Abstract
Purpose
To review the different imaging modalities utilized in the diagnosis of Intracranial Atherosclerotic Disease (ICAD) including their latest development and relevance in management of ICAD.
Methods
A review of the literature was conducted through a search in google scholar, PubMed/Medline, EMBASE, Scopus, clinical trials.gov and the Cochrane Library. Search terms included, “imaging modalities in ICAD,” “ICAD diagnostic,” “Neuroimaging of ICAD,” “Evaluation of ICAD”. A summary and comparison of each modality's basic principles, advantages and disadvantages were included.
Results
A total of 144 articles were identified and reviewed. The most common imaging used in ICAD diagnoses were DSA, CTA, MRA and TCD. They all had proven accuracy, their own benefits, and limitations. Newer modalities such as VWI, IVUS, OCT, PWI and CFD provide more detailed information regarding the vessel walls, plaque characteristics, and flow dynamics, which play a tremendous role in treatment guidance. In certain clinical scenarios, using more than one modality has been shown to be helpful in ICAD identification. The rapidly evolving software related to imaging studies, such as virtual histology, are very promising for the diagnostic and management of ICAD.
Conclusions
ICAD is a common cause of recurrent ischemic stroke. Its management can be both medical and/or procedural. Many different imaging modalities are used in its diagnosis. In certain clinical scenario, a combination of two more modalities can be critical in the management of ICAD. We expect that continuous development of imaging technique will lead to individualized and less invasive management with adequate outcome.
Keywords
Introduction
Symptomatic intracranial atherosclerotic disease (ICAD) is among the most common causes of acute ischemic stroke (AIS). 1 Its worldwide prevalence is reported to be around 10% 2 in ischemic stroke patients and has been reported to be highest among minorities. 3 In some ethnic groups, such as Asians, it has been reported to be as high as 30 to 50% regardless of known risk factors such as diabetes, smoking and hypertension. 4
Our understanding of the management of symptomatic ICAD is evolving. Trials such as the Comparison of Warfarin and Aspirin in Symptomatic Intracranial Arterial Stenosis (WASID) 5 and Stenting versus Aggressive Medical Management for Intracranial Arterial Stenosis (SAMMPRIS) provided some initial guidance in the management of patients with AIS secondary to ICAD. 6 However, guidance for select cases of recurrent stroke despite maximal medical management remains controversial with endovascular therapy having an increased role. 7 ICAD can be diagnosed with a variety of different modalities such as catheter angiography, computed tomography, and magnetic resonance imaging. Other less frequently used modalities such as transcranial doppler (TCD), computational flow dynamics (CFD), and optical coherence tomography (OCT), may also have a role in categorizing higher risk lesions identified by previously mentioned modalities.
This paper is a narrative review of the concept of multimodal imaging approach for ICAD diagnosis and a statement on the current and future relevance of these imaging modalities in ICAD management.
Digital subtraction imaging (DSA)
DSA remains the gold standard modality for diagnosing and quantifying the severity of ICAD. 8 Since des Plantes described the idea of suppressing surrounding tissues to better visualize blood vessels in 1935, 9 Ovitt et al. reported the first initiative for the development of a digital video subtraction system for intravenous angiography in humans in 1978. 10 The physical principle behind DSA is the use of advanced radiologic technology to obtain a televised fluoroscopic image. It is then converted into digital data. In this process, an image is obtained prior to contrast injection and another image is obtained after. The two are superimposed, then surrounding tissues are digitally subtracted, leaving the space containing contrast only. The final process consists of logarithmically amplifying the image and then displaying it on a screen.11,12 The reason why DSA remains gold standard is because of its high spatial resolution, valuable anatomic and hemodynamic information provided.13,14 Although other non-invasive tests might provide similar details, the DSA provides much more spatial manipulation capabilities which is essential (Figure 1). However, Nicolas et al. reports that despite such amazing spatial resolution, 2D DSA might underestimate intracranial vessel luminal diameter due to its limited number of projections.15,16 This study, however, did not take into account high resolution 3D rotational angiography which eliminates this particular weakness. 17 Another interesting feature of DSA is the ability to use the Time To Peak (TTP) and the peak opacification to create single color-coded parametric perfusion images 18 (AngioViz) that result in improved visualization of perfusion or vascular flow. (Figure 2)
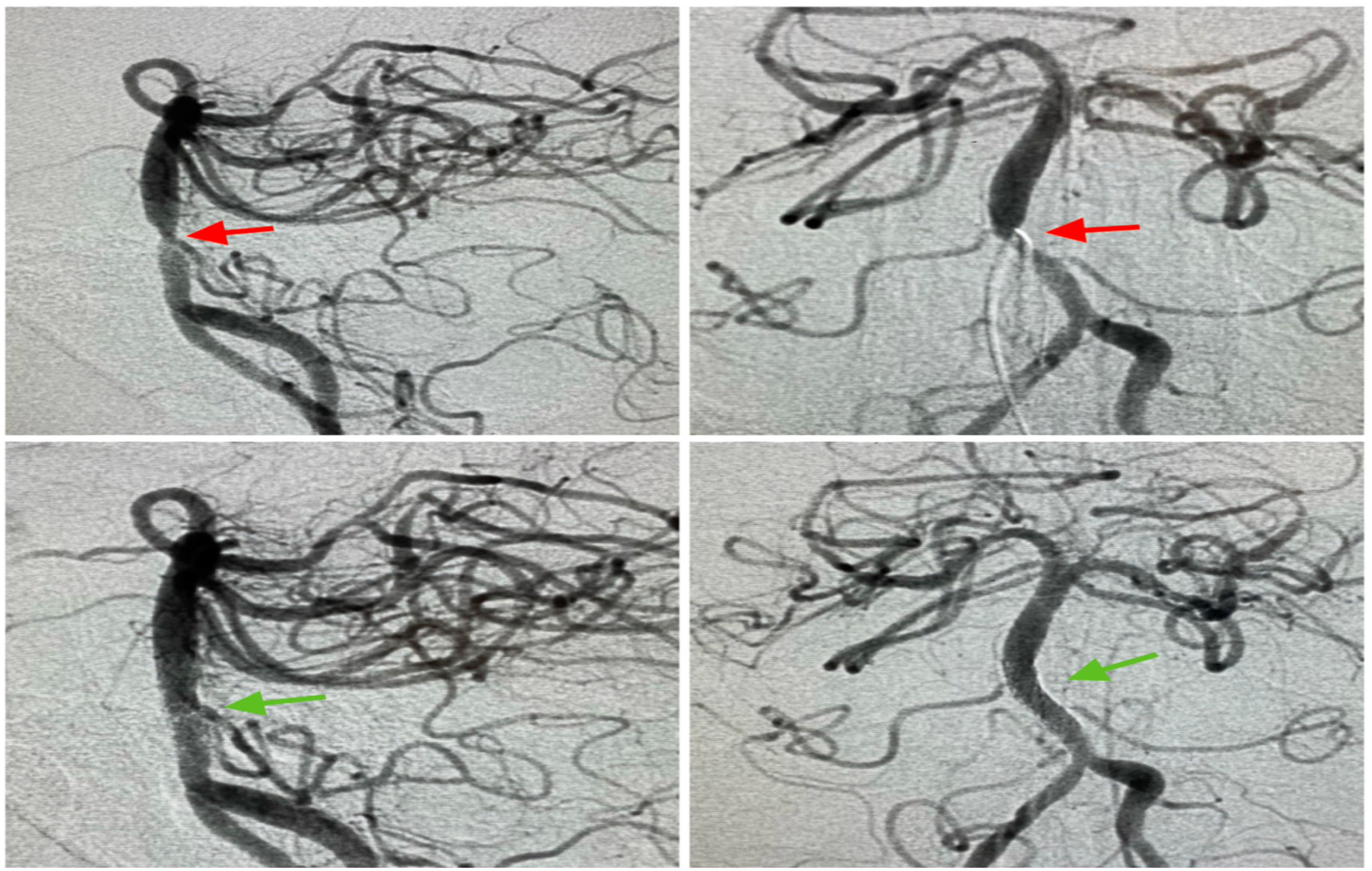
Mid basilar stenosis pre and post stenting on DSA.
