Abstract
Background
The Silk Vista Baby (SVB, BALT) is a first-in-class flow-diverter device delivered using a 0.017” microcatheter, designed for the treatment of intracranial aneurysms, including those in small diameter vessels. This study reports a systematic literature review (SLR) to evaluate the safety and efficacy of using SVB to treat intracranial aneurysms in vessels less than 3.5 mm in diameter.
Methods
We performed a PRISMA-compliant SLR to evaluate the outcomes of SVB in the treatment of aneurysms in small intracranial vessels. Primary outcomes were occlusion status and major stroke, and secondary outcomes included all-cause mortality, procedure-related neurologic death, and post-operative aneurysm rupture. Data were expressed as descriptive statistics only.
Results
A total of four studies, including 163 patients with 173 intracranial aneurysms, were included. The most common aneurysm locations were the anterior cerebral artery (24.9% [43/173]), the middle cerebral artery (24.3% [42/173]), and the anterior communicating artery (23.1% [40/173]). Parent artery diameter ranged from 0.9 mm to 3.6 mm, and 29% were acutely or previously ruptured aneurysms. Overall, complete or near-complete occlusion was 72.1% on early-term follow-up. Mortality rate among the studies was 2.5%, with 3 instances adjudicated as neurologic deaths (1.8%). Major stroke was noted in 1.2% of cases, and branch occlusion or stent thrombus formation in 5.5%.
Conclusion
Our review suggests that SVB is a safe and effective treatment for intracranial aneurysms in small vessels. Further prospective and comparative studies with patient outcome data specific to aneurysm location are needed to confirm the safety and efficacy of SVB.
Introduction
The treatment of intracranial aneurysms has been reshaped over the years with the refinement of endovascular tools. 1 Compared to alternative techniques, flow diverters (FDs) represented a breakthrough therapy due to their distinct mechanism of action, allowing treatment of lesions that were not previously amenable to endovascular therapy. 2 Initially, the use of FDs was reserved for the management of large and giant aneurysms in the anterior circulation, but recent studies demonstrated that the device can be safely used for the treatment of small and medium aneurysms.3,4 Nonetheless, evidence of the safety and efficacy of FD treatment for intracranial aneurysms in small parent vessels remains unclear. 5 Technical constraints on the treatment of aneurysms in small parent vessels included challenges in navigating the delivery system and catheter size compatibility, as well as unavailability of smaller devices.
The Silk Vista Baby (SVB) FD stent (Balt SAS, Montmorency, France) is the first FD that can be deployed using a 0.017” microcatheter, permitting navigation through challenging vessel anatomy and management of previously untreatable lesions. It was designed for use in intracranial vessels, including small vessels ranging from 1.5 mm to 3.5 mm in diameter. SVB received the CE mark in May 2018, but it has not yet been approved for commercial use in the United States. While several single-center studies on SVB have been published,6–14 no literature review has been performed to date. In this study, we performed a systematic review to assess the safety and efficacy of the SVB to treat aneurysms in small cerebral vessels.
Methods
Literature search
We conducted a PRISMA-compliant systematic literature review using the AutoLit platform (Nested Knowledge, St Paul, MN, nested-knowledge.com). Using the Application Program Interface (API) in AutoLit, we searched the PubMed database for the following terms 1) (silk OR SV) AND balt AND baby, 2) “flow diverter” AND “baby” AND “silk”, and 3) aneurysm AND Silk Vista Baby, on September 28, 2021.
Screening process
Articles were included if they reported clinical or angiographic outcomes from the use of SVB to treat intracranial aneurysms in small vessels (defined as vessels <3.5mm). Each study was screened by a single reviewer. Titles and abstracts were screened for relevance, followed by further assessment of full texts of the initially included articles. Clinical trials, retrospective studies, and prospective studies were included. Editorials, letters, guidelines, pre-clinical studies, and studies with less than five patients were excluded. Additionally, reports that did not report on effectiveness and safety endpoints were excluded. Reference lists of included articles were scanned as an additional means of identifying articles.
Data extraction
Data from each study were extracted by one reviewer and confirmed by two independent reviewers. Data were extracted and reviewed using the AutoLit platform (Nested Knowledge, St Paul, MN, nested-knowledge.com).
Baseline and procedure characteristics
Baseline characteristics collected included number of cases, aneurysm location, aneurysm rupture status, aneurysm morphology, aneurysm size, and parent artery diameter. Aneurysm size was defined as the largest reported dimension, usually dome height or dome width. For studies that did not provide aneurysm-level data, the dimension with the largest mean was used. Procedure characteristics collected included additional stent deployment, angioplasty, device shortening, and technical success. Technical success was defined as successful SVB device deployment with adequate aneurysm neck coverage using one or more devices.
