Abstract
Purpose
Various adjunctive devices have been reported to be helpful during the endovascular coiling of complex, wide-necked cerebral aneurysms. This study aimed to describe the utilization of a recently introduced intrasaccular neck-bridging implant specifically designed to facilitate coil embolization.
Methods
We retrospectively reviewed eight consecutive patients who underwent treatment for intracranial aneurysms with the Nautilus intrasaccular bridging system between February 2022 to May 2022. We evaluated the angiographic appearances of treated aneurysms at the end of the procedure and early follow-up, the clinical status and complications.
Results
Eight patients with eight cerebral aneurysms (3 men and 5 women, mean age 52.8 years (range 39–66) were analyzed. Five of the treated aneurysms were managed in an acute case scenario. There were no technical difficulties while executing the technique. No other procedural adverse events were documented. Immediate complete occlusion of the aneurysm was seen in 6 patients, neck remnant in 1 and stable aneurysm reperfusion in one case. Early radiological follow-up was available in five patients, and the observed occlusion rates were – RR class I in all radiologically screened aneurysms.
Conclusion
Nautilus-assisted coil embolization represents a rapid and safe solution for complex cerebral aneurysms. In our opinion this device symbolizes a significant step forward in treating intracranial aneurysms. We believe that the technique's actual value consists of the fact that the device is fully compatible with the modern low-profile microcatheter technology and does not involve the need for antiplatelet medications.
Introduction
The technical arsenal available for the endovascular treatment of intracranial aneurysms is noticeably evolving. With the intent to technically address a broader array of aneurysms, the endovascular neurosurgery has entered a new span that marks the progressive expansion of its armamentarium. 1 It is now possible to circumvent the limitations of conventional endovascular coiling with the utilization of balloons and endoluminal stent scaffolding.2,3 However, a small subset of aneurysms with specific geometrical features like the wide-necked bifurcation aneurysms (WNBA) often require a permanent endoluminal reconstruction across the aneurysm orifice with double stents in "Y" or "X" configurations.4–8 Such approaches may sometimes be technically complicated and experience demanding, which indicates the need for better treatment options. 9
Intrasaccular neck-bridging is a conceptually new approach that aims to provide a mechanical reconstruction of the aneurysm neck from within the aneurysmal sac. A promising new technique that leaves room for intriguing device engineering that could mitigate the need for dual antiplatelet therapy (DAPT).
The Nautilus intrasaccular bridging system (EndoStream Medical, Israel) is a recently introduced and dedicated intrasaccular tool designed to facilitate coil embolization of intracranial WNBAs. The Nautilus device harbors a unique design configuration that mechanically reconstructs the aneurysm/neck interface, regardless of the aneurysmal dome's size, shape, and morphology. The self-expanding nitinol-based implant is entirely compatible with the modern 0.017-inch microcatheter technology. The device is fully retrievable and can be repositioned by retracting it into the delivery microcatheter at any point during or after being already fully deployed. The supporting framework for the system has no intraluminal components that could interfere with the hemodynamic flow at parent vessel bifurcations. Due to its nitinol nature, the Nautilus exhibits two closely related and unique technical properties: shape memory effect and superelasticity. During insertion, the device imitates a soft embolization coil until its fully deployed. Once entirely unsheathed at body temperature and inside the aneurysmal dome, the phase transformation of the nitinol configures the device into a flat, circular and disk-like shape (Figure 1). The subsequent coiling of the aneurysm is meant to be performed after a second microcatheter is navigated through the implant's intended central pore. The superelastic nature enhances the sealing capabilities of the Nautilus implant, thus providing room for more coils to be safely inserted into the aneurysm. Additional coiling of the dome would accelerate the thrombosis of the aneurysm and the biological healing of the aneurysm. CE (0297) mark has already been granted for the device.
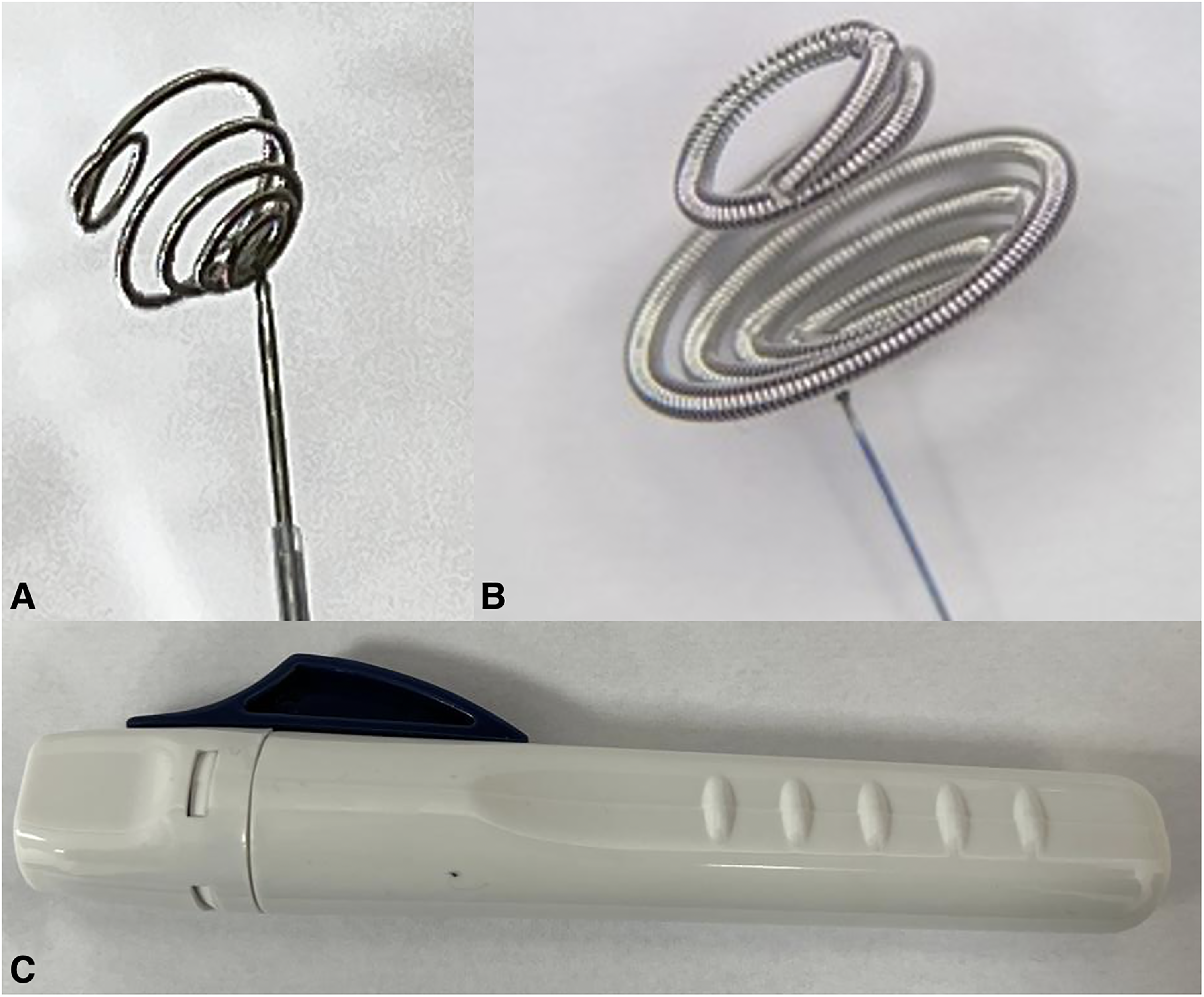
Nautilus intrasaccular bridging system is a self-expanding implant consisting of a spiral element and a distal ring (A). The device is made of a single nickel-titanium alloy (Nitinol) wire, covered with a platinum alloy coil. The Implant is offered in multiple sizes determined by the diameter of the spiral (B) element (e.g. 4 mm, 5 mm, 6 mm, and 7 mm). The device is pre-attached to a detachable delivery system. Once fully deployed inside the aneurysm sac, the device can be separated from the delivery wire via the detachment handle (C). The device received the CE mark (0297) in 2021.
We report our preliminary experience with this novel endosaccular system as a technical note with illustrated case examples and provide a detailed description of utilizing this adjunctive device to achieve successful angiographic and clinical results.