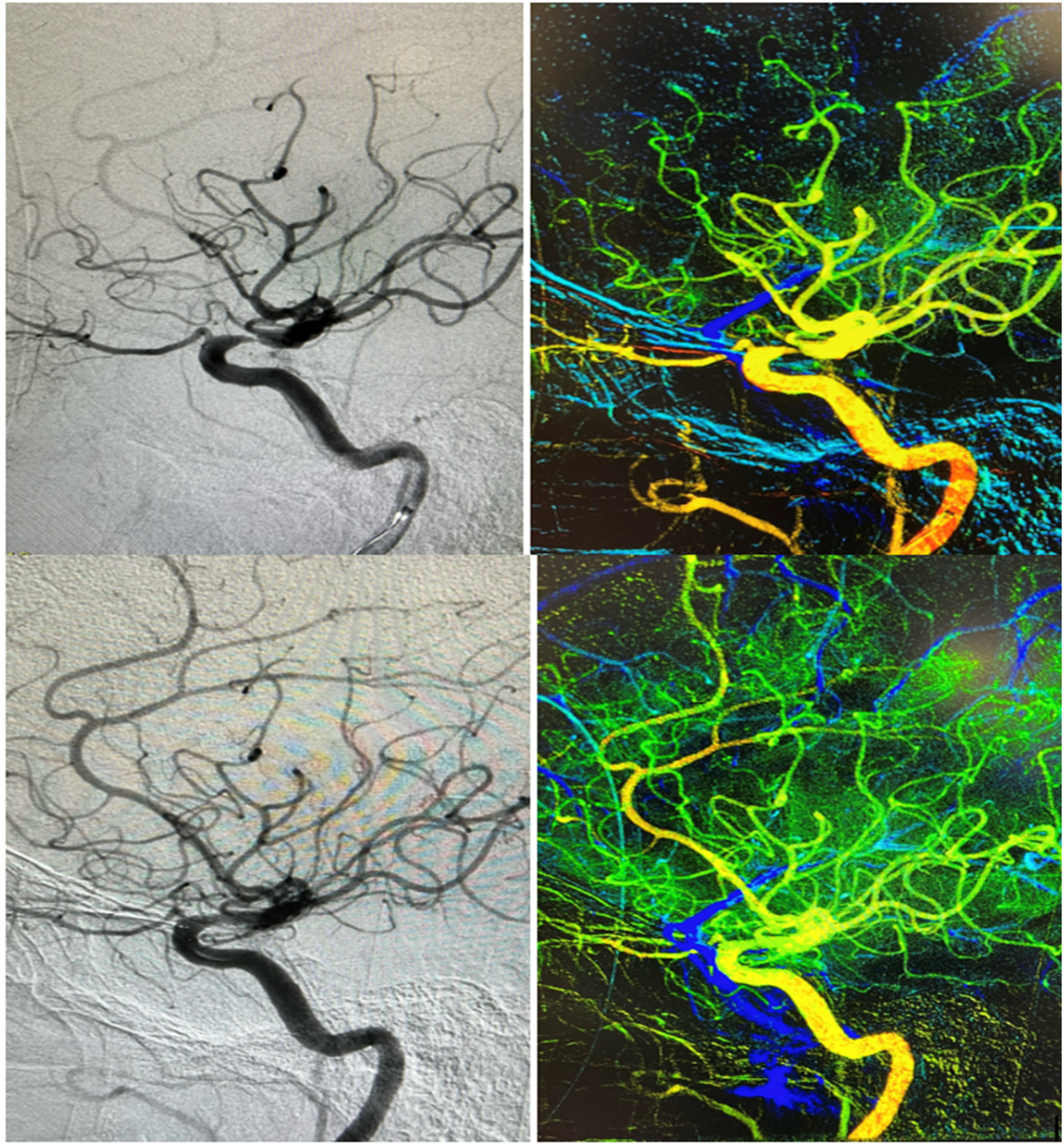
C5-6 ICA stenosis (above) treated with Wingspan stenting system seen via AngioViz feature.
DSA is also associated with a minimal, but not unheard of, risk of embolic stroke or vascular dissection(0.5%) resulting in transient neurological deficit (0.5%) or even permanent disability (0.7%), radiation exposure, contrast allergy (0.5%), access sites hematoma (4%), and acute kidney injury.19,20 These complications are rare but do occur often in older patients with cardiovascular risk factors, challenging tortuous anatomy such as a type 3 arch, or if the fluoroscopic time is longer than 10 min 21 and tend to occur less with more experienced operators. 22 Improvements in techniques such as using radial access make complications like a life-threatening retroperitoneal hematoma almost nonexistent. However, since DSA is not a risk-free procedure, non-invasive imaging studies are thus preferred in some cases. 23
Computerized tomography angiogram (CTA)
CTA was introduced in clinical practice around 1986 and was considered a maturing, less desirable technique compared to MRA and angiography for the diagnosis of vascular diseases. 24 There are three important metrics in CTA: spatial resolution, temporal resolution, and volume coverage. Advancement in technology has enabled the optimization of these metrics and allowed significantly finer imaging. 25 CTA can detect objects as small as 1 millimeter, can make a gantry rotation in one second,25,26 and can cover up to 30 cm per second. 25 These abilities make CTA a good tool to obtain vessel imaging. However, to date, there is no consensus on the value of CTA in ICAD. The Stroke Outcomes and Neuroimaging in Intracranial Atherosclerosis (SONIA) study reports that CTA has a negative predictive value (NPV) of 84% in terms of accurately identifying severe intracranial stenosis compared to DSA. 27 Other studies report a sensitivity of 97% and a specificity of 99.6% in identifying ICAD. In such cases, CTA is depicted as capable of accurately ruling in and ruling out stenosis as effectively as DSA and can be used to guide clinical management (Figure 3). 28 It has been demonstrated as well that CTA can outperform DSA in the posterior circulation in some instances such as the presence of very low or balanced flow states due to severe stenosis. 29 It is reported to be significantly reliable if arterial lumen is about 0.8 mm or more. Below that, CTA is unable to show the vessels or requires source images to show them. 30 Additionally, as previously reported, the invasiveness of DSA is the reason why other non-invasive tests are even considered. In a prospective study, Hirai et al. reports that the combination of CTA and MRA accurately Identifies stenosis in 98% of cases. 31
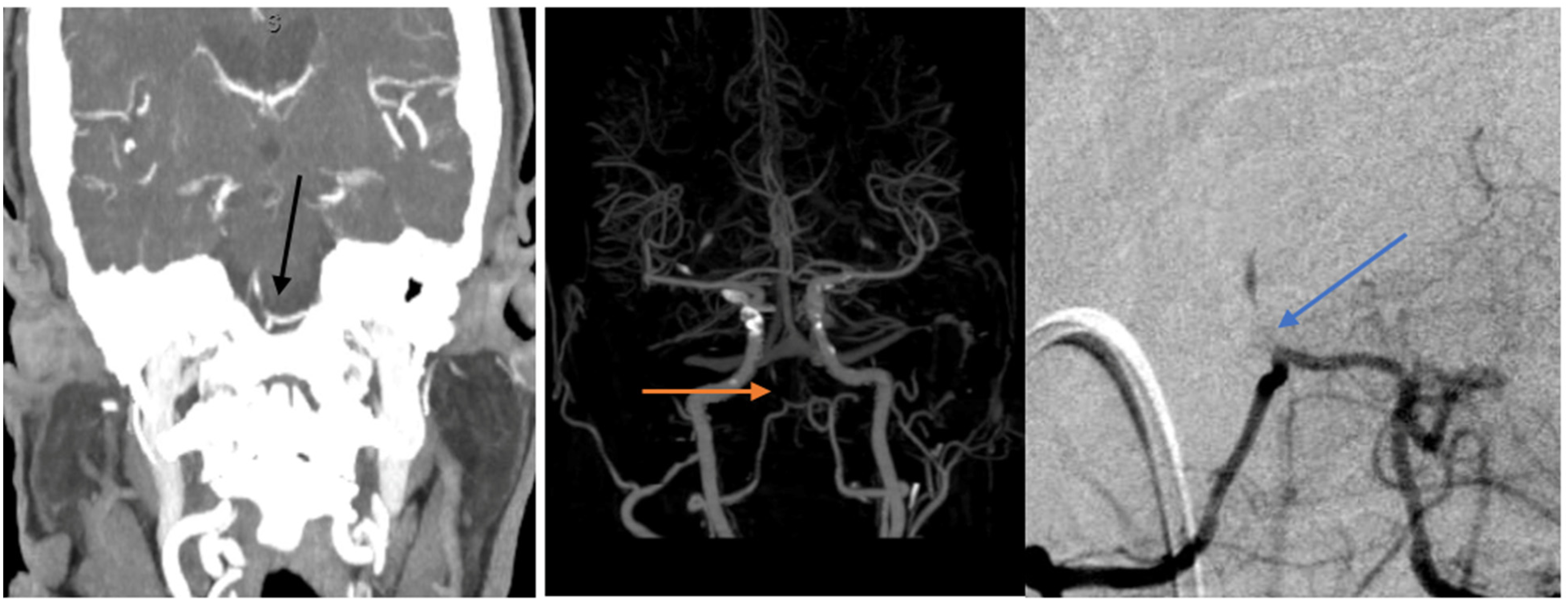
75YO F presenting with intermittent right sided weakness, vertigo, and emesis. Found to have severe proximal basilar artery stenosis on CTA coronal cut (left), then CTA reconstruction (center) then on angiogram (right).
The main advantage of CTA is non-invasiveness. Some of its disadvantages include: radiation exposure, contrast allergy and acute kidney injury (Table 1). Hirai et al. emphasize that in almost 20% of cases, CTA did not accurately delineate the vessels’ lumen when there is circumferential calcification and in the cavernous internal carotid artery (ICA), mainly due to equally enhanced cavernous sinus and when there is vessel kinking. 31 Skutta et al. reported similar concerns in regards of moderate stenosis for the petrous segments of the Internal Carotid Artery (ICA). 30
Some of these issues are mitigated with the use of reconstruction software developed with the help of advanced technology. Maximum Intensity projection (MIP) and surface and volume rendering imaging can eliminate surrounding noises, such as bone, and present a well-defined 3D reconstructed image. 32 Zhang et al. argue that this method of using iterative reconstruction algorithm software with low tube voltage as well as low concentration contrast solution provides excellent quality 3D imaging of the head and neck vessels with reduced ionizing radiation. 33 In interventional cardiology, the utilization of artificial intelligence and advanced computational analytics led to the development of Fractional Flow Reserve CT (FFR-CT) that provides high quality 3D images with precise anatomic and hemodynamic assessment of coronary arteries in a color coded fashion, thus limiting the need for catheter angiography. 34 This can potentially be extrapolated for cerebral imaging in the future.
Magnetic resonance angiography (MRA)
MRA visualizes hemodynamic flow through imaging techniques that discriminate flowing spins in moving particles from stationary particles. If an image is acquired from the amplitude of the moving spins, it is called time-of-flight (TOF) method. Whereas, if an image is acquired from the phase accumulated by the flowing spins it is called phase contrast method (PC-MRA). 35 The SONIA trial demonstrated that MRA can be used to accurately rule out intracranial stenosis since it has a negative predictive value (NPV) of 91%. 36 However, it has been more than a decade since this trial was published, and there is evidence of more advanced technological development in the field of non-invasive imaging, making such results no longer accurate. 8 Other studies report that MRA, compared to DSA, has an accuracy as high as 91% as the SONIA trial reported, but when combined with CTA, this accuracy increased to 98%. 31
