Abstract
Wide-necked bifurcation aneurysms (WNBAs) are challenging lesions to treat via both open surgical and endovascular techniques. Presently, there are 3 intrasaccular devices available to address many of the limitations of prior techniques, all of which are at different phases of approval for human use around the world. These devices include the Woven EndoBridge (WEB
Introduction
Wide-necked bifurcation aneurysms (WNBAs) have posed a significant challenge for endovascular treatment since the advent of detachable coils.1,2 An aneurysm with a neck greater than 4 mm or having an aspect ratio (dome-to-neck) of less than 1.6 is considered a WNBA. 3 At this size, coils tend to prolapse out of the aneurysm into the parent vessel, which can pose both embolic and thrombotic risk. In recent years, multiple adjunctive treatment methods have been developed to help mitigate these issues with WNBAs, including temporary techniques, such as balloon-assisted coiling or retrievable scaffolding devices, as well as implantable devices, such as stents. However, WNBAs add further difficulty as 2 vessels must be adequately protected. While complex stent constructs or deployments can be utilized or use of various balloon techniques can be used to protect both vessels, these measures increase the risk of the procedure.2,4 With these concerns, other options, such as flow diversion or intra-saccular flow disruption devices, have been sought for the safe treatment of WNBAs.
Principles of flow diversion
In large aneurysms, blood flow is a complex co-existence between inflow and outflow from the neck, although predictable. 5 A small high-velocity inflow jet enters the aneurysm through the distal aspect of the neck; the blood swirls within the aneurysm and then exits via a broader, slower outflow area of the neck. 5 The path of blood is much like the path a wire takes when utilizing the “around the world” technique to access distal vessel during the treatment of large or giant aneurysms.
Flow diversion aims to modulate this balanced flow dynamic to initiate blood stagnation within the aneurysm and ultimately thrombosis. 6 With the increased resistance to flow caused by the low porosity of the flow diverting stent, the inflow jet decreases in velocity and increases in size. 7 As a result, the outflow region decreases in size and, therefore, blood pools within the aneurysm. This correlates with the contrast stasis seen within an aneurysm after flow diverter placement. 8 Interestingly, when radiolucent coils are deployed within a simulated aneurysm, a similar contrast stasis is seen.
Aneurysm healing by any endovascular treatment begins with an intervention causing flow disruption within the aneurysm dome that results in thrombus formation. 9 A process of neointimal formation across the face of the thrombus provides long term exclusion of the aneurysm from the arterial circulation. In the setting of a coil embolization, this process occurs due to the random alignment of coils within the aneurysm causing turbulence in flow, resulting in areas of stagnation. 9
Flow diverting stents can be an effective tool for branch vessel aneurysms, where the whole neck can be covered by deployment in a parent vessel to cause the flow disruption described above. 10 However, with the neck of WNBAs often involving both vessels, deployment in one vessel may not offer adequate neck coverage, and thus, alternative treatments need be selected. 1
Intra-saccular flow disruption
Intra-saccular flow disruption devices were developed as an endovascular solution to a number of challenges that alternate treatment strategies for WNBA face.11–13 These devices employ various designs to obtain a high fine mesh density at the interface between the aneurysm neck and parent vessel. This configuration facilitates the alteration of the inflow and outflow, much like a flow diverting stent, while limiting metal along the parent vessel(s) and not placing intraluminal metal, as needed in for Y-stent assisted coiling. The intra-aneurysmal position of the device does not require dual-antiplatelet therapy, as intra-luminal stents presently require. A lack of dual-antiplatelet therapy can be a significant advantage to intra-saccular devices, particularly in the setting of subarachnoid hemorrhage that may require ventriculostomy or other procedures. Another advantage to many of the intrasaccular devices over coils is preserved visualization within the aneurysm to assess contrast stasis and better evaluate for recurrence.
