Abstract
Background
Flow diverters are an increasingly used treatment option for intracranial aneurysms. A recent addition to the European market is the DERIVO®2 Embolization Device (DED2), promising improved radiopacity. We aimed to assess the safety and efficacy of the DED2 regarding angiographic and clinical outcomes in ruptured and unruptured cerebral aneurysms.
Methods
We performed a multicenter trial at six interventional centers. Data were prospectively collected and all patients treated with the DED2 were included. The primary endpoint was angiographic aneurysm occlusion at 6 months as assessed by the O’Kelly Marotta (OKM) grading scale with a favorable outcome definition of OKM C + D. Clinical outcome was evaluated according to the modified Rankin scale (mRS).
Results
Between August 2020 and July 2021, 37 patients were treated with the DED2 and were included in our analysis. Five patients presented with ruptured aneurysms. Median age was 60 years, 27 patients were female, and 10 male. Median mRS was 0 (range 0–4). Mean aneurysm size was 8.9 ± 7.1 mm with a mean neck size of 6.5 ± 6.1. The DED2 fully opened at deployment in all cases. Clinical follow-up was available for 30 patients (81%). Twenty-five (83%) had an mRS of 0 or 1. Three patients with ruptured aneurysms died during the follow-up period. No treatment-related major morbidity was observed. Follow-up imaging was available in 27 (90%) patients, with 23 patients (85%) showing satisfactory aneurysm occlusion OKM grade C-D.
Conclusion
In this small cohort, the DED2 provided safe and effective treatment of ruptured and unruptured intracranial aneurysms.
Introduction
For the treatment of intracranial aneurysms flow diverters (FDs) have become a widely used and accepted alternative to other endovascular techniques, with a good safety and efficacy profile.1–5 FDs occlude the aneurysm by diverting the blood flow along the lumen of the parent vessel. This leads to the reconstruction of the parent vessel by formation of a neo-intima and intra-aneurysm thrombosis, eventually causing decoupling between parent vessel and aneurysm.6–11
The DERIVO®2 Embolization Device (DED2) is a novel self-expanding device consisting of up to 64 nitinol wires depending on device diameter promising to provide better X-ray visibility as well as a more homogeneous distribution of radial force and porosity. 12 It is covered with a thin surface layer of oxides and oxynitrides with the aim to reduce thrombogenicity. The second generation of the device was introduced in 2021, providing improved radiopacity due to a higher rate of platinum-iridium core wires. Unlike the first generation, it does not need distal and proximal device markers. The concept of the transport wire marker with a radiopaque transport wire tip has remained unchanged. A good safety and efficiency profile for the DERIVO® 1 has been reported in the literature.13–16 To date there are no clinical angiographic data on the technologically improved DED2.
The objective of the current study was to report clinical and angiographic outcomes of the novel DED2 at 6 months. We evaluated prospectively kept multicenter data on the DED2 regarding safety and efficacy in the treatment of ruptured and unruptured aneurysms of the anterior and posterior cerebral circulation.
Material and methods
The analysis was approved by the local ethics committee (19/21). A retrospective chart review of all patients treated with the DED2 from August 2020 to July 2021 was performed. Patient clinical data, demographics, aneurysm characteristics, procedural details, and follow-up data were extracted from prospectively collected data bases. Clinical outcome was graded by the modified Rankin scale (mRS), with favorable outcome defined as mRS 0–2 and major morbidity as mRS 3–5 or a deterioration by two points. Aneurysm occlusion was assessed using the O'Kelly Marotta (OKM) grading scale (A: total filling, B: subtotal filling, C: entry remnant, D: no filling).