Another method that can be used to improve accuracy of TOF-MRA is the contrast enhanced MRA (CE-MRA) with gadolinium. Gadolinium increases T1-weighted signal of the vasculature by significantly decreasing the relaxation time of the blood, and thus providing a better quality image. 35 Studies suggest CE-MRA perform at the same level or higher than TOF-MRA in many instances.37–39 Choi et al. reported that higher magnetic field strength (MRI) improves accuracy when compared to DSA. 3T TOF-MRA has an accuracy nearing 99% for high grade and complete occlusion with an interobserver agreement of 0.81. 16 Simply put, TOF-MRA and CE-MRA have demonstrated good PPV, NPV and accuracy in the diagnostic of ECAD and ICAD when compared to the standard reference.37,38,40
Relatively recently, zero echo time MRI (zTE-MRI) has been developed. It follows the same principles as ultrashort echo time (UTE) MRI. 41 UTE is MRI with very short relaxation time, T2, where spatial encoding and data acquisition are done as quickly as possible in order to achieve both high spatial resolution and signal-to-noise-ratio. In case of zTE, T2 is equal to 0. 42 Although initially designed for other parts of the body, Zhang et al. conducted a feasibility study comparing performance of zTE-MRA versus TOF-MRA in accurately detecting ICAD. This study showed that zTE MRA images were significantly higher in quality when compared to TOF-MRA (Wilcoxon rank test, p = 0.017) and zTE-MRA had higher sensitivity, specificity (78% vs 67%), and accuracy (96% vs94%) in identifying ICAD seen on DSA. 43
In PC-MRA, the flow velocity is encoded in the phase of MR signal allowing visualization of flow and at the same time allowing to quantify it. This leads to the development and the utilization of quantitative MRA (qMRA) in the study of cerebral vasculature. 35 Key information provided by qMRA is about hemodynamics within the stenotic segments and collateral circulation instead of a mere estimation of the degree of stenosis. Therefore, in many instances, qMRA is utilized as a predictive tool in regards to future risk of stroke as well as in the assessment of post intervention flow such as in post stent placement.44,45 For instance, in one retrospective study, patients who underwent stent placement for ICAD were followed up with qMRA and DSA. It was found that a 20% reduction of blood flow on qMRA correlates with more than 50% in stent stenosis on DSA. 46 In a case series, Serulle et al., using Non-Invasive Optimal Vessels Analysis software (NOVA), suggest that the utilization of qMRA can help stratify the patients who will likely benefit from revascularization or not. 47
Benefits inherent to MRA are non-invasiveness and the absence of ionizing radiation. 38 Disadvantages include longer time for acquisition, some pacemaker incompatibility and overestimation of stenosis in ICAD.36,39,40,48 This is especially true with tortuous anatomy where vessel kinks appear to be areas of focal stenosis. In some instances, this discrepancy can be significant (as high as 30%). 49 When combined with CTA, such overestimation is mitigated considerably. Finally, U-king-Im et al. showed that, when offered the choice, patients usually prefer to undergo MRA instead of DSA. 50
Transcranial Doppler (TCD)
Transcranial doppler(TCD) is an ultrasound technique, based on the Doppler effect principle, used mainly to study the blood flow in the arteries of the base of the skull.51,52 Miyazaki and Kato were the first in 1965 to use ultrasound to study blood flow in extracranial vessels and subsequently, this technique was used for intracranial vessels intraoperatively and in children with open fontanels. 53 Due to the difficulty to study the blood flow in the intracranial vessels in adults because of the high density of the skull, it was not until 1982 where, for the first time, TCD technique was experimented by Professor Aasild et al. on 50 adults.52–54 The Doppler effect principle makes it possible to obtain a frequency difference called “Doppler shift frequency” between the waves emitted by the probe on the skull and the waves reflected on the intravascular moving red blood cells. This shift frequency being directly proportional to the speed of displacement of the red blood cells, makes it possible to estimate the velocity of the intravascular blood flow by spectral analysis.52,53,55–57 A frequency of 2 MHz is necessary for good penetration of the skull through the so-called acoustic window zones which are transtemporal, suboccipital (transforaminal), transorbital, and submandibular (retromandibular) depending on the arteries targeted in the brain.52,58 Several indices are studied in TCD with the Mean Flow Velocity (MFV) being the main parameter. An increase MVF indicates either a stenosis, a vasospasm, or a hyperdynamic flow. While a decreased MVF reflects an ICP, hypotension, or cerebral death.