Presently, there are 3 intra-saccular flow disruption devices available worldwide.11–13 Two are variations on self-expanding mesh balls, including the Woven EndoBridge (WEB
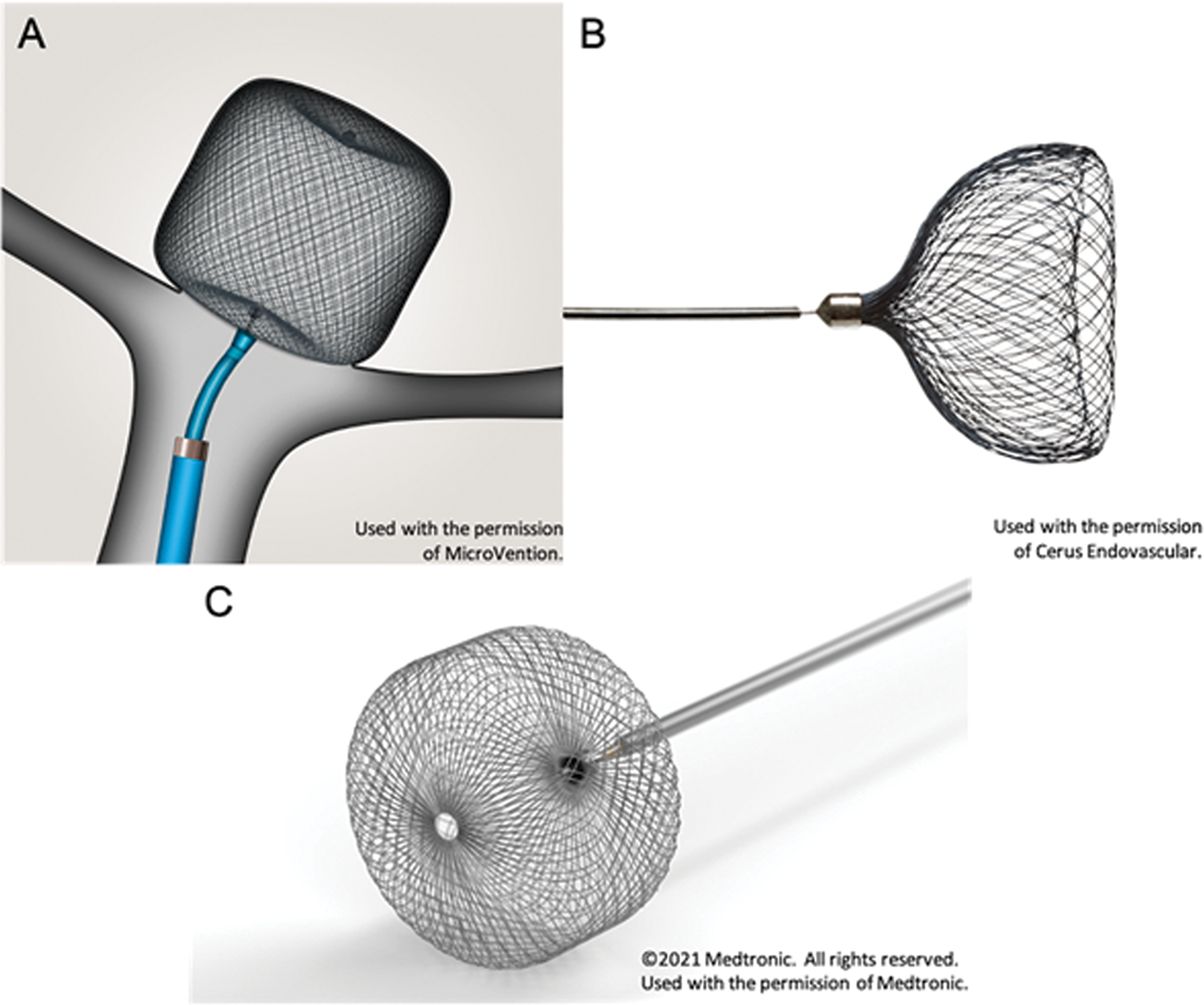
Images of the Woven EndoBridge (WEB
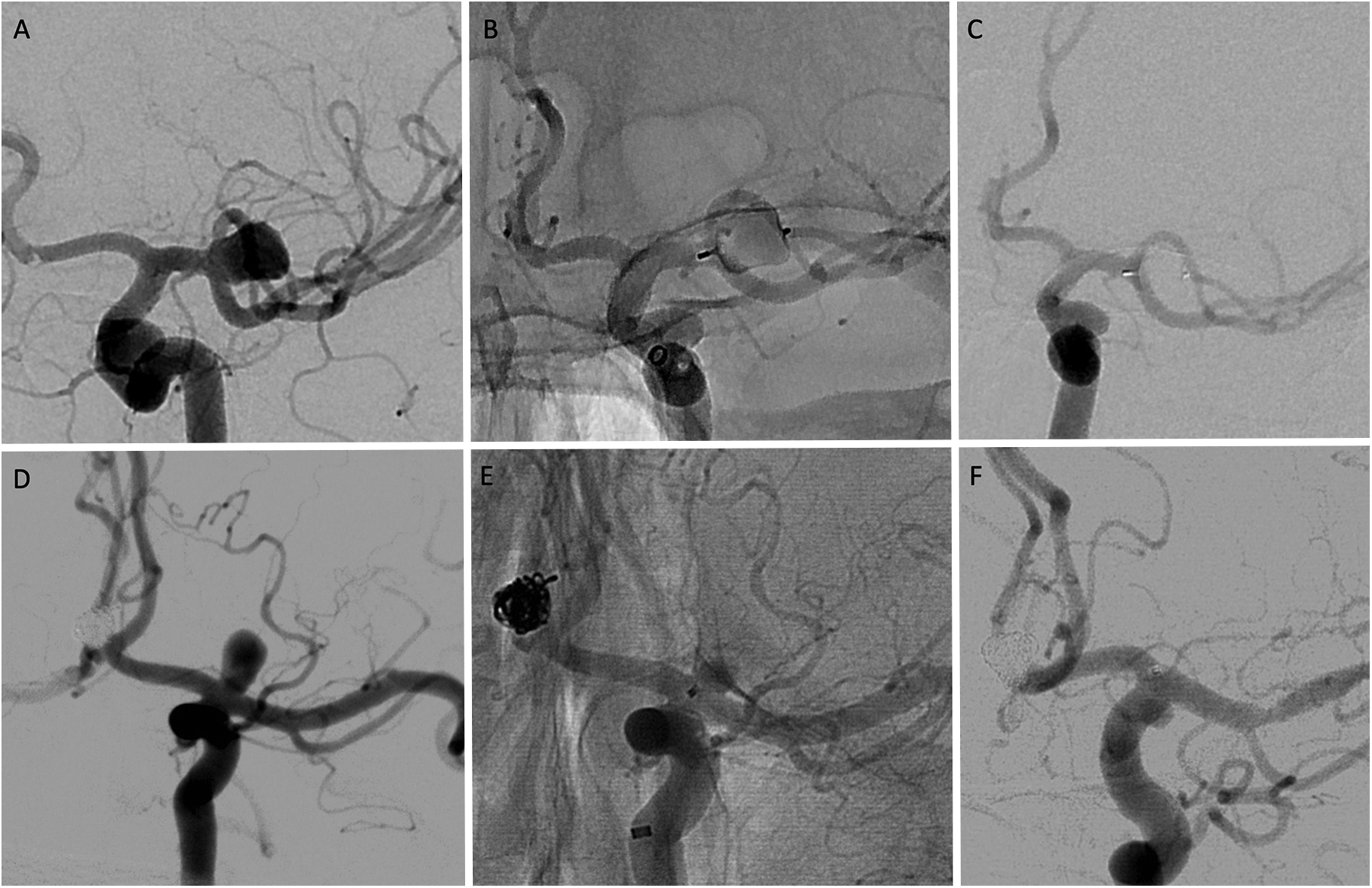
(A) digital subtraction angiogram of a left middle cerebral artery aneurysm prior to treatment. (B) Unsubtracted cerebral angiogram following WEB placement. (C) Six-month follow-up angiography demonstrating complete aneurysm occlusion. (E) Digital subtraction angiogram of a left internal carotid artery terminus aneurysm prior to treatment. (B) Unsubtracted cerebral angiogram following Contour placement. (C) Six-month follow-up angiography demonstrating complete aneurysm occlusion.
