Abstract
Background
Two major studies, The International Subarachnoid Aneurysm Trial and the Barrow Ruptured Aneurysm Trial, compare the long-term outcomes of clipping and coiling. Although these demonstrated coiling’s initial benefits, rebleeding and retreatment rates as well as converging patient outcomes sparked controversy regarding its durability. This article will critically examine the available evidence for and against clipping and coiling of intracranial aneurysms. Critics of endovascular treatment state that the initial benefit seen with endovascular coiling decreases over the duration of follow-up and eventually functional outcomes of both treatment modalities are similar. Combined with the increased rate of retreatment and rebleeding, these trials reveal that coiling is not as durable and not as effective as a long-term treatment compared to clipping. Also, due to the cost of devices following endovascular treatment and prolonged hospitalization following clipping, the financial burden has been considered controversial.
Summary/Key Messages
Short-term outcomes reveal better morbidity and mortality outcomes following coiling. Despite the higher rates of retreatment and rebleeding with coiling, there was no significant change in functional outcomes following retreatment. Furthermore, examining more recent trials reveals a decreased rate of recurrence and rebleeding with improved technology and expertise. Functional outcomes deteriorate for both cohorts over time while recent results revealed improved long-term cognitive outcomes and levels of health-related quality of life after coiling in comparison to clipping. The expense of longer hospital stays following clipping must be balanced against the expense of endovascular devices in coiling.
Introduction
Bare platinum coils were introduced in 1990 for the endovascular treatment of intracranial aneurysms. 1 Since then, several randomized controlled trials have been conducted to compare the outcomes between conventional surgical clipping and endovascular coiling of aneurysms. The largest and most significant studies were the International Subarachnoid Aneurysm Trial (ISAT) 2 and the Barrow Ruptured Aneurysm Trial (BRAT). 3 The initial results of these trials have contributed to the expansion and utilization of endovascular coiling. Despite both trials reporting improved functional neurologic outcomes following coiling early during the studies, controversy remains regarding the longer-term durability and the need for retreatment with coiling. This is highlighted by the increased rate of recurrence, retreatment, and rebleeding seen in patients who received coiling in subsequent follow-up studies.4, 5
This article aims to assess the arguments for and against coiling and clipping in circumstances where both treatments are technically possible and to determine whether the data from these trials support them. Results from these trials will also be considered alongside more contemporary studies to better reflect modern endovascular practice.
Overview of the randomized trials
ISAT was the first major randomized trial comparing the outcomes of aneurysmal subarachnoid haemorrhage treated with coiling or clipping. Patients were recruited between 1994–2002. ISAT aimed to compare outcomes in patients with aneurysms amenable to treatment with both modalities. These “ISAT aneurysms” were most commonly good clinical grade (World Federation of Neurosurgical Societies score 1–2), small (<10mm), and located in the anterior circulation. 2 Only 22.4% (n = 2143/9559) of patients screened were eligible for inclusion, which affected the study’s generalizability. The results of ISAT (Table 1) are often extrapolated to the treatment of non-ISAT aneurysms. 6 Ten-year results indicated coiling was associated with a significant survival benefit. At the end of follow-up, coiling was associated with a slightly higher risk of rebleeding, but this did not translate into significant clinical deterioration. 4
Summary of the results of the mentioned randomised trials.