Outcomes
The primary outcomes of this review were occlusion status of the target aneurysm and incidence of major stroke (ischemic or hemorrhagic). An event was considered a major if significant morbidity remained on follow-up or if adjudicated by studies’ authors as major. The secondary outcomes were all-cause mortality, neurologic death associated with the procedure, post-operative aneurysm rupture, thromboembolism, and other procedure-related complications. Angiographic occlusion was reported using the Raymond-Roy (RR) grading system (class 1, complete obliteration; class 2, neck residual; class 3, aneurysm residual) or the O’Kelly Marotta (OKM) grading system (class A, total filling [>95%]; class B, subtotal filling [5–95%], class C, entry remnant [<5%]; class D, no filling). Angiographic outcomes were combined into two main groups for analysis as follows: RR-1 and OKM-D were considered complete occlusion, whereas RR-2 and OKM-C were considered near-complete occlusion. Occlusion was collected at the longest time point reported in each study.
Statistical analysis
Continuous data were reported as mean ± standard deviation (SD) or median (interquartile range [IQR]) unless otherwise noted. Categorical data were summarized using rates and percentages. Due to heterogeneity in treatments used and outcomes reported, inferential statistics were not performed, and data were expressed as descriptive statistics only. Pooled values are unadjusted rates across studies.
Results
Literature search results
A total of 11 articles were identified through our searches. No duplicates were found. Of the 11 articles screened, six were selected for full-text review. Of these, four were included in the qualitative summary, all of which were retrospective case series.6–9 Of the studies excluded, five had fewer than five patients, one was pre-clinical, and one duplicated patient from an included study. PRISMA diagram is displayed in Figure 1.
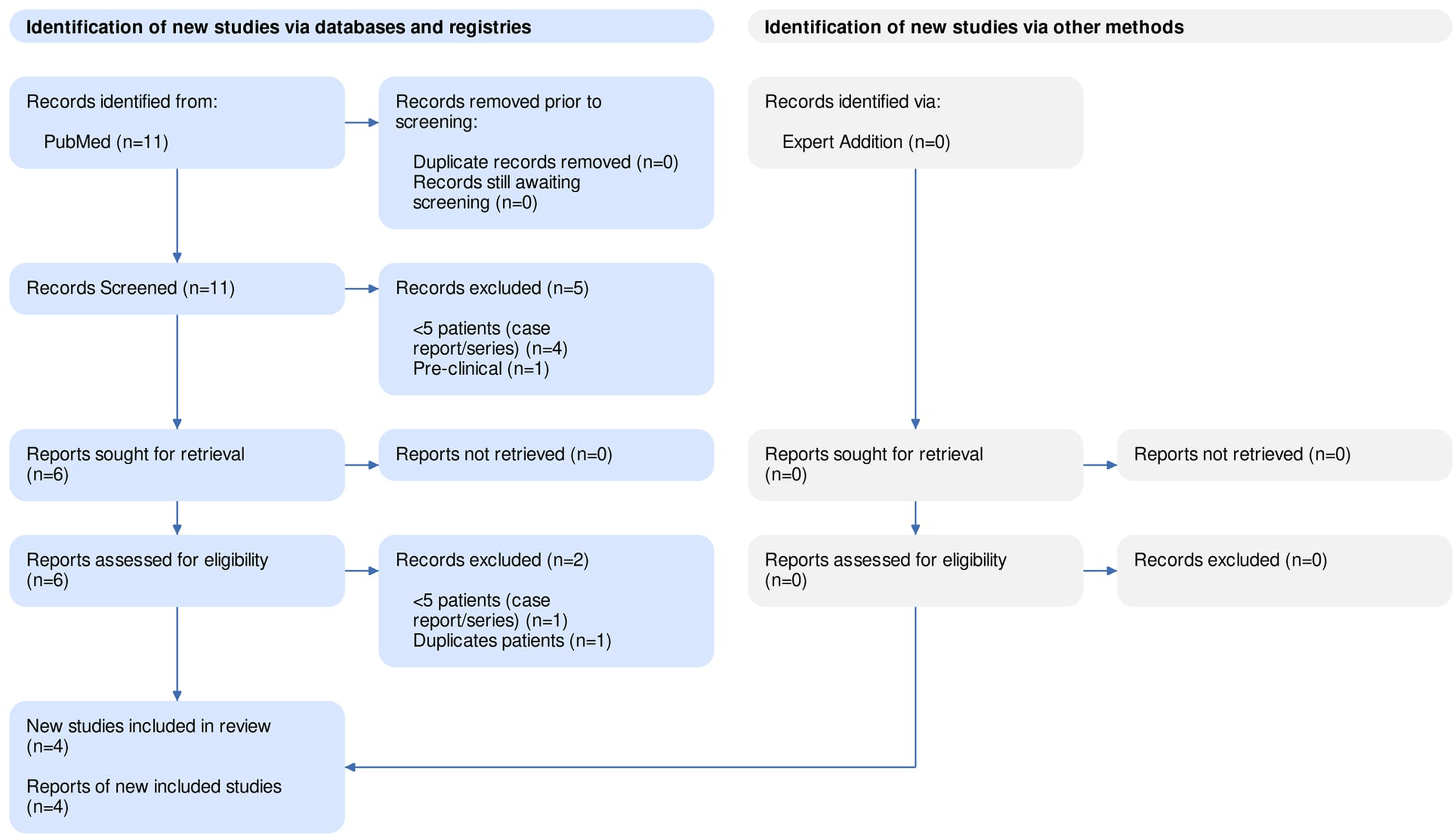
PRISMA diagram detailing the search strategy.
