Abstract
Background
The Low-profile Visible Intraluminal Support device (LVIS Jr) has become a commonly used intracranial stent for the treatment of intracranial aneurysms. However long-term stability and effectiveness remains to be seen. The purpose of the study was to assess the long-term efficacy, safety and durability of LVIS Jr. in a retrospective multicenter registry.
Methods
Patients with saccular aneurysms treated at centers across Canada using LVIS Jr for intracranial aneurysms were included in this retrospective registry between the dates of January 2013 and April 2019. Self reported outcomes were collected and used to assess both perioperative and long term safety and effectiveness. Both univariate and multivariate analysis were performed.
Results
Total of 196 patients (132 Women; mean age of 57.6 years) underwent endovascular aneurysm treatment with at least 1 LVIS Jr. stent. Mean aneurysm dome size was 7.4 mm, and mean neck size of 4.3 mm. Mean clinical and imaging follow up were 950 and 899 days respectively. Class I/II was achieved in 85% on long term follow up. Periprocedural morbidity and mortality was 4.6% and 2% and additional delayed morbidity and mortality was 3% and 2.5%. Aneurysm size >10 mm was independent predictor of periprocedural complication (OR 2.59, p = 0.048) while an increased dome to neck ratio >1.5 was independent predictor of increased delayed complications (OR 3.99, p = 0.02).
Conclusion
The LVIS Jr. intracranial stent is an effective device in the treatment of intracranial aneurysms. Satisfactory long term occlusion rates can be achieved safely with stent-assisted coil embolization.
Introduction
Intracranial stents are often used in the treatment of intracranial aneurysms, lessening angiographic recurrence rates and expanding the complexity of aneurysms considered amenable to endovascular treatment. They are most used in the treatment of wide neck intracranial aneurysms and can be used as a primary means of ensuring parent vessel patency or as a bail-out manoeuvre following attempted balloon assisted or primary coiling. The use of intracranial stents in the treatment of aneurysms has been shown to reduce angiographic recurrence rates. 1 There are several types of self expanding intracranial stents currently used for intracranial aneurysms, each with technical variations, limitations and improvements.2–4 Flow diverting stents offer a variation of intracranial stenting, with unique risk versus benefit profiles and have further increased the complexity of aneurysms able to be treated endovascularly.5,6
The LVIS Jr device is a nickel-titanium self expanding retrievable intracranial stent (LVIS Jr, MicroVention-Terumo, Tustin, California, USA). Its low-profile design allows it to be placed through 0.017 inch lumen microcatheter (Headway 17 microcatheter or Scepter balloon occlusion catheter, Microvention-Terumo, Tustin, California, USA). Improved visibility is achieved through a set of 3 tantalum markers on both the proximal and distal tines, as well as 3 radiopaque tantalum strands within the stent body. Diameters of 2.5–3.5 mm are intended for use in smaller parent vessels. The braided closed cell design results in a larger cell size of 1.5 mm, improving ease of catheter placement through the stent to facilitate coiling. With a metal surface area coverage of 17–23%, LVIS Jr is significantly higher than laser-cut hypo-tube stents, but remains lower than flow diverting stents.7,8
While safety and efficacy of the LVIS Jr device has been evaluated previously, long term studies demonstrating stent durability and persistent aneurysm occlusion are limited due to the relatively new nature of the device and the more recent advent of its widespread use. The purpose of our study is to assess the long-term effectiveness, safety and durability of LVIS Jr. in a retrospective multicenter registry. The current study expands upon the previously published Canadian Registry of LVIS Jr for Treatment of Intracranial Aneurysms (CaRLA), with increased duration of follow up and number of patients across multiple centers and operators. 9
Materials and methods
The Canadian Registry of LVIS Jr for Treatment of Intracranial Aneurysms (CaRLA) is a multicenter retrospective registry of consecutive patients undergoing stent-assisted coil embolization of intracranial aneurysms across Canada. Approval was obtained from research ethics board at individual centers. Six neurointerventional centers across Canada contributed to the registry. Anonymized case report forms were completed by practitioners at individual sites. Clinical and imaging follow up was according to standard of care and collected by the centers. Case report forms were submitted and reviewed at the core lab where results were consolidated and analyzed.