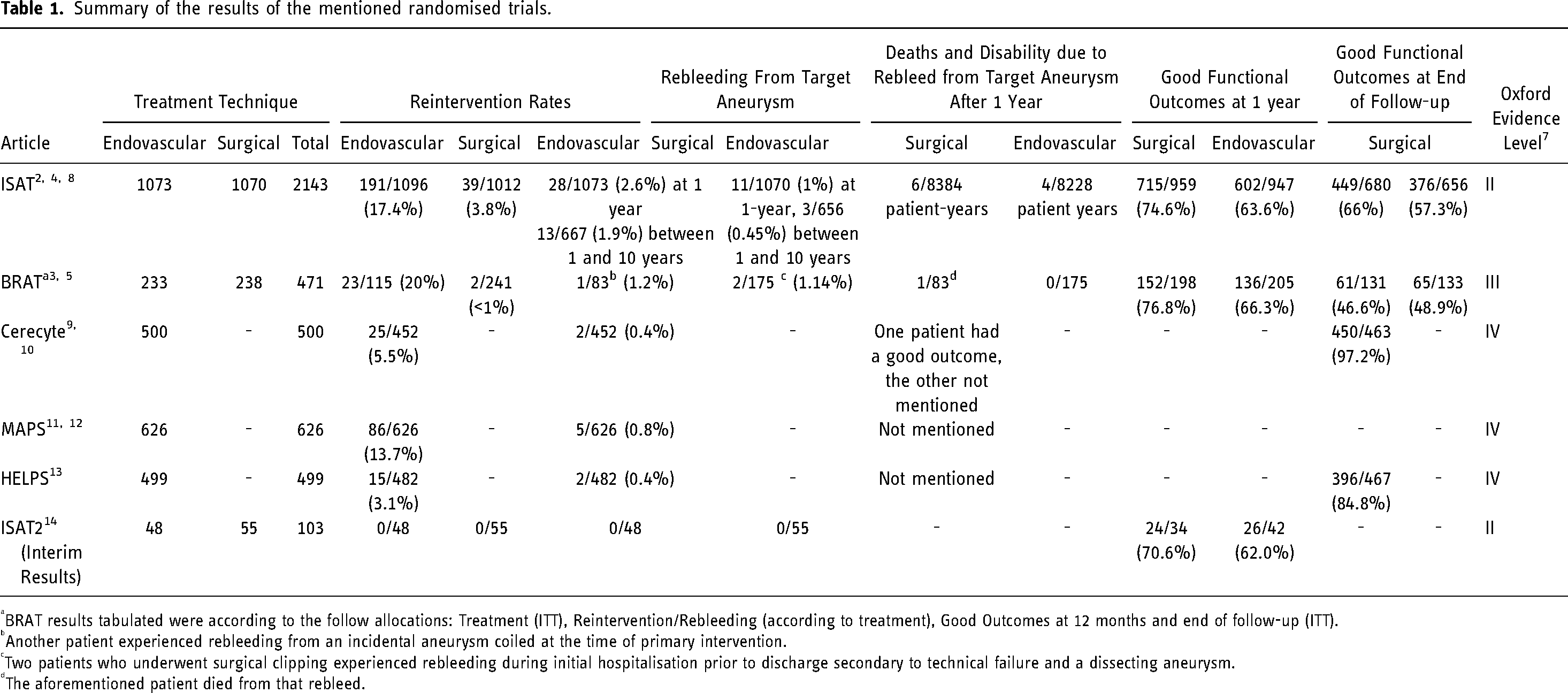
BRAT results tabulated were according to the follow allocations: Treatment (ITT), Reintervention/Rebleeding (according to treatment), Good Outcomes at 12 months and end of follow-up (ITT).
Another patient experienced rebleeding from an incidental aneurysm coiled at the time of primary intervention.
Two patients who underwent surgical clipping experienced rebleeding during initial hospitalisation prior to discharge secondary to technical failure and a dissecting aneurysm.
The aforementioned patient died from that rebleed.
The BRAT study recruited patients between 2003–2007, 3 with follow-up to 10 years. 5 It attempted to address the issue of inclusivity seen in ISAT by randomizing patients into treatment groups prior to screening, then altering allocation after determining whether patients were amenable to the assigned treatment. Analysis was mainly done on an “intention-to-treat” (ITT) basis i.e., according to randomization regardless of the treatment completed. However, substantial crossover affected true randomization. Coiling to clipping crossover was much more common than vice versa (n = 75 vs n = 4). 3 At 1-year, patients assigned to coiling had significantly better outcomes, however at 10 years their outcome was slightly worse (discussed below). Coiling was also associated with a lower long-term occlusion rate and a higher rebleeding rate. 5
Other endovascular randomized trials discussed in this review are the cerecyte coil trial, matrix and platinum science (MAPS) trial and hydroCoil endovascular aneurysm occlusion and packing study (HELPS). These are more contemporary, comparative endovascular trials between bare platinum coils and bioactive coils without a surgical clipping arm, conducted after the conclusion of ISAT and BRAT. Unlike the other 2 trials, Cerecyte excluded patients who received stenting. However, these studies had shorter follow-up periods and included patients with ruptured and unruptured aneurysms.9, 11, 13
