Abstract
Introduction
Current methods for angioplasty and stenting of the intracranial vasculature for neurointervention are limited. The Wingspan Stent System is Food and Drug Administration (FDA) approved with human device exemption for a limited patient group and despite numerous prospective registries and trials demonstrating reasonable safety, still carries warnings from the FDA for its use. Given these limitations, we present the technical nuances and outcomes of the off-label use of the Resolute Onyx drug-eluting stent (DES) for neurointerventional purposes.
Methods
Retrospective chart review of all patients undergoing a neurointerventional procedure with the Resolute Onyx DES was done from January 2017–2021. The Resolute Onyx is a coronary balloon-mounted drug-eluting (zotarolimus) single wire laser cut stent. Technical details and procedural outcomes were collected.
Results
In total 40 patients had attempted placement of the Resolute Onyx DES with procedural success in 95% of patients. The most common vessel stented was the basilar artery, 30% (12/40). The most common indication was intracranial atherosclerotic disease in 62.5% (25/40) patients, followed by acute stroke in 17.5% (7/40) of patients. The technical and procedural outcomes were excellent with only one technical complication (2.5%).
Conclusions
This series describes the initial technical safety and utility of utilizing a new generation balloon-mounted drug-eluting stent for neurointerventional purposes. This stent offers the potential for improved navigability, delivery, and outcomes compared to current neurointerventional options and warrants further study.
Keywords
Introduction
Stenting of intracranial stenosis and occlusion is challenging with the currently available neurointerventional devices. This was highlighted by the high rates of periprocedural complications in the Stenting versus Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis (SAMMPRIS) trial. 1 In SAMMPRIS, the 14.7% rate of 30-day complications in the stenting group far outweighed the 5.8% rate seen in the medical management group with this significantly higher rate of stroke and death being driven by procedural complications of the stent placement, which accounted for 75.8% of those 30-day complications. 1 SAMMPRIS required the use of the Gateway percutaneous transluminal angioplasty (PTA) Balloon Catheter and Wingspan Stent System (Boston Scientific), which utilizes angioplasty followed by an exchange to the stent system and relies on the outward radial force of the stent to open the lesion. 2 This leads to potential technical failures where the stent fails to deploy in significantly stenotic lesions. 3 Furthermore, the Wingspan Stent System is prone to high rates of in-stent stenosis.4–6
Early balloon-mounted drug-eluting stents (DES) were significantly limited by their stiffness and poor trackability. Second-generation drug-eluting stents, which incorporate newer alloys and polymers, utilize systems that substantially improve their deliverability. These newer coronary stent systems offer the benefit of high navigability and drug-eluting design thus decreasing the risk of in-stent stenosis.7,8 The additional benefit of a balloon-mounted stent requires practitioners only to cross the stenotic lesion once and ensures adequate stent deployment. However, these balloon-mounted DES have not been widely tested for neurointerventional procedures. Further potential benefits of the Resolute Onyx DES (Medtronic, Minneapolis, MN) include: zotarolimus polymer coating for the prevention of restenosis, single wire laser cut design enhancing navigability, and excellent angiographic visualization due to the platinum iridium core.9–11
To date, only one series limited to 18 patients addresses the potential utilization of this stent for neurointervention. 12 Furthermore, it only includes patients with symptomatic intracranial atherosclerotic disease. 12 Given the limited literature on the Resolute Onyx DES in neurointervention, we sought to describe our institutional experience. The purpose of this study was to demonstrate the technical nuances and procedural safety of the Resolute Onyx DES for neurointerventional procedures.
Methods
Patient selection
All patients who had a Resolute Onyx drug-eluting stent placed or attempted to be placed during their neurointerventional procedure at a single institution between January 1st, 2017 to January 1st, 2021 were included in this study. Institutional Review Board approval was obtained. All patients who underwent stenting utilizing the Resolute Onyx drug eluting stent for a neurointerventional procedure were included in the study. The Resolute Onyx stent was used off- label in all cases. It is institutional practice to use the Resolute Onyx stent for patients requiring intracranial stenting for symptomatic ICAD, stenosis, or in the setting of acute stroke.