Patients were treated between January 2013 through April 2019. Results of the first 100 patients of shorter follow up has been previously reported. 9 Similar to previously described, only patients with saccular aneurysms were included. Single stents were most often deployed; however, multiple stents were used in various (X, YT-, Type and telescoping) configurations in 36 patients.
Clinical decision making was at the full discretion of the operator. Consecutive patients treated with LVIS Jr were included regardless of indication. This allowed diverse data collection including elective versus ruptured; and planned versus bail-out scenarios. Dual antiplatelet regimens were at the discretion of the operator and institutional standard of care. The majority included a dual antiplatelet regimen of aspirin (ASA) (81–325 mg) and Plavix (75 mg) for 5 days prior to an elective cases. Cases requiring emergency stent placement (ruptured or unplanned stent placement for unintended complication management) were typically loaded with 650 mg ASA and 300 mg Plavix. Dual antiplatelet were typically continued for at least 3 months, followed by a single agent (usually ASA) for life.
Stents were most placed using the Headway 17 catheter. Coils were placed either through the same microcatheter used for stent deployment (microcatheter advanced through the stent) or through a separate microcatheter which had been placed in the aneurysm prior to stent deployment (trapping/jailing technique). Coil selection was at the discretion of the individual operator and varied across operators and sites.
Baseline clinical information collected included patient demographic information, clinical presentation, originating site, operator, equipment selection, aneurysm characteristics and antiplatelet regimen. Initial angiographic results and periprocedural complications were noted. Clinical and imaging follow up varied across sites regarding timing and imaging modality. Clinical follow up for delayed complications and modified Rankin scale (mRS) assessment was used throughout. Conventional angiographic and MRA follow up were both included. Treatment results were categorized as complete, neck residual or residual aneurysm according to the commonly used Raymond classification. 10
Statistics
Descriptive statistics and chi-squared tests were used to compare the different proportions for categorical variables. Logistic regression was used to calculate odds ratio for binary variables in univariate analysis. Variables that showed significant association with either successful occlusion of aneurysms as well as perioperative and delayed complications were included in the multivariate analysis using logistic regression. A p < 0.05 was considered significant. Stata 13.0 statistical package was used for statistical analysis.
Results
A total of 196 patients (132 Female; mean age – 57.6 years; range- 20–80 years) were included in the study. Patient, aneurysm and follow up details are provided in Table 1. Aneurysms ranged in size from under 2 mm to as large as 22.5 mm; with a mean of 7.4 mm. The majority were wide necked and most commonly located at the anterior communicating or basilar arteries. The majority were incidental, with approximately 13% presenting with clinical symptoms. Greater than 80% of symptomatic aneurysms presented with subarachnoid hemorrhage. An additional 27 patients underwent stent assisted coiling for retreatment of previously ruptured aneurysms. The mean clinical and imaging follow up were approximately 2.5 years, with a slightly longer clinical follow up.
Summary of patient demographics, aneurysm characteristics and follow up.

A summary of treatment details and outcomes are provided in Table 2. Stent related periprocedural complications occurred in a total of 27 patients, with stent related thrombosis being the most common in 17 (63%), and stent migration was seen in 4 patients. There were 13 stent related strokes, though the majority (9) resulted in non-disabling symptoms. Of the 17 patients having periprocedural stent related thrombosis, 4 had initial presentation of subarachnoid hemorrhage with only 1 of these patients being pre medicated with ASA and Plavix, the remaining 3 being bail out stent placement with an inherent higher rate of thrombus. Increased number of stents was associated with a higher incidence of in stent thrombosis, with 6 of the 17 (35%) patients having more than one stent. Of the remaining 190 patients, only 30 (15%) required more than one stent. Of the 17 patients with periprocedural in stent thrombosis, 7 were located in the anterior communicating artery and 6 within the basilar. Of these basilar aneurysms, 4 required more than one stent. Of the 13 unruptured patients that developed in stent thrombosis, 9 were on ASA and Plavix for at least 5 days prior; 2 were started on integrellin followed by ASA + Plavix or Prasurgel following unplanned stent placement.
Treatment details: technical considerations in treatment, complication and outcome summary.
