Abstract
Objective
p64MW HPC is a new low-profile flow diverter with reduced thrombogenicity due to hydrophilic coating. The purpose of this study was to evaluate its safety and efficacy in Mongolian patients under dual antiplatelet therapy.
Methods
Consecutive patients with unruptured anterior circulation aneurysms were prospectively enrolled. All patients received aspirin and clopidogrel before and six months after the procedure, followed by lifelong aspirin medication. High platelet reactivity (VerifyNow) did not trigger further action. The safety and efficacy endpoints were clinical outcome and aneurysm occlusion.
Results
In 29 patients (26 female, median age 57 years), 46 aneurysms (neck width 3.3 mm, fundus diameter 3.7 mm, median) were treated. Dual platelet function inhibition was confirmed in eight patients (28%). The response to Clopidogrel was between 100 and 239 P2Y12 reaction units (VerifyNow) in 13 patients (45%). Non-response to at least one drug was found in 8 of 29 patients (28%). One collapsed p64MW HPC required balloon angioplasty. No other periprocedural thrombus formation occurred. Postprocedural MRI revealed lesions with diffusion restriction in 3 of 29 patients. Digital subtraction angiography after three months for 42 of 46 (91%) aneurysms showed an adequate aneurysm occlusion in 25 (60%). Distal p64MW HPC migration of 3 implants was retreated with another p64MW HPC. Follow-up digital subtraction angiography of 26 of 46 (57%) aneurysms after six months showed adequate aneurysm occlusion in 22 (85%). Significant in-stent stenosis or thrombosis, morbidity or mortality was not encountered.
Conclusion
p64MW HPC implantation in patients under dual antiplatelet therapy with or without dual platelet function inhibition has a low procedural complication rate. The early aneurysm occlusion rate is high.
Keywords
Introduction
During the decade from 2008 flow diversion became a generally accepted and widely used treatment strategy for selected intracranial aneurysms. 1 The first generation of devices such as Silk (Balt Extrusion), Surpass (Stryker), Pipeline (Medtronic), and FRED (MicroVention) require 0.027 in inner-diameter (ID) catheters (e.g., Marksman; Medtronic). These implants have significant thrombogenicity, which contributed to the rate of thromboembolic complications.2,3 Both aspects have been addressed during the development of the new p64MW HPC flow diverter. This novel flow diverter requires only a 0.021 in ID microcatheter. The hydrophilic surface coating using a glycan polymer simulates features of the glycocalyx, thus reducing thrombogenicity. 4 We sought to evaluate this so far untested flow diverter under real world clinical conditions in the treatment of unruptured anterior circulation aneurysms in patients under dual antiplatelet therapy (DAPT). In the single institution involved, both prior to and outside this study, patients undergoing flow diverter treatment are pre- and post-medicated with aspirin (ASA) and clopidogrel without response testing. It was agreed by the ethics committee that the investigators would perform response tests for all patients in the study without changing their medication regimens. It was also agreed that eptifibatide IV and IA would be used as a salvage strategy in the case of thrombus formation. The concept to perform response measurements for antiaggregant drugs without drawing conclusions from what has been measured is innovative since it simulates the widespread practice of non-testing, and addresses the associated risks. This ongoing study may eventually answer the question: can p64MW HPC be implanted under ASA and clopidogrel without response tests without an undue incidence of thromboembolic and hemorrhagic complications?
The immediate purpose of this study was to assess the safety and efficacy of p64MW HPC under DAPT irrespective of the level of platelet inhibition actually measured.
Materials and methods
This is an ongoing prospective registry. Eligible patients meet the following inclusion criteria:
at least one unruptured sidewall aneurysm in the anterior circulation, no implant (e.g., stent) in the target vessel segment, age ≥18 years and ≤80 years, not pregnant and in women of childbearing age, on oral contraception for two years following the procedure, no participation in another trial, no concomitant disease limiting the life expectancy to <2 years, no allergy to non-ionic contrast medium or to ASA and P2Y12 receptor antagonists, no other neurovascular disorder in the same vascular territory requiring treatment in the foreseeable future, ability and willingness to comply with the medication requirements within the study, ability to understand the goal and risks of this study.
