Abstract
Purpose
Catheter-based intra-arterial chemotherapy (IAC) has revolutionized the treatment of retinoblastoma (RB). Variability in ophthalmic artery (OA) flow, either retrograde from external carotid artery branches, or anterograde from the internal carotid artery, necessitates multiple IAC techniques. We evaluated the direction of OA flow and identify OA flow reversal events over the course of IAC treatment as well in comparison to OA flow direction in non-RB children.
Materials and Methods
We performed a retrospective analysis of OA flow direction in all RB patients treated with IAC, along with an age-matched control group who underwent cerebral angiography at our center from 2014 to 2020.
Results
IAC was administered to a total of 18 eyes (15 patients). Initial anterograde OA flow was demonstrated in 66% (n = 12) of eyes. Five OA reversal events were identified (3/5 anterograde-to-retrograde). All five events were in patients receiving multiagent chemotherapy. No correlation was found between OA flow reversal events and the initial IAC technique. A control group of 88 angiograms representing 82 eyes (41 patients) was utilized. The anterograde flow was observed in 76 eyes (86.4%). Our control group included 19 patients with sequential angiograms. One OA flow reversal event was identified.
Conclusion
OA flow direction is dynamic in IAC patients. Anterograde and retrograde OA directional switches do occur and may necessitate delivery technique variation. In our analysis, all OA flow reversal events were associated with multiagent chemotherapy regimens. Both anterograde and retrograde OA flow patterns were observed in our control cohort, suggesting bidirectional flow can occur in non-RB children.
Introduction
Retinoblastoma (RB) is the most common primary intraocular malignancy of childhood, predominantly affecting individuals under 5 years of age. 1 The incidence of RB is estimated to be one case per 15,000–20,000 live births, equating to approximately 300 new cases in the United States annually.2,3 RB can affect one or both eyes, and if left untreated, RB can grow to invade local structures including the brain, metastasize, and ultimately prove fatal. However, modern treatments have led to survival rates approaching 100%.4,5 While enucleation has historically been the definitive treatment for intraocular RB, refinement in therapeutic techniques has led to increased emphasis on the secondary goals of globe preservation and vision optimization.6,7 Systemic intravenous chemotherapy (IVC) has been a mainstay of RB treatment since the 1990s, 8 but the development of intra-arterially delivered chemotherapy (IAC) in the early 2000s led to paradigm changes in our management of this disease9–11. IAC is based on superselective catheter-based delivery of chemotherapeutic agents directly to the orbital arterial circulation. This reduces the need for high systemic doses of chemotherapeutic agents necessary to achieve a therapeutic effect and offers superior efficacy to IVC in terms of tumor control and globe salvage.9,10,12
Secondary to the increased recognition of IAC as a safe and effective therapy in RB, increased focus has been applied to the angiographic anatomy of the ophthalmic artery (OA) and its anastomoses with branches of the external carotid artery (ECA).13,14 Normal anatomic variation and temporary hemodynamic considerations can result in an OA filling pattern that is predominantly anterograde, via the internal carotid artery (ICA), or retrograde, via anastomotic branches of the ECA. 15 These anatomic considerations, combined with occasional technical challenges in catheterizing the OA, have led to the development of several endovascular techniques to optimize drug delivery. 15 These techniques include direct catheterization of the OA,16–18 balloon occlusion of the ICA just distal to the OA origin to prevent distal delivery of the chemotherapeutic agent,10,19 balloon occlusion of the ECA with simultaneous ICA injection of the chemotherapeutic, 20 and direct catheterization of the ECA-originating orbital branch often via the MMA. 17 In this retrospective analysis, we reviewed our institution's consecutive series of RB patients treated with IAC, along with an age-matched control group to determine anatomic patterns of OA filling and to describe our drug delivery technique. We also sought to identify and describe any OA flow reversal events detected on sequential angiograms which necessitated the utilization of an alternative drug delivery technique.