Procedures
Treatment decisions were made at the discretion of the treating physician and confirmed by a neurovascular board. All procedures were performed by a board-certified neuroradiologist under general anesthesia. Systemic heparinization was at the discretion of the interventionalist. The DED2 was delivered through a 0.017–0.039 inch (micro-)catheter. The additional use of coils or balloon angioplasty was at the discretion of the interventionalist. Transform balloon Stryker or Scepter C Balloons were used for remodeling. Anti-platelet regimen standard of care was a loading dose with ASA (acetylsalicylic acid) 100 mg/d and Clopidogrel 75 mg/d for 7 days prior to intervention and for 6 months after treatment. In cases with insufficient platelet reaction in either Multiplate or VerfiNow testing, antiplatelet inhibition was changed to ASA and Prasugrel or Ticagrelor. For acute cases, either Tirofiban or Eptifibatide was used in combination with ASA.
Follow-up
Follow-up was performed upon discharge of the patient and at 3, 6, or 12 months, according to local guidelines. Aneurysm occlusion was assessed independently by the local interventionalist. MR angiography was accepted as follow-up when angiographic follow-up was not advisable.
Statistical analysis
Clinical data and endpoints were evaluated in a descriptive manner. Relative frequencies were calculated by the total number of patients in each category with non-missing values. Periprocedural complications were assessed descriptively.
Results
Patient baseline characteristics
A total of 37 patients were included in the analysis. Twenty-seven were female and 10 were male. Median age was 60 years (range 16–88 years). Twenty patients (54%) were asymptomatic at presentation. Five aneurysms were ruptured at the time of treatment. One ruptured aneurysm had been treated before. Eight patients presented for elective retreatment of previously treated aneurysms. Median baseline mRS was 0, with 25 patients (68%) with an mRS of 0, 10 (27%) with an mRS of 1–2, and 2 with an mRS > 2 (Table 1).
Baseline patient and aneurysm characteristics.

Aneurysm characteristics
Twenty-eight aneurysms (76%) were located at the sidewall. The most frequent location was the internal carotid artery (ICA) (76%), followed by vertebrobasilar (11%), and middle cerebral artery (MCA) (8%). There were 26 saccular (78%) and 11 non-saccular aneurysms. In eight cases (22%), a branch originated from the aneurysm sac. The mean aneurysm size was 8.9 ± 7.1 mm, with a mean neck size of 6.5 ± 6.1 mm (Table 1).
Treatment characteristics
All FD deployments were carried out successfully. One patient received two FDs in the same session due to proximal displacement of the device. Thirteen cases required resheathing maneuvers (range 0–3). Eleven patients received adjunctive coiling. Mean time from first maneuver to final implantation was 7 ± 12 min. One case of ICA dissection occurred while probing the vessel. One case of intraluminal thrombus and two cases of parent vessel stenosis were observed. Twisting of the device occurred due to technical difficulties in one case (3%) in which the FD could not expand properly to its nominal or vessel diameter due to a stenosis on the level of the aneurysm neck. A high vessel curvature was an additional factor presumably causing the observed twisting. The first FD (4.5 × 15 mm) was therefore not implanted and replaced by a 5 × 15 mm device, which was deployed successfully. A slight rebleed as determined by follow-up CT occurred in a patient with a ruptured aneurysm (3%). One patient experienced a hand paresis due to a small precentral infarction, secondary to acute in-stent thrombosis. The thrombus was treated with tirofiban, with nearly complete resolution at follow-up visits. No dislocations of the implanted devices during retrieval of the carrier were observed. Immediately after deployment, 21 patients (57%) showed an OKM grade of C or above. Treatment information is summarized in Table 2. Exemplary patient cases are depicted in Figures 1–3.