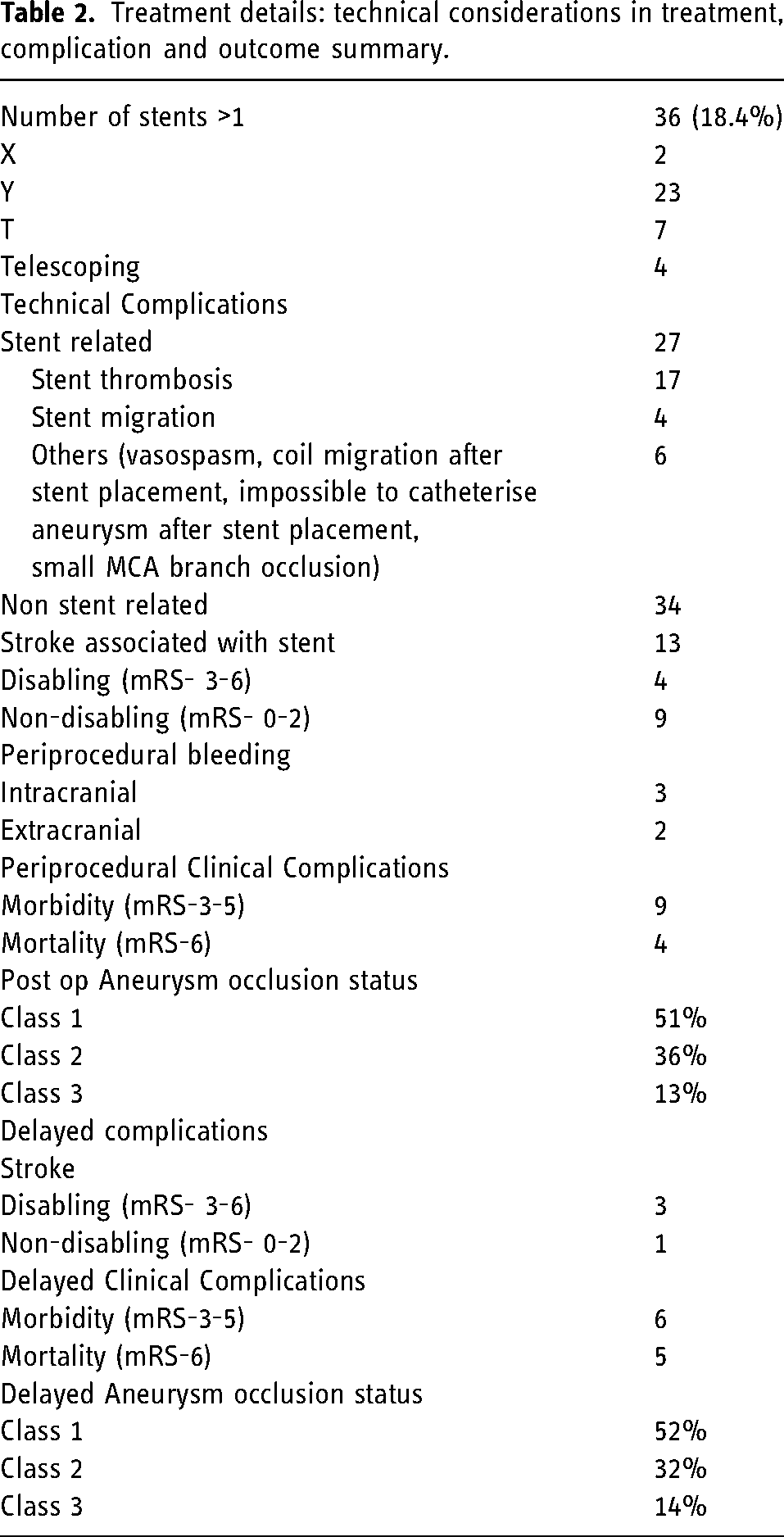
Of the procedure related deaths, 1 was attributed to refractory stent thrombosis in the basilar artery; and 1 related to a fatal intraprocedural aneurysm rupture. The remaining 2 periprocedural deaths were more complicated scenarios and not attributable directly to the stent placement. In the first, an LVIS Jr. stent was deployed in an attempted bail-out maneuver, in an attempt to open a migrated flow diverter which had occluded. The second, followed successful placement of an LVIS Jr. stent for treatment of protruding coil loops that were occluding an A2 branch. This patient had presented with a WFNS grade 5 subarachnoid hemorrhage.