Methods
This retrospective chart review was performed with the approval of the Vanderbilt University Medical Center Institutional Review Board 190171. All patients who underwent IAC at our institution between 2014 and 2020 were included in this analysis. Collected demographic and treatment information included patient age at the time of treatment, gender, number of IAC treatments and treatment technique utilized (Direct catheterization of OA, ICA balloon, ECA balloon with direct OA catheterization), laterality of treated eye, and chemotherapeutic agents administered. An age-matched control group was collected from the pool of all pediatric patients who underwent cerebral angiography for any indication during this same time period. All angiograms were assessed by neuroendovascular experts (SGR, HCG, MJF, MTF) with discrepancies arbitrated by neuroendovascular faculty (MTF). Observation of choroidal blush was used to define primacy of ECA versus ICA circulation. Reviewers were blinded to patient clinical information but not the OA access technique given the obvious nature of the treatment methodology on angiogram review. All available injections for each patient were reviewed, which included ICA and OA injections for all patients undergoing direct catheterization, and ECA and ICA injections for all other patients. All OA catheterization included docking of microcatheter at the OA ostium, with no wire or catheter navigation distally, as is standard at our institution. The standard setup for direct OA catheterization was 4 French dilator sheaths for femoral access and a marathon microcatheter for navigation and vessel access utilizing a “pull back” technique. Only gentle hand injections were performed. Statistical analysis was performed in Microsoft Excel for descriptive statistics (Microsoft) and Jamovi for analysis of variance (The jamovi project (2021). jamovi (Version 2.3) [Computer Software]. Retrieved from https://www.jamovi.org).
Results
IAC was administered to a total of 18 eyes across 15 patients, including three patients with bilateral disease processes (Table 1). A total of 66 sequential treatment angiograms were assessed. The number of IAC treatments ranged from 1 to 7 sessions per treated eye. Percentages of each treatment technique utilized are depicted in Figure 1. Of