Data and analysis
Patient data included age, sex, diagnosis, and post-procedural complications. Procedural and radiological data included access system, vessel stented, stent size, proximal and distal lumen diameters, diameter of stenotic segment, dual antiplatelet regimen, number of stents deployed, and post-stenting diameter. Any immediate periprocedural imaging was reviewed as well. The primary outcome assessed was technical success of the procedure as well as any periprocedural complications. Categorical variables were described as frequencies and percentages, while continuous variables were described as mean ± standard deviation. A student's T test was used to compare pre- and post-stenting vessel lumen diameter.
Device details
The Resolute Onyx Zotarolimus-eluting stent is a balloon-mounted coronary stent deployed via rapid exchange system or over-the-wire technique. Both methods utilize a 0.014-inch microwire. The stent is made from a single wire shaped into a sinusoidal wave and subsequently helically wrapped and laser fused. The regular shape is used to enhance the ability to recross the stent. The stent itself consists of a platinum iridium core to enhance visibility as well as a cobalt alloy shell and zotarolimus polymer coating. It comes in a range of sizes from 2.0 to 5.0 mm in diameter with increasing 0.25 mm and 0.5 mm increments with a half millimeter expansion capability. 9 The 2.0 to 4.0 mm stent systems have an outer diameter of 2.7 French, while the 4.5 to 5.0 mm systems have an outer diameter of 3.2 French. The maximum system length is 140 centimeters. The system requires a 5F/0.056in guide catheter for use. To enhance delivery, the device has 2 radiopaque markers. The over-the-wire system is not available in the largest 4.5 mm and 5.0 mm diameters. In the coronary setting, the stent is approved for one month of dual antiplatelet therapy (DAPT); however, given the risk of in-stent stenosis in neurointerventional procedures, all patients were planned to maintain dual antiplatelet therapy for at least 6 months.
Procedural details
This series included all patients who underwent stenting with the Resolute Onyx DES; indications ranged from recurrent stroke due to intracranial atherosclerotic disease (ICAD), acute stroke with immediate re-occlusion or inability to open the target vessel, arterial dissection, and in-stent stenosis from another device. When possible, patients were placed on DAPT for at least 5 days prior to the procedure and confirmed to be therapeutic via Accumetric P2Y12 testing. If the stent was placed in the acute setting, early cases in the series received intraprocedural Eptifibatide (Integrilin), while the current practice at our institution is to initiate an intravenous Cangrelor (Kengreal) infusion at the time of stenting. Guide catheter varied per procedure but was always at least 0.071in diameter. An intermediate catheter was used for all intracranial cases and the stent was deployed over either a Synchro2 (Stryker) or Transcend EX (Boston Scientific) microwire. Stent size was chosen based on 80% of the proximal parent vessel diameter, while stent length was chosen based upon the shortest possible stent to cover the diseased region. In cases of acute occlusion, a microcatheter was initially passed through the stenotic segment and microangiography was performed to ensure the distal end of the device would be deployed into the true lumen. If multiple stents were placed, overlap was minimized and avoided when possible. During stent placement, the stent was deployed in a fashion to avoid overlap over branching vessels. Balloon inflation was not always to nominal pressure, but instead done by visualization and halted once the stent was successfully expanded to a reasonable diameter when compared to the adjacent normal parent vessel. The stent was deployed and detached from the delivery system by deflating the balloon. After stent deployment, micro cone-beam computed tomography angiography (DYNA CT) (Siemens Healthineers AG, Erlangen, Germany) was used to assess stent apposition and patency. (Table 1)
Patient demographics.

Results
Patient details
A total of 38 patients underwent stenting. Two patients had attempted but ultimately failed stenting. One patient had severe ICAD in both the vertebral and internal carotid artery and underwent stenting of both vessels. 32.5% of patients were women with a mean age of 64.1 years. The most common indication for stenting was recurrent stroke in the setting of ICAD in 62.5% of patients (25/40). Other indications for stent placement included acute ischemic stroke with inability to recanalize the vessel using aspiration or stent retriever (17.5%), arterial dissection (2.5%), vertebral origin stenosis (7.5%), or in-stent stenosis of a prior flow diverter (7.5%). The most common vessel stented was the basilar artery (30%) followed by the internal carotid artery (25%).