A single deviation from the inclusion criteria prevented participation in this study.
Informed consent was obtained from all patients enrolled in written form.
So far, 29 patients (26 women, median age 57 years, range 40–77) with 46 unruptured sidewall aneurysms in the anterior circulation have been enrolled. Aneurysms have been located as follows: cavernous internal carotid artery (ICA; n = 2), paraophthalmic ICA (n = 13), superior hypophyseal artery (n = 16), paraclinoid ICA (n = 2), supraclinoid ICA (n = 6), posterior communicating artery (PcomA; n = 2), anterior choroidal artery, (AchoA; n = 2), anterior cerebral artery (ACA; n = 1), and middle cerebral artery (MCA; n = 2).
Patients received 1× 100 mg ASA (Aspirin cardio 100; Bayer Vital) and 1× 75 mg clopidogrel (Plavix; Sanofi-Aventis) orally (p.o.) daily for at least five days before the scheduled procedure. Platelet inhibition level was measured with VerifyNow (Accriva) for all patients, supplemented by Innovance PFA-200 (Siemens) tests for 13 patients. For the VerifyNow test, an aspirin reaction unit <550 was considered to indicate sufficient platelet inhibition. Based on the institutional and personal experience of the senior author the VerifyNow P2Y12 reaction unit (PRU) was interpreted as such: >240: non-responder, 100–239: possible responder, <100: certainly sufficient P2Y12 receptor platelet inhibition. For the Innovance PFA-200 test, the thresholds were set at >193 s for collagen/epinephrine (ASA) and >159 s for P2Y12 (clopidogrel). If at least one of the two tests showed significant platelet inhibition for the medication concerned, the patient was considered to be a “responder”. High platelet reactivity on ASA and/or clopidogrel was acknowledged but did not trigger any further action.
All procedures were carried out under general anesthesia via a right femoral artery access using a Philips Allura biplane digital subtraction angiography (DSA) system. Patients were heparinized with 5000 IU unfractionated heparin (IVCO Healthcare LLC, Mongolia) IV. Access to the cervical artery was achieved using an Envoy DA (Cordis), FargoMax (Balt) or 6F Neuron (Penumbra). Intermediate catheters were not required. A Rebar-18 microcatheter (Medtronic) was used with a pORTAL (phenox) microguidewire to deploy the flow diverters. A working projection was chosen on the basis of 3D DSA images. The dimensions of the aneurysms (neck width, fundus width, fundus depth) and the diameters of the target vessels proximal and distal to the aneurysms were measured on calibrated 2D and 3D DSA images. The diameter and length of the p64MW HPC was chosen by the operator to be as close as possible to the diameters of the landing zones, allowing for a minimum of 8 mm distal and proximal anchoring and avoiding—as far as possible—diameter under- or oversizing.
The flow diverters were deployed with moderate device compression, avoiding any distal migration, device invagination or stretching. The endovascular procedure ended with DSA runs in the working projection followed by whole-head runs.
Within 48 h after the intervention, MRI including fluid attenuated inversion recovery (FLAIR) and diffusion weighted imaging (DWI, b1000) was performed and ischemic lesions were registered in detail. Early MRI follow-up after flow diverter implantation is part of our clinical routine and a means to detect clinically silent DWI (i.e., ischemic) brain lesions. 5 Follow-up DSA was scheduled for 3 or 6 months, 12 and 24 months after the treatment, according to the availability of the patients.
The evaluation included safety endpoints based on clinical evaluation and MRI examinations (after the initiation of treatment: ischemic or hemorrhagic stroke, deterioration of the clinical condition by ≥1 mRS point, groin hematoma). Efficacy endpoints were based on follow-up DSA examinations and included the degree of aneurysm occlusion during follow-up examinations (complete occlusion, neck remnant, aneurysm remnant), occurrence of in-stent stenosis and implant occlusion.
Results
Within a dataset of 29 patients, a total of 46 unruptured aneurysms of the ICA and MCA were treated with a p64MW HPC. The medians of neck width and maximum fundus diameter were 3.3 mm (range: 1.5–12 mm) and 3.7 mm (range: 1.3–19.1 mm), respectively (Table 1).