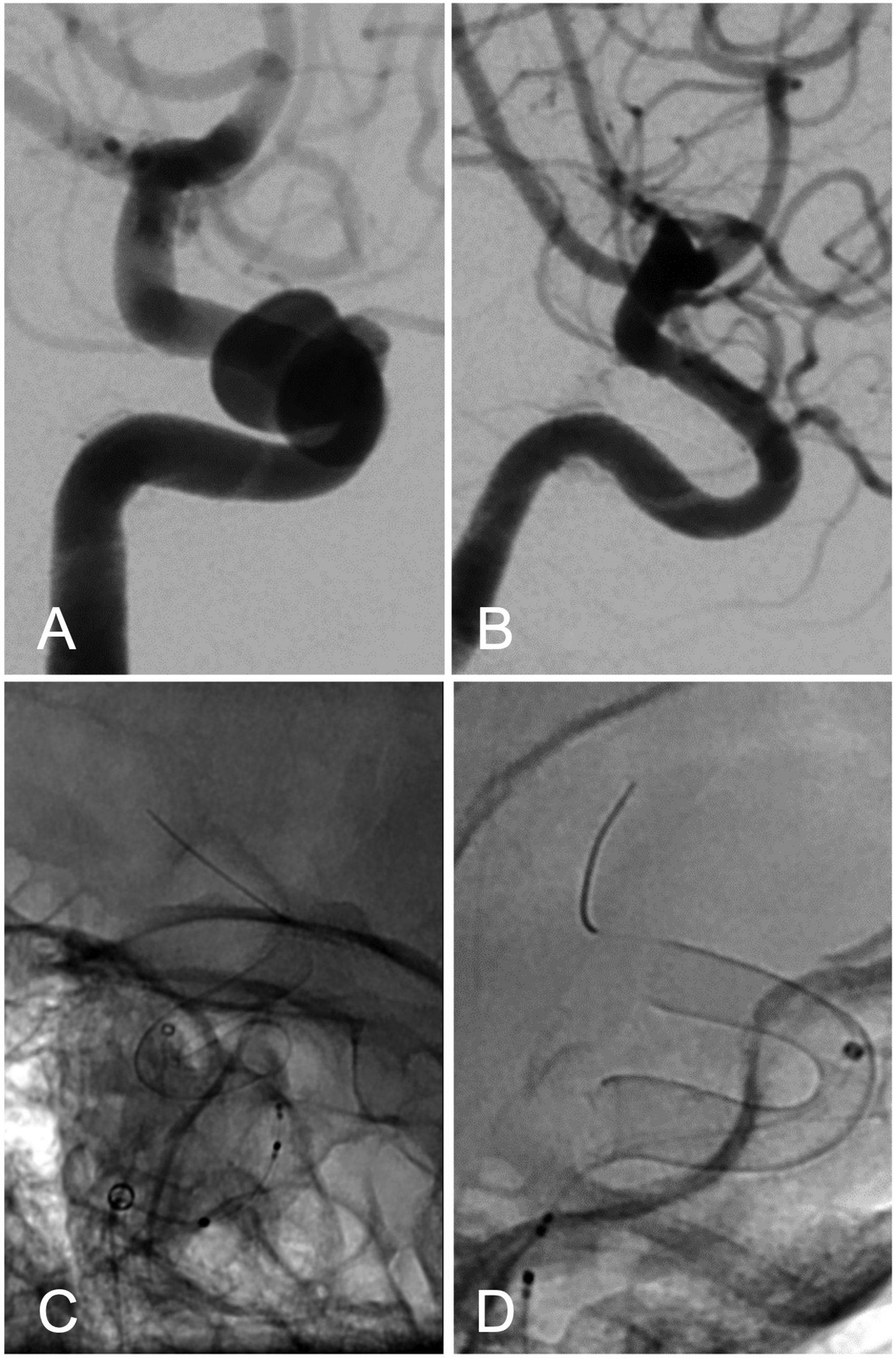
(A) Lateral angiogram prior to intervention showing a paraophthalmic aneurysm of the right carotid artery. (B, C) Fluoroscopic images lateral and a.p. view of a DERIVO 2 4.5 × 15 mm directly after implantation. (D) Six months follow-up confirming complete aneurysm occlusion.