Online access to data
All data gathered for this review, including the PRISMA diagram detailing our search strategy, can be found at https://nested-knowledge.com/nest/439.
Aneurysm location
A total of 163 patients with 173 aneurysms were included. The most common aneurysm location was the anterior cerebral artery (ACA, 24.9% [43/173]), followed by the middle cerebral artery (MCA, 24.3% [42/173]). The anterior communicating artery (ACoA) was also a common aneurysm location, representing 23.1% (40/173) of reported aneurysms. All four included studies reported at least one aneurysm in each of these three vessels (Table 1). Other aneurysm locations included the superior cerebellar artery (SCA), the anterior circulation, the posterior cerebral artery (PCA), the basilar artery (BA), the internal carotid artery (ICA), the posterior inferior cerebellar artery (PICA), and the vertebral artery (VA).
Baseline characteristics.

Continuous data is expressed as mean ± SD (range) and categorical data is expressed as % (n/N) unless otherwise noted.
Abbreviations: ACA = anterior cerebral artery; ACoA = anterior communicating artery; BA = basilar artery; ICA = internal carotid artery; MCA = middle cerebral artery; PCA = posterior cerebral artery; PICA = posterior inferior cerebellar artery; SCA = superior cerebellar artery; VA = vertebral artery; N/A = not applicable.
Parent artery diameter
Parent artery diameter was reported in three of the four studies.6,8,9 Mean diameter ranged from 2.28 mm 8 to 2.49 mm. 9 One study reported both the smallest and largest diameter with a range of 0.9 mm to 3.6 mm. 8 (Table 1). In the study that did not report parent artery diameter, mean diameter of the parent artery reported by other sources was used as a proxy. All reported aneurysms in this study were in arteries whose mean diameter + standard deviation was less than 3.5 mm, 7 including the ACA, 15 the ACoA, 16 the MCA, 17 and the PCA. 18
Other aneurysm characteristics
Aneurysm rupture status at baseline was reported in three of the four studies.6–8 Overall, 21.4% (27/126) aneurysms were acutely ruptured at baseline. In additional cases, previously ruptured aneurysms underwent retreatment with the SVB (Table 1). The majority of aneurysms reported were saccular (81.8% [139/170]), both within each study and overall. Other aneurysm morphologies reported included fusiform, 8 dissecting,6,8 blister, 6 and other.6,9 Aneurysm size, defined as the largest reported dimension, varied widely. One study reported the smallest and largest size, with a range of 1 to 36 mm. 6 The mean aneurysm size ranged from 5.46 mm 9 to 9.5 mm 8 (Table 1).
Procedure characteristics and outcomes
Technical challenges included additional stent deployment, angioplasty, and device shortening. Additional stents were deployed in at least one case in all four studies, with additional stents used in 6.4% (3/47) 9 to 12.2% (6/43) 8 of aneurysms. One study reported a single case where angioplasty was used. 8 Two studies reported instances of device shortening.6,9 Overall, 32.9% (57/173) of the aneurysms managed with the SVB had previously been treated, mainly using coiling. Moreover, adjunct coiling use ranged from 2.1% to 30.2%. Technical success was achieved in the majority of aneurysms for all studies, with two of the four studies reporting technical success in all aneurysms.7,8 Detailed information is depicted in Table 2.
Procedure characteristics and outcomes.