Location and size of the aneurysms treated with p64MW HPC.
ACA: anterior cerebral artery; AchoA: anterior choroidal artery; ICA: internal carotid artery; MCA: middle cerebral artery.

In three of four patients with a two-digit PRU result, a PFA-100 test was available and confirmed the dual platelet function inhibition (DPFI). In 5 of 20 patients with a PRU between 100 and 239, the PFA-200 test demonstrated a P2Y12 receptor inhibition. In 2 of 20 patients, despite a PRU of 193 and 219, PFA-200 did not show a P2Y12 receptor inhibition.
A total of 36 p64MW HPC were implanted. No periprocedural thrombus formation was observed. MRI (DWI) within 48 h revealed some level of diffusion restriction in 3 of 29 patients (10%) and lesions with a diameter >5 mm in 1 of 29 patients (3%). A clinical deterioration at discharge was found in no patient. No permanent neurological morbidity or mortality was encountered (Table 2). The first follow-up DSA after 104 ± 23 days (median ± SD) for 42 of 46 (91%) aneurysms showed complete aneurysm occlusion, neck remnant or aneurysm remnant in 23 of 42 (55%), 2 of 42 (5%), and 17 of 42 (40%) aneurysms, respectively (Figure 1). The second follow-up DSA of 26 of 46 (57%) aneurysms after 182 ± 44 days (median ± SD) confirmed complete aneurysm occlusion, neck remnant or aneurysm remnant in 22 of 26 (85%), 0 (0%), and 4 of 26 (15%) aneurysms (Figure 2). In one patient, an immediate postprocedural device occlusion due to a collapsed proximal flow diverter was successfully treated with balloon angioplasty (Figure 3). Due to distal FD migration, three aneurysms (6%) had to be retreated (Figure 4). This was again done using a p64MW HPC without any issues. No hemodynamically significant in-stent stenosis or thrombosis was encountered. During a follow-up period of 205 ± 69 days, in 29 patients no secondary clinical deterioration or death occurred.
Clinical and radiographic outcome.
ACA: anterior cerebral artery; AchoA: anterior choroidal artery; ICA: internal carotid artery; MCA: middle cerebral artery; NA: not applicable; PcomA: posterior communicating artery.
aIntraprocedural complications—ICA paraophthalmic left/ICA superior hypophyseal left (1 patient): (1) FD occlusion (p64MW HPC 400/18) due to a collapsed proximal flow diverter was treated with a compliant balloon (Scepter, MicroVention) and 7.3 cm3 eptifibatide (1 cm3 IA, 6.3 cm3 IV) was given. A second p64MW HPC 400/12 was implanted in telescopic fashion. ICA paraophthalmic right: (1) bulging of the FD at the aneurysm neck. ICA superior hypophyseal left: (1) First FD (p64MW HPC 400/18) foreshortened distally and a second FD (p64MW HPC 400/12) was implanted in telescopic fashion. ICA PcomA right: (1) distal marker detached during FD deployment. No clinical sequel. ACA A2 right: (1) misplacement of the FD. Position corrected with an Acclino stent.
bPostprocedural complications—ICA paraophthalmic left: (1) due to distal FD (p64MW HPC 500/18) migration a second FD (p64MW HPC 500/18) was implanted in telescopic fashion. Retreatment (126 days after intervention) due to distal FD migration. ICA superior hypophyseal right: (1) due to distal FD (p64MW HPC 400/18) migration a second FD (p64MW HPC 450/21) was implanted in telescopic fashion. Retreatment (126 days after intervention) due to distal FD migration. (2) Due to distal FD (p64MW HPC 500/30) migration a second FD (p64MW HPC 500/30) was implanted in telescopic fashion. Retreatment (after 127 days) of right ICA due to distal FD (p64MW HPC 500/30) migration. Implantation of a second FD (p64MW HPC 500/30) in telescopic fashion.
cRROC: Raymond–Roy occlusion classification; RROC I: aneurysm occlusion; RROC II: neck remnant; RROC III: aneurysm remnant.