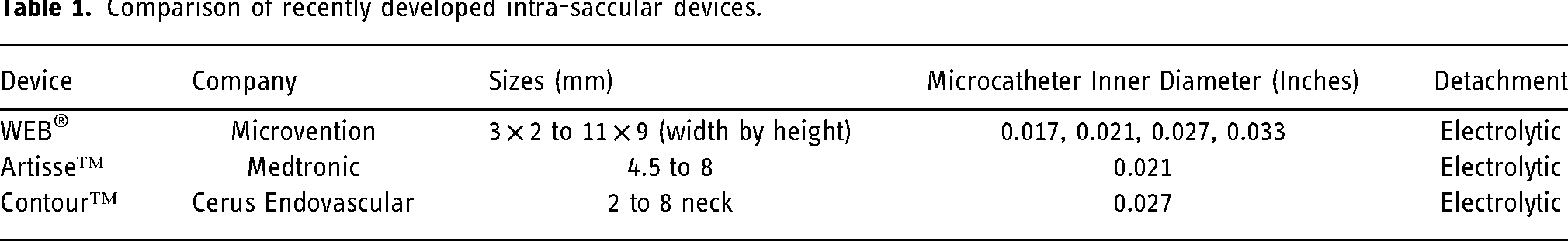
Comparison of recently developed intra-saccular devices.
Woven endoBridge (WEB®)
The WEB
Three European studies, the WEBCAST I & II and the “French Observatory study,” were the first large prospective series demonstrating the technical feasibility and occlusion outcomes of the WEB
Recent WEB® studies .

The WEB-IT trial was a prospective, multi-center, core laboratory adjudicated, single arm study to evaluate the occlusion rates of WEB
Artisse™ Embolization Device
The Artisse™ Embolization Device (Medtronic, Dublin, Ireland) is a self-expanding, double-layered, braided mesh device that comes in 2 configurations: spheroid and flared, the latter of which is shaped like an acorn.
12
The device is made of nitinol with platinum radio-opaque markers, ranges in diameter from 4.5–8.0 mm, is delivered through a 0.021 inch microcatheter, and is electrolytically detached. The largest clinical series (using its former name, LUNA) was conducted in Europe and published in 2018.
26
Sixty-three patients were enrolled from 2011–2013. In contrast to the WEB
Contour Neurovascular System™
The Contour Neurovascular System™ (Cerus Endovascular, Fremont, California, USA) is a another dual-layered mesh device. However, it is flat in its unconstrained form, akin to a mesh steamer basket. The device is deployed within an aneurysm and conforms to the aneurysm neck, forming a tulip shape. Another difference from the 2 prior devices is that the mesh itself is radio-opaque. The device is sized to treat aneurysm necks from 2 to 8 mm, delivered through an 0.027-inch microcatheter, and electrolytically detached. Two European single-center small case series have been published on the use of the device.13,27 Of the 14 patients reported in these studies, 12 had one-year follow-up imaging, which demonstrated 7 (58%) aneurysms with complete occlusion, 4 (33%) with residual neck filling, and 1 (8%) with residual aneurysm filling attributed to a displaced device.13,27 While there were no mortalities or major morbidities in these 2 studies, there were 3 minor strokes without lasting sequale.13,27 The CERUS study, which began enrolling in 2018, will be prospectively evaluating the Contour device in 30 patients, and is yet to be completed.
Conclusion
WNBAs are challenging lesions to treat via both open surgical and endovascular techniques. A variety of intrasaccular endovascular devices have been developed in the past decade to address many of the limitations of prior methods of treatment. While our understanding of the long-term occlusion rates is being elucidated at various stages, the relatively good safety profiles and short-term success rates provide optimism for these innovative intrasaccular devices. A proposed multicenter randomized trail comparing intra-saccular flow disruption devices with best conventional management options (surgical or endovascular) would further help us understand the safety and efficacy profile of these intra-saccular devices in comparison to other available options. 28
Footnotes
Acknowledgements
The authors would like to thank Tufail Patankar, MBBS, PhD (Department of Neuroradiology, Leeds Teaching Hospital, Leeds, United Kingdom) for providing case images of the Contour Neurovascular System, and Andrew J. Gienapp (Neuroscience Institute, Le Bonheur Children's Hospital, Memphis, TN and Department of Neurosurgery, University of Tennessee Health Science Center, Memphis, TN) for copy and technical editing, preparation of the manuscript, tables, and figures for publishing, and publication assistance.
Declaration of conflicting interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ASA is a consultant for Microvention. DMH, NG, VI, and CMN have no conflicts to disclose.
Ethical approval statement
N/A. The follow report is a review of an FDA-approved device, describing its technical aspects as a part of standard care. The case examples have been completely anonymized to protect the privacy of the individuals.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