the 18 treated eyes, 66% (n = 12) demonstrated baseline anterograde OA flow patterns. A total of 5 OA flow reversal events were identified, each in unique eyes, with each instance necessitating alteration in drug delivery technique (Figure 2). All 5 events were observed in patients receiving multiagent chemotherapy, which constituted 5/16 (31%) eyes that received multiple chemotherapy agents. All 5/5 (100%) eyes had received previous multiple-agent chemotherapy. No correlation between a number of prior multiagent treatments and flow reversal was seen. No correlation was found between OA flow reversal events and the initial endovascular treatment technique. A total of 88 angiograms representing 82 eyes across 41 patients were evaluated as a control group (Table 1). Indications for angiography are depicted in Figure 3. Baseline anterograde OA flow was seen in 76 eyes (86.4%). Our control group included 19 patients (37 eyes) who underwent more than one sequential angiogram. A single flow reversal event was identified in a patient treated for an ECA arteriovenous fistula not in the region of the OA (Figure 4).
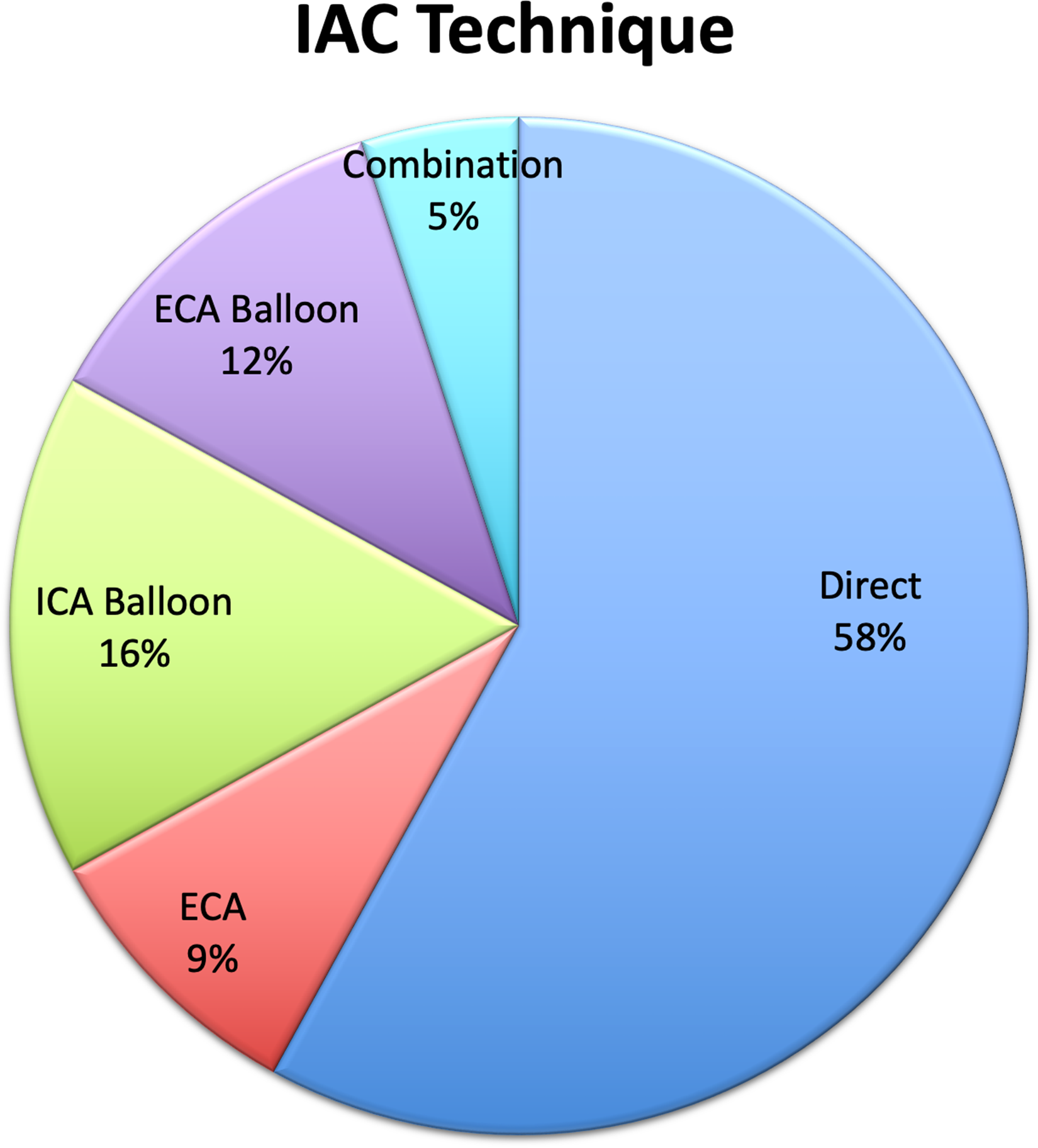