dGraduation for the DWI lesions: 0 = none; I = 1–5 DWI spots; II = >5 DWI spots; III = watershed infarct; IV = subterritorial infarct; IVa = non-eloquent brain region; IVb = eloquent brain region; V = territorial infarct, 1 supply territory; VI = territorial infarct, 2 supply territories; VII = hemispheric infarct.
eModified Rankin scale (mRS): 0: no symptoms/normal; 1: no significant disability despite symptoms; able to carry out all usual duties and activities; 2: slight disability; unable to carry out all previous activities, but able to look after own affairs without assistance; 3: moderate disability; requiring some help, but able to walk without assistance; 4: moderately severe disability; unable to walk without assistance and unable to attend to own bodily needs without assistance; 5: severe disability; bedridden, incontinent and requiring constant nursing care and attention; 6: dead.
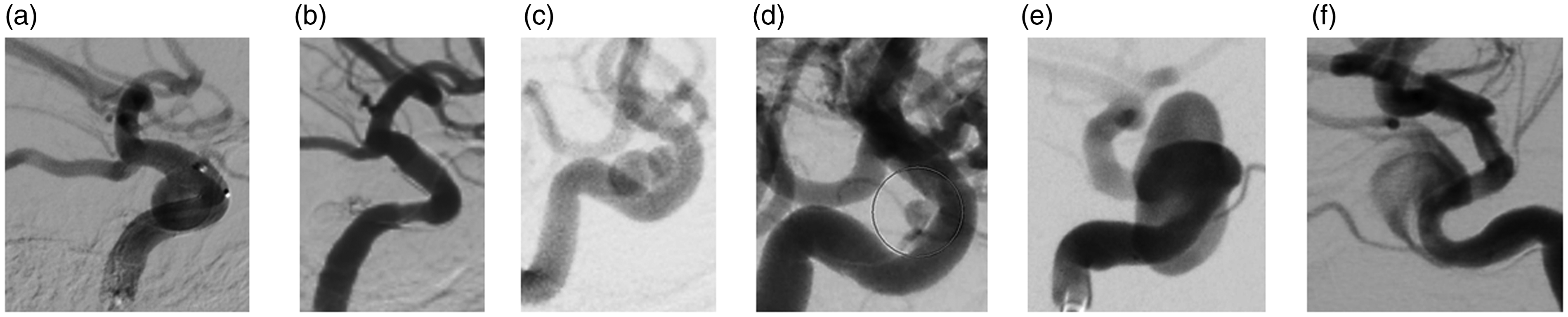
Examples for early follow-up DSA results three months after the flow diverter implantation with complete occlusion (a, b), neck remnant (c, d) and aneurysm remnant (e,f), as observed in 55%, 5% and 40% of the aneurysms, respectively.
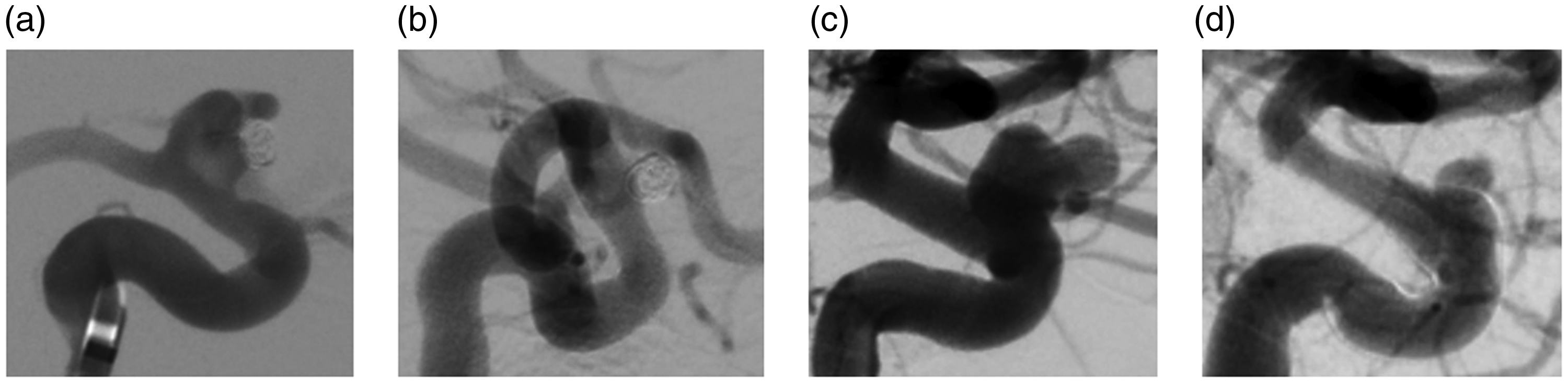
Intermediate follow-up with complete occlusion (a, b) after 180 days or aneurysm remnant (c, d) after 230 days, found in 67% and 33% aneurysms, respectively.