Due to its ease even at the bedside, low cost, and rapidity in the evaluation of cerebral blood flow, TCD reveals to be a diagnostic tool of first intention in the elimination of ICAD with an IIB level of evidence.55,59 TCD seems to perform better in high degree stenosis as opposed to low degree stenosis. 60 Nonetheless, due to its low positive predictive value estimated at 36–75%, confirmation by other imaging examinations is always required. 61 Additionally, on an international multicentric study, Zhao et al. demonstrated that TCD demonstrated good inter-reader agreement in accurately identifying intracranial stenosis using the SONIA criteria. 62 The main advantage of TCD is noninvasiveness and the main limitations are low PPV, and its operator related dependency. Those can be addressed by coupling TCD with other imaging modalities such as 3T black blood MRI, as demonstrated by Perren et al. 63 Operator-related dependency is currently mitigated with the use of robotic TCD integrated with automatic highly accurate algorithms. 64
High-Resolution MRI (HD-MRI)/vessels wall imaging (VWI)
Most of the methods used to diagnose ICAD are luminography-based, meaning that they provide information regarding the internal diameter of the vessels. 65 Therefore luminal studies are unable to discriminate many vasculopathies that have similar appearance on imaging like primary angiitis of the central nervous systems (PACNS), Reversible cerebral vasoconstriction syndrome (RCVS), other vasculitis, ICAD, infections, and many more. 66 As a direct consequence, high resolution MRI for vessels wall imaging (VWI) had emerged.
VWI has been around since 1982 and was known as black blood MRI (BBMRI) after Kaufman noticed changes in MRI signal of a moving target, such as blood. 67 Since then, others have used it to diagnose atherosclerotic changes in extracranial blood vessels as well as other peripheral vasculature.68,69 Aoki was one of the first to report the utility of using VWI for intracranial pathologies by emphasizing the abnormal vessel wall enhancement in the geriatric population with atherosclerotic disease. 70
Specific pulse sequences are required for optimal vessel wall imaging, according to the American Society of Neuroradiology, and are not widely available. However, existing scanners’ parameters (hardware and software) can be adjusted to obtain sufficient quality VWI for clinical use. 71 Such quality comes from an intricate combination of high spatial resolution techniques, careful 2D or 3D acquisition and adequate suppression of intraluminal and extraluminal components to ensure good signal-to-noise-ratio (SNR), and contrast-to-noise-ratio (CRN). Because blood vessel walls are thin (0.2–0.3 mm), 71 blood flow and surrounding CSF suppression is of capital importance to delineate the walls. Techniques such as Spin-Echo, spatial pre-saturation, double and quadruple inversion recovery preparation and 3D imaging techniques, like Delay Alternating with Nutation for Tailored Excitation (DANTE) and Motion Sensitized Driven-Equilibrium (MSDE), are used to overcome these problems.66,71–75
Since the main utility of VWI is not accurate stenosis evaluation, few studies compare VWI with reference standard (DSA) in the evaluation of ICAD. Al-smadi et al. reports in a retrospective study that VWI was 100% sensitive and 99.8% specific in accurately reporting intracranial occlusion using DSA as comparison. 76 The interobserver agreement was k = 0.63 (good) for T1 imaging and was 0.89 (excellent) after contrast injection. One of the important advantages of VWI over other invasive or non-invasive tests is the ability to provide unique information on atherosclerotic vascular lesions. That information includes but is not limited to plaque location, degree of plaque enhancement, fibrous cap (FC), lipid-rich necrotic core (LRNC), intra-plaque hemorrhage (IPH) or thrombus, surface irregularity, and vessels’ remodeling pattern.65,77–82 Each of the provided information has specific clinical relevance. Plaque location can help guide endovascular intervention by identifying whether there is risk for perforators occlusion. 83 Plaque enhancement is an indication of plaque vulnerability (culprit plaque) and suggest high risk of distal artery-to-artery embolization70,84 and has been demonstrated to be an indicator of risk of stroke recurrence. 85 Active plaque hemorrhage is associated with active symptoms.86,87 Interestingly, DSA has been reported to miss those findings. De Havenon et al. reports a case of recurrent stroke and transient ischemic attacks (TIA) despite being on anticoagulation. 88 VWI was considered and revealed a non-stenotic active plaque. Treatment was switched from anticoagulation to antiplatelet and all symptoms were subsequently resolved. Simply put, VWI can be used as an adjunct to DSA at the very least, since it provides critical aspects of stenotic vessels that have substantial values in guiding therapy.