Common arguments
Argument 1
“Increased rebleeding and retreatment rates following coiling suggest that coiling is not as durable and effective”
Over the 10-year follow-up in BRAT, coiling was associated with higher rebleeding and retreatment rates and lower complete obliteration rates. 5 At 1 year, the retreatment rate was significantly higher after coiling (12/113 vs 11/245; p = 0.03); however, coiled patients had a lower rebleeding rate (0/113 vs 2/245) and a statistically significant improved outcome. 3 Between 1 and 10 years postoperatively, 2/83 patients who were allocated to the coiling group and received coiling experienced rebleeding (which led to death in both cases) while 0/175 clipped patients experienced rebleeding (p = 0.04). Only 1 patient experienced rebleeding from the target aneurysm; the other had recanalization of, and rebleeding, from an incidental basilar aneurysm which was coiled at the time of initial treatment. Similarly, 93% (n = 50/54) of clipped aneurysms were completely obliterated compared to 22% (n = 5/23) of coiled aneurysms (p < 0.001). The retreatment rate (Figure 1) was <1% (2/241) in patients treated with clipping compared to 20% in the coiled group (23/115, including 3 patients who received recoiling twice) (p < 0.001). 5 When closely examining these results, it is important to consider that most endovascular procedures in BRAT were conducted using first generation Matrix coils which were subsequently recalled due to high recurrence rates reported in several studies. 3 Furthermore, only 30% of both groups completed angiographic follow-up to assess for occlusion. 5 Whilst the difference between treatment arms at 10 years was quite significant, these observations call into question the reliability and generalizability of conclusions regarding recurrence and retreatment of coiling from BRAT.
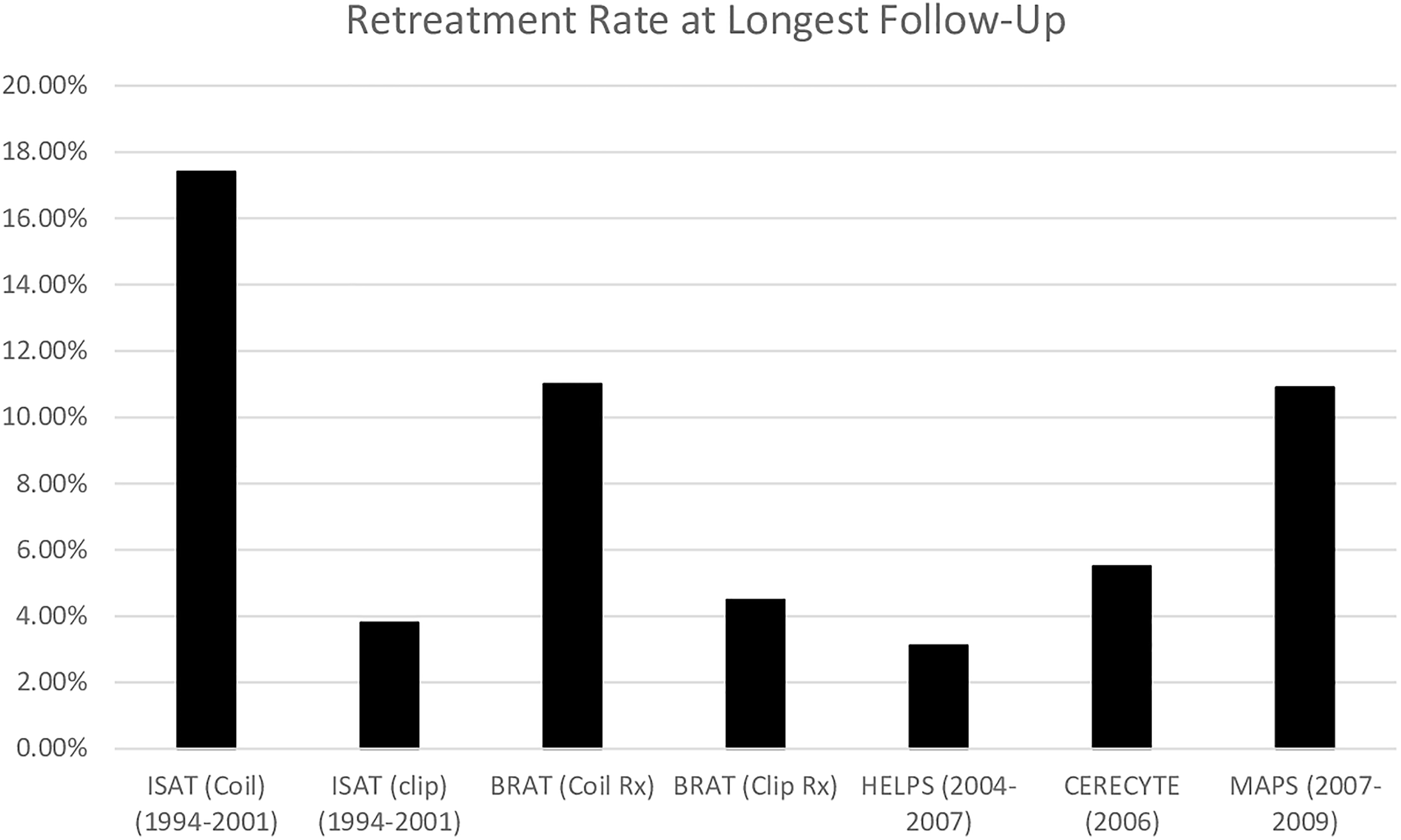
Rates of reintervention at longest follow-up point.