Materials and methods
We retrospectively reviewed consecutive patients who underwent endovascular treatment for either ruptured or unruptured cerebral aneurysms with Nautilus-assisted coil embolization between February 2022 to May 2022. For every included participant, demographic and clinical data were gathered. Radiological data, aneurysm geometric characteristics, procedural details, and, if available, results from early clinical and radiological follow-ups were also collected and analyzed.
Evaluation of the angiographic outcome at the end of the procedure and available follow-up was performed according to the simplified Raymond-Roy occlusion scale (ROC). 10
The decision to facilitate coil embolization of the aneurysm with the Nautilus implant was taken at the discretion of both the junior and the senior author. If considered appropriate for the reported technique, all information regarding the intervention, the proposed technique, and all available alternatives were explained to the patients or theirs’ legally present authorities. Local ethics committees approved this study, and the requirement for the patient consent for publication was waived due to the study's retrospective nature.
Antiplatelet and anticoagulation regimen
No dual antiplatelet therapy (DAPT) was assigned to any patients treated and presented in this study. An antiplatelet regime was thought not to be necessary because the Nautilus does not have any extrasaccular/intraluminal parts. Intraprocedural anticoagulation (50 IU/KG) was initiated upon insertion of the femoral sheath.
Endovascular technique
Written informed consent was obtained from all patients before each procedure. Procedures were conducted in accordance with routine care and local hospital policy in a European mid-volume neurovascular situated in Sofia, Bulgaria. Under general anesthesia, all procedures were performed in a dedicated neuroendovascular suite equipped with a biplane angiographic unit (Philips Azurion Neuro 7 B20/15). After completion of diagnostic angiography, the diagnostic catheter was exchanged over an exchange-length wire for a 6F guiding catheter (Chaperon, MicroVention). A three-dimensional rotational angiogram (3D RA) was performed once the guiding system was positioned in the pre-petrous segment of the internal carotid artery or distal cervical vertebral artery segment. Once the aneurysmal morphology was evaluated and carefully reconstructed for the needed procedural measurements, an appropriate working angiographic projection was selected that pictures the aneurysm/neck interface and the proximal arterial anatomy. Microcatheter work used for the implant delivery and deployment was done by Headway17 Advanced (MicroVention). The 3D RA analyse allowed Nautilus sizing and access planning. As per our protocol and experience with the device, we advocate oversizing the selected Nautilus implant with at least 0.5 mm over the diameter of the aneurismal neck. The correctly sized Nautilus was then inserted carefully into the delivery microcatheter and deployed inside the equatorial plane of the aneurysmal sac. Once the frisbee-like shape of the device is formed, a slow retraction of the whole system was performed to adjust the flexible layers at the level of the neck. A continuous and pressurized flush setup was attached to the proximal end of the Nautilus to prevent any biological material interference between the delivery wire and the hypotube of the implant. If any readjustments to the position of the implant are needed, the device could be resheated completely and redeployed multiple times until the desired location has been obtained. Before inserting the second microcatheter into the cerebral vasculature, an angiographic run was performed to assess the device placement and rule out any immediate complications. The tip of the coiling microcatheter was then navigated and positioned 2–3 cm away from the central pore of the Nautilus implant. Catheterization of the aneurysm dome through the implant's central opening was then attempted and obtained, followed by positioning of the coiling catheter as deep inside the aneurysm as possible without releasing any tension. The Headway 17 microcatheter was then gently retracted back 2–3 cm away from the distinct marker of the Nautilus implant over the delivery wire to allow the system to release the additional tension generated during the entire manipulation. This allows further adaptation and increased compliance of the mechanical discus during coiling. Embolization of the aneurysm was performed through the coiling catheter with coils sized to the maximum diameter of the aneurysm. Due to the solid mechanical barrier at the neck level, long and standard framing coils could be easily inserted at the start. Once adequate embolization of the aneurysm has been radiologically confirmed, the coiling microcatheter was carefully retracted out of the aneurysm. Subsequently, the Nautilus device was mechanically detached, and the delivery microcatheter was also removed. The final angiogram was performed to assess the surrounding vasculature and the occlusion rate of the aneurysm. This procedure was carried out in every described patient (Figure 2). Early clinical and radiological follow-up was scheduled for three months later.