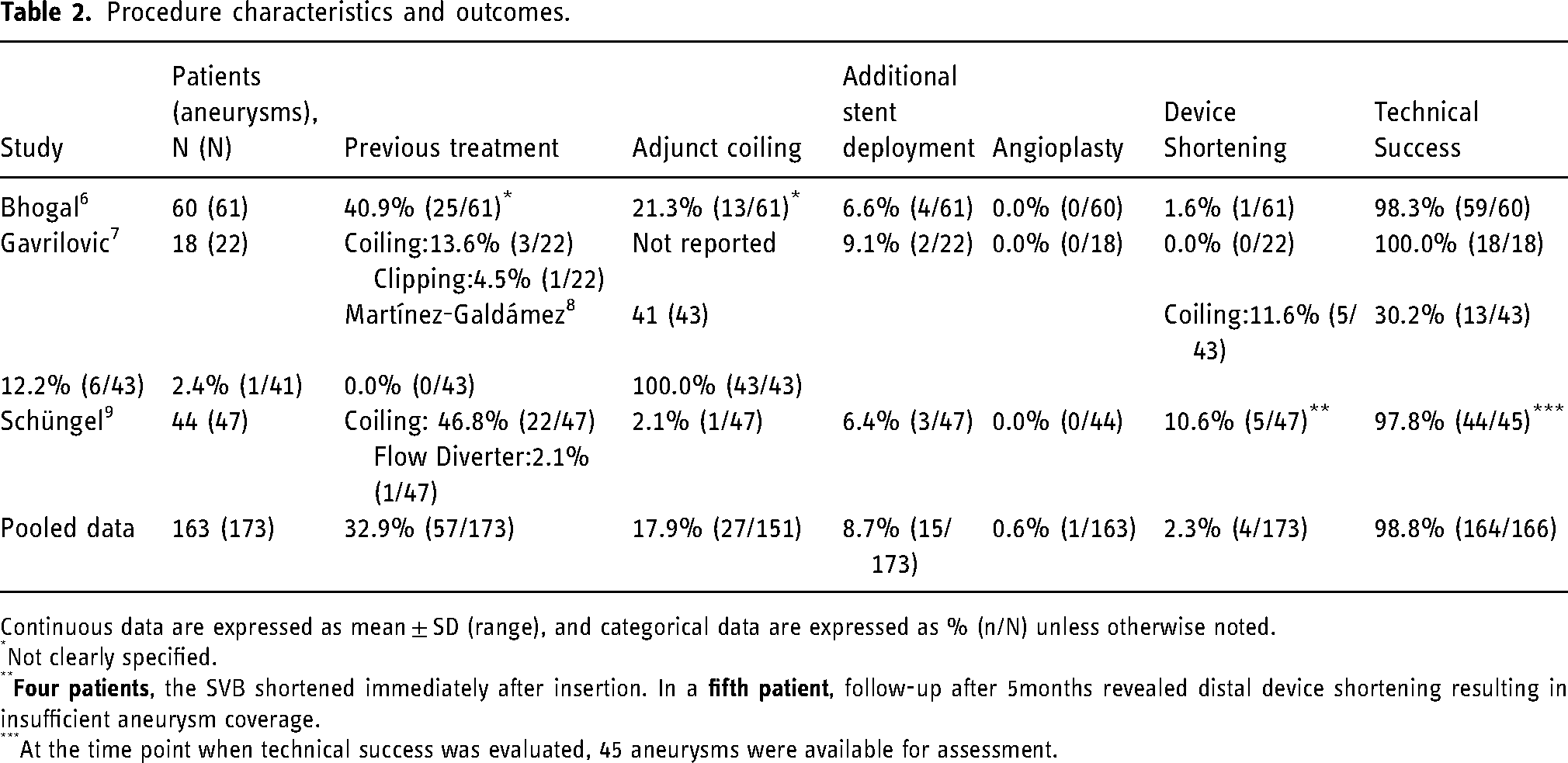
Continuous data are expressed as mean ± SD (range), and categorical data are expressed as % (n/N) unless otherwise noted.
Not clearly specified.
At the time point when technical success was evaluated, 45 aneurysms were available for assessment.
Occlusion
Time points and reporting methods for aneurysm occlusion varied across studies. Mean time for the longest follow-up time point for occlusion ranged from 0 months (immediately post-treatment) 8 to 10.6 months. 9 Two studies reported occlusion using the RROC, and complete occlusion was the most common outcome, with over half of the reported aneurysms in both studies.6,7 The other two studies reported occlusion using OKM,8,9 one of which only reported results immediately after treatment. Overall, pooled data of studies with available early-term follow-up demonstrated that 60.6% of aneurysms (60/99) achieved complete occlusion (RROC-I or OKM-D). Additionally, near-complete occlusion was reported in 12.1% of cases (12/99), resulting in a rate of complete and near-complete occlusion in the early-term follow-up of 72.7%. Detailed information is available in Table 3.
Occlusion outcomes.