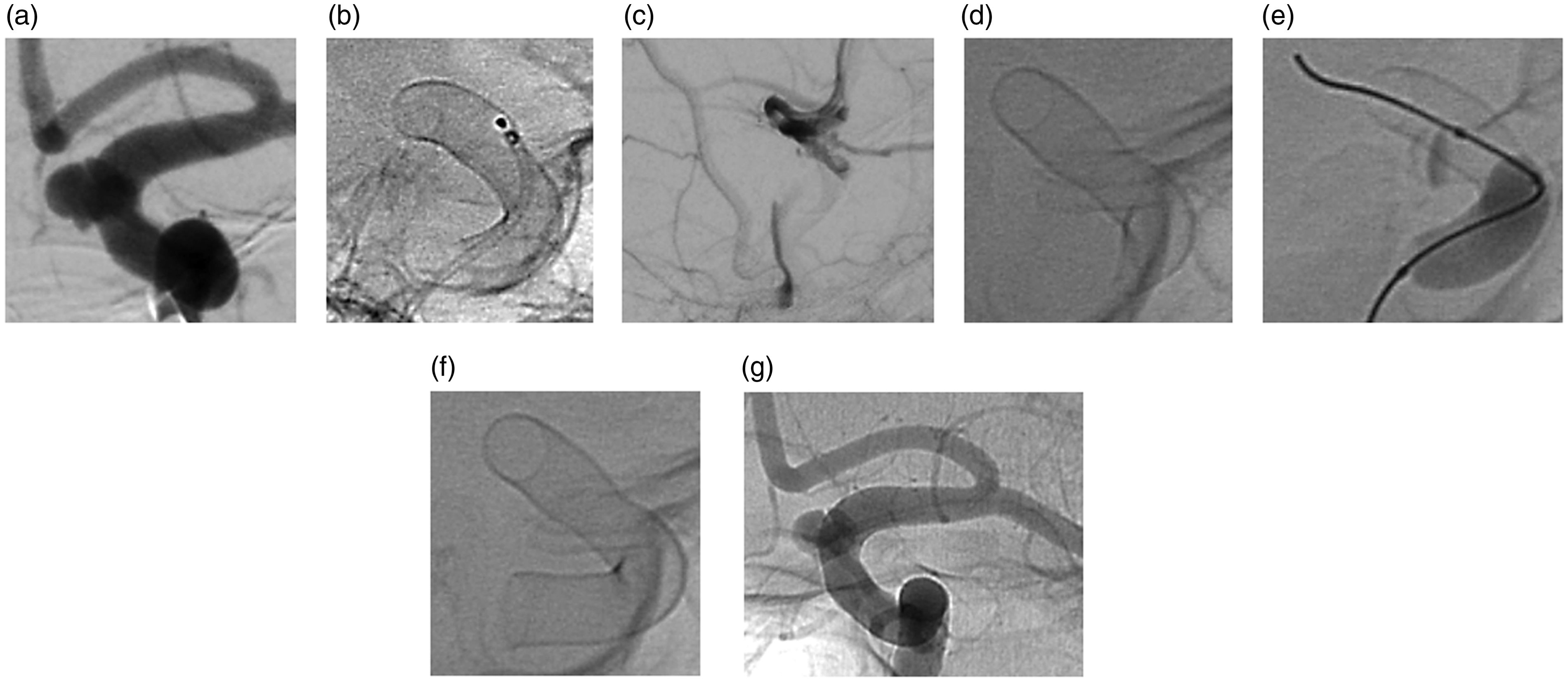
Proximal device collapse with parent artery occlusion. A small paraophthalmic and an opposite superior hypophyseal artery aneurysm were located on an ICA with a diameter of 4 mm (a). A p64MW HPC 400/18 was implanted (b). Twelve hours later the patient complained about a right-hand weakness and became aphasic. DSA showed an occlusion of the left ICA at the proximal end of the implanted FD (c). The FD was proximally collapsed (d). A compliant Scepter balloon (MicroVention) was gently inserted into the FD and inflated (e). Once the proximal end was opened a second p64MW HPC 400/12 was implanted proximally (f). The final DSA run demonstrated the complete reconstruction of the lumen of the left cavernous and intradural ICA. The patient recovered clinically within two days.