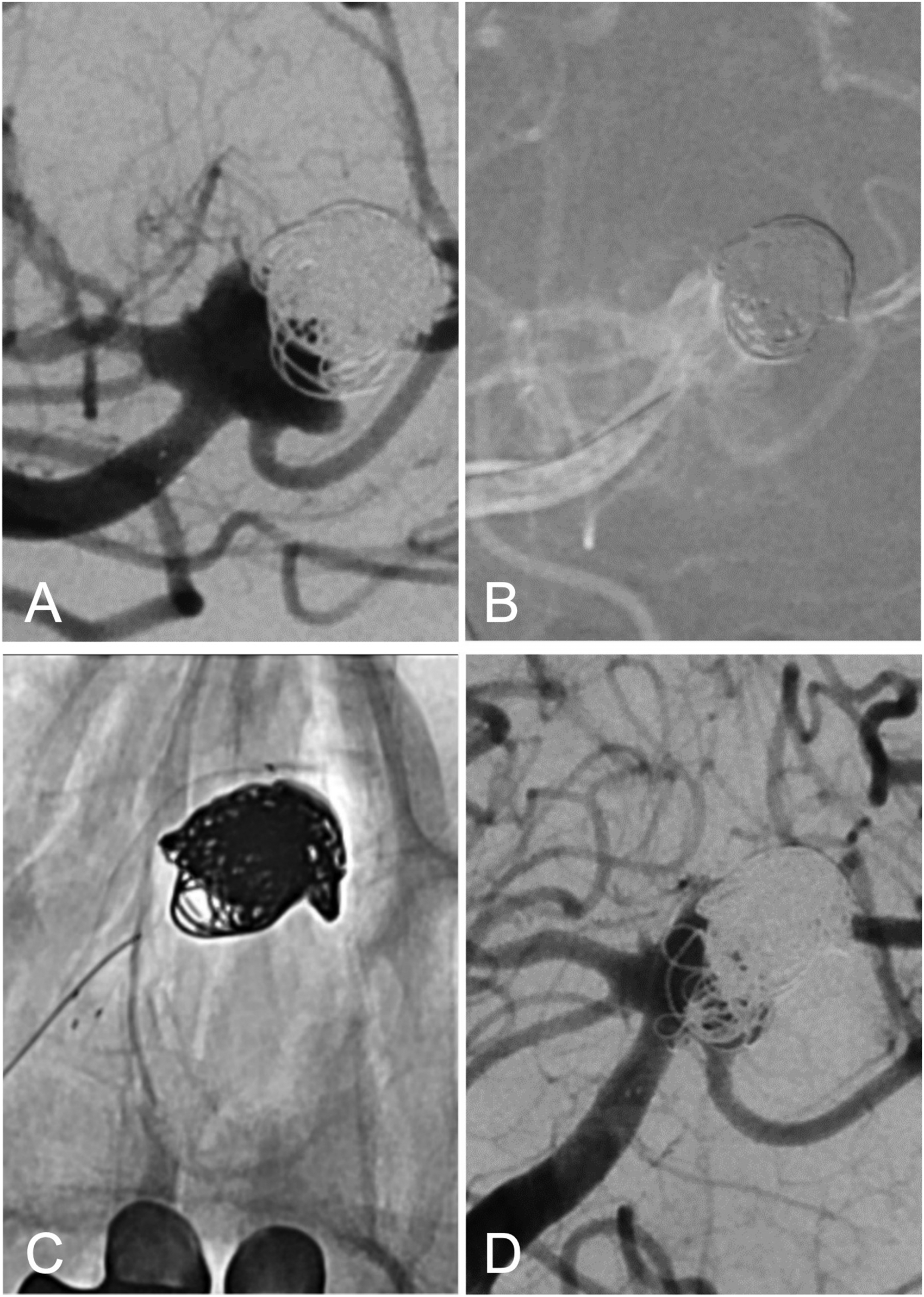
(A) A.p. road image of a symptomatic basilar tip aneurysm remnant which was treated with stent-assisted coiling before. (B) Road map a.p. image of the same aneurysm prior to catheterization of the left posterior cerebral artery (PCA) and the aneurysm sack. (C) A.p. fluoroscopic image after implantation of a DERIVO 2 3.5 × 15 mm. (D) Final angio run after DERIVO 2 deployment and additional coiling showing near complete occlusion of the aneurysm.