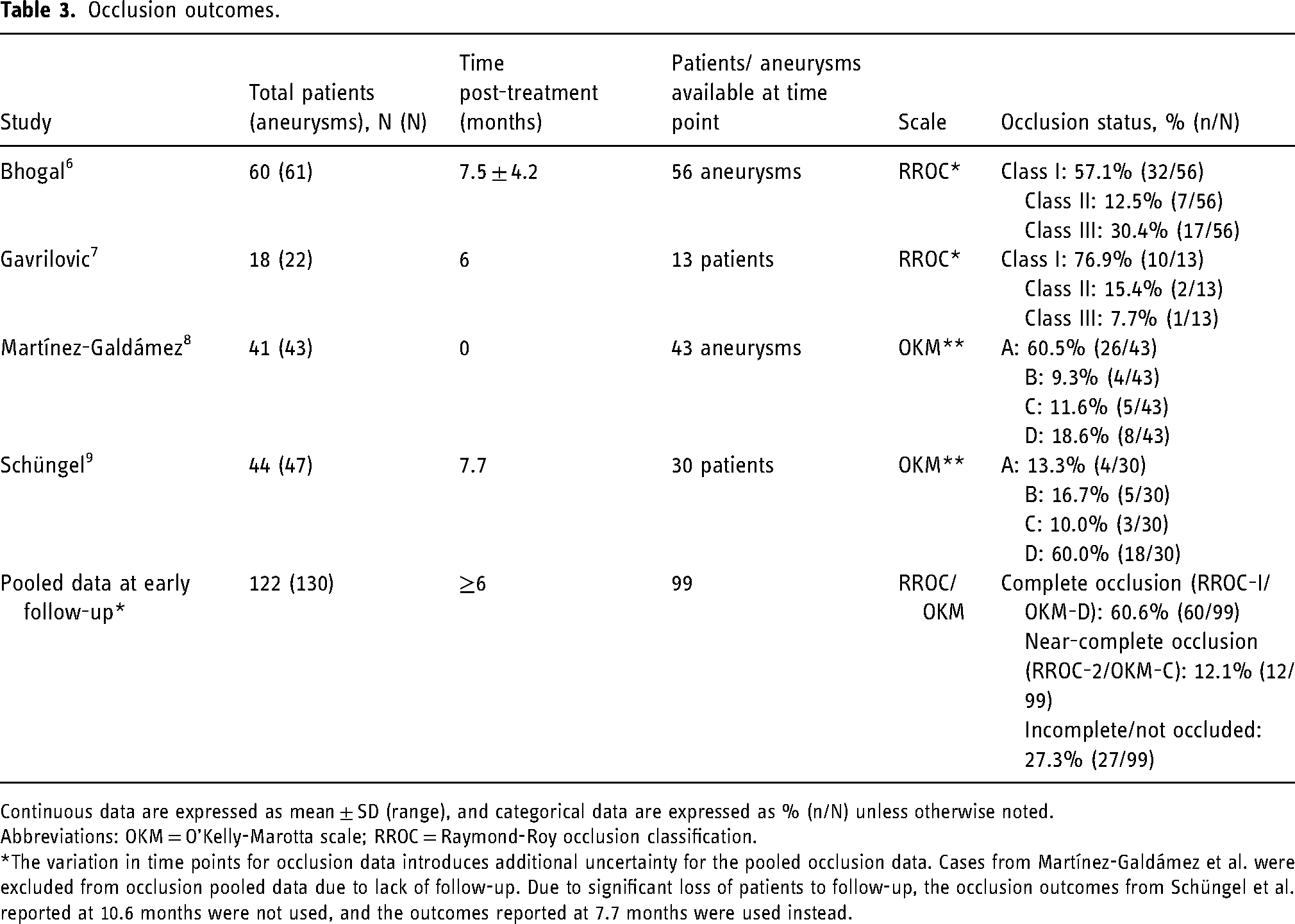
Continuous data are expressed as mean ± SD (range), and categorical data are expressed as % (n/N) unless otherwise noted.
Abbreviations: OKM = O’Kelly-Marotta scale; RROC = Raymond-Roy occlusion classification.
*The variation in time points for occlusion data introduces additional uncertainty for the pooled occlusion data. Cases from Martínez-Galdámez et al. were excluded from occlusion pooled data due to lack of follow-up. Due to significant loss of patients to follow-up, the occlusion outcomes from Schüngel et al. reported at 10.6 months were not used, and the outcomes reported at 7.7 months were used instead.