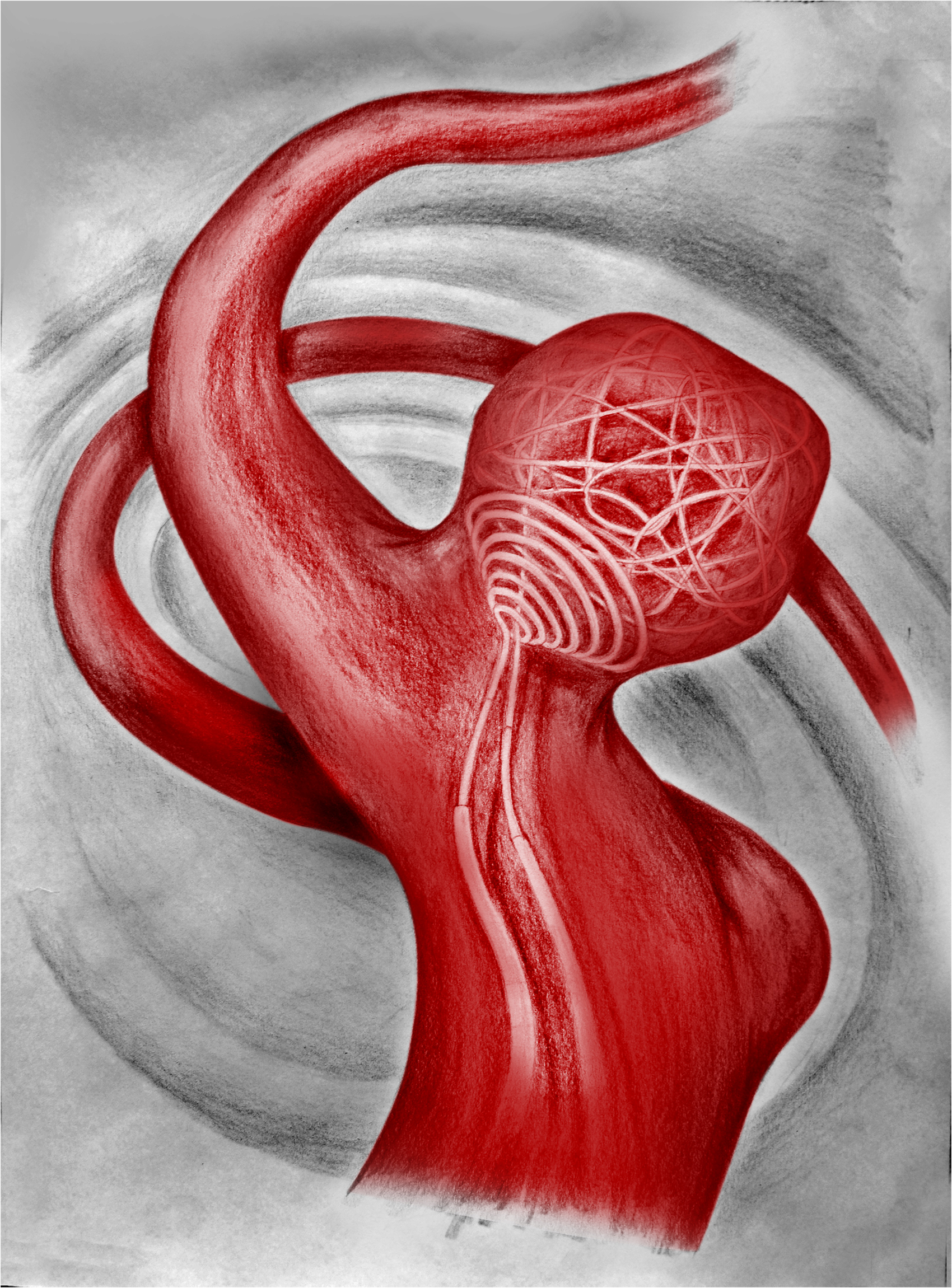
Illustrative drawing of Nautilus-assisted coiling: in this schematic, a wide-necked aneurysm has been treated with the recently introduced endosaccular neck-bridging device. © Anton Robov, used with permission, all rights reserved.
Procedural measurements and Nautilus device selection
The implant is offered in multiple sizes determined by the diameter of the spiral element (e.g. 4mm, 5mm, 6mm, and 7mm). The most appropriate size of the Nautilus device was selected according to the diameter of the aneurysmal neck. To be precise, the diameter of the spiral element of the Nautilus was aimed to be larger, with at least 0.5 mm over the length of the neck. Sizing was conducted according to the instructions for use provided by the manufacturer. The aneurysm width, usually the most critical parameter for coil embolization, can be easily ignored for the device selection. However, while gaining experience with the device, we found out that having a larder dome would only provide more space for the Nautilus deployment and further facilitate the embolization process. We believe that other aneurysmal configurations like axis kinking or bottle-neck configuration of the aneurysm/neck interface could not compromise the neck-briding capabilities of the Nautilus due to the superelasticity of the nitinol and flexible, compliant layers.
During conventional and non-assisted coil embolization, the first coil should be carefully selected to frame a solid basket for the following coil mass. We advocate using long and non-undersized framing coils as the first few picks. This should allow the formation of a stable structure of coil mass inside the aneurysm that adapts and anchors to the Nautilus even more to the aneurysmal neck.
Results
Eight patients (3 men and 5 women, mean age 52.8 years (range 39–66)) with eight cerebral aneurysms were considered for treatment with Nautilus-assisted coiling. Three patients have been treated on an elective basis for incidentally found aneurysms, with the remaining five treated under subarachnoid hemorrhage due to rupture of their aneurysm. None of the treated patients had previous endovascular treatment.