Of the 5 patients with hemorrhagic complications, 2 were ultimately fatal. Two patients with extracranial hemorrhages were related to puncture site/retroperitoneal hematoma. Of the 3 patients having intracranial hemorrhage only 1 made a complete recovery. Of the two fatal intracranial hemorrhages, one patient had an intraprocedural rupture with a frontal hematoma requiring emergency evacuation. They were discharged to a regional palliative care center with mRS of 5; the other suffered an intraprocedural rupture and passed prior to discharge.
There were 5 deaths during the follow up period after discharge. Two of these patients suffered fatal in stent thrombosis, noting details regarding antiplatelet adherence at the time of in stent thrombosis were not clear. One patient passed from respiratory failure, and the remaining 2 died shortly after discharge – the exact details of which are not clear.
Cumulatively, 9 deaths occurred in this patient group over long term follow up, with significant neurological morbidity (mRS >2) in 21 patients. Of the deaths, 3 were related to in stent thrombosis; while 2 occurred after discharge, all occurred within 3 months of the procedure. Details regarding antiplatelet strategies, adherence to therapy and underlying comorbidities were not available. Dual antiplatelet strategies in patients where information was available were ASA and Plavix in 89% of patients, with ASA monotherapy, Ticagrelor + ASA or ASA + Prasugrel in the remaining; with 4% listing unknown antiplatelet therapies. Duration prior to therapy was most commonly 5 days; with 3 month follow up – noting variability in practices and limited reporting of duration. The routine use of antiplatelet response testing was not documented, though this is not routinely available at all sites included in this study and has only become more readily available recently. Of the remaining patients, 2 had documented dependence prior to procedure, while 8 patients had clinically significant strokes.
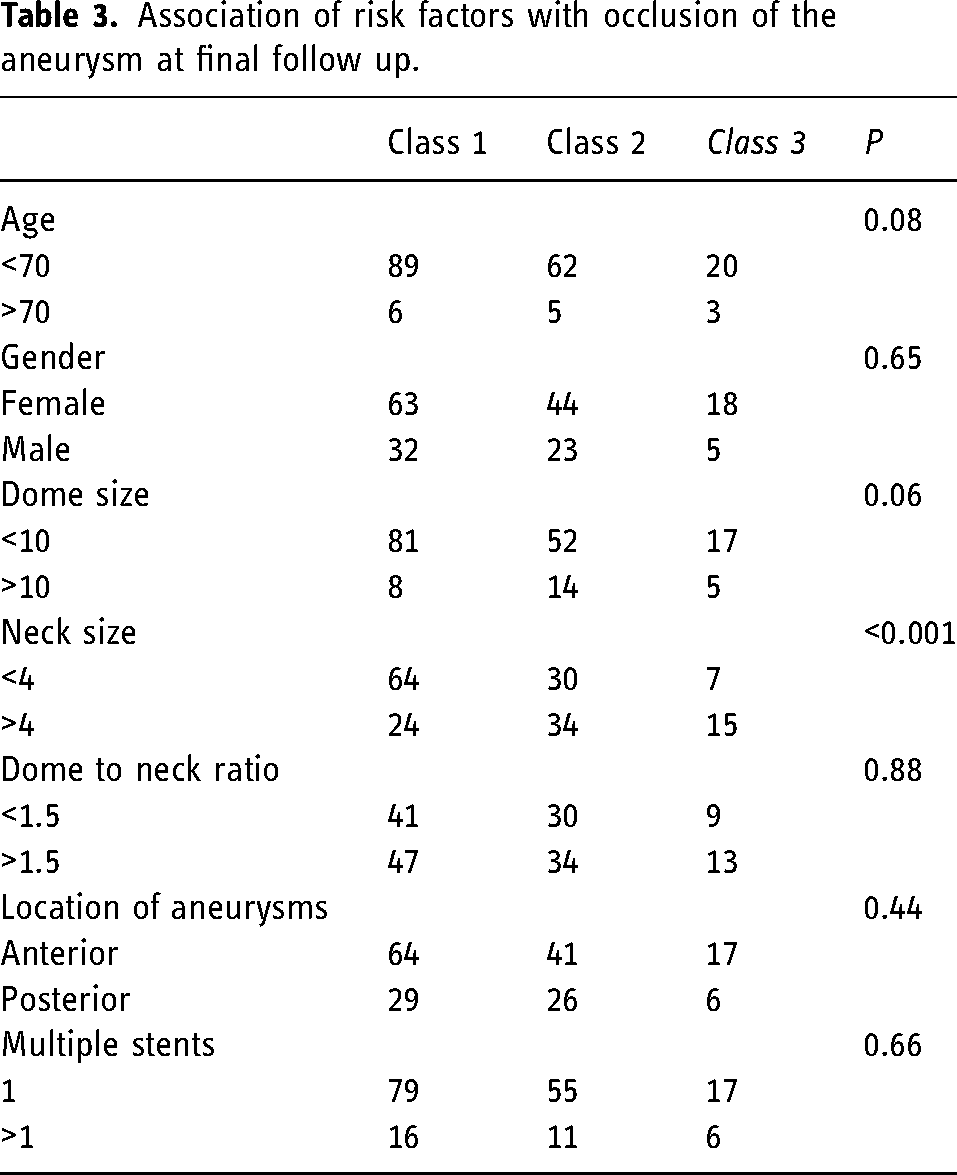
Treatment outcomes were favorable over our long term follow up, with complete occlusion in 52% of patients; and the majority having satisfactory (class 1 or 2) outcomes in 85% of cases. Factors associated with long term aneurysm occlusion are provided in Table 3. Patient age less than 70 and aneurysm size less than 10 showed a trend towards more complete aneurysm occlusion; however, only an aneurysm neck size of less than 4 mm was attributed with a statistically significant improvement in aneurysm occlusion.
Association of risk factors with occlusion of the aneurysm at final follow up.