Technique used for IAC delivery in the setting of RB.
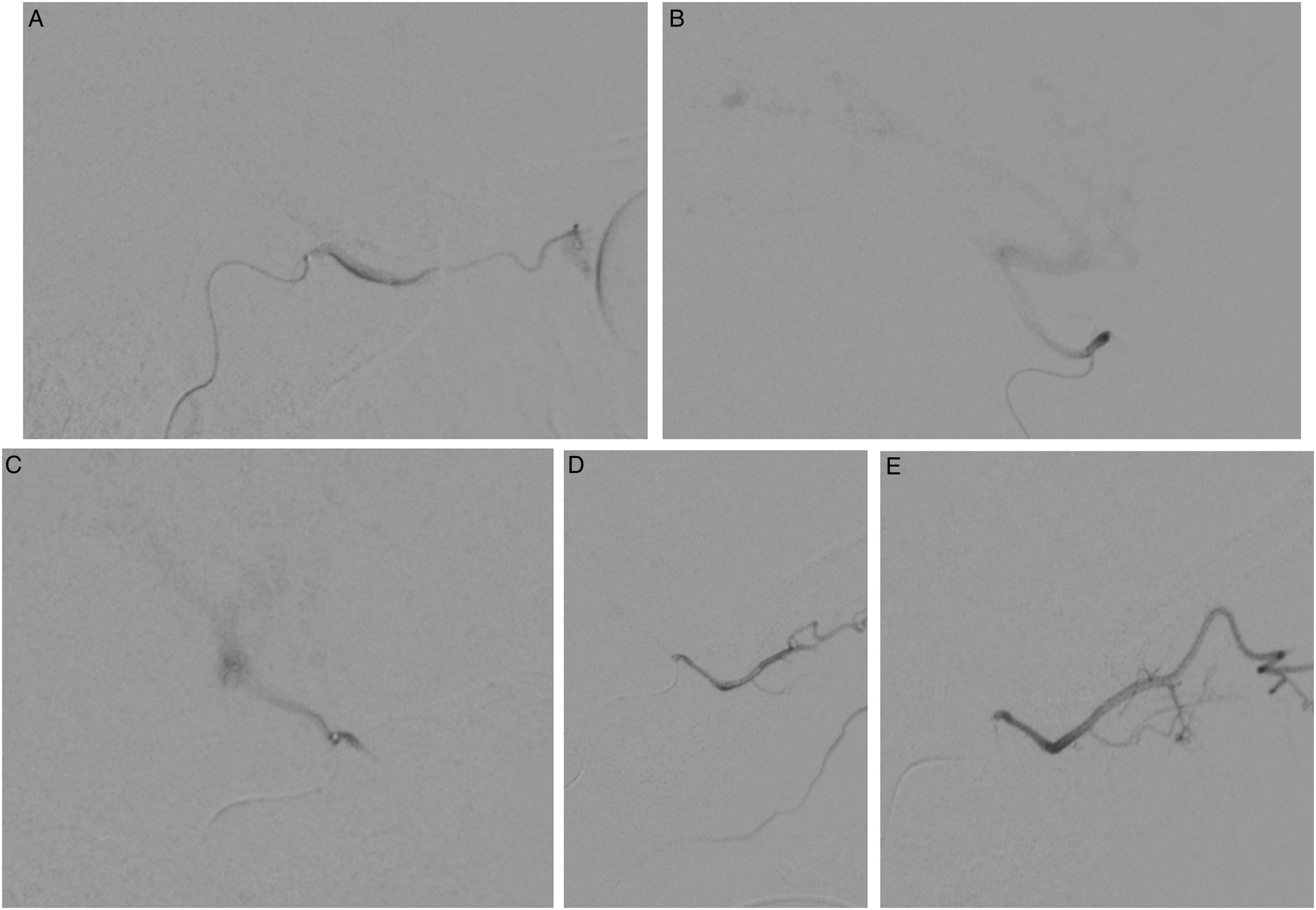
Ophthalmic artery angiogram with a demonstration of anterograde flow (A), and subsequent reversal of flow to retrograde in the same patient (B) demonstrated by ophthalmic artery reflux into the ICA. A second patient was initially noted to have retrograde ophthalmic flow (C), which required balloon occlusion of the ECA to induce anterograde ophthalmic flow (D). Ophthalmic artery angiogram in subsequent treatment demonstrated anterograde flow reversal (E).
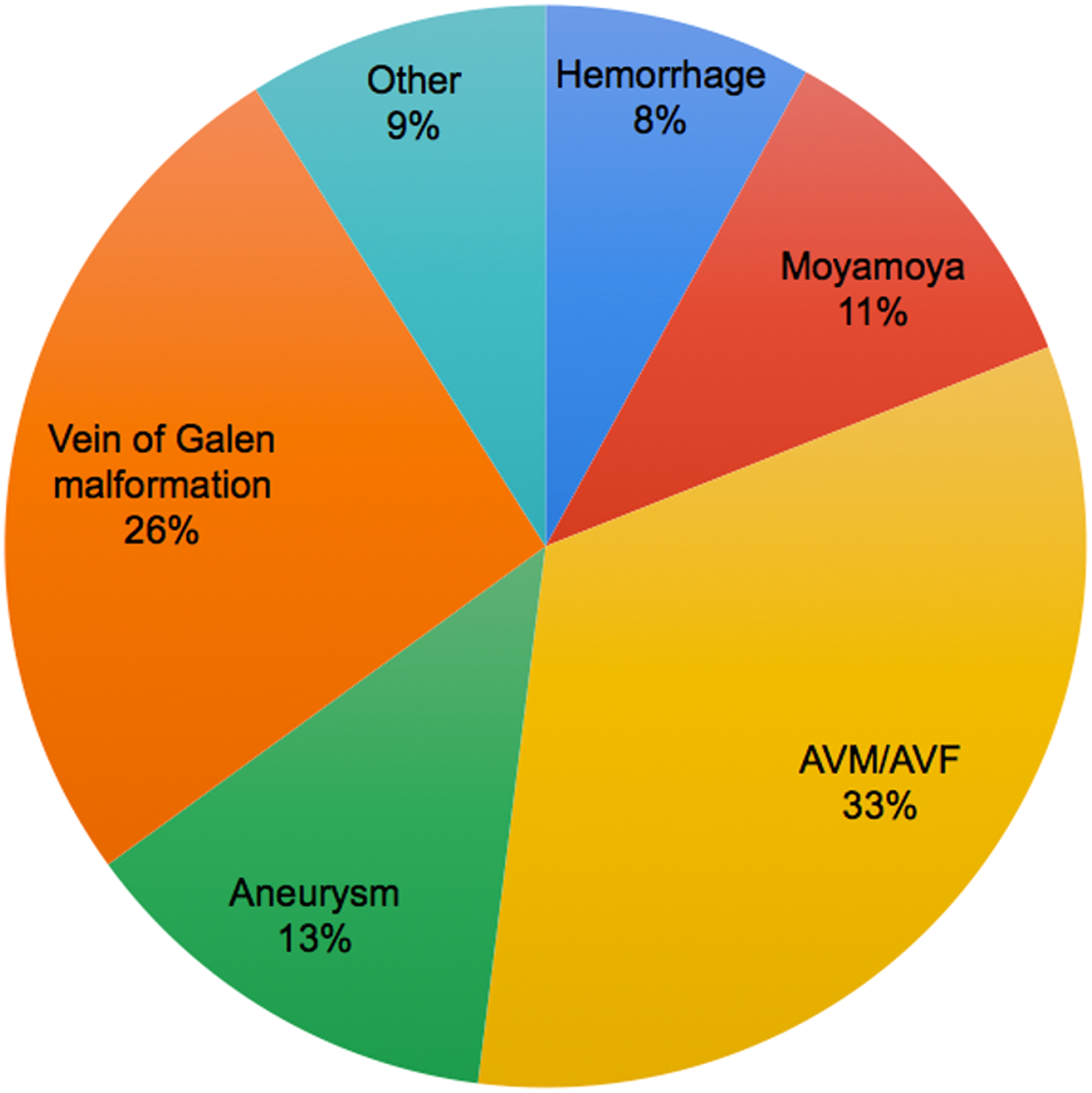
Indication for control angiograms.
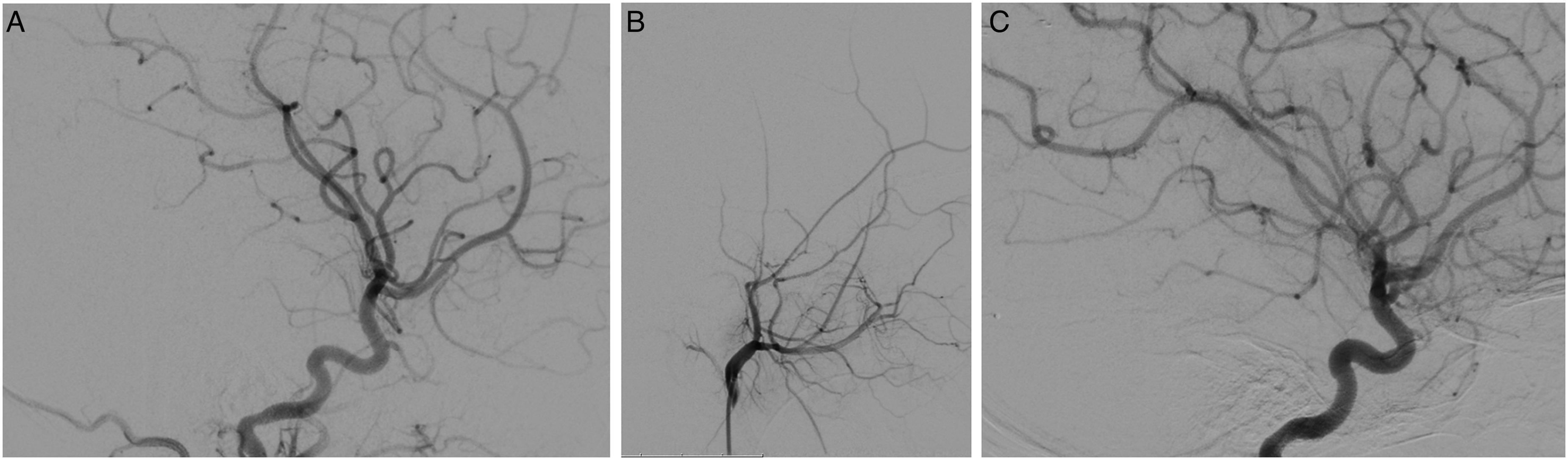
(A): Initial left ICA angiogram with a demonstration of anterograde ophthalmic artery filling. (B). L ECA injection after previous embolization of fistula from the left occipital artery and bilateral middle meningeal artery, with a demonstration of retrograde ophthalmic artery flow, and ECA flow from the anterior deep temporal branch (C). L ICA injection with a demonstration of cessation of anterograde ophthalmic artery flow, and retrograde flow to the left ophthalmic artery.