Aneurysms and clinical characteristics
In total, three middle cerebral artery (MCA) aneurysms, one anterior communicating artery (AcomA), two posterior communicating artery (PcomA) aneurysms, one aneurysm at the tip of the basilar artery (BA) and one internal carotid artery (ICA) aneurysm were treated. The mean aneurysm dome was 6.5 mm (range 4.1–9.1mm), mean aneurysm height was 8.9 mm (range 5.2–12.4mm), with mean neck size of 5.2mm (range 4.5–6.2mm). All elective patients had a pretreatment modified Rankin scale (mRS) of 0. According to the Hunt and Hess grading system (HH), at the point of treatment, two of the patients with ruptured aneurysms had scores of one, two patients had scores of two and one patient had a score of three. The patient with a score of three was presented with extended SAH due to a ruptured MCA aneurysm with present adjacent intraparenchymal hematoma.
Rationale for treatment and case examples
All aneurysms met our presumed definition of wide-necked aneurysms and were felt suitable for the proposed technique. The presented aneurysms were with saccular morphology, with five of them located at the level of arterial bifurcation. There was incorporation of an arterial vessel to the aneurysmal neck in the remaining three. The main objective was to utilize the ability of the Nautilus to reconstruct the wide-neck morphology mechanically with subsequent optimal coil embolization. We sought to combine the semi-flow modulation effect of the Nautilus at the aneurysm neck with the excellent ability of the implant to provide room for biological healing of the aneurysm/neck interface via endothelial overgrowth across its nitinol scaffold.
Benefits of the Nautilus-assisted embolization included the absence of intraluminal parts; therefore, the antiplatelet regimen was considered unnecessary. Secondly, the fact that the technique is compatible with 0.017-inch microcath technology.
Case example 1
A 51-year-old male patient (Figure 3, patient 3) presented with a ruptured anterior communicating artery aneurysm. Angiography revealed a saccular aneurysm with a dome width × height of approximately 10 × 8.3 mm. The neck of the aneurysm was measured 4.6 mm in diameter. Endovascular occlusion of the aneurysm with conventional coil embolization was considered unsuitable due to the unfavorable neck-to-dome ratio of the ruptured aneurysm. Based on the 3D RA, an oversized Nautilus device was carefully selected. After implanting the device and eight embolization coils, stable occlusion of RR class III was observed at the final angiography. The patient was discharged from the hospital with procedural or SAH related complications. Angiographic follow-up at three months revealed complete aneurysm occlusion with slight shrinkage of the aneurysmal sac (Figure 3).
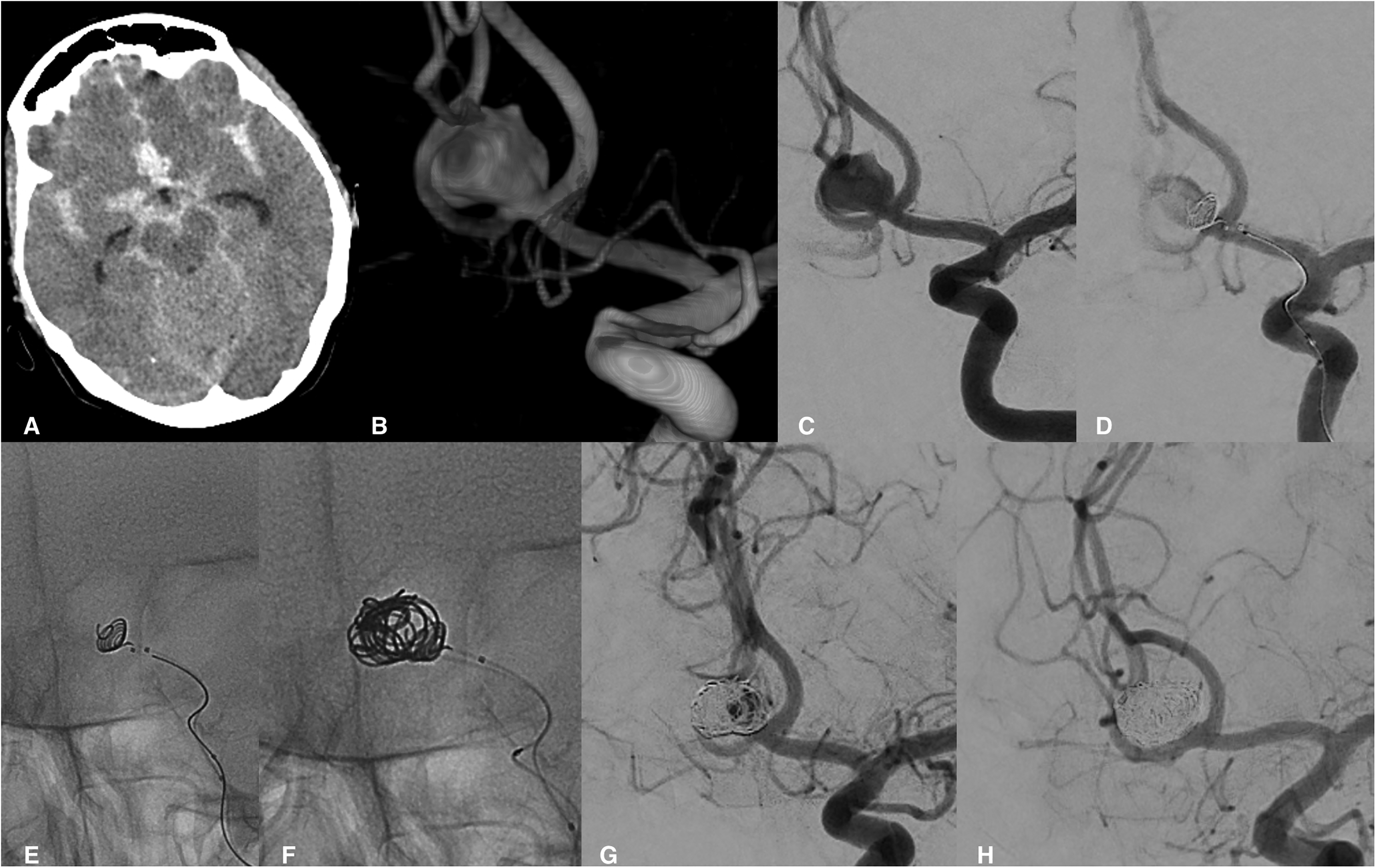
Nautilus assisted coil embolization of an acutely ruptured AcomA aneurysm. Computed tomography documented the presence of extensive SAH (A). Three-dimensional reconstruction allowed for better planning of the embolization strategy (B), including measurements for the Nautilus implant and adequate angiographic view. Working projection (C) demonstrated the vascular angioarchitecture of the AcomA complex. 6 mm Nautilus implant was safely deployed inside the aneurysm and slowly retracted to the neck interface, followed by navigating a second catheter through the central opening (D, E). Coil embolization was initiated (F) with stable occlusion observed at the final angiography - RR class III (G). Early angiography demonstrated the complete occlusion of the aneurysm (H).