Similarly, over the course of ISAT, coiling was associated with significantly higher rates of recurrence and retreatment, and a significantly lower rate of aneurysmal obliteration. The total retreatment rate following coiling was 17.4% (n = 191/1096) compared to 3.8% (n = 39/1012) following clipping. Retreatment can be divided into 2 groups: early and late. Early retreatment is related to technical failure whereas late retreatment is more likely to be due to true recurrence. The late retreatment rate following coiling was 9.0% (n = 99/1045) compared to 0.9% (n = 9/1063) following clipping (p < 0.00001). 8
In ISAT patients who underwent follow-up angiography, a higher proportion of coiled aneurysms required retreatment compared to clipped aneurysms (10.6% of 881 coiled vs 2.0% of 450 clipped, Odds Ratio (OR) = 5.85, 95% CI 2.92–11.71, p < 0.0001). Rebleeding accounted for 7.4% (n = 7/94) of retreatments of initially coiled aneurysms compared to 33.3% (n = 3/9) of clipped aneurysms. This is likely due under-sampling given lower rates of angiographic follow-up in the clipping group. Regardless, the difference in retreatment rates is still significant albeit not as stark. 8 Furthermore, the late retreatment rate in ISAT (9.0%) is lower than the post-discharge retreatment rate in BRAT (20%) which could partially be due to the types of coils utilized.5, 8
Although coiling was associated with a greater retreatment rate, this does not necessarily translate into a rebleeding event or adverse outcome. In ISAT, 65 coiled patients were retreated with coiling, and this was associated with 6 procedure-related transient neurological symptoms and no changes to long-term outcomes up to 12 years. 8 A multicentre prospective study revealed that from 352 retreatment coiling procedures in 311 patients, there was a 1.28% risk of death or permanent disability. 15 This indicates that retreatment of previously coiled aneurysms is inherently safe. The small risk that accompanies retreatment is offset by short and long-term improvements in functional status and quality of life associated with coiling.
During the study period (excluding first year follow-up), ISAT revealed a nonsignificant higher rebleeding rate with coiling (13 rebleeds/8351 patient-years, incidence per 1000 patient-years 1.56; 95% CI 0.69–2.4) compared to clipping (4 rebleeds/8228 patient-years, incidence per 1000 patient-years 0.49; 95% CI 0.01–0.96). Death or increased disability (modified Rankin Scale [mRS] 3–6) following rebleeding was seen in 6 patients treated with coiling compared to 4 patients treated with clipping [incidence 0.72 (95% CI 0.14–1.29) vs 0.49 (0.01–0.96) per 1000 patient-years]. 4 There was no significant difference at the end of follow-up between cohorts in terms of rebleeding, disability, and death.
The rebleeding rate has been decreasing in randomised trials over the years (Figure 2). The 5-year follow-up results of MAPS revealed that rebleeding occurred in 0.79% (n = 5/626) of patients. 12 All instances occurred in patients who were known to have Raymond class III angiographic result where there is residual aneurysm perfusion and is highly linked with aneurysm recurrence and rebleeding. Three cases occurred within 4 days of intervention following aneurysm rupture while the remaining 2 occurred in unruptured aneurysms at days 344 and 541 postoperatively. 12 When comparing the intermediate term MAPS results (5 years) to BRAT (6 years), the post-discharge rebleeding rate was higher in MAPS (2/626 vs 0/114) while the retreatment rate decreased (13.7% vs 16.4%).12, 16 The rebleeding rate in the MAPS, Cerecyte, and HELPS trials were lower than those in ISAT at their primary endpoints (Figure 2).2, 10, 12, 17 This suggests that technological advances and improvements in coil design may partially account for the declining rebleeding risk. However, the relatively shorter follow-up durations of these studies limit direct comparability with ISAT and BRAT for long-term outcomes.