VWI has also performed well in discriminating different diagnoses that were all seen on DSA as luminal stenosis (Figure 4). For instance, it reveals to be superior to DSA in diagnosing posterior circulation arterial dissection. 78 Ahn et al. also demonstrated that while DSA reports invariably focal and non-focal stenosis or complete occlusion of examined vessels, VWI did final demarcation by identifying Moyamoya disease, dissection and inflammatory vasculitis. 89 Many other studies have reported similar utilities in differentiating intracranial vasculopathies that would otherwise be mislabeled by other luminal studies like DSA.89–94
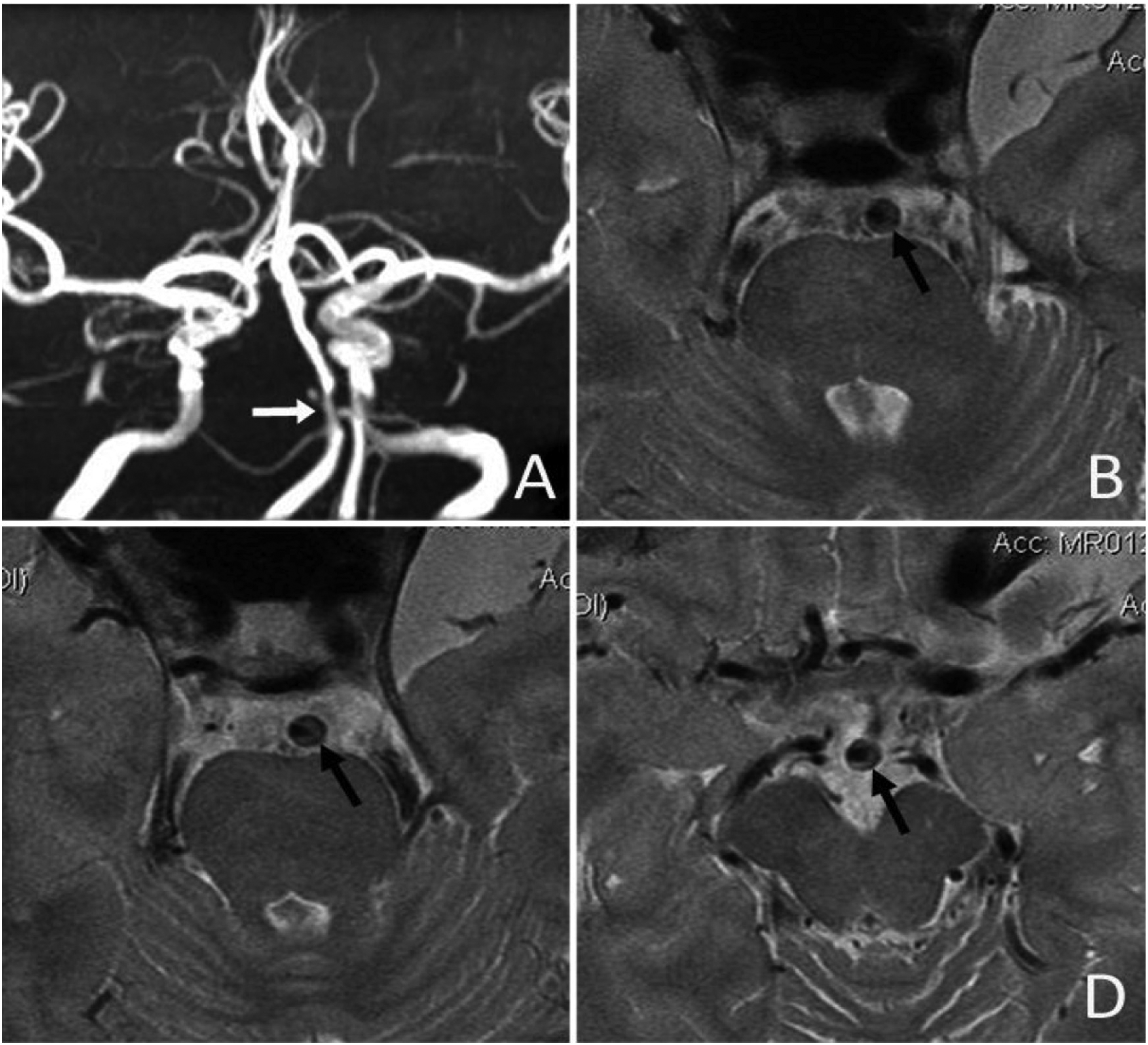
Intracranial plaques seen on VWI (With permission from Liebeskind).
The idea of combining VW-MRI with other imaging modalities has been entertained before as a multimodal imaging approach. For instance, it has been postulated that Positron Emission Tomography (PET) associated with HR-MRI can yield incomparable data on the anatomy, physiology, and metabolism of diseased intracranial vessel segments. In such amalgam, the MRI would provide volume and motion corrections that would improve the localization of PET signal. 95 Such information would be used to guide therapeutic intervention on an individualized level. Although PET/MRI has been widely used in pre-clinical settings only, many challenges related to hardware and techniques are currently being addressed. 96
Intravascular ultrasound (IVUS)
Currently, three sonographic modalities have been used in cerebrovascular imaging studies, which are TCD, brightness modulation ultrasound (B-Mode US), and Intravascular Ultrasound (IVUS). 97 IVUS has been widely used in interventional cardiology for many years as it provides invaluable details of the nature of coronary artery lesions that subsequently help intervention planning and result predictions.98,99 Mechanistically, IVUS is a moderate to high frequency piezoelectric transducer mounted on a catheter that produces acoustic waves upon electrical stimulation. Those waves propagate through the vessels and reflect after collision with surrounding tissues based on their acoustic properties. 100 The reflected waves are subsequently received by the miniaturized transducer and transferred to a computerized unit as sonographic data that is later transformed in photographic 2D images to be displayed. Like HR-MRI, 101 IVUS inform about plaque components, presence of IPH or ulceration and other characteristics, but in an invasive way.100,102 Another added unique benefit of IVUS is peri procedural guidance in a sense that it allows selection of optimal endovascular device sizes, assessment of in vivo stent apposition, expansion and guide balloon inflation pressure. 97 Because of ongoing software development and advanced computer based data analysis, the idea of Virtual Histology (VH) came to existence with the use of IVUS. 103 Bartorelli et al. 104 reports an accuracy as high as 96% when IVUS VH is compared to actual histopathology of intravascular atherosclerotic plaques. Similar findings were reported by Majidi et al. using 7T MRI. However, both modalities have demonstrated limitations such as inadequate mechanical properties that would allow IVUS to adapt to the tortuosity of vessels in the anterior circulation. Such limitations are expected to be resolved with newer generation of devices.105,106
IVUS has yet to establish its acceptance into the neuro-endovascular world. Meanwhile, many authors have reported, through case reports, its potential underappreciated utilities in the diagnosis and management of ICAD and ECAD. Seung reported a case of bilateral vertebral artery dissection, initially seen on MRA as stenosis, that was successfully treated with stent-assisted angioplasty under IVUS guidance. 107 Doing so was critical, given the dissection was found to be unstable and was unnoticed even on angiography. Angiography, unfortunately, accurately depicts dissection pathognomonic findings in less than 10% of cases depend on the affected segment.108,109 Another author reports similar utilization and argues that IVUS can help with ICAD lesion evaluation and proper device placement. 110 In fact, Meyers et al. were the first to demonstrate that such an intervention is feasible and safe in a patient with recurrent stroke in the setting of ICAD that failed medical therapy. 111 This showed that IVUS can identify plaques with high risk features and appropriately guide therapy. Its universal feasibility and safety profile needs to be proven with further investigation.