Case example 2
Presentation with an acute SAH from a saccular left MCA bifurcational aneurysm. Aneurysmal characteristics confirmed on the 3D RA were the following - a dome width × height precisely 5.2 × 5.2 mm. The neck was measured 4.5 mm in diameter. The presence of arterial incorporation into the aneurysm neck was clearly visible. To avoid permanent stenting in the setting of SAH, the Nautilus-assisted coiling was considered appropriate. Endovascular treatment was performed, and the final angiography demonstrated complete aneurysm occlusion. Ninety-six days later, the follow-up examination angiography confirmed the permanent and total obliteration of the aneurysm (Figure 4, patient 1).
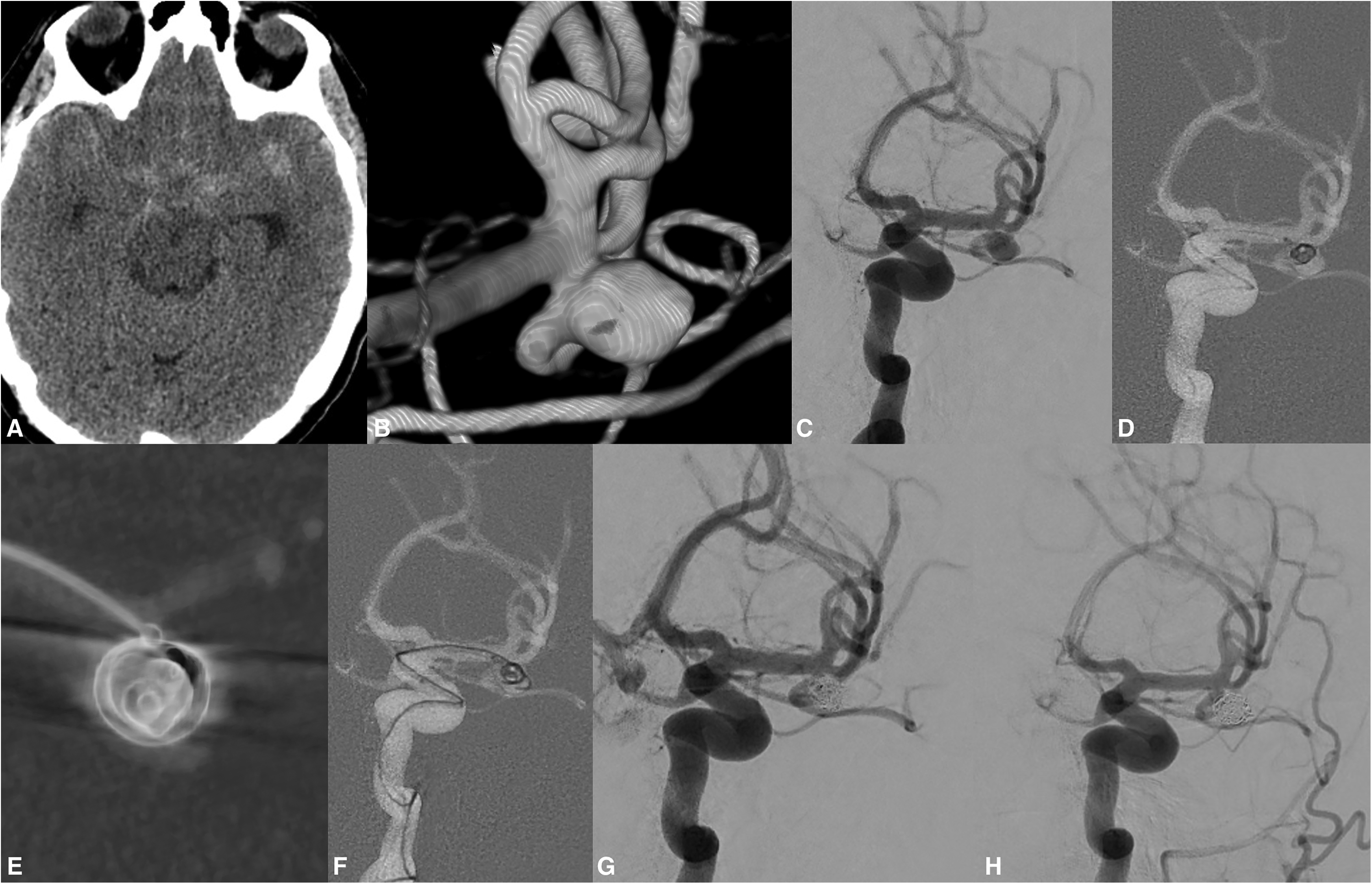
Patient 1 presented with SAH due to a ruptured left MCA saccular aneurysm (A). The aneurysm was unfavorably located on the lower M2 trunk with arterial incorporation at the aneurysm/neck interface (B). 6 mm device was considered a proper candidate to mechanically bridge and reconstruct the complex aneurysm neck. The selection of the Nautilus implant was based on the 3D RA. A working projection was obtained (C), and the Nautilus implant was successfully deployed and positioned at the operator's desired location. The complete neck reconstruction and the patency of the MCA vasculature were confirmed on the VasoCT (Philips Healthcare, Best, the Netherlands) (E). Coil embolization was initiated once the second microcatheter was navigated inside the aneurysm (F, G). Complete occlusion was observed on the early radiological follow-up (H).