Technical and radiological outcomes
The procedure was a technical success in 95% (38/40) of cases. In one case, vessel tortuosity limited the ability of the delivery system to reach the target M2 vessel and in the other case, the stent was unable to reach the target lesion due to length limitation. In total, 97.5% of procedures were not associated with a complication. There was one periprocedural technical complication: patient developed dissection upon placement of the access system that subsequently led to vessel rupture when the balloon was inflated, and the stent was deployed leading to a cavernous carotid fistula. The dissection was only noted on post-procedural review of the images. The most common guide sheath for the semi-elective cases was a 5F Fubuki, which was used in 45% (18/40) of all cases, while for acute stroke was the 6F BMX used in 57.1% (4/7) of stroke cases (Table 2). An intermediate catheter system was used in 82.5% of cases (33/40), most often a Sofia EX 115 cm 42.5% (17/40). The most common wire used for delivery was the Synchro2 microguidewire (82.5%), although in 17.5% of cases a Transcend EX Extra Support microguidewire was utilized.
Procedure and technical details.
Mean vessel diameter was 3.1 mm with a mean stenotic diameter of 0.7 mm. Mean percent stenosis was 77.9%, which improved to 16.5% after stenting (P < 0.05). The percent change in diameter of the stenotic segment increased significantly post stenting by 59.6%. Post-stenting angioplasty was required in a single case, while pre-stenting angioplasty with the Maverick balloon was done in 3 cases. Only 4 patients required more than one stent be placed at the time of the procedure. Of the 33 patients who underwent stent placement semi-electively, 81.8% (27/33) underwent post-angiography MRI. There were no large new infarcts on MRI; however, 33.3% of patients (9/27) had MRI demonstrating limited number of small punctate foci with restricted diffusion (Table 3). Interestingly, one patient had reduced number of foci of restricted diffusion from pre-stenting to post-stenting MRI. Two patients with vertebrobasilar ICAD had a worsening of their neurological exam post procedure. One was transient and returned to baseline prior to hospital discharge. The other patient had a small punctate medullary infarct leading to swallowing difficulty requiring a gastrostomy tube with no other neurological deficits.
Complication details.
Case example 1
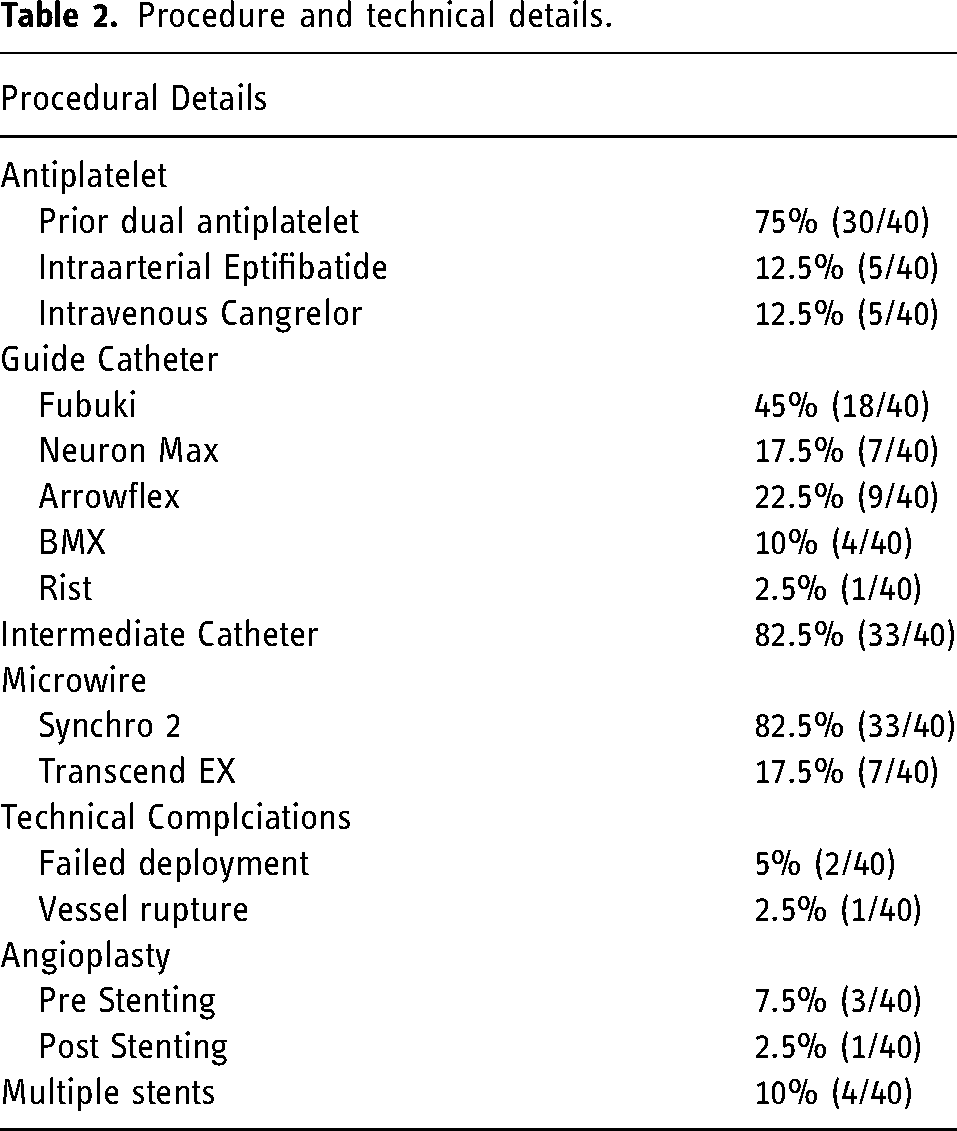