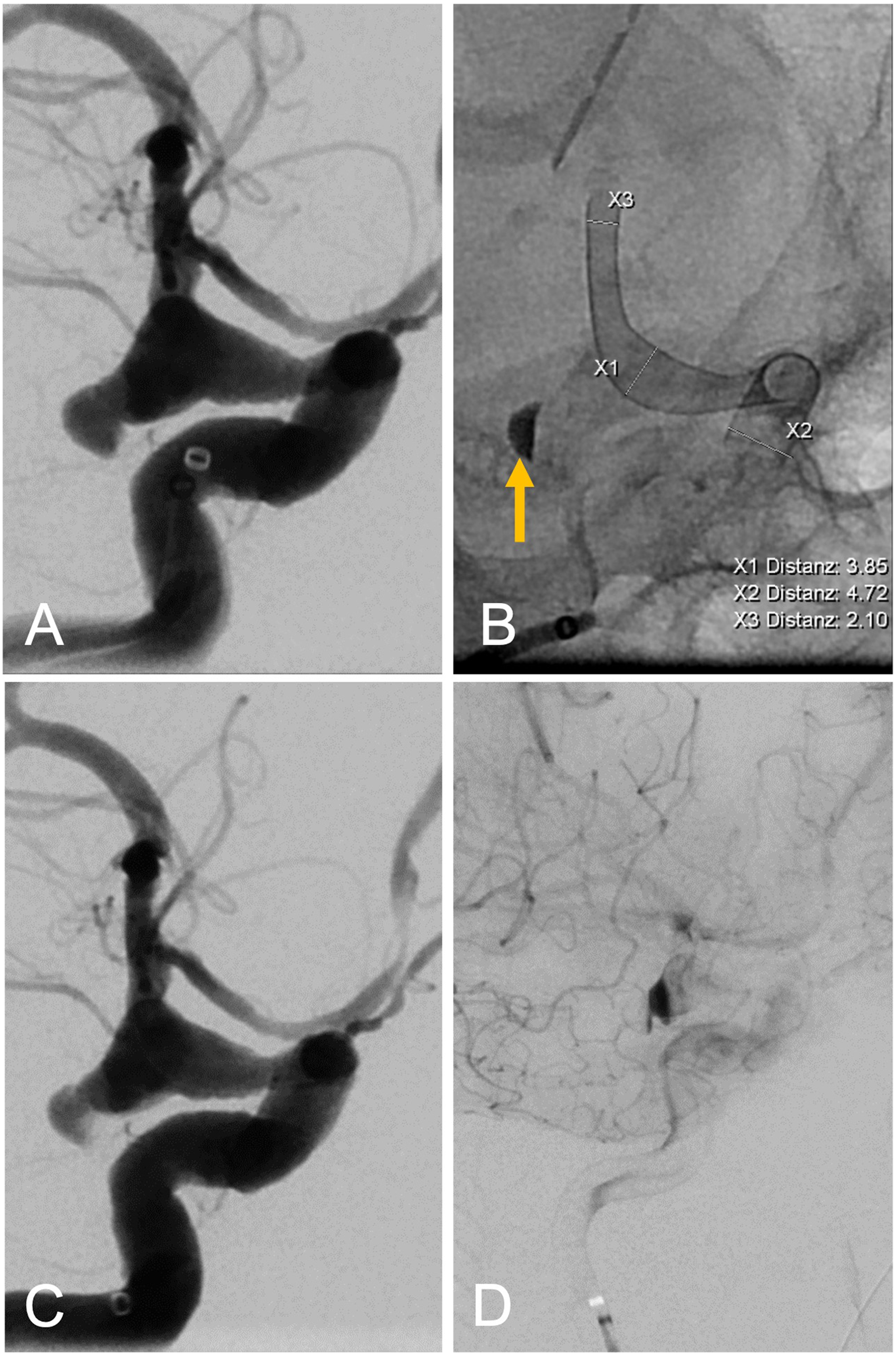
(A) A.p. view of an acutely ruptured, dysplastic distal carotid aneurysm of the right side. (B) A.p. fluoroscopic image directly after implantation of a DERIVO 2 5 × 20 mm showing initial contrast stasis in the aneurysm dome (arrow). (C) A.p. run after implantation showing incomplete filling of the dome (arrow). (D) Late arterial phase a.p. run depicting contrast stasis in the aneurysm (arrow).
