Abstract
Introduction
Wearable sensors are increasingly applied to rehabilitation for arm movement analysis. However, simple and clinically relevant applications are scarce.
Objectives
To investigate the feasibility of single smart watch-based parameters for functional assessment in upper limb rehabilitation for musculoskeletal injuries using a commercial smart watch.
Method
Ten patients with unilateral shoulder pain and range-of-motion limitations were enrolled. They wore Galaxy Watch® and performed three sets of upper extremity tasks consisting of gross activities-of-daily-living tasks, Wolf Motor Function Test (WMFT), and Upper Extremity Functional Index (UEFI), and the acceleration and angular velocities were acquired. The motion segment size (MSS), representing motion smoothness from a clinical perspective, and various sensor-based parameters were extracted. The correlation between the parameters and clinical outcome measures were analyzed. The percent relative range (PRR) of the significant parameters was also calculated.
Results
For overhead and behind body activity task set, mean MSS for elbow flexion/extension axis significantly correlated with WMFT score (R = 0.784, p = .012). For planar tasks, mean MSS for the forearm supination/pronation (R = 0.815, p = .007) and shoulder rotation (R = 0.870, p = .002) axes significantly correlated with WMFT score. For forearm and fine movement task set, mean MSS of the elbow flexion/extension angle showed significant correlation with WMFT (R = 0.880, p < .001) and UEFI (R = 0.718, p = .019). The total performance time (R = −0.741, p = .014) also showed significant correlation with WMFT score. The PRR for mean MSS in forearm supination (71.5%, planar tasks) and mean MSS in x-direction (49.8%, forearm and fine motor movements) were similar to the PRR of WMFT (58.5%), suggesting sufficient variation range across different degree of impairments.
Conclusion
The commercial smart watch-based parameters showed consistent potential for use in clinical functional assessments.
Introduction
Functional evaluation is a key step in the rehabilitation process of upper extremity impairments. Accurate and detailed functional evaluations are necessary to develop individualized rehabilitation plans, and serial functional evaluations can help assess patient progress. Therefore, objective and consistent functional evaluation tools are required. Many task-based functional evaluations tools are used clinically, most of which have been developed for patients with stroke, such as the Action Research Arm Test (ARAT) (Hsieh et al., 1998), Wolf Motor Function Test (WMFT) (Wolf et al., 2001), and modified Barthel Index (MBI) (Shah et al., 1989). Patients with industrial injuries predominantly experience multiple musculoskeletal injuries, including complex or comminuted fractures. These fractures commonly involve the joint space and soft tissue injuries, resulting in severe pain. Traditionally, bony union and improvements in the range-of-motion (ROM), and pain are indicators of recovery. However, despite structural recovery, affected patients have difficulty returning to work, but performing objective evaluations of their functional status is difficult.
During the last decade, significant breakthroughs have been made in utilizing the wearable technology in rehabilitation (Toh et al., 2023) and also measuring and evaluating limb function using sensor technology and machine learning approaches. Multiple inertial measurement unit (IMU) sensors on the arms, trunk, and legs have been frequently used to track movement and calculate joint ROM, frequently in conjunction with cameras, image sensors, and digital simulation methods (Bernaldo de Quiros et al., 2022; Held, Klaassen, et al., 2018). One of the most widely used tools is the ActiGraph sensor and software, which provides daily activity data. Researchers used actigraphy sensors on both wrists of patients with stroke to detect limb neglect by calculating the acceleration vectors (Toba et al., 2021). However, ActiGraph-based evaluations typically require data collection periods exceeding 24 hours. Accelerometers have also been used in longitudinal studies to detect stroke recovery over time (Vatinno et al., 2022). Mobile applications using smartphone- and smartwatch-derived data were recently developed to facilitate application-guided home exercises (Burns et al., 2020; Chen et al., 2020). In above mentioned analyses, machine learning methods has been applied to detect and classify the movements.
Regarding upper limb functional evaluations using sensors, previous studies predominantly focused on stroke. They usually estimated clinical functional evaluation scores, such as the Fugl-Meyer Assessment score (Fugl-Meyer et al., 1975) and WMFT, usually employing machine learning algorithms (Otten et al., 2015; Oubre et al., 2020). For non-central nervous system disorders, two sensors on the upper arm and wrist were used to identify upper extremity tasks in patients with adhesive capsulitis (Chang et al., 2020). Another study utilized two sensors on the chest and upper arm to measure the shoulder ROM and recommended home exercises using a mobile application (Chen et al., 2020). Although the research has provided meaningful quantitative data, its successful incorporation into routine clinical practice has been difficult for the following reasons: (1) the sensor settings and calibration are complex, time-consuming, and require large spaces; (2) the sensor type, sensor mounting location, and data acquisition and processing methods varied among laboratory settings; (3) the data provided by the sensors cannot be confidently used by clinicians, and (4) machine learning approaches cannot identify sufficient causality between parameters, precluding their clinical feasibility. Moreover, compared with imaging-based evaluations, IMU sensors have innate limitations, including drift, gimbal-lock phenomenon, calibration problems, and decreased accuracy (Hoglund et al., 2021; Mourcou et al., 2015).
Therefore, a simple, straightforward, and universal device for functional evaluations is highly required. A commercial smartwatch may be promising owing to improved adherence, convenience, and low cost. Our research question was whether a single wrist sensor-derived parameter could represent the upper limb functional status despite the inevitable measurement errors. We previously analyzed upper extremity motion using multiple IMU sensors during the functional assessment for hemiplegic stroke patients and proposed a novel sensor-based parameter, motion segment size (MSS), to represent smoothness from a clinical perspective, and the parameter was significantly correlated with the ARAT score (Nam et al., 2022).