A 70-year-old man presented to an outside hospital with left hemiparesis and dysarthria with an unknown last well-known time. CT Angiography (CTA) Head and Neck and CT Perfusion (CTP) demonstrated vertebrobasilar stenosis with elevated Tmax throughout the posterior circulation. He was subsequently transferred to our institution for further management. Upon arrival he had NIHSS 14 with worsening 3 h later to NIHSS 23 from profound lethargy and was subsequently taken for emergent cerebral angiography. Diagnostic angiography revealed diffuse intracranial vertebrobasilar atherosclerosis (Figure 1). The right vertebral artery was occluded at the level of the foramen magnum with near occlusion of the left vertebral artery at the vertebrobasilar junction. The right vertebral artery was dominant. Given the severity of the disease, the patient underwent balloon angioplasty and stenting to achieve adequate recanalization.
(A) Computed tomography perfusion demonstrating significant time to maximum elevation in the posterior circulation. (B) Prepocedure angiography. Dual injection through the intermediate catheter in the distal vertebral artery as well as the microcatheter which is in the top of the basilar artery. Clear evidence of distal vertebral artery occlusion as well as diffusion ICAD in the vertebrobasilar system. (C) Final Resolute Onyx stent construct. (D) Post procedure angiography showing a now patent verterbrobasilar system. (E) Post procedure QMRA NOVA showing good flow in the vertebrobasilar system. ICAD: Intracranial atherosclerotic disease; QMRA NOVA: Quantitative magnetic resonance angiography non-invasive optimal vessel analysis.
A 6F ArrowFlex sheath was advanced into the right vertebral artery followed by a 5F Sofia EX intermediate catheter. Given the occlusion, aspiration was attempted twice with no evidence of reperfusion. A Headway Duo microcatheter was then navigated over a Synchro2 microguidewire into the distal basilar artery. The microcatheter was removed while keeping a Synchro2 exchange length microguidewire in place. Over the Synchro2 microguidewire a total of 3 Resolute Onyx stents were placed from the distal basilar artery to the distal right vertebral artery. Each balloon was inflated to 8 atm to successfully deploy the stent. On post-procedure angiography, the basilar artery was patent with no residual stenosis and significantly improved flow, though the right superior cerebellar artery was occluded. Post-procedure MRI demonstrated diffuse regions of diffusion restriction in the posterior circulation and an expected infarct in the right superior cerebellar artery distribution. The patient's neurological examination post-extubation demonstrated NIHSS 18, which subsequently improved to 13 upon discharge. The patient was discharged on dual antiplatelet therapy.