Additional outcomes
The incidences of major stroke, all-cause mortality, procedure-related neurologic mortality, post-operative aneurysm rupture, and other complications were reported by all four studies (Table 4). Two studies reported major strokes (ischemic and hemorrhagic), with one incident of stroke each.6,9 Overall mortality among the studies was 2.5% (4/163), with 3 cases adjudicated as neurologic deaths (1.8%).6,9 One was the previously described case of a ruptured MCA aneurysm that progressed with a stroke. The second case was a recurrent, previously ruptured PCA aneurysm (initially treated with alternative flow diverters); retreatment took place with the SVB device, but the patient presented three months later with a new subarachnoid hemorrhage. 6 The third case occurred in a patient who presented with severe SAH related to an acutely ruptured dissecting aneurysm of the vertebral artery. 9 Two studies reported no incidences of stroke, aneurysm rupture, or mortality.7,8 No incidents of thromboembolism were reported in any study.
Additional outcomes.
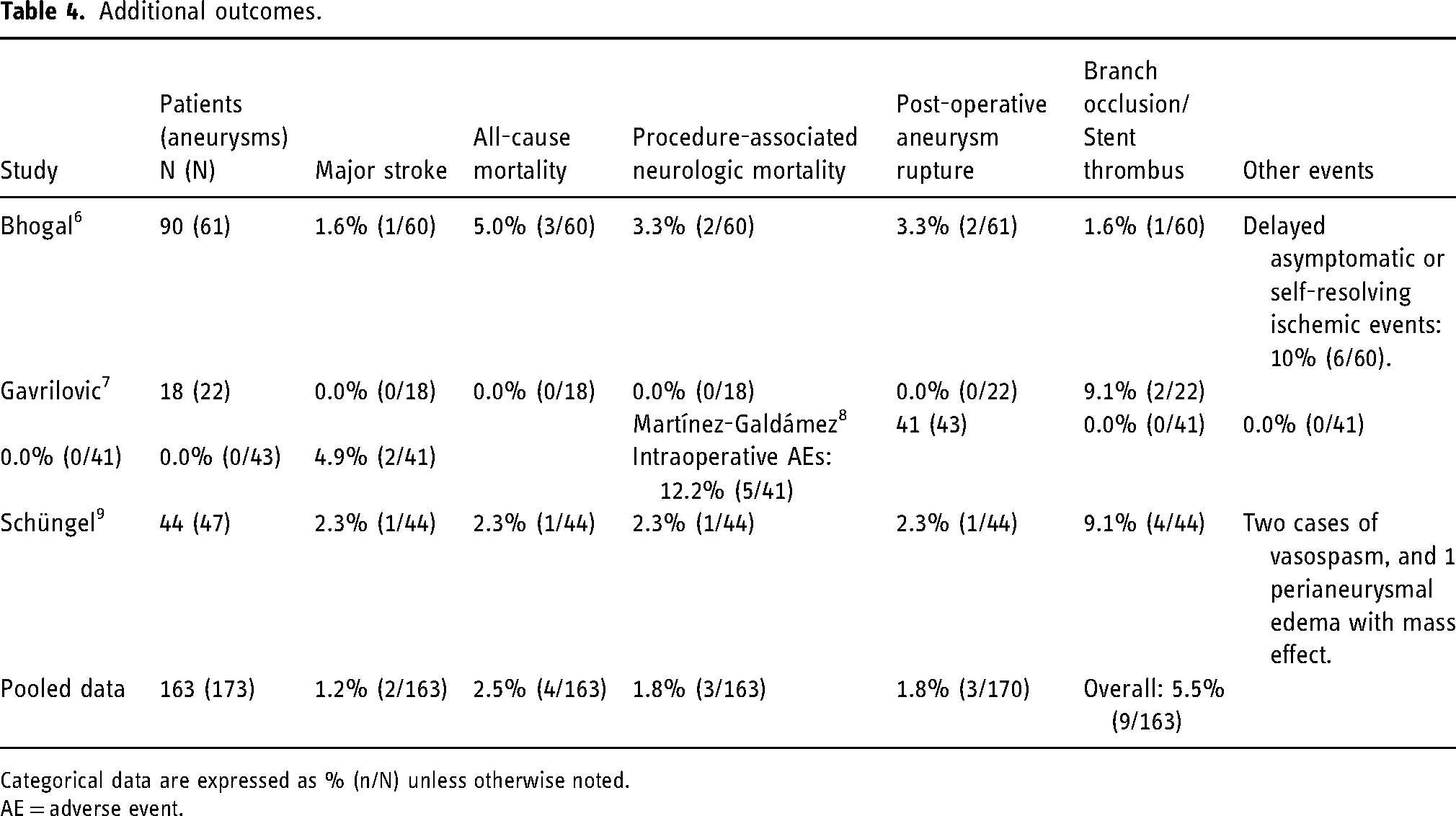
Categorical data are expressed as % (n/N) unless otherwise noted.
AE = adverse event.