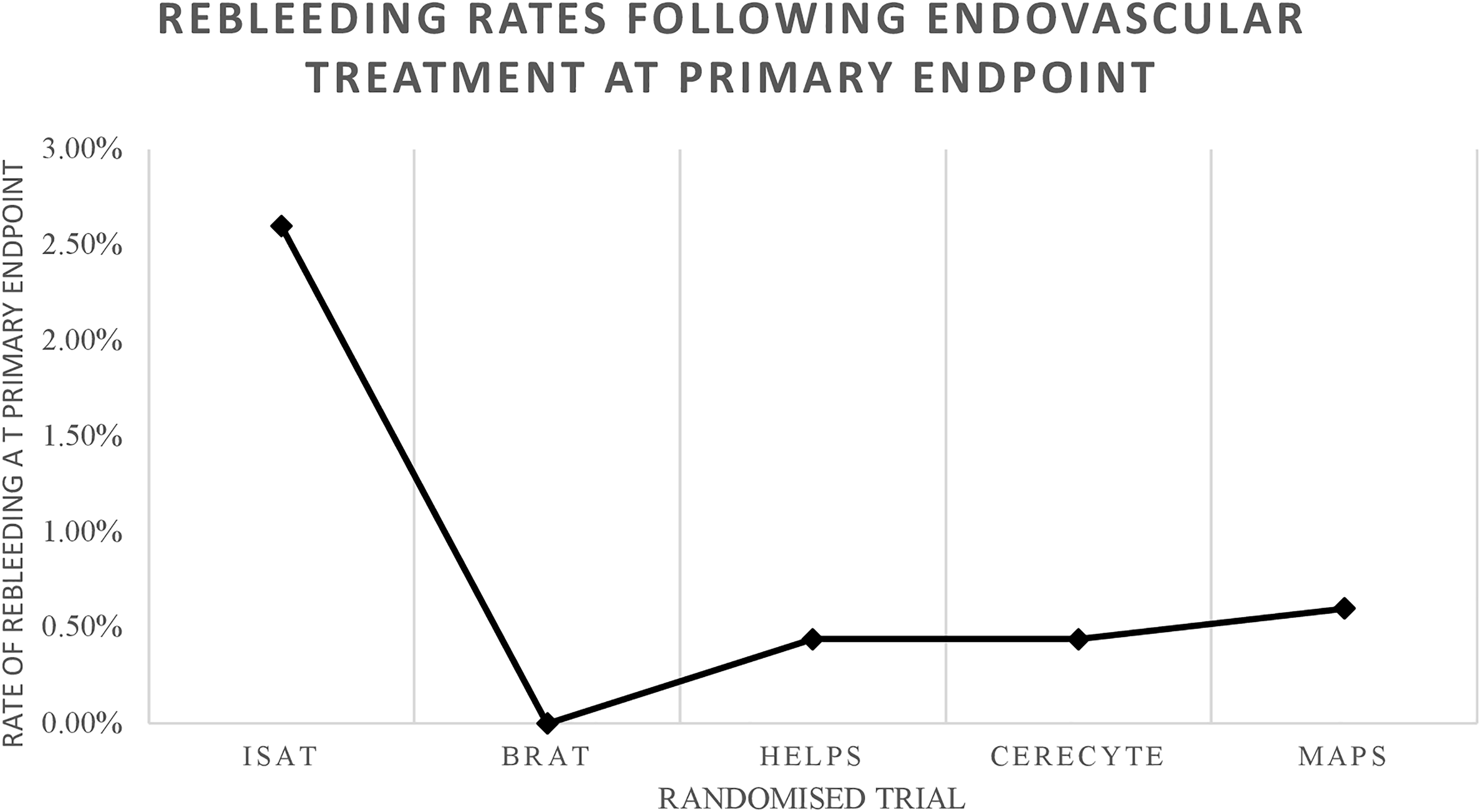
Rebleeding rates following endovascular treatment at primary endpoints.