Case example 2
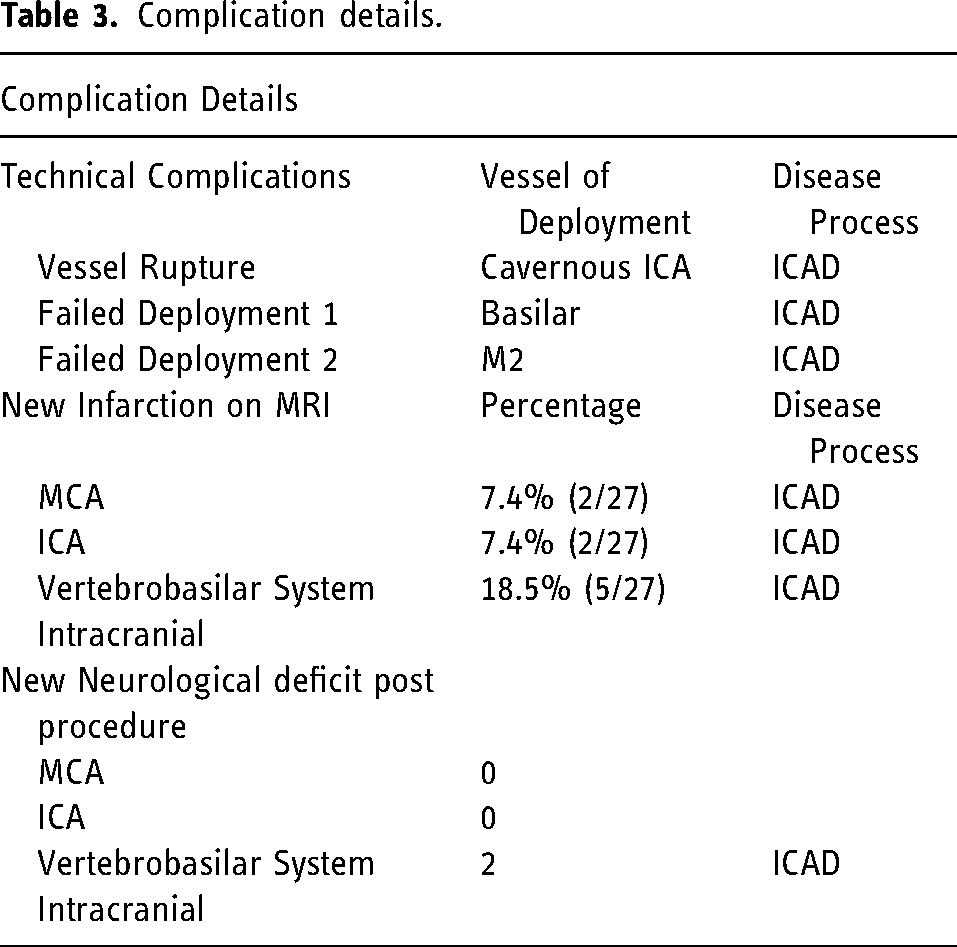
A 76-year-old man presented to our hospital with last well-known of one day with headache, confusion, dysarthria, and trace left-sided weakness with NIHSS 2. CTA Head and Neck and CTP demonstrated a right M1 focal stenosis with elevated Tmax in the right MCA territory. The patient was placed on DAPT and observed with permissive hypertension. MRI demonstrated low flow on quantitative MRA (QMRA) and diffusion weighted imaging (DWI) restriction throughout the right MCA vascular territory (Figure 2). During the patient's hospital course, he had multiple transient episodes of worsening left hemiparesis and neglect, prompting diagnostic cerebral angiography for assessment of the M1 stenosis with possible intracranial stenting.
