in 99% (95% CI: 98–100%, I2 = 0%; Figure 2A) of cases. One case was technically unsuccessful, 11 one was aborted due to extreme tortuosity of the proximal MMA, 20 one was aborted due to sudden bradycardia after lidocaine injection into the parietal branch of the MMA, 20 one was aborted due to prominent orbital branches coming off the MMA with difficulty accessing MMA distally, 31 and one was aborted due to high-risk anatomical anastomosis. 8

Proportion meta-analysis of studies using liquid embolic materials. Forest plots for (A) technical success, (B) all complications, (C) major complications, (D) mortality, (E) hematoma size improvement, (F) complete resolution, (G) recurrence, and (H) reoperation.
The rate of all complications was 1% (95% CI: 0–5%, I2 = 0%; Figure 2B), and of major complications 0% (95% CI: 0–0%, I2 = 0%; Figure 2C). Complications included an access site hematoma, 7 a thromboembolic event (a transient episode of altered mental status and slurred speech after MMAE), 7 a retained microcatheter, 31 a small extradural hematoma secondary to rupture of one of the distal MMA branches, 31 and a postprocedural facial droop from Onyx infiltrating the meningeal branches of the skull base and resulting in facial nerve damage. 11 In our institutional experience, we encountered one wrist hematoma and a small temporoparietal infarct.
The mortality rate was 1% (95% CI: 0–6%, I2 = 0%; Figure 2D). All four reported deaths were unrelated to MMAE; one due to worsening congestive heart failure and aspiration pneumonia 1 month after MMAE, 20 one due to COVID-19 pneumonia on the seventh postoperative day, 31 and two without reported cause. 10 All deaths in our institutional experience were also unrelated to the MMAE.
SDH size was reduced in 97% (95% CI: 73–100%, I2 = 0%; Figure 2E) and completely resolved in 64% (95% CI: 33–87%, I2 = 54%; Figure 2F) of cases. The radiographic recurrence rate was 3% (95% CI: 1–7%, I2 = 0%; Figure 2G), and the reoperation rate was 3% (95% CI: 1–7%, I2 = 0%; Figure 2H).
Sensitivity proportion analyses on each liquid embolic agent separately (nBCA, Onyx, and Squid), are shown in Supplemental Table S5. Sensitivity proportion analyses on each indication separately (upfront, concurrent, and recurrent) are shown in Supplemental Table S6.
Liquid embolic agents versus particles
No statistically significant differences were found between liquid embolic agents and particles regarding success (RR 1.01, 95% CI: 0.99–1.02, I2 = 0%), all complications (RR 0.51, 95% CI: 0.10–2.46, I2 = 4%), mortality (RR 0.47, 95% CI: 0.13–1.67, I2 = 0%), improvement in SDH size (RR 1.02, 95% CI: 0.89–1.17, I2 = 0%), recurrence (RR 0.75, 95% CI: 0.30–1.85, I2 = 0%), and reoperation (RR 0.49, 95% CI: 0.21–1.13, I2 = 0%; Supplemental Table S7).
Sensitivity comparison analyses on each indication separately (upfront, concurrent, and recurrent) are shown in Supplemental Table S8. They revealed that liquid embolic agents were associated with lower reoperation rates than particles in upfront MMAE (RR 0.13, 95% CI: 0.02–0.95, I2 = 0%).
Discussion
This systematic review and meta-analysis of 18 studies (plus our institutional experience) shows that MMAE with liquid embolic agents is safe and effective, comparable to particles. Specifically, MMAE with liquid embolic agents was technically successful in 99% of cases, with a rate of all complications at 1%, major complications at 0%, and a mortality rate of 1%. CSDH size reduced in 97% and completely resolved in 64% of cases, but radiographically recurred in 3%, and was reoperated in 3% of cases. No significant differences were found between liquid embolic agents and particles, except that liquid embolic agents were associated with a decreased risk of reoperation compared with particles in upfront MMAE. To date, this is the first systematic review and meta-analysis to evaluate the safety and efficacy of MMAE specifically with liquid embolic agents of a high number of patients with CSDH and to compare them with particles. Given the emergence of this minimally invasive technique in the armamentarium of neurovascular surgeons and the absence of prospective randomized studies, this study offers valuable information.