Aneurysm characteristics and follow-up results.
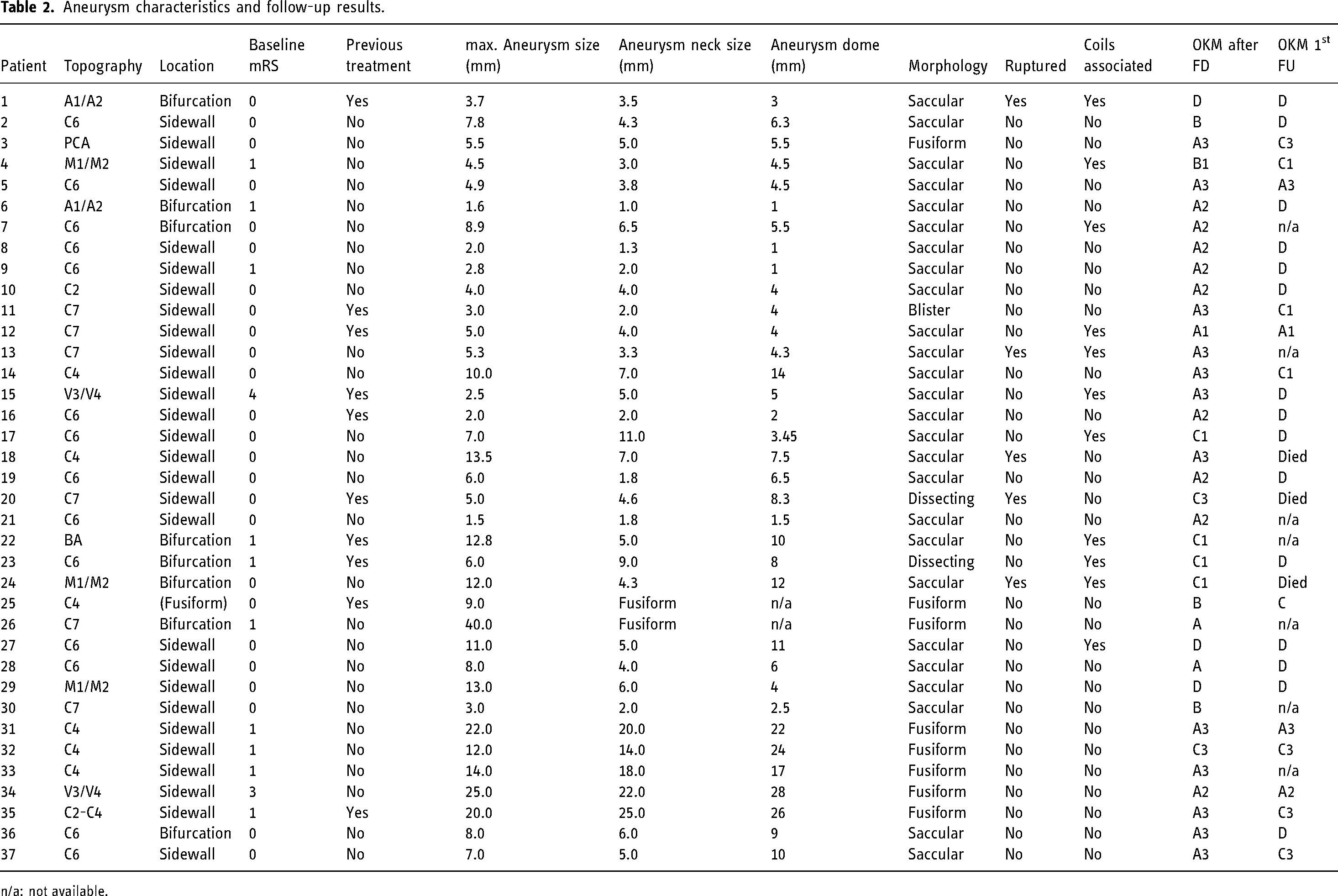
n/a: not available.