Univariate and multivariate statistical analysis was performed to assess the association of risk factors with increased rates of complication (Table 4). Univariate analysis suggested higher periprocedural (less than 30d from procedure) complications with increased aneurysm size, and more than one stent being used; whereas delayed complications were more frequent with increasing patients age, posterior circulation aneurysms, increasing aneurysm size, increased dome to neck ratio and more than one stent being used. Upon multivariate analysis, in the periprocedural period an aneurysm dome size >10 mm (OR 2.59, 1.01–6.66); and on delayed follow up a dome to neck ratio of >1.5 (OR 2.35, 1.12–4.95), were found to be the independent predictors of complications.
Univariate and multivariate analysis for association with perioperative mRS (<30 days) and long term follow up mRS.
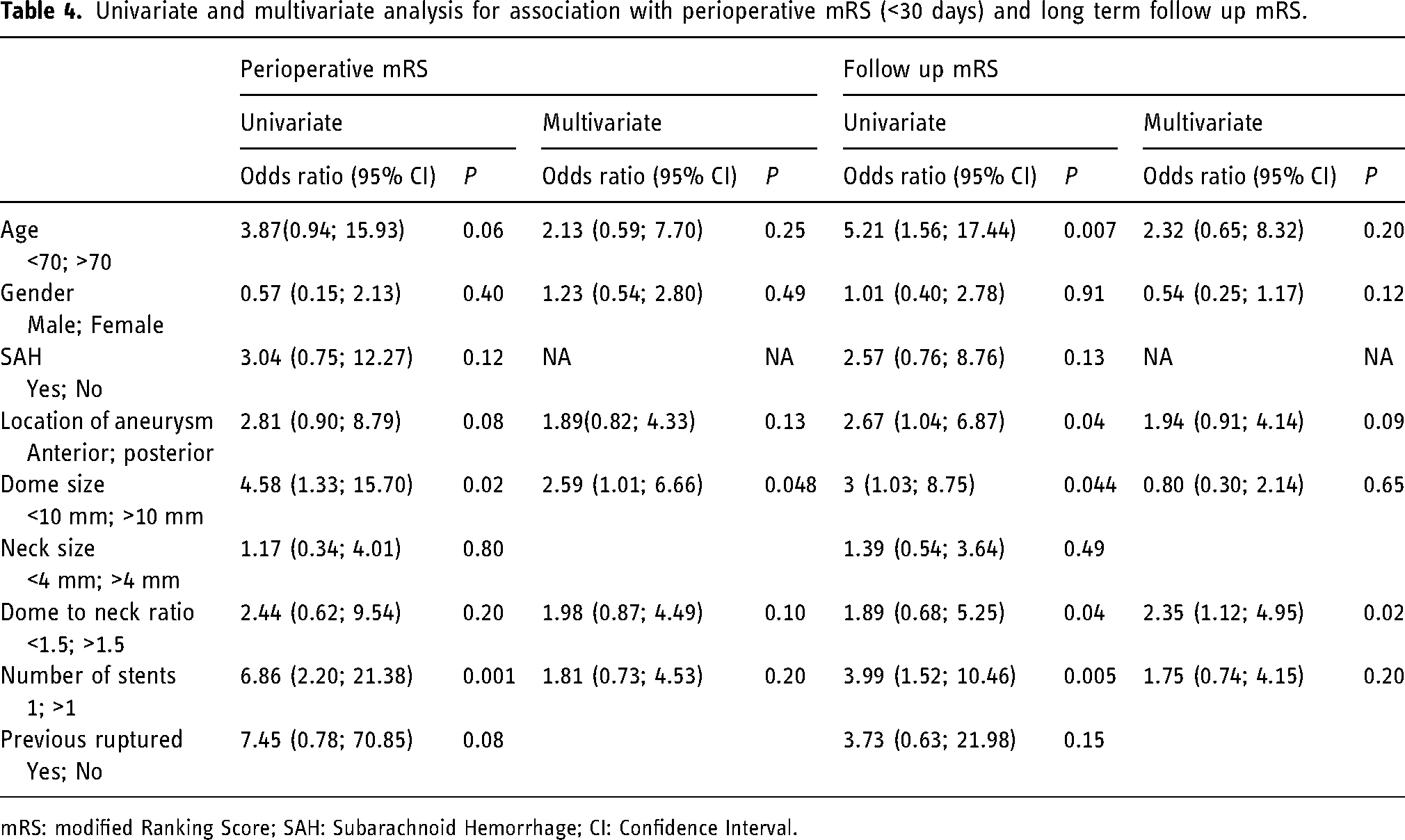
mRS: modified Ranking Score; SAH: Subarachnoid Hemorrhage; CI: Confidence Interval.
Discussion
While the safety and efficacy of the LVIS Jr device in the treatment of intracranial aneurysms has already been established,11,12 data on long term efficacy and durability are less robust due to its relatively recent development. The current study reports on the long-term effectiveness and safety of LVIS Jr from a large multicenter registry. We found stable class 1 and 2 results in 85% of patients. Of the 196 patients included, periprocedural morbidity and mortality was 4.6% and 2% and additional delayed morbidity and mortality was 3% and 2.5%.
The use of intracranial stents in the treatment of intracranial aneurysms has become a cornerstone adjunct in endovascular treatment. This is particularly true of wide neck, complex and even dissecting aneurysms.13–15 Successful treatment of intracranial aneurysms of essentially every location have been reported.16–18 The advent of flow diverters and reliability of balloons has not replaced the need for intracranial stents in many of these cases. Rather, technological advances and increased operator experience have contributed to the enduring use of these devices. 19 Varied techniques including the barrel and shelf techniques have also improved utility.20,21
Intended for use by a single operator, the LVIS Jr offers a low profile which allows it to be advanced through smaller microcatheters than larger intracranial stents. This is particularly advantageous in small, distal and often tortuous vessels; and in the treatment of very small aneurysms. 22 Added markers at the flared ends of the stent, and increased number of radiopaque wires within the stent body improves visibility. The widely adopted “C” generation of device offers more radial force, imparting higher technical success rates for deployment. Multiple studies have documented high success rates for stent deployment, often reaching or approaching 100%. 7 Our study is in keeping with other studies high technical success rates for stent deployment, with only 4 instances of stent migration across 196 patients treated with 232 LVIS Jr. and no documented cases of unsuccessful stent placement.9,23,24