The HELPS trial reported a 0.44% rebleeding rate (n = 2/457). Both instances occurred in patients with unruptured aneurysms within 3 days of coiling. 17 Similarly, 0.44% of patients (n = 2/452) in the Cerecyte trial experienced rebleeding; one on the day of coiling, another delayed at 5 months post-treatment. The latter was retreated with coiling and had a good outcome. 10
Argument 2
“Despite initial benefit with coiling, patient outcomes converge with time.”
Functional outcomes
Patient outcomes in BRAT were mainly analysed as per ITT. Patients allocated to the clipping ITT group were more likely to have adverse functional outcomes (mRS>2) at 1 year postoperatively (OR = 1.68, 95% CI 1.08–2.61, p = 0.02). 3 We believe that analysis ought to have been performed on a per protocol basis, i.e., solely for patients who completed the treatment arm they were randomized to rather than including patients who were crossed over. This is because more patients were crossed over from coiling to clipping (75 vs 4) and 36% (n = 64/178) of patients in the coiling ITT cohort did not receive coiling. 3 The treatment groups were not fully independent of each other to allow for accurate ITT analysis and the crossover group’s poorer outcomes might underestimate the treatment effect of coiling. Per protocol analysis allows adherence to randomization as patients crossed over from coiling to clipping are not randomizable. Although this would have ultimately resulted in a smaller cohort for analysis, it would have avoided confusion or “crossover” bias which plagues the current BRAT data.
Furthermore, there is little consistency between clinicians in deciding on the optimal modality for aneurysm repair. 18 The only patterns noted were that specialists had high levels of confidence in their choice of treatment and the majority were likely to select the same option again (>80%). There was weak consensus amongst specialists regardless of speciality, experience, location of practice, or capability groups (kappa 0.210, 95% CI 0.158–0.276). This highlights the difficulty in having consistency in allocating patients for randomised studies. 18
In addition, the presentation of data solely as ITT without explicitly reporting the per-protocol results at 10 years can be problematic as patients who were assigned to coiling but were crossed over to clipping eventually had poorer functional outcomes. Fourteen coil-clip patients had hematomas requiring evacuation which may partially account for these poorer outcomes. 3 The following analysis will focus on saccular aneurysms as these were the focus of the BRAT 10-year results.
At 12 months, the rate of poor outcomes (mRS>2) in the coil-clip crossover group was higher than in the clip-clip group [37% (n = 22/65) vs 31% (n = 48/155); p = 0.4]. 19 At 3 years, the coil-clip group had significantly worse outcomes than the clip-clip group [47% (n = 25/53) vs 31% (n = 46/149), p = 0.03]. At 6 years, the coil-clip group had persistently inferior outcomes, but the difference was not statistically significant [50% (n = 26/52) vs 38% (n = 55/145), p = 0.16]. 19 Conversely, the coil-coil group had significantly better outcomes at the 1, 3, and 6-year time points than the coil-clip group [20% vs 37% p = 0.02, 24% vs 47% p = 0.005, and 29% vs 50% p = 0.01 respectively]. 19 Presenting the 10 year outcomes as ITT therefore fails to capture the more nuanced picture of the results as the inclusion of the coil-clip crossover group is likely to have skewed the functional outcomes of the coil-coil group. When taking all this into account, the ITT analysis at 10 years with a small non-significant difference must be interpreted with caution and should not inform decision making regarding durability of functional outcomes.
When examining patients in a per protocol fashion, the OR of an adverse outcome 1 year after clipping increases for all aneurysm types to 2.28 (95% CI 1.03–4.13, p = 0.005). 3 At 6 years, there was a greater likelihood of a poor functional outcome following clipping compared to coiling in saccular aneurysms; however, this was no longer statistically significant (OR 1.50, 95% CI 0.85–2.61, p = 0.16). 19 At the 10-year mark, there was no significant difference in adverse outcomes between the coil-assigned group (53.4%, n = 70/131) and the clip-assigned group (51.1%, n = 68/133) (p = 0.71). 5 Patient outcomes gradually deteriorated across both treatment groups throughout the study as all-cause mortality expectedly increased over the course of a decade.