Follow-up data
Clinical and angiographic follow-up data were available for 30 (81%) and 27 (73%) patients, respectively (Table 3). Median follow-up time was 6 months (range 3–12 months). Twenty-five patients (83%) had an mRS of 0 or 1, with two patients with prior mRS >2 showing no clinical deterioration. For those patients with missing follow-up mRS, 6 out of 7 patients had an mRS of 0 at discharge and one patient with a baseline mRS of 1 did not show clinical deterioration. Three patients died during the follow-up period, all of which had ruptured aneurysms initially. No deaths were observed in patients with unruptured aneurysms. No treatment-related major morbidity was observed. Follow-up imaging was available in 27 patients (90%, 24 angiographic follow-ups, 2 MRA, 1 vaso-CT), with 23 patients (85%) showing satisfactory aneurysm occlusion OKM grade C-D (Table 3). Two patients were scheduled for retreatment, one due to coil occlusion of the distal contralateral V4 segment, the other due to insufficient aneurysm occlusion (A3) on follow-up.
Treatment characteristics.

Discussion
The aim of this study was to assess the safety and efficacy of the novel DED2 with regard to angiographic and clinical outcomes. Implantation of the device was successful in all cases. Treatment of intracranial aneurysms with the DED2 resulted in an overall occlusion rate of 85%, with no treatment-related major morbidity observed in our cohort. All three deaths in our cohort had ruptured aneurysms initially. No deaths were observed in patients with unruptured aneurysms (Table 4).
Periprocedural complications.

For the DERIVO 1 FD, two prospective multicenter studies have been published in recent years. Taschner et al. enrolled 96 patients with unruptured intracranial aneurysms. 3 The aneurysm occlusion rate at 18 months as assessed by Kamran 3 and 4 (Kamran grade 3: Neck remnant, Kamran grade 4: complete occlusion, comparable to OKM Scale C + D), was 89%, with a permanent neurologic morbidity rate of 3%. Only patients with a baseline mRS of 0–1 were included. The mean aneurysm size was 14.2 ± 16.9 mm, slightly larger than in our cohort. Forty-seven patients (49%) received additional adjunctive coiling. The study was paralleled by a prospective Brazilian cohort by Trivelato et al. with 146 patients with 183 aneurysms. 15 Baseline mRS was >2 in 20 patients (14%). Six aneurysms (3%) were ruptured. The mean aneurysm size was 6.7 ± 5.1, smaller than in our cohort. The reported complete aneurysm occlusion rate at 12 months was 89%. In a retrospective multicenter study on the DERIVO 1 on 49 unruptured and 10 ruptured aneurysms, Goertz et al. reported a satisfactory aneurysm occlusion rate as defined by OKM C + D of 89% at 6 months and a permanent morbidity rate of 2%. 13
A recent meta-analysis on the DED by Monteiro et al., including the three abovementioned studies, reported a complete angiographic occlusion rate of 81% in 481 patients. 17 The follow-up interval was 9–18 months. Adjunctive coiling was used in 26%.