Case example 3
A 53-year-old woman presented with an incidental finding of an 5.1-mm wide-neck internal carotid artery aneurysm. The patient reported a recent event of massive bleeding from a gastric ulcer. The aneurysm was obliterated via Nautilus-assisted coiling to avoid permanent stenting and the mandatory DAPT. Oversizing, the implant with 1 mm over the neck diameter provided excellent reconstruction that facilitated the coil embolization of the aneurysm, as seen in Figure 5 – patient 4.
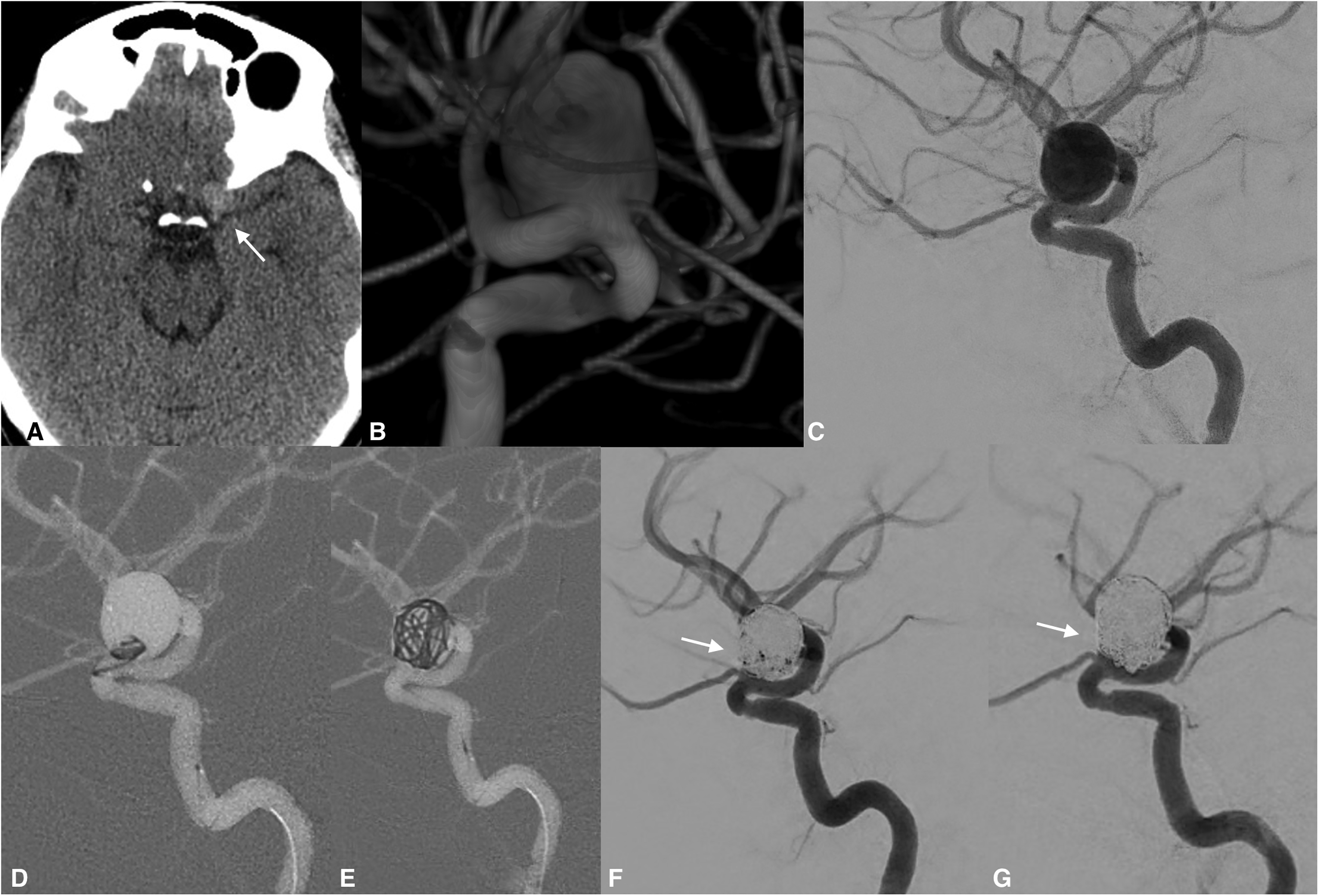
An incidental hyperdense (A, arrow) finding was noted on a routine non-contrast computed cranial tomography. The same week the patient underwent endovascular embolization of the target aneurysm. 3D RA demonstrated the large saccular internal carotid artery aneurysm (B). DAPT was contraindicated due to hemorrhagic gastric comorbidities, and Nautilus-assisted coil embolization was considered an appropriate strategy (C, D). The solid mechanical barrier of the implant prevented any coil protrusion during the obliteration of the aneurysm (E). Final angiography demonstrated stable occlusion - RR class II with some contrast layering within the aneurysm neck (F, arrow). Note the complete obliteration of the aneurysm observed during the early follow-up (G, arrow).
Technical and clinical outcome
The Nautilus devices used in our series were successfully deployed in the desired location and configuration. Complete neck coverage was confirmed in all aneurysms once the device was deployed and retracted back at the neck level. Navigation of the second microcatheter was performed without any issues in all cases. We failed to document any technical issues while performing this technique. There were no device migrations, detachment issues or dreadful coil interactions or device protrusion in this case series. Post embolization angiography documented complete aneurysmal obliteration in 6 patients, neck remnant in 1 and stable aneurysm perfusion in one case. Early radiological follow-up was available in five patients, and the observed occlusion rates were - RR class I in all radiologically screened patients. We failed to document any presence of aneurysm recanalization during the follow-up period.
Additionally, none of the available follow-up imaging reported here showed any device movement in any of the cases, suggesting the implant is stable once deployed and detached even if the aneurysm is not fully embolized. There have been no cases of related procedural complications. Overall, no new neurological symptoms developed after the treatment for all reported patients. Summary of specific patient’s characteristics, procedural data, and outcomes are provided in Table 1.