MMAE is used both as a stand-alone treatment and as an adjunct to surgical evacuation of CSDH,9,32 but the optimal embolic agent and timing of MMAE are yet to be determined. Compared with MMAE alone, surgical evacuation (alone or combined with MMAE) seems to achieve better hematoma resolution in the immediate and early follow-up periods, but similar radiographic outcomes in late follow-up periods.22,33 Although MMAE and conventional management result in similar follow-up modified Rankin Scale scores, 34 MMAE is associated with decreased odds of reoperation compared to surgery. 32
Efficacy
Our efficacy results seem comparable with a recently published systematic review and meta-analysis by Ku et al., 32 including 22 studies with 382 patients undergoing MMAE and 1373 undergoing surgery for CSDH. They reported separate outcomes for each embolic agent, with rates close to ours, but without directly comparing liquid embolic agents to particles. 32 Abdollahifard et al. 12 reported a systematic review and meta-analysis on MMAE for CSDH, focusing on particles and comparing results to conventional treatment. Their results were comparable to ours regarding efficacy variables. Specifically, MMAE with particles was successful in 100% (95% CI: 19–100%) of cases, while 99% (95% CI: 98–100%) for liquid embolic agents in our study. SDH improved in size in 85% (95% CI: 66–94%) and completely resolved in 66% (95% CI: 39–86%) particle cases, whereas our results for liquid embolic agents were 97% (95% CI: 73–100%) and 64% (95% CI: 33–87%), respectively. Finally, they reported a radiographic recurrence rate of 5% (95% CI: 3–8%) and a reoperation rate of 5% (95% CI: 3–9%) for particles, while our numbers for liquid embolic agents were both 3% (95% CI: 1–7%). Our results support that liquid embolic agents for MMAE are as effective as particles.
When we compared liquid embolic agents with particles, we found no differences in success, hematoma size improvement, and reoperation. However, in upfront MMAE, liquid embolic agents significantly decreased the risk of reoperation by 87% compared with particles. Catapano et al. 21 and Kan et al. 5 found no differences in hematoma size change between particles and liquid embolic agents. Scoville et al. 11 also found no differences in retreatment after comparing liquid embolic agents with particles for each MMAE indication. Similarly, Krothapalli et al. 8 found no differences in outcomes after comparing liquid embolic agents with particles. Samarage et al. 35 found no differences in improvement rates between different embolic agents. Finally, Shehabeldin et al. 4 compared Onyx with polyvinyl alcohol particles (PVA). They found no differences in unplanned surgical evacuation, even after adjustment for baseline characteristics. Hematoma thickness at late follow-up was significantly smaller in the PVA group, but not persisting with the nonimputed multivariable model. This difference did not result in better clinical outcomes. 4 It should be noted that our comparison sensitivity analysis included only two studies, and even though I2 was zero, the number of studies was too small. Also, sensitivity analyses for the other two indications were not possible.
Safety
In our meta-analysis, the rate of all complications was 1% (95% CI: 0–5%), of major complications 0% (95% CI: 0–0%), and mortality 1% (95% CI: 0–6%). In the meta-analysis of Abdollahifard et al. 12 for particles, the complication rate was 4% (95% CI: 2–9%), and the mortality rate was 1% (95% CI: 0–3%). Even though complications seem more frequent with particles, as in the meta-analysis by Ku et al., 32 we found no differences.