Our study reports Raymond class 1 or 2 occlusion rate of 85%. Aneurysm occlusion rates with intracranial stent assisted coiling have a wide reported range, which is in part related to technological differences in stents, their evolution over time and varied use. A meta-analysis of intracranial self expanding braided stents, which included the LVIS devices (both LVIS and LVIS Jr) and Leo stents (both Leo and Leo Baby) (Balt Extrusion, Montmorency, France) demonstrated overall complete/near complete aneurysm occlusion rates of almost 80%. 23 For LVIS Jr specifically, this rate was 86%, comparable to the Raymond 1/2 rates of occlusion of 85% on delayed follow up in this study. LVIS Jr. specific studies have proven similar efficacy.11,25,26
Overall complication rates for intracranial stent placement are variably reported. Recent meta-analysis found thromboembolic complications in 4.7% of patients and periprocedural hemorrhage in 1.5%; with overall permanent morbidity of 2.1% and mortality of 1.1%. 15 Braided stents were found to have permanent morbidity rates of 1.6% and 1.1% respectively, noting this included non-LVIS Jr. braided stents. 15 The LVIS device specific TRAIL study (which included both LVIS and LVIS Jr. devices) was a multicenter observational study, that reported on 90 patients. 27 Overall reported procedure specific mortality rates of 3.3% and permanent morbidity rates of 5.6% with stent specific morbidity of 2.2%. 27 The US LVIS Pivotal trial which included a heterogenous group of patients and presentations best reflecting “real life” practice reported major strokes in 6.5% of patients, with overall mortality of 2%. 7
The current study reports on a similar heterogenous group of patients across multiple sites, attempting to emulate data from clinical practice. Overall procedural stent related complications of 14% appears high but is reduced to a 7% periprocedural risk of clinically significant events. Antiplatelet response testing was not routinely utilized; and antiplatelet strategies were not standardized across sites. Inclusion of patients presenting with ruptured aneurysms and unplanned stent placement in patients not premedicated with dual antiplatelets may be contributing to these numbers. A loading period of at least 5 days, followed by long term dual therapy of at least 3 months is commonly used, though others advocate longer intervals. Comprehensive results of long-term morbidity and mortality of 11% are taken in context of the entire patient population and reporting method. Of the 9 deaths, 3 could be reasonably attributed directly to stent related thrombosis. Patient suffering an intraprocedural rupture is difficult to attribute solely to the use of a stent, while another was used in an attempt to open an occluded and migrated flow diverter. Two