Occlusions rates of 84% according to Raymond–Roy Occlusion Classification 1 were reported at 375 ± 73 days for the p64 flow diversion device in the prospective multicenter Diversion-p64 study with 420 patients. The reported morbidity rate was 2%. 18 For the Pipeline device complete occlusion rates were 87% at 12 months in the prospective multicenter Pipeline for Uncoilable or Failed Aneurysms study. 19 A retrospective study using the Flow-Redirection Intraluminal Device including 531 patients found an occlusion rate of 83% at 180 ± 20 days with a permanent morbidity rate of 0.8%. 20
Florez et al. reported an aneurysm occlusion rate of 80% in a meta-analysis using the Silk FD device. 21 A study on the Surpass FD device including 165 patients with 190 aneurysms showed near-complete or complete occlusion in 80% of patients at 6 months. 22 In a large meta-analysis including 1451 patients by Brinjikji et al., the 6-month occlusion rate was 76% with a procedure-related morbidity rate of 5%. 2 The analyzed studies used different FD devices; the DED was not included. Similarly, Zhou et al. reported an overall occlusion rate of 82% with a morbidity rate of 6% 23 using FD devices. The most commonly used devices were the pipeline embolization device and the SILK device. The DED was not included.
A slight subarachnoid rebleed after treatment occurred in one patient (3%). This is in line with the results published by Taschner (1/96) and Trivelato (1/146) on the DERIVO 1.3, 15 Other trials reported rates of intracranial hemorrhage of 3%.2, 22 No delayed ischemic stroke was reported in our cohort. We also did not observe any adverse events when switching from dual antiplatelet therapy to monotherapy during the follow-up period. The rate of in-stent thrombosis in our cohort did not differ significantly from the rates published earlier.
While the overall mortality in our cohort is high compared to other studies, all deaths occurred in patients with ruptured aneurysms. No deaths were observed in patients with unruptured aneurysms. The rate of ruptured aneurysm in our cohort was 16.2%, higher than in the study by Trivelato et al. (3%) and in line with the study by Goertz et al. (16.9%). We intentionally decided to include cases with ruptured aneurysms to demonstrate the usability of the device in both ruptured and unruptured aneurysms. Given our small sample size, the mortality rate within the ruptured aneurysm subgroup may not necessarily be attributed to the device or treatment with FDs in general, but rather mirror the severity of specific patient cases.
While comparisons across studies and devices are difficult due to different inclusion criteria and technical setups, our efficacy and safety results are within the range published for competing FDs and the DERIVO 1. However, while the FD did not open in 3%, displaced in 6%, and showed fish-mouthing in 12% of cases in the prospective trial by Taschner et al., 3 we observed somewhat lower rates, with 0%, 5%, and 3%, respectively. Adjunctive coiling was used in 30% of our cases, compared to 49% in the study by Taschner et al. The meta-analysis by Monteiro et al. reported rates of adjunctive coiling similar to ours. 17 The rate of balloon remodeling in our cohort was 11%, corroborating results on the DERIVO 1 (13%). 17 Our data does not allow us to determine whether this is due to the improved design of the DED2. Trivelato et al. reported four cases of improper DED expansion. 15
Limitations of our study are its retrospective nature and the relatively small sample size. Patient selection was at the discretion of the treating physician and may be subject to selection bias. Our follow-up of 6 months is relatively short and follow-up data was not available for all patients. Long-term outcomes of the DED2 will need to be assessed in further studies. Angiographic outcomes were assessed by interventionalists themselves and not by a core laboratory. We did not perform a matched-pair comparison with other aneurysm treatments. Post-interventional anti-platelet therapy was not standardized. Our sample size is too small to investigate the influence of different dual anti-platelet regiments on occlusion rates.
Conclusion
Our study demonstrates that the treatment of intracranial aneurysms with the novel DED2 is feasible with a safety and efficiency profile comparable to other FDs. Six months follow-up shows a high rate of satisfactory aneurysm occlusion and low rates of permanent major morbidity.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