Summary of specific patients’ characteristics, procedural data and radiological outcomes of Nautilus-assisted coil series.
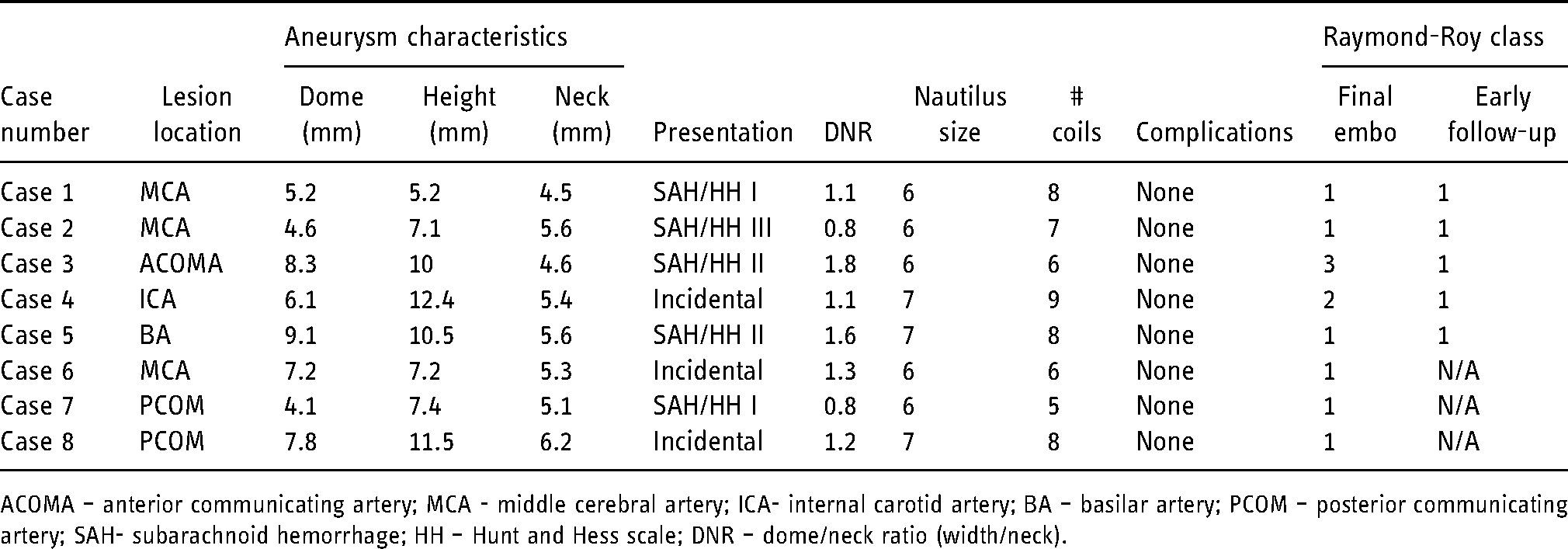
ACOMA – anterior communicating artery; MCA - middle cerebral artery; ICA- internal carotid artery; BA – basilar artery; PCOM – posterior communicating artery; SAH- subarachnoid hemorrhage; HH – Hunt and Hess scale; DNR – dome/neck ratio (width/neck).
Discussion
A common fraction of the intracranial aneurysms are present at the level of arterial bifurcations, and wide-necked bifurcation aneurysms (WNBAs) remain a notorious challenge for the neurointerventional practitioner. 11 Usually, such aneurysms are technically difficult to be treated with primary coiling and often demand an adjunctive device facilitating the embolization. 12 To overcome the technical challenges associated with the wide neck and unfavorable neck-to-dome ratio, the endovascular community has established various techniques, including balloon remodeling and flow diversion, flow disruption and endoluminal stenting. Stent reconstructions include compositions in various geometrical configurations, including waffle-cone stenting, parallel stenting, Y-stenting, and T-stenting. 13 The basic principle of providing mechanical neck bridging mitigates coil protrusion and enhances coil packing inside the aneurysmal sac. 14 A growing body of evidence has been published demonstrating a promptly improving level of clinical safety and encouraging results. Dedicated devices for mechanical neck bridging, such as the PulseRider (Pulsar Vascular, California, USA) and the recently introduced pCONus 2 (Phenos GmbH, Bochum, Germany), aim to facilitate the volumetric displacement of the aneurysm with coils and have been successfully studied in WNBAs.15,16 Technically speaking, these devices are modified waffle-cone techniques that artificially reconstruct the aneurysm neck from within but harbor an intraluminal component extending into the parent artery. A recent systematic review and meta-analysis demonstrated that PulseRider-assisted coiling in wide-necked aneurysms could yield up to 91% adequate occlusion rate with a 5% complication rate. 17 Aguilar - Salinas et al. reported less encouraging results as they found complete occlusion rates of 61.3% with slightly higher complication rates up to 7.9%. 18 The pCONUS first and second generation devices remain the most extensively reported and used neck-bridging device.19,20 The device has been reportedly used in both unruptured and ruptured case scenarios. 21 In their meta-analysis, Sorenson et al. highlighted that the pCONUS device represents a unique addition to available endovascular tools that yield high technical success rates with reasonably low complication and mortality rates. 22 Even though both devices are designed and manufactured with a meagre amount of metal and antithrombotic surface modifications are available for the pCONUS implant, antiplatelet therapy remains an important necessity for the safety of this technique. 20
Flow modulating endoluminal implants have completely revolutionized the treatment of intracranial aneurysms in general. 23 This technique has long been studied with proven durability and safety profile, especially for treating wide-necked and sidewall internal carotid artery aneurysms. 24 The use of flow diverters to manage WNBAs remains debatable and is considered an "off-label". 25 These stents operate from within the parent artery by providing a dense metallic scaffold for neoendothelization, remodeling the aneurysm/neck interface, and healing the lesion. Due to the increased metallic density, the fate of the covered side branches was still uncertain and remains a solid argument against the applications of FDs for the treatment of bifurcation aneurysms. 26 Covering one or more branches at the level of an aneurysm at bifurcations can be dangerous given the risk of blood flow alterations and spontaneous occlusion.27,28 Distal vasculature and complex arterial anatomy further limits the application of this technique, and incomplete neck coverage of the target aneurysm can often be seen. Discrepancies in the diameter of the parent artery can sometimes pose a technical challenge that could cause implant twisting, incomplete opening or foreshortening. Initial results demonstrated relatively low aneurysmal occlusion rates with a questionable safety profile. 29 Improvements in clinical operator expertize and stent braiding technologies that resulted in the development of new lower profile flow diverters led to increased safety, efficacy and better occlusion rates. Bhogal et al. used commercially available FDs to treat 13 patients with bifurcational MCA aneurysms. 30 The authors’ experience yielded encouraging obliteration results were 91.7% of the treated aneurysms were either completely occluded or showed only a small residual. There were no clinical and technical complications reported. Concerning covered branches of the bifurcations, 50% were fully patent at follow-up, a reduction in caliber was observed in 41.7%, and a complete occlusion in only one case. The authors discussed that symptomatic remodeling of the covered side branch occurs relatively rarely. Similar results were documented by Michelozzy et al. in their flow diversion study for bifurcational aneurysms. 31 It is important to note that dual antiplatelet therapy (DAPT) is typically employed to mitigate thromboembolic complications after flow diversion. The use of antiplatelets seriously limits the indications of flow diversion to unruptured aneurysms.