patients’ deaths were at least in part related to underlying high grade subarachnoid hemorrhage; and details surrounding the deaths of two patients after their discharge (mRS = 0) are missing. Mortality directly related to stent associated complications in long term follow up would therefore range from 3/196 (1.5%) to 5/196 (2.6%) based on the inclusion of patients in whom details are not available. Similarly, of surviving patients with pre-treatment baseline function of >2 mRS; long term outcome of permanent disability is a comparable 8/187 (4.2%). This data is clinically significant as it provides a large number of patients with increased follow up to support previously published smaller trials of shorter duration. This supports the clinical use of the LVIS Jr stent and provides unbiased information on safety and effectiveness across patient and practice profiles, i.e., the heterogenous reality outside of clinical trials.
Similar to other studies on intracranial stents, increased complication rates were associated with larger aneurysm size, with perioperative complications slightly increased in in larger aneurysms (>10 mm) and long-term complications higher with increased dome to neck ratios on multivariate analysis. 28 Compliance with antiplatelet medications thought to be one of the most important cause of some of these delayed complications. Our study further emphasizes of the need of patients’ compliance to antiplatelet medication to prevent complications in the long term after use of intracranial stents. This has been highlighted in studies with the use of flow diverters.6,29,30
Limitations of this study include retrospective design and self reporting of outcomes. The absence of routine antiplatelet response testing and standardized dual antiplatelet regimens across sites is an additional limitation. Lack of an independent review process/central reading to objectively assess outcomes and assess complication adds another layer of potential bias. Varied reporting methods and operator/site specific decisions regarding treatment and follow up are potentially confounding. However, our group had been reporting roughly uniform.
Conclusion
The LVIS Jr. intracranial stent is an effective device in the treatment of intracranial aneurysms. Satisfactory long term occlusion rates can be achieved safely with stent-assisted coil embolization.
Footnotes
Contributors
James McEachern; conception, data acquisition, data analysis and interpretation, drafting, revising, and approving the manuscript. Daniella Iancu, Brain van Adel, Brain Drake, Zul Kaderali, Michael Spirou, Howard Lesiuk, Alain Weill, Daniel Roy, and Jean Raymond; conception, data acquisition, revising and approving the manuscript. Jai Shankar; conception, data interpretation, revising and approval of manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