Demographics—RB patients treated with IAC compared to controls.
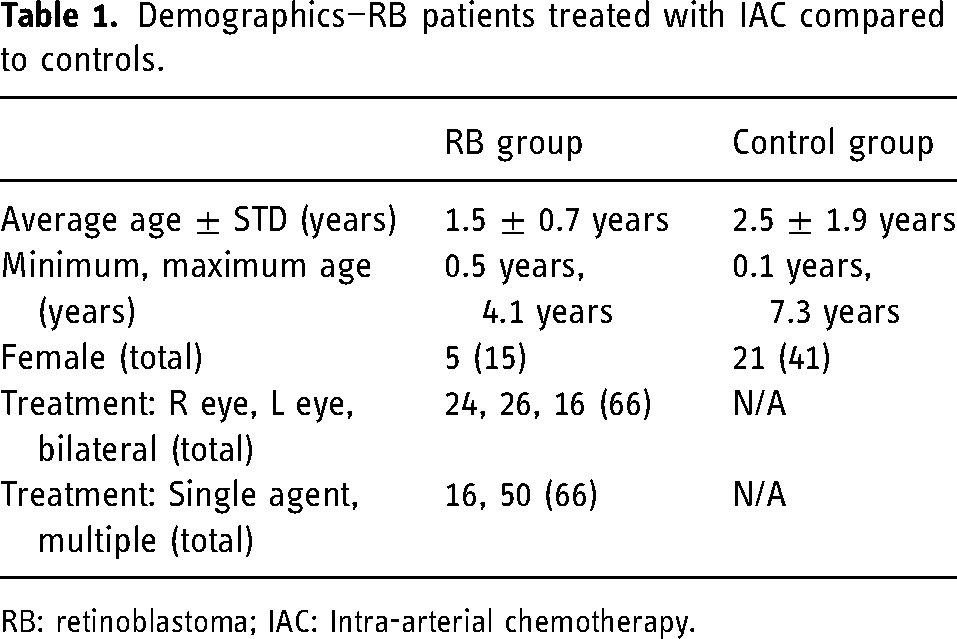
RB: retinoblastoma; IAC: Intra-arterial chemotherapy.
Discussion
In this study, we demonstrate the variability of OA flow patterns, either ICA (anterograde) or ECA (retrograde) dominant, in pediatric patients being treated for RB with IAC and in an age-matched control population. Importantly, we demonstrate that the OA flow pattern can reverse in both the IAC and control populations. These findings emphasize the importance of understanding OA anatomy and having command of the various IAC treatment techniques. Variation in OA flow patterns did not correlate with direct OA catheterization.