Potential advantages and disadvantages
Although most studies have utilized particles, both liquid embolic agents (including nBCA, Onyx, Squid, and absolute alcohol) and particles have potential advantages and disadvantages.4,11 Our results suggest no significant difference between the two groups, except probably for upfront MMAE. Liquid embolic agents permanently devascularize the target vasculature,3,7,11 as they are not resorbed, thus avoiding delayed recanalization that may occur with particles.3,4,21 However, there is limited evidence that the resorption of particles over time may result in higher recanalization and recurrence compared to the permanent liquid embolic cast.4,8
Liquid embolic agents block proximal vessels along with the neovascular channels within the CSDH. 11 In contrast, because blood flow directs particles to their target, a proximally placed microcatheter may be required, increasing the risk of inadvertent embolization and the choice to use coils for proximal blockade. 11
The ideal microcatheter position when using liquid embolic agents is a distal “wedged” location without antegrade blood flow, minimizing reflux around the catheter tip. 11 The microcatheter must be positioned distal to any retinal anastomosis and the petrosal MMA branch to avoid permanent facial droop and blindness.4,11 Liquid embolic agents can achieve more diffuse and deeper membrane penetration when injected under pressure with a wedged catheter or balloon microcatheter. 7 Deeper penetration may prevent collateral vessels formation. 21 Because particles propagate with the flow depending on the size of the vessels, elderly patients who tend to have smaller vessels may benefit from liquid embolic agents that can penetrate smaller dura vessels distally. 4
Liquid embolic agents can be infused faster. 12 They can be directly and more easily visualized without the need for a delivery and visualization medium,3,4,11 helping to prevent reflux into dangerous anastomoses and making it easier to control the distribution and depth of embolization at the distal neovasculature.3,7,8,11
However, liquid embolic agents can lead to catheter retention and are more expensive, 3 although the more frequent use of coils with particles may narrow the cost difference. 11 In addition, dimethyl sulfoxide (DMSO) injection before Onyx is painful, requiring general anesthesia or prior lidocaine injection.3,4,21 Both Onyx and Squid can cause imaging artifacts, but Squid offers better visibility than Onyx for longer injections. 36 Absolute alcohol is widely available and of low cost but has not been widely used for MMAE. 28 Particles offer multiple size options and more consistent penetration 3 that blocks distal branches receiving collateral perfusion, 8 but may require longer fluoroscopy and procedure times compared to nBCA. 3
Limitations
Our study has several limitations, and any conclusions should be interpreted with caution. Most included studies were retrospective with potential selection bias, as a limited number of prospective and randomized trials were available in the literature. However, over ten ongoing clinical trials estimated to finish during the next 4 years aim to evaluate the safety and efficacy of MMAE using liquid embolic agents, assess the best timing of performing MMAE, and compare MMAE to conventional management. Between and even within studies, there is the potential significant confounder of individual operator variability for different approaches and techniques and a learning curve that can influence outcomes. Additionally, there is notable heterogeneity among the included studies regarding essential outcomes such as recurrence, the number of cases, bilateral CSDH, adjunctive coiling, embolization indications, and follow-up durations, as many studies did not report long-term outcomes. This heterogeneity makes the use of existing and future studies and trials in meta-analyses problematic. We compared all liquid embolic agents and all particles together for the primary analysis, although there are differences between the embolic materials; however, sensitivity analyses were conducted. Insufficient data were available regarding antiplatelet and anticoagulant use, symptoms and functional outcomes, microcatheter location, and the embolization of frontal or parietal branches. Finally, there was insufficient data to compare MMAE with medical treatment alone or surgical evacuation.
Conclusion
This systematic review and meta-analysis found that MMAE with liquid embolic agents is both safe and effective in terms of CSDH size reduction and resolution, recurrence, reoperation, complications, and mortality. Outcomes were comparable to those achieved using particles, but liquid embolic agents were associated with lower recurrence needing reoperation after upfront MMAE. However, well-designed clinical trials are necessary to support and validate our findings and determine any potential difference between embolic materials.
Supplemental Material
sj-docx-1-ine-10.1177_15910199231183132 - Supplemental material for Liquid embolic agents for middle meningeal artery embolization in chronic subdural hematoma: Institutional experience with systematic review and meta-analysis
Supplemental material, sj-docx-1-ine-10.1177_15910199231183132 for Liquid embolic agents for middle meningeal artery embolization in chronic subdural hematoma: Institutional experience with systematic review and meta-analysis by Georgios S Sioutas, Juan Vivanco-Suarez, Oleg Shekhtman and Irina-Mihaela Matache, Mohamed M Salem, Jan-Karl Burkhardt, Visish M Srinivasan, Brian T Jankowitz in Interventional Neuroradiology
Footnotes
Author contributions
All authors made substantial contributions. Study conception/design: GSS and BTJ. Data acquisition: GSS, OS, JVS, IMM, and MMS. Data analysis: JVS and GSS. Data interpretation: all authors. Manuscript drafting: GSS. Manuscript critical revision for important intellectual content: all authors. Approval of final version of manuscript: all authors. Guarantor of the study: BTJ.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics committee approval
Penn IRB Protocol: #850264
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