This study aimed to investigate the clinical feasibility of MSS using a commercial smartwatch rather than multiple sensor systems or research-purpose sensors that require specific hardware and software for upper limb functional assessments in patients with musculoskeletal injuries. We also aimed to extend the target population to patients with various musculoskeletal injuries causing upper extremity functional impairments by using a different set of tasks primarily extracted from the WMFT and Upper Extremity Functional Index (UEFI) (Chesworth et al., 2014; Wolf et al., 2001).
Materials and methods
Participants
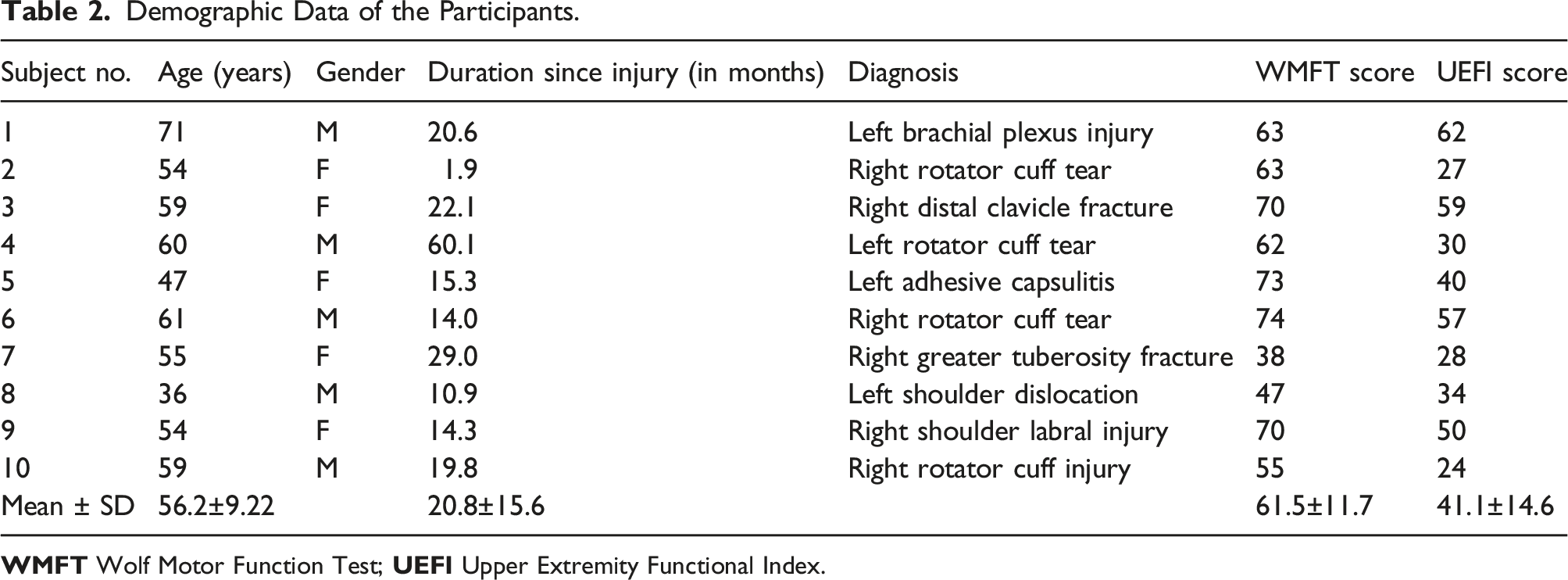
Ten patients with functional impairments of the unilateral proximal upper extremity due to traumatic injuries, such as fractures, tendon injuries, and nerve injuries, with shoulder pain and/or range-of-motion (ROM) limitations were enrolled in this study between October 2019 and December 2019, from the rehabilitation clinic in a workers’ compensation hospital which provides rehabilitation for patients with workplace injury. The patients were screened by a rehabilitation specialist physician and selected as per the inclusion and exclusion criteria. The inclusion criteria were as follows: (1) functional impairment of the unilateral upper extremity; and (2) age of 20–75 years. Each participant was considered as having upper limb functional impairment upon conveying difficulty in performing one or more daily or vocational activities that were regularly performed before the injury, as confirmed by the attending physician upon examination. The exclusion criteria were as follows: (1) motor power of grade 2 or lower in manual muscle testing of the affected arm; (2) cognitive impairment causing difficulty following the tasks or the inability to understand and provide written informed consent; (3) the inability to wear wearable devices due to trauma or surgery, and (4) ineligibility to participate in the study, as deemed by the investigator. The study protocol was approved by the Institutional Review Board of a tertiary hospital (1907-176-1050).
Performance tasks
Task Description for Each Task Set.

ADL activities of daily living; WMFT Wolf Motor Function Test; UEFI Upper Extremity Functional Index.
Measurements and data acquisition
For clinical functional level evaluations, the WMFT [0–75] and UEFI [0–80] scores were measured by an occupational therapist using standard methods (Chesworth et al., 2014; Wolf et al., 2001). For both evaluation scales, 0 represents the worst function and the highest point represents normal function. The participants were then equipped with two Galaxy Watch® devices (Samsung Electronics Co., Ltd., Suwon, Korea) on each wrist. Considering the findings of several step-count comparison studies, in which most IMU