All studies reported at least one case of branch occlusion, stent thrombosis, or delayed aneurysm filling, with an overall rate of 5.5% (9/163). Most events were transitory, successfully managed, or asymptomatic due to good collateral circulation. In two cases, occlusion was associated with major events, and one with a minor neurologic deficit.6,9 The overall rate of post-operative aneurysm bleeding across the studies was 1.8%, all cases occurring in previously ruptured aneurysms or acutely ruptured at baseline.6,9 Briefly, a giant previously ruptured PCA aneurysm initially treated with alternative flow diverters underwent retreatment with SVB due to worsening mass effect and aneurysm enlargement; new localized bleeding was noted three months later. The second case was a ruptured AcoA aneurysm initially treated with coiling that underwent retreatment with coiling and the SVB device implantation; a new hemorrhage occurred 11 months later. The third case was an acutely ruptured dissecting vertebra-basilar aneurysm treated with SVB with a significant episode of SAH associated with uncontrollable intracranial hypertension.
Additional adverse events and complications included one study reporting that, besides a case of a major stroke, other six patients (10%) experienced delayed clinical or radiological evidence of ischemia, with all cases demonstrating spontaneous resolution of symptoms. 6 Intraoperative complications occurred in five cases (12.2%) in one study, all of which were successfully managed without sequelae. 8 These include one branch occlusion, two vasospasm, one distal branch perforation, and one proximal-end stent clot formation. 8 Local mass effect after device deployment was reported in one case, with complete resolution of the symptoms after treatment. 9
Discussion
The results of this systematic literature review suggest that SVB may be used to safely and effectively treat intracranial aneurysms in small vessels (<3.5 mm). The device has been used to successfully manage aneurysms with parent artery diameters as small as 0.9 mm. 8 Additionally, our review emphasizes a high rate of technical success, including treating ruptured aneurysms, which corresponded to 1 of every 4 lesions treated with the SVB device. Major stroke, post-operative aneurysm rupture, and mortality occurred infrequently in our review of the literature. Other complications included symptomatic ischemic events, side-branch occlusion, and in-stent thrombosis, which were also uncommon. The studies included ruptured and unruptured aneurysms and different aneurysm types, including not only saccular but also dissections, fusiform, and blister aneurysms. Some of these conditions present a higher morbimortality profile that was not observed in this review with SVB.
The necessity of using a 0.021” or larger microcatheter delivery system for navigation in tortuous anatomies and the narrow range of available sizes options were commonly technical constraints encountered with previous flow diverters. Comprehension of flow dynamics and optimization of flow diverter technology was accompanied by evolving of stent designs. 19 The feasibility of endoluminal reconstruction for managing aneurysms located at distal vessels has been previously suggested. In an early experience with the pipeline embolization device (PED; Medtronic, Irvine, California, USA) for the treatment of distal anterior circulation aneurysms, Lin et al. reported rates of complete occlusion of 77.8% at a follow-up average of 7.7 months and an overall complication rate of 10.8%, including two periprocedural ischemic events (7.1%) adjudicated as clinically significant. 20 Cagnazzo et al. analyzed in a systematic review the outcomes of flow diverters among patients with unruptured distal anterior circulation aneurysms, defined as MCA branches, anterior communicating artery, and distal anterior cerebral artery (A2 or beyond). In a sample originating from 27 studies, the rate of adequate long-term occlusion was 82.7% (295/364; 95% CI, 77.4%–87.9%; I2 52%). The overall complication rate was 12.5%, and ischemic/thromboembolic and hemorrhagic events were noted in 9.9% (54/418; 95% CI, 7%–12.7%; I2 6.6) and 2.6% (7/418; 95% CI, 1.1%–4%; I2 0%) of cases, respectively. 21 A multicentric cohort by Ravindran and colleagues on 46 distal aneurysms successfully treated with flow diverters reported a 78.2% adequate aneurysm occlusion rate, with a higher likelihood of occlusion in lesions located in the posterior circulation. 22 They reported two cases (4.3%) of perforator vessel stroke but no hemorrhagic complications. 22 Limbucci et al. assessed the expanded indications for flow diverters, including distal aneurysms treatment, and described a rate of adequate aneurysm occlusion in the literature ranging from 60 to 90% and treatment-related complications from 4% to 17%. 23 When comparing the safety and efficacy outcomes of the different devices, heterogeneity within and between the studies must be considered owing to significant variability in aneurysm location, vessel dimensions, inclusion criteria, technical approaches, and outcomes definition.