Optical coherence tomography (OCT)
OCT is considered an analogous imaging technique to IVUS that uses near infrared light instead of sound waves. It uses low coherence interferometry to produce 2D images from scattered near infrared light coming from biological microstructure such as blood vessels. 112 It confers the advantages of axial and lateral resolutions that are up to ten times higher than IVUS due to its low penetration capabilities. 98 Like IVUS, it is widely used in coronary arteries and provides similar information as IVUS. OCT helps identify high risk plaques characteristics such as thin-capped fibroatheroma, calcification and lipid contents (Figure 5).113,114 Its high resolution also helps with more accurate virtual histology, thus identifying features of stable versus unstable plaques, neoangiogenic activities as well as macrophage accumulation in the vessel walls.115,116
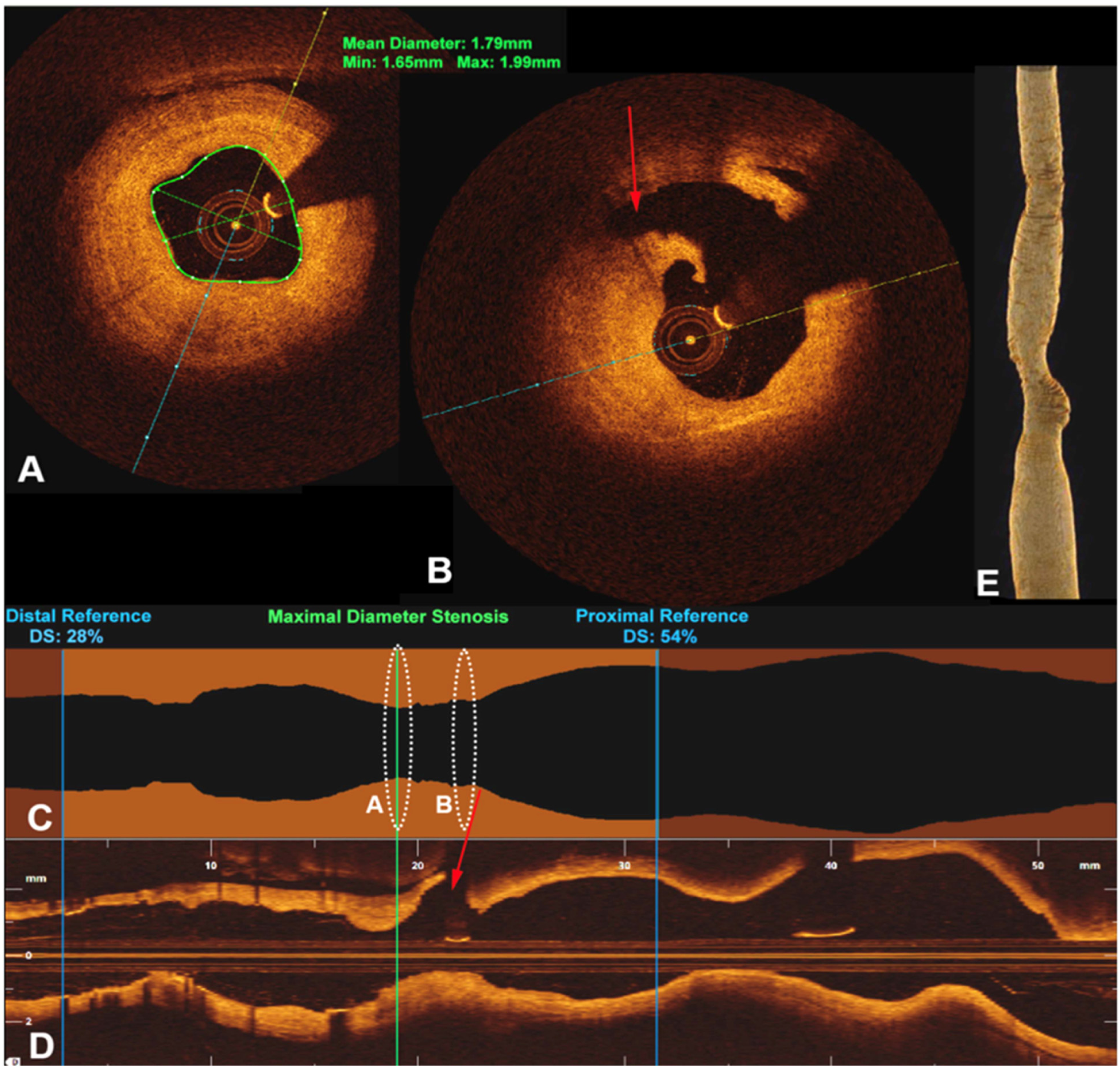
Cross sectional view of OCT images of the R vertebral artery at the level of (A) PICA. (B) Intimal thickening seen. (C) and (D) longitudinal view. (E) 3D reconstruction of the artery (special permission obtained from BMJ case report team).
Size of the instrumentation in OCT limits its utilization in intracranial vasculature. Mathews et al. conducted a feasibility study to address this to see the obstacles in using OCT intracranially. They found that the existing OCT device lacked the size, the biomechanical, optical, and radiological properties to be used in such cases. They were able to make arrangements, creating a hybrid, more flexible and maneuverable device using neuro-endovascular guidewires and OCT device to access the cavernous ICA using standard angiographic techniques. 117
Frequency Domain OCT (FDOCT) has been used during interventional procedures to assess re-occlusion, malposition, apposition, displacement of endovascular devices and even perforators occlusion. One group reported using this technology during deployment of a Wingspan stent to treat symptomatic bilateral vertebral artery stenosis.98,118 Others reported being able to diagnose a dissection after mechanical thrombectomy of a basilar occlusion which was not apparent on DSA. OCT confirmed the presence of false lumen, mural hematoma and other findings that were not obvious on DSA. 119 In a case series, Xu et al. demonstrated that OCT can be efficiently and safely utilized in posterior circulation for interventions like stent angioplasty. 120 Additionally, in a different surveillance study, they reported identifying the reason for medical therapy failure and stent restenosis, which was microcalcification seen on OCT. 121
Nonetheless, OCT is not ready for prime time as the device is not yet suitable for full neuro-endovascular use mostly due to size and other inherent ergonomics. Despite many authors demonstrating that it can be used in the proximal anterior and posterior circulation provided that the anatomy is not tortuous. It also can provide additional information regarding plaque histology making it a reasonable adjunct, at the very least, to DSA in qualifying cases. It is promising that some preclinical studies, such as the one conducted by Puri et al., argued that a newer generation of OCT, like High Frequency OCT (HF-OCT), has been built to be more suitable for neuro-endovascular utilization. 122
Computational fluid dynamic (CFD)
Computational Fluid Dynamic uses modern computational power, applied mathematics and physics to create a model that can assist in analyzing and predicting the behavior of fluid dynamics in real life. 123 It is utilized in various scientific fields 124 and has been revealed to be recently useful in the biomedical science widely due to development of high performing computer hardware and softwares. 125 In the medical use of CFD, blood is considered to be an incompressible Newtonian fluid and the Navier-Stokes equation is solved to predict the behavior of the blood inside the vessels. 126 CFD has been widely used in cardiology as a simulation tool that provides hemodynamic metrics such as Wall Shear Stress (WSS), Velocity, Pressure gradient (PG), Shear Strain Rate (SSR), mesh geometry, 126 and helps in patient selection for intervention.