Endosaccular fluid dynamic disruption is a novel competing for technology, wherein the device occupies the aneurysm rather than the parent vessel, thus promoting progressive or immediate occlusion. 32 The technique aims to provide a mechanical barrier at the shared boundary across the aneurysm and its neck, theoretically eliminating the possibility of recurrence with conventional coil obliteration. The concept of intra-aneurysmal flow disruption was developed for the treatment of WNBAs. Several devices have been developed that share quite a typical geometrical configuration braided in globular, round spherical or conical shapes, thus providing an artificial carina at the level of the neck-aneurysm interface. 33 The current flow disruption arsenal includes a handful of dedicated devices such as the Women EndoBridge system (WEB), Luna Aneurysm Embolization System (LUNA AES), Medina Embolic Device (MED) and the latest frontier, the Contour Neurovascular System. 34 These devices have been comparatively well studied, with several prospective and multiple other and smaller case series in patients predominantly harboring bifurcational cerebral aneurysms. The WEB clinical assessment of intrasaccular aneurysm therapy (WEBCAST), the French Observatory, WEBCAST 2, WEB-IT, ARTISSE ISD and the contour neurovascular system european pre-market unruptured aneurysm study.35–38 Preliminary results demonstrated a high level of procedural safety and technical success. Complete occlusion rates vary between devices but remain consistent with 52% to 69% immediate total obliteration rates and documented adequate occlusion in up to 81% at follow-up. Despite the growing enthusiasm and the paradigm shift that the flow disruption technology has caused, it is essential to highlight the relatively scant data on the long-term outcomes, recanalization and retreatment rates. Limited data reflect a learning curve for some devices. However, this technique has been looking with great promise due to its independence from dual antiplatelet therapy. 32
In comparison, our study shows immensely surprising complete occlusion rates. All the aforementioned devices should not be directly compared to the Nautilus implant. The device represents a borderline concept between the neck-bridging and intrasaccular device armamentarium. A hybrid device as it combines the design of a detachable coil with only endosaccular capabilities intended to facilitate coil obliteration of WNBAs. A significant advantage over the other available endosaccular devices is that it is comparatively easy to size. 39 With the Nautilus implant, a slight oversizing of the device (usually 0.5 to 1mm) over the diameter of the neck is enough to obtain full neck reconstruction. The implant is available in sizes from 4 to 7 mm, making most the aneurysms suitable for the technique.
The system is fully compatible with the 0.017-inch microcatheter work, making it accessible and easily applicable in distal vasculature. The excellent radiopacity (Figure 3) of the system allows for precise placement and further facilitates the navigation of the second catheter through the central opening. Indeed, for the proper execution of the technique, after optimal device deployment has been obtained, the central pore should ideally remain visible on one of the projections to ensure safe and successful catheterization. A remaining downside of the technique is the necessity of the simultaneous usage of two microcatheter - the second being used for the coil insertion. Fortunately, we failed to document any technical difficulties while performing and utilizing this technique in this study.
The disc-like configuration and the compliant nature of the flexible layers provide a noticeable coil retaining effect that we believe is behind the surprising obliteration results. The mechanical sealing at the neck offers room for endothelial migration with further overgrowth that could heal the aneurysm. As seen in figure 3, there is a notable volumetric reduction and shrinkage of the aneurysmal sac during the short follow-up period. Based on this limited experience and in light of our patient group, we believe that this same features of the device have led to the progressive thrombosis of the aneurysms that were not entirely obliterated during the initial treatment.
We did not observe any compaction or device deformities during the available follow-up period. There was no radiological sign of aneurysmal recanalization on the analyzed follow-up data.
The device does not have any parent vessel or intraluminal component by design, making the DAPT completely unnecessary. This allowed us to theorize that this device can be safely applied in ruptured case scenarios. In this case series, we did not observe any device-related immediate or short term thromboembolic complications.
Limitations
We acknowledge that the small sample size and the study's retrospective nature are significant limitations. There was no external validation of our angiographic results, leading to discrepancies. Secondly, the lack of long-term clinical and radiological follow-up could majorly impair the interpretations of our obtained results and limit the value of this new device. However, the primary purpose of this study was to provide a technical note and technical evidence of this new. The unexpectedly high rates of occlusion and the fact that the implant does not need any DAPT could potentially catch the attention of some colleagues who might want to try this new approach.
Conclusion
Our limited and early experience with the Nautilus device in treating WNBAs yielded encouraging results. We believe that this device offers a considerably high safety profile while reducing the complexity of the treatment in general. The simple device design promotes precise implant selection and sizing, making procedural planning and coil obliteration relatively easy. Long-term follow-up data are needed to assess this novel technology fully.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