The Flow Re-Direction Endoluminal Device Jr (FRED Jr, MicroVention, Tustin, California, USA) was the first flow diverter dedicated to small vessels (≤3 mm). It is a dual-layer stent comprised of integrated nitinol, and it is delivered through a 0.021-inch microcatheter. The device has the delivery wire finishing inside the stent during deployment, providing a technical advantage of not dealing with possible distal wire problems when navigating small, tortuous vessels. In a multicentric experience including all but one case of unruptured aneurysms, Möhlenbruch et al. reported a rate of complete or near-complete occlusion at 6 months follow-up in 77% in patients treated with FRED Jr. 24 Safety outcomes occurred in 7% of the cases, represented by one disabling ischemic stroke, one minor stroke with complete recovery at discharge, and one. Jesser et al. observed similar rates of adequate occlusion on early follow-up, with a 16% overall complication rate, 11% of which related to thrombotic-ischemic events. 25 The dual-layer concept has raised concerns about increased thrombogenicity, and larger prospective studies are needed to better assess this issue.
The p48 flow diverter (Phenox, Bochum, Germany) is delivered through a 0.021-inch microcatheter and was also designed to be accommodated in small parent vessels, with rates of complete aneurysm occlusion varying from 47.9% to 75% on early angiographic follow-up.26–28 The complications encountered in previous experiences using the device included hemorrhagic and ischemic events, with reported morbidity ranging from 2.5% to 13.6%.26–28
The SVB is a 48 self-expandable, braided construct composed of drawn filled tubes (DFTs) of platinum-filled nitinol, with the latter component contributing to elasticity and shape memory. 29 The mechanical properties and design of the stent facilitate visualization of the device, navigability, and controlled deployment. These characteristics are of great utility during the treatment of aneurysms located in small vessels, as some technical limitations are unique to this subset of aneurysms. The SVB is the first in class FD to be delivered through a 0.017” delivery microcatheter, permitting better trackability and facilitating distal access, and allowing for decreased vessel trauma, induced vasospasm, and possible vessel perforation. Our results demonstrated that technical success of SVB device placement was reported in 98.8% of the cases, and complete or near-complete aneurysm occlusion was obtained in 72% of cases at early-term follow-up. The optimization of the technology, like device trackability and reduced system dimensions, also facilitated the treatment of previously judged not amenable lesions, including a wide range of aneurysms located in anterior and posterior circulation distal vessels. Safety events represented by major stroke (1.2%) and neurologic death (1.8%) were low. SVB mortality and complications were similar or lower than those reported for other FD stents used in the treatment of unruptured anterior circulation aneurysms. 21 SVB mortality and complications were also comparable to preliminary outcomes reported for the LVIS Jr.30,31 This emphasizes the safety profile of the SVB device in the management of intracranial aneurysms.
Limitations
Our study had several limitations. The data were not stratified by aneurysm location or vessel size, preventing correlations between outcomes and vessel size. While exact vessel size is ideal for future analysis, location can be used as a proxy for vessel size, so in the interim, location-specific analyses could be used to examine performance in small vessels. The lack of angiographic follow-up in one study, differences in follow-up time, and absence of long-term results limit the internal validity and generalizability of the results. Another major limitation was that SVB is currently only reported in retrospective case series, with no clear head-to-head evaluation versus other devices, where failed procedures or conversion to different strategies happened secondary to limitations of catheter size and larger profile FD delivery system, not captured in retrospective published series. We have presented unadjusted pooled values for comparison, but these values have high uncertainty due to variation in patient populations across studies. A further prospective trial with intention-to-treat analysis and success rate of device implantation is necessary to determine the performance of SVB compared to other treatments, particularly other devices designed for use in small vessels.
Conclusion
SVB may be a safe and effective option to treat intracranial aneurysms in small vessels. Initial results from retrospective case series suggest that SVB is at least comparable to other FD stents. Future randomized, comparative studies will be needed to directly compare the safety and efficacy of SVB to other treatment options.
Footnotes
Acknowledgements
The authors acknowledge Karl Holub, Stephen Mead, Jeffrey Johnson, and Darian Lehmann-Plantenberg for their design and support of the Nested Knowledge meta-analytical software.
Data availability
Data are available upon reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RAH is a consultant for Medtronic, Stryker, Cerenovous, Microvention, Balt, Phenox, Rapid Medical, and Q'Apel. He is on advisory board for MiVI, eLum, Three Rivers, Shape Medical and Corindus. Unrestricted research grant from NIH, Interline Endowment, Microvention, Stryker, CNX. Investor/stockholder for InNeuroCo, Cerebrotech, eLum, Endostream, Three Rivers Medical Inc, Scientia, RisT, BlinkTBI, and Corindus. ES is employed by Superior Medical Experts. DS is employed by Nested Knowledge. JP is employed by and has ownership interest in Superior Medical Experts and Nested Knowledge, Inc. VMP is a consultant for Stryker, Medtronic, Penumbra, Neurovasc, and Balt. All other authors report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Balt, USA supported the study through an unrestricted grant, (grant number N/A).