A 76-year-old male who experienced recurrent right MCA infarcts and was found to have flow-limiting stenosis on his initial imaging evaluation. Pre-procedure MRI demonstrated watershed infarct pattern on MRI. (A) Angiography demonstrated severe stenosis of the right M1. (B) Syngo iFlow analysis of the right ICA injection demonstrating the time density color map of the right MCA territory. Compared to the ipsilateral ACA, there is significantly decreased flow. (C) QMRA NOVA showing flow-limiting stenosis of the right MCA stenosis. (D) Post-procedure angiography demonstrating resolution of prior stenosis after placement of the Resolute Onyx stent. (E) Post-procedure iFlow analysis showing resolution of time density curve flow limitation on angiography. (F) Post- procedure QMRA NOVA showing improved volumetric flow after placement of the right MCA stent. MRI: Magnetic resonance imaging; ICA: Internal carotid artery; MCA: Middle cerebral artery; ACA: Anterior cerebral artery; QMRA NOVA: Quantitative magnetic resonance angiography non-invasive optimal vessel analysis.
A 5F Fubuki was placed into the right ICA followed by a 5F Navien intermediate catheter. Angiography demonstrated severe stenosis of the proximal right M1 up to the MCA bifurcation. A Synchro2 wire was navigated into the right M2 and a 2 × 8 mm Resolute Onyx stent was placed into the M1 segment. Post-procedure angiography and micro-DYNA CTA demonstrated good stent apposition with no evidence of in-stent thrombosis. The patient was neurologically stable with trace left hemiparesis post-procedure and the subsequent MRI demonstrated improved DWI restriction with improved flow on QMRA.
Discussion
The management of recurrent stroke in the setting of intracranial atherosclerotic disease (ICAD), and persistent occlusion during mechanical thrombectomy remain significant neurointerventional challenges. Currently, the only Food and Drug Administration (FDA) approved device for the treatment of symptomatic ICAD is the Wingspan Stent System with Humanitarian Device Exemption (HDE) approval; however, numerous other endovascular treatments of ICAD have been proposed.1,13–19 We present our technical results with the off-label use of the Resolute Onyx DES for neurointerventional procedures. Our series clearly demonstrates the diverse utility of this balloon-mounted DES as well as the technical safety. In our series, we encountered one technical complication likely from vessel dissection during vessel access and not due to the device itself. Furthermore, its use in the setting of acute ischemic stroke and in-stent stenosis offers another technically safe tool in the endovascular armamentarium to use as a bail-out technique. The Resolute Onyx DES clearly has a role in the acute treatment of failed thrombectomy as well as in the management of intracranial steno-occlusive disease given its technical applicability and reliability.
Currently, the Wingspan Stent System (Stryker, Kalamazoo, MI) is approved through the HDE pathway for the treatment of intracranial atherosclerosis. To deploy the stent, the stenotic lesion must first be treated with angioplasty using the Gateway PTA Balloon (Stryker, Kalamazoo, MI). After angioplasty, the systems require practitioners to remove the balloon and then recross the stenotic lesion to deploy the stent. Furthermore, since the stent itself is not balloon-mounted, some lesions require post-stenting angioplasty to completely open the stent. Beyond the need to exchange the system multiple times, the polyolefin tip of the delivery system can make stent deployment difficult over tight angles. 20 In SAMMPRIS, the majority of strokes occurring within the first 30 days of the procedure highlighted the periprocedural risks of this device system. More recent data from the Wingspan One-year Vascular Events and Neurologic Outcomes (WOVEN) and Wingspan Stent System Post Market Surveillance (WEAVE) trials does demonstrate that safety of the device in carefully selected patients in the hands of high-volume practitioners; however, data from the WOVEN trial still demonstrates high rates (17.4%) of in stent restenosis.13,21 In comparison to the Wingspan system, the Resolute Onyx is a high navigability balloon-mounted stent requiring practitioners to only cross the stenosis a single time.