CFD brings out additional previously unknown complexity of the pathophysiology of ICAD related strokes. For instance, Leng et al. 127 demonstrated that risk of recurrent stroke is not only related to the level of stenosis. They reported that hemodynamics parameters such as SSR ratio and the Velocity ratio are the two most predicting factors of recurrent stroke in 1 year in ICAD patients. 127 Schirmer et al. found that in stenotic intracranial vessel segments, wall shear can reach as high as 2050 Dyne/cm and a Shear rate of more than 19,000/s. These appear to perpetuate the pathophysiological cycle leading to atherosclerosis and contribute independently to platelets aggregation and thrombus formation that leads to subsequent distal embolization.128,129
As a matter of fact, Zaverio et al. and other authors previously demonstrated that physiologic activation-independent platelets adhesion and aggregation are possible and can happen with Shear Stress Rate as low as 6000/s, which is three times higher than physiologic level.130,131
Liebeskind et al. studied other simulated parameters using CFD, such as blood pressure fluctuation in the SAMMPRIS population. They discovered that only 40% of severe ICAD (70–90%) are hemodynamically significant. Even more interestingly, when hypotension is simulated, fractional flow worsens in 6% of the patients, while fractional flow improves in 5% of the patients with simulated hypertension. 132 This potentially constitutes a mean to select candidates for interventions and also can guide blood pressure management in ICAD patients. In fact, Schirmer and Malek showed flow restoration and normalization of WSS and SSR after carotid stenting as clear benefits. 133 However, some complex underlying vessel geometry can play a role in ICAD beyond just stenosis. Some studies showed that even moderate stenosis can be dangerous in the presence of tortuous geometry by altering pressure gradient, WSS, SSR and platelets function.134,135 In terms of a multimodal approach, CFD has shown additional utility in predicting carotid plaques progression and risk of stroke in asymptomatic patients in a prospective study, when combined with MRA. 125
Some advantages related to CFD are that it can be obtained from prior imaging such as CTA and DSA and it does not require an invasive procedure (Figure 6). 127 Inherent limitations are that it heavily relies on vessels geometry provided by other imaging techniques such as CTA and DSA, and that CFD does not take into account some parameters such as cerebral autoregulation, blood pressure, viscosity, flow rate and vascular resistance, which makes its reproducibility questionable. 136
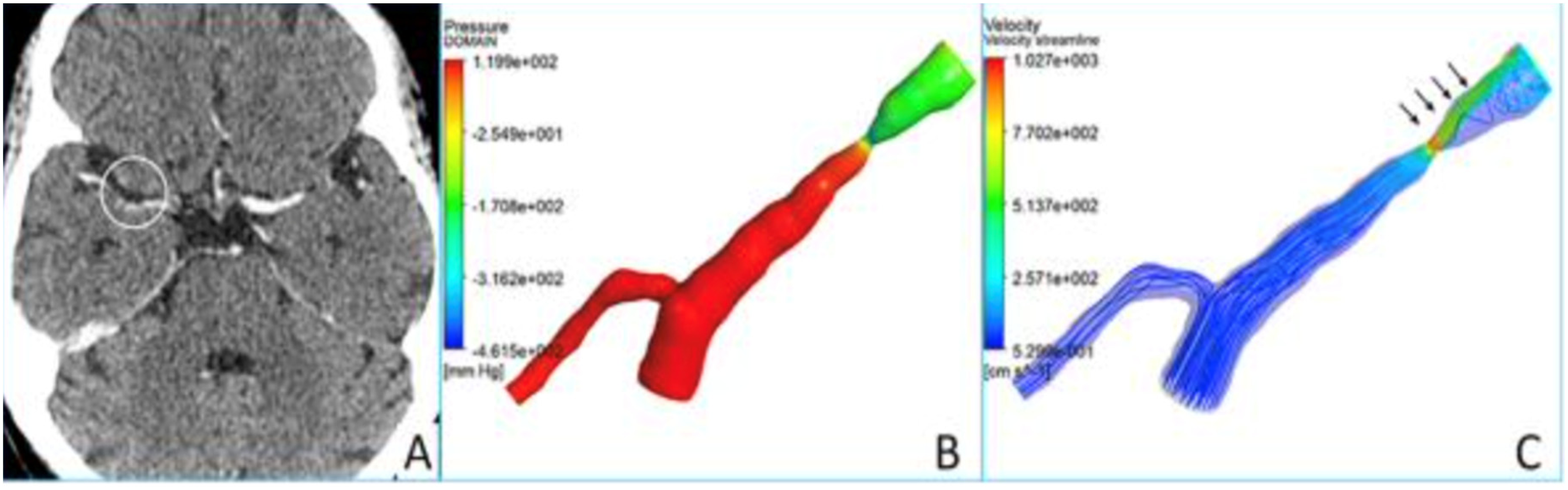
CFD analysis of a RMCA stenosis (with permission from Liebeskind).
Perfusion weighted imaging (PWI)
Perfusion is nutrient delivery to biological tissues via blood and perfusion imaging provides an estimate of how much blood that reaches a peripheral tissue, thus informing about the presence of a deficit or mismatch (Figure 7). Many imaging modalities have been utilized to assess perfusion deficit. Those include PET, SPECT (single photon emission computed tomography), CT, and MRI perfusion. By far, CT and MRI perfusion are the most widely utilized. CTP is based on the central volume principle and uses software assisted complex deconvolution algorithms to produce a perfusion map with qualitative and quantitative values. 137 On the other hand, MRI perfusion exploits signal changes of a moving paramagnetic contrast agent to yield information on blood volume and flow in a specific anatomic region. 138 Three main types of information provided by perfusion imaging are: cerebral blood flow (CBF), Cerebral blood volume (CBV) and Mean transient time (MTT). 139
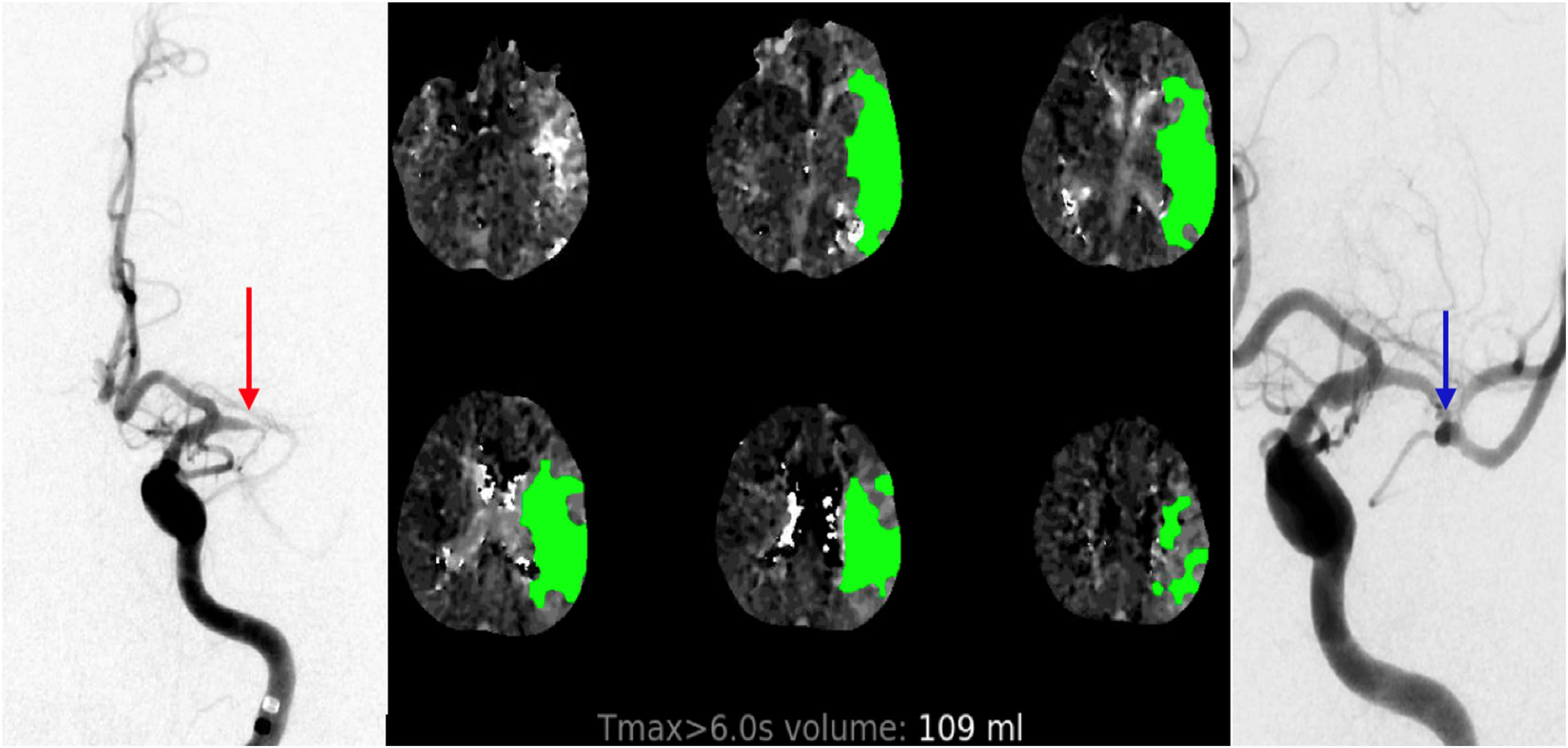
Left M1 critical stenosis on DSA that correlates with CTP perfusion deficit. Same segment seen post angioplasty.