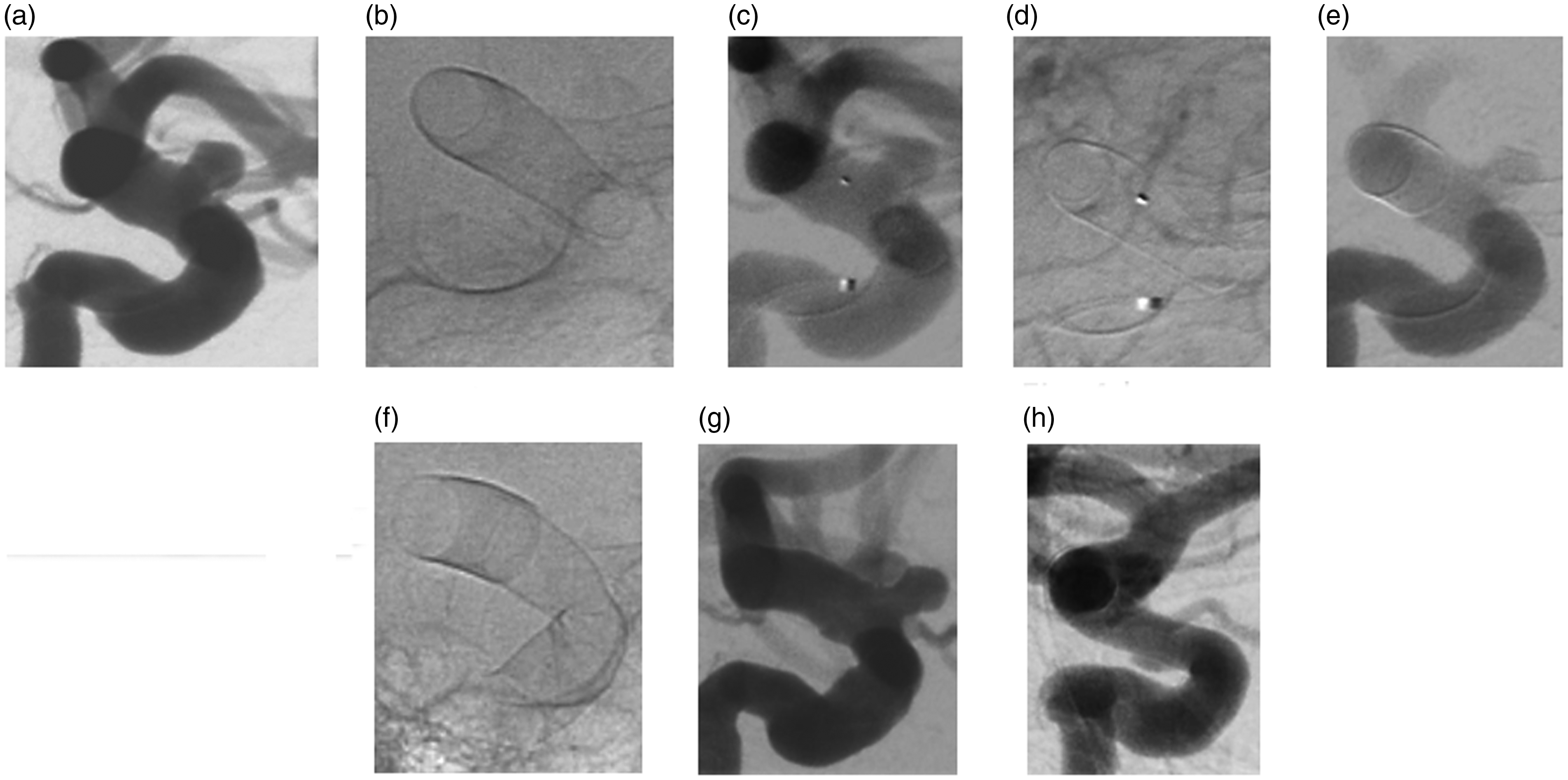
Distal migration of an oversized FD implanted into the supraclinoid segment of the left ICA. A left ICA paraophthalmic aneurysm (a) was covered with a p64MW HPC 500/18. The digital radiogram of the device showed that the concerning segment of the left ICA was distally wider than proximally (b). Consecutive images of the same DSA run confirmed the coverage of the aneurysm immediately after the FD deployment (c, d). Follow-up DSA 115 days later revealed that the proximal 2 of 3 of the FD had migrated distally, leaving the aneurysm uncovered (e). A second p64MW HPC 500/18 was implanted proximally in a telescoping fashion (f, g), now covering the paraophthalmic aneurysm. Follow-up DSA three months later showed the complete occlusion of said aneurysm (h).
Discussion
The data presented herein are the result of an interventional “teaching mission” to Mongolia with the aim of eventually enabling our colleagues to take over the neuroendovascular service for Ulaanbaatar and the entire country. 6 The hospital infrastructure (outpatients´ department, pre- and postprocedural monitoring, ICU) and the technical equipment (biplane DSA) are already up-to-date. The focus of our efforts is on detail teaching of the endovascular procedures and to establish adequate follow-up routines.
The incidence of stroke in the Mongolian population is high, with 87% ischemia, 13% hemorrhagic stroke and almost 5% of subarachnoid hemorrhage (SAH). 7 The mortality rate following ICU admission in Mongolia is about 25%, with the majority of deaths related to stroke, liver failure, or brain trauma. 8
In this scenario, modern methods for the treatment of intracranial aneurysms, ischemic stroke, and less frequent neurovascular disorders are required. Flow diverter implantation, although expensive, has the advantage of being a relatively straightforward and safe endovascular technique, especially in the treatment of anterior circulation non-giant saccular aneurysms. While the implantation of flow diverters usually has a steep learning curve, 9 the logistical circumstances are more demanding than those for straight coiling or clipping. When employing conventional, uncoated and therefore highly thrombogenic flow diverters, dual inhibition of platelet function is imperative in order to avoid a thrombus being induced by the implant. ASA combined with a P2Y12 receptor antagonist (e.g., clopidogrel) is nowadays considered standard of care. 10 For all potential drugs, however, cases of patient non-responsiveness have been described, which is a potential reason for acute thrombotic flow diverter occlusion.11–13 This issue of effective DAPT is even more relevant for Mongolian patients. A loss-of-function polymorphism of the CYP2C19, the enzyme responsible for the bioactivation of clopidogrel, has been described in Mongolians. 14 Drugs produced in Mongolia are not always meeting high-quality standards. 15 These circumstances make using flow diverters with reduced thrombogenicity desirable.