There has only been a single series describing the use of the Resolute Onyx DES for neurointerventional procedures. 12 The prior series by Hassan et al. clearly demonstrated an excellent safety profile and low rate of in-stent stenosis with the Resolute Onyx stent in a small group of patients with symptomatic ICAD. 12 The current series adds to these previously reported results and continues to demonstrate the safety and ease of use of this device in the setting of ICAD. Furthermore, our results support its use in failed recanalization after attempted thrombectomy in the setting of acute ischemic stroke due to large vessel occlusion. Older generations of DES have previously been described with one meta-analysis demonstrating a perioperative risk of 6.0%. 22 Perhaps the most significant reason for adoption of DES is the high rate of in-stent stenosis reported with the Wingspan device, which has been reported between 24.6% - 29.7%.23,24 Early generation coronary DES have shown excellent improvements in rates of in-stent stenosis, which have inevitably led to their off-label adoption in the field of neurointervention.22,25–27 The current literature unequivocally supports that the use of DES lowers the rate of in-stent stenosis significantly. 22 However, these early generation DES systems suffered from poor trackability and required large delivery systems, making navigation into the intracranial vasculature challenging. 28
Newer generation DES such as the Resolute Onyx stent have been associated with even lower rates of in-stent stenosis in the cardiac literature and their new design significantly improves navigability. 29 The Resolute Onyx stent is made from an 81-um diameter wire making it thinner than the prior generation stent increasing navigability and deliverability. Furthermore, the platinum iridium core increases both its radial force and its visibility on digital subtraction angiography.30–32 Furthermore, the Resolute Onyx DES has been tested in very small vessels with the Resolute Onyx 2.0 mm Clinical Study demonstrating safety and efficacy in coronary vessels averaging 1.91 mm. In the Onyx ONE trial, the Resolute Onyx stent was shown to deliver better angiographic and technical success when compared to other DES procedures.10,33 This is an extremely promising tool to adapt to the neurointerventional field.
Failure to achieve reperfusion when attempting a mechanical thrombectomy for a large vessel occlusion has been documented in DWI or CTP Assessment with Clinical Mismatch in the Triage of Wake-Up and Late Presenting Strokes Undergoing Neurointervention with Trevo (DAWN), The Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke (DEFUSE) 3, and trials evaluated in the highly effective reperfusion evaluated in multiple endovascular stroke trials (HERMES) meta-analysis to occur in 16–29% of patients.34–36 Salvage procedures for inability to achieve reperfusion with aspiration alone or a stent-retriever are limited and generally include balloon angioplasty, stenting, or infusion of intra-arterial thrombolytics. Their use has been well-described, most often in large vessel occlusions (LVO) occurring in the setting of ICAD.37–39 Because of these salvage techniques, rates of reperfusion in these patients have been reported to be as successful as in patients without ICAD ultimately resulting in similar patient outcomes. 40 However, these procedures are not without risk and delayed re-occlusion is known to occur. 40 This is the first series to report our experience with the Resolute Onyx for immediate re-occlusion after mechanical thrombectomy for LVO with ICAD as the underlying mechanism. In our series of patients, successful reperfusion was achieved in all cases with no post-procedural reperfusion hemorrhage. This treatment has the potential to provide durable reperfusion in these challenging patients.
There are obvious limitations of this study, including its retrospective nature and the small number of patients included in the study. However, its purpose is meant to demonstrate the technical feasibility of utilizing this stent in the intracranial circulation. In our series, we only had a single complication likely not associated with the stent deployment itself and 2 cases where the stent was not successfully delivered. Interestingly, one case was because the stent delivery system was not long enough to reach the target M2 vessel. The purpose of this series is also to demonstrate the Resolute Onyx stent's technicality and therefore does not report long term neurovascular or clinical follow up. More studies are needed to further assess this device and its utility in neurointervention given its technical promise and potential to improve the current treatment of classically challenging cerebrovascular diseases.
IRB: Human Research Protection Program, Institutional Review Board – Feinstein Institutes for Medical Research, Northwell Health: IRB # 20-1194.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