In prior studies, the dominance of ICA flow to the OA has been well-established in adults using ultrasonography of the orbit. However, OA flow patterns in children with RB have been described as being much more variable. In one study by Bertelli et al., directly accessing the OA allowed for successful angiographic choroidal blush and chemotherapy administration in 87% of IAC treatments. 15 This same study also mentions the variability of flow within the OA and concludes that these changes are likely not secondary to melphalan administration. Other studies have suggested that direct catheterization of the OA with subsequent vasospasm, along with the vasotoxic effects of chemotherapy, may alter flow patterns and induce dependence on alternate anastomotic pathways. 21 Building on Bertelli et al.'s work, we did not find that directly catheterizing the OA was associated with a subsequent reversal in OA flow. We observed a trend between multiagent chemotherapy and subsequent OA flow reversal events. The use of carboplatin and/or topotecan, which differ in toxicity as compared to traditional melphalan, may be associated with changes in OA flow reversal events not seen in Bertelli's cohort as these are more modern additions to our chemotherapeutic armamentarium. Our observed rate of OA flow reversal was somewhat higher than the 17% seen by Bertelli's group, who also did not find a significant correlation between single versus double agent chemotherapy and flow reversal events.
Although both anterograde and retrograde OA flow patterns have previously been observed in children with RB, no prior studies have sought to identify these flow patterns in a normal pediatric population. To that end, our finding that 13.6% of eyes in the control group demonstrate baseline retrograde OA flow suggests that there are innate differences in pediatric orbital arterial blood flow as compared to the adult population. The finding of an OA flow reversal event in a patient without RB is suggestive that these events may occur in the general pediatric population (with the caveat of this single reversal event was observed after embolization of bilateral distal ICA and ECA branches in the setting of a complex arteriovenous fistula separate from the OA). It should be noted that OA flow patterns in adults having previously undergone IAC as children remain unclear.
In humans, the OA arises from complex anastomoses between the primitive ventral and dorsal ophthalmic arteries, and ultimate regression of stapedial and hyoid branches resulting in the middle meningeal artery and inferolateral trunk as well as the OA. Thus, given the redundant and complex ICA and ECA vascular supply to the embryonic globe, the resultant mixed ICA and ECA orbital supply is embryologically unsurprising. 8 Our findings highlight the lack of understanding of the OA flow transition from the late embryonic mixed anastomoses to adult steady-state ICA supply. The OA flow variations seen in RB may represent normal transitions as opposed to sequelae of disease and/or IAC administration, as has been presumed throughout the development of IAC treatment.15,22 Despite this, variations in OA flow patterns are particularly notable during IAC treatments, and providers should be adaptable and prepared to alter treatment techniques between sessions; even more so given our findings of lack of predictability of flow reversal based on prior treatment characteristics. Reassuringly, there does not seem to be a treatment outcome difference for patients related to the IAC technique. 15
Our findings are not without limitation. The included case number is low given the rarity of RB, which limits the power of our findings, in particular the frequency of OA flow reversal events as well as correlated treatment factors. Furthermore, given the rarity of angiography performed in the pediatric population, our control group is heterogeneous and rife with vascular pathology. Notably, our singular finding of OA flow reversal in a control patient was in the setting of a complex arteriovenous fistula undergoing treatment. Nevertheless, the observation of both anterograde and retrograde flow dominance in this control population is striking and suggests embryologic origin. Further investigation into these OA flow patterns will likely help bridge our understanding of flow from the early post-embryonic anastomotic phase to adult-oriented OA anterograde pattern, and could potentially open doors to OA-based treatment of adult ophthalmic disease processes.
Footnotes
Author Contributions
MJF and MTF designed the study, MJF, HCG, and SGR performed the experiments, MJF, HCG, SGR, and NM analyzed the data, and MJF, HCG, SGR, NM, HCV, ABD, and MTF prepared the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the National Eye Institute grant NIH/NEI K08EY027464 [ABD], and by an unrestricted departmental grant from Research to Prevent Blindness to the Vanderbilt Department of Ophthalmology and Visual Sciences. ABD has a patent with Vanderbilt University Medical Center. ABD has received research funding from the NIH, an Alcon Research Institute Young Investigator Award unrelated to the material presented in this article, and research funding from Acrotech Biopharma through an investigator-initiated study separate from the data presented in this article. MTF is a consultant for Genentech, Cerenovus, Balt USA, and Oculus, and receives research funding from NIH, Genentech, Medtronic, Stryker, and Siemens.