Like BRAT, ISAT revealed significantly better functional outcomes following coiling compared to clipping at 12 months (mRS>2 23.5% vs 30.9% respectively, absolute risk reduction = 7.4%, 95% CI 3.6–11.2, p = 0.0001). 2 This gain was maintained at 10-years whereby coiling had a significantly greater rate of independent survival (OR 1.34, 95% CI 1.07–1.67) and a non-significant trend towards greater independence (82% (n = 435/531) vs 78% (n = 370/472); OR 1.25; 95% CI 0.92–1.71). 4 The significance of the former has been debated since more patients had preoperative bleeding due to the delay in neurosurgical intervention (1.7 vs 1.1 days: 28 vs 17 bleeds). 2 Excluding these results from the 1-year analysis shows a statistically significant absolute risk reduction of 6.7% instead of 7.4% (p = 0.0004), but this was not observed at 5 or 10 years if the preoperative bleeding events were excluded.
Conversely, a recent substudy by the ISAT authors stated that when changing analysis of outcomes from binary (mRS 0–2 vs 3–6) to ordinal, coiling gained superiority to clipping at 10 years for survival and independence (
A limitation of both ISAT and BRAT is that neither study was geared to provide adequate information regarding the management of high grade aneurysmal intracranial haemorrhage. Under 5% of ISAT patients had World Federation of Neurosurgical Societies (WFNS) grades >III. 2 Patients with high grade aneurysmal subarachnoid haemorrhage generally have poorer functional outcomes and higher mortality rates. There is a dearth of high-quality evidence regarding the optimal treatment modality for high grade aneurysmal subarachnoid haemorrhage. A systematic review and meta-analysis conducted by Zhao et al. 22 which assessed 4506 patients across 85 studies revealed that microsurgical and endovascular treatments have similar functional outcomes after 6 months follow-up, while microsurgical treatment had lower 6 month mortality rates. Their findings supported the need for ultra-early intervention (<48 h) to improve functional outcomes and reduce mortality. 22 Microsurgical centres are increasingly favouring this approach, overcoming the challenges of brain oedema and aneurysm neck localisation. 23 Endovascular treatment usually avoids these issues but is not favoured when large haematomas are present. 22 Zhao el al’s 22 review has several limitations including significant heterogeneity, largely consisting of retrospective studies, and the absence of randomised trials and comparative studies from the literature. Factors limiting the ability to conduct randomised studies in this cohort is the severity of the physical condition in which these patients present, making enrolling them in a trial very difficult. Furthermore, factors such as presence of hematomas >50mL requiring evacuation or the need for drainage necessitates microsurgical intervention. 24 Furthermore, patient outcomes are compared without the ability to assess for aneurysm location and patient baseline. This highlights the need for more prospective, comparative, and randomised studies to improve the quality of the current evidence.
Neuropsychological outcomes and quality of life
Most studies emphasize physical disability as measured by scales such as mRS; however, this offers an incomplete picture as neuropsychological outcomes are not assessed.25, 26 An ISAT substudy focused on reporting neuropsychological outcomes 1-year postoperatively in patients with mRS 0–2. 27 Cognitive impairment, defined as at least 2 impaired test scores in 6 assessed cognitive domains, was present in a third of all patients. It was significantly less common in patients treated with coiling compared to clipping [26.7% (n = 70/262) vs 38.7% (n = 82/212), OR 0.58, 95% CI 0.38–0.87, p = 0.0055]. 27 Additionally, epilepsy had a significantly lower incidence in the coiling group independent of cognitive impairment (7/262 vs 18/212, OR = 0.30, 95% CI 0.11–0.77, p = 0.005). 27 Hua and colleagues 20 found that patients in the UK ISAT cohort who were treated by coiling had significantly higher life years and quality-adjusted life years (QALYs) at 1, 5, and 10-years. At 10 years, coiling was associated with gains of 0.36 QALYs (95% CI 0.04–0.66) and 0.32 life years compared to clipping (95% CI 0.06–0.57). 20
Argument 3
“Endovascular treatment is not cost-effective.”