p64MW HPC is a new self-expanding flow diverter, consisting of 64 drawn, filled tube wires braided together as one unit. At the nominal diameter, a coverage of 23–35% is achieved. The device is technically similar to the p48MW HPC flow diverter. 16 Anecdotal experience has shown that the p48MW HPC can be implanted under ASA or prasugrel single antiplatelet therapy (SAPT).17,18 The same can be expected for the 64-wire variant. The data presented here confirm that the p64MW HPC flow diverter can be safely implanted in patients taking ASA and clopidogrel as DAPT and with a wide variety of measurable response levels. We achieved adequate aneurysm occlusion after three and six months in 60% and 85%, respectively. These numbers are at least equal to those reported for other devices.19,20 The 0% morbidity and mortality in this series is explained by the fact that the inclusion criteria for this study did not allow to treat patients with aneurysms carrying increased risks (e.g., fusiform aneurysm, posterior circulation, recent rupture).21,22
The procedural issues encountered have been related to the deployment technique (device invagination) or due to inaccurate diameter measurements (distal device migration). Distal flow diverter migration is a rare phenomenon, which has been encountered by other authors as well.23,24 There are several possible mechanisms to be considered, which might cause flow diverter migration, including undersizing of the device relative to the diameter of the target vessel, significant diameter discrepancies of the target vessel diameters, poor wall apposition of the implant, and proximal stretching of the device during implantation together with distal device fixation. The p64MW HPC and quite similar also the p48MW (phenox) have a tendency to foreshorten more than other flow diverter stents. This has to be considered during the size selection of the implant. The three instances of distal device migration in this series are related to the “learning curve” of the operators, dealing with a new device.
None of those adverse events had permanent, clinically relevant sequel.
MRI (DWI and FLAIR) served as a surrogate marker for ischemic stroke. 25 The frequency and nature of the encountered lesions confirmed the good safety margins of the procedures performed and the devices implanted. A high rate of DWI lesions after flow diversion has been reported. 5 It is too early and the number of treated patients is too small to tell, if the use of p64MW HPC will prevent this phenomenon.
A mild in-stent stenosis has been observed on several follow-up DSA images. Currently there is no large-scale follow-up data available for p64MW HPC. The substrate of this stenosis is not well understood. It could be hyperplastic intima or layers of wall adherent thrombus. Experience with other flow diverter stents has shown that this is a frequent and usually benign phenomenon. In the far majority of patients, no hemodynamic effect is observed. If antiplatelet medication is continued these instent-stenoses resolve within 3–6 months. 26
The limitations of this study are the small number of patients and the still pending long-term follow-up DSA results. The results, both angiographic and clinical, were self-adjudicated, with no external independent monitoring.
Future efforts will include more patients under DAPT with ASA and clopidogrel. DAPT through ASA plus prasugrel or ticagrelor has been found suitable for flow diversion. 27 ASA adds little impact compared to prasugrel alone 28 and is subject to several drug interactions and situational effects such as fever, trauma, SAH and pregnancy, which may significantly reduce the efficacy of ASA at standard dosage. It therefore appears logical—and has been proposed during recent presentations—to use prasugrel as a SAPT when implanting surface-modified flow diverters. The instructions for using a p64MW HPC allow the implantation of this device under SAPT if justified by clinical circumstances. Time will tell if SAPT with prasugrel rather than DAPT with ASA and a P2Y12 antagonist has the potential to make flow diversion even safer, with less challenging logistics and lower costs due to removing some of the response testing, medication tailoring, etc. inherent in DAPT. 29 Safe and efficacious SAPT for flow diversion remains the goal and prasugrel could be the solution. Initial experience with p64MW HPC under prasugrel SAPT has been encouraging, also for ruptured aneurysm in the acute post-SAH phase.
Conclusion
In Mongolian patients receiving ASA and clopidogrel as a DAPT, VerifyNow and PFA-200 frequently showed high platelet activity on medication. The endovascular treatment of unruptured intracranial aneurysms with the new antithrombogenic p64MW HPC was nevertheless possible with no undue incidence of thromboembolic complications and with a high rate of aneurysm occlusion within six months.
Footnotes
Acknowledgements
Study Identifier (ClinicalTrials.gov); COated Mongolian Aneurysm Treatment Study #1; COMATS1; NCT04305626, STUD-192206. We used the STROBE cohort checklist when writing this report.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Andrey Petrov and Elina Henkes have proctoring and consulting agreements with phenox GmbH. Sara Jagusch is an employee of phenox GmbH. Hans Henkes is co-founder and shareholder of phenox and has a proctoring and consulting agreement.
Ethical approval
p64MW HPC obtained a CE mark. The Ethics Committee responsible approved this clinical study. All patients were informed about the circumstances of this clinical study. Treatment using a conventional, uncoated device was offered to all patients as an alternative. All patients enrolled agreed to their participation in this study in written form.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: phenox GmbH is the sponsor of this study.