One potential use of CTP in ICAD, besides the evaluation of acute stroke in certain situations, is differentiating ICAD related large vessels occlusion (LVO) versus cardioembolic-related LVO. Haussen et al. reported that Tmax>4 s/Tmax>6 s ratio of 2 or greater was independently associated with ICAD instead of embolic occlusion. 140 They argue that because of usual collateral circulation established in the long term, ICAD predisposes to a smaller perfusion deficit on imaging compared to an acute embolic event. Similar findings were reported by Sacchetti et al., where a Tmax> 6 s in patients presenting with acute stroke due to ICAD predict that they are five times more likely to have recurrent stroke at 90 days follow up (p = 0.001) compared to patients with a lower Tmax. 141
Distal post stenotic blood flow in ICAD can be of equivocal significance and may require more evaluation. In a case report, a patient with distal perfusion deficit presenting with acute stroke showed symptom improvement after blood pressure augmentation, however, exhibited clinical deterioration on maximal medical management. 142 Other retrospective studies argue that hypoperfusion can reliably predict stroke recurrence. 48
Conclusion
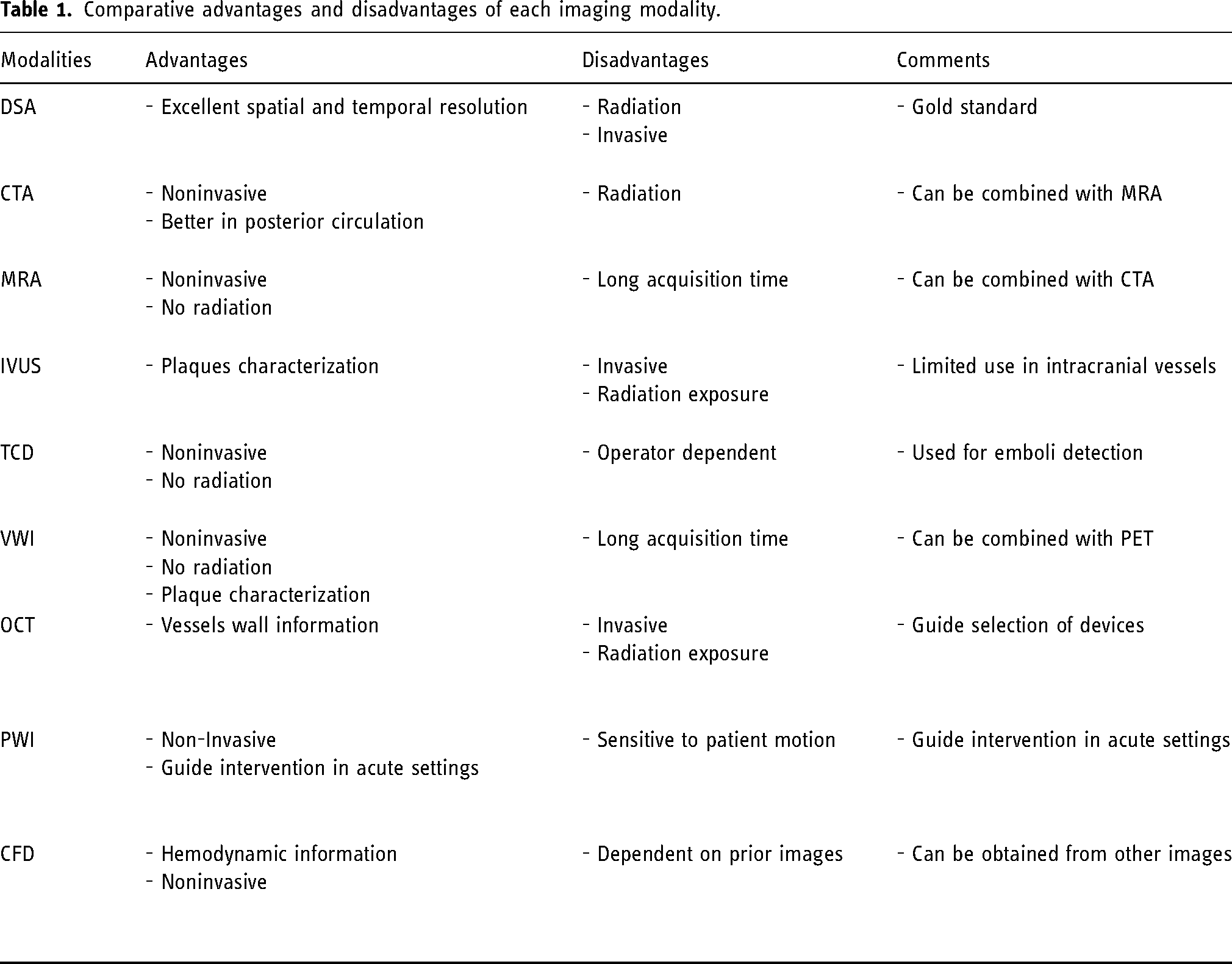
ICAD is one of the most common etiologies of acute ischemic stroke in the world and a major risk factor for stroke recurrence. The imaging tools utilized for the diagnostic of ICAD have significantly evolved over the past few decades. The tendency is to transition from luminographic studies to studies with hemodynamic and vessels wall analysis capabilities. DSA, CTA, MRA and TCD have proven to be effective in identifying intra luminal stenosis at various accuracy while VWI, IVUS and OCT provide excellent information regarding vessel walls and plaque characteristics that can help in guiding management. PWI and CFD can provide valuable hemodynamic features that would otherwise be missed by other luminal studies. Each of the above stated modalities have their own advantages and disadvantages (Table 1). Each of them can be used in a tailored manner based on the patient need and the clinical scenario. In certain situations, coupling two or more modalities can be extremely helpful and provide a more complete picture of pathophysiology and guide an individualized treatment. With such advances, one can only expect an increase in accuracy and a decrease in invasiveness of the methods utilized in the diagnostic of ICAD.
Comparative advantages and disadvantages of each imaging modality.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