A recent systematic review and meta-analysis found that there was no overall significant cost difference between coiling and clipping for ruptured intracranial aneurysms. 28 Most of the cost associated with endovascular management is the material cost of devices such as coils and stents while most expenditure in clipping is the cost of staffing and bed occupancy over a significantly longer hospital admission. 28 Patients treated by coiling are less likely to require intensive care unit admissions. Length of hospitalization following coiling has decreased over time while it has been relatively stable following clipping. 29 In South Korea and China, clipping was significantly cheaper, likely secondary to the lower cost of staffing. 28 In the United States, the cost of both procedures was comparable. 28 Clipping was associated with higher one-year cumulative costs despite the additional follow-up angiographic imaging with coiling. 28 ISAT and BRAT indicated significantly better functional outcomes at 1-year with coiling. There is likely an increased need for rehabilitation and a slower recovery and return to work contributing to the increased cost 1-year following clipping.
Endovascular treatment necessitates longer angiographic follow-up due to the greater risk of recurrence and retreatment. There is significant heterogeneity in follow-up between centres and there is potential for follow-up stratification by risk of recurrence to optimize cost-effectiveness further. 14 Retreatment rates have been decreasing but are still significantly higher than those of clipping.10, 12, 17 A recent study of unruptured aneurysms reported no significant difference in cost at 2 or 5-years. 30 Studies with longer follow-up are needed to assess the financial effects of retreatment after coiling and the lower QALYS after clipping. 4
Future trials
ISAT-2 is a multicentre randomized trial currently underway aiming to explore the optimal treatment for patients with aneurysms amenable to both clipping and coiling i.e., not including ISAT aneurysms for which endovascular treatments are the preferred modality. 31 This will include a wider range of aneurysms than ISAT due to the incorporation of newer technologies and the increased scope of endovascular treatment over the past 2 decades. 31
Such newer devices include the Woven EndoBridge Device (MicroVention, Aliso Viejo, California) which has good efficacy, low morbidity, and low recurrence rates following the treatment of wide-necked aneurysms which previously required complex stenting-coiling procedures,32, 33 and flow diversion which is increasingly used in the treatment of aneurysms previously deemed too difficult to coil, such as dissecting and blister aneurysms.34, 35 However, to date, there has been no head-to-head trial comparing the clinical efficacy of either of these devices to surgical clipping or endovascular coiling.
This will also permit assessment of whether the benefits at 1-year holds when factors such as haemorrhage risk due to dual-antiplatelet therapy following stenting are considered. 31 Analysis will be conducted as ITT and the primary endpoint is the incidence of poor functional outcome at 1 year. 31 There are several secondary outcomes including all-cause morbidity and mortality at 1-year, suboptimal occlusion, and length of hospitalization. 31 Interim results reported mRS>2 in n = 16/42 patients allocated to clipping (38%; 95%CI 25%-53%), and in n = 10/34 patients allocated to coiling (29%; 95%CI 17%-46%). Results by treatment received were similar. Satisfactory occlusion (modified Raymond-Roy occlusion classes I and II) was also similar at 1 year at 96.3% (n = 26/27) following clipping and 92.6% (n = 25/27) following endovascular intervention (p = 0.55) with 1 patient in each group with uninterpretable imaging. 36 Clipping was associated with a significantly longer stay in hospital. 36
Conclusion
The findings of the ISAT and BRAT landmark trials remain fundamental in shaping current practice. An in-depth analysis of the data within the context of the studies reveals that, for aneurysms amenable to both endovascular and surgical techniques, coiling is effective and quite durable; evidenced by low rates of rebleeding, disability, and mortality after long-term follow-up. The benefits to quality of life and neurological outcomes need to be balanced against the higher rate of reintervention following coiling, despite the low complication rate. Further, carefully designed, randomized studies are necessary to assess the efficacy of contemporary treatment methodologies as they evolve into complementary roles to ensure optimal patient outcomes.
Footnotes
Author contributions
MN is responsible for drafting and subsequently reviewing the manuscript. JM, JH, GM, HK, AJ, and DMB contributed by reviewing the manuscript as well as providing the INR perspective. JR and PS contributed by reviewing the manuscript as well as providing the neurosurgical perspective which was critical in detecting and minimising bias. HA is the overall project supervisor who was responsible for the conceptualisation of the paper as well continued supervision and guidance throughout the writing of the manuscript. HA also reviewed the manuscript and gave his approval for submission.
Authors' Note
Jonathan Hall, Interventional Neuroradiology Unit, Monash Imaging, Monash Health, Clayton, Victoria, Australia and Department of Radiology, Interventional Neuroradiology Service, Austin Health, Heidelberg, Victoria, Australia.
Declaration of conflicting interests
The author(s) declare that the article content was composed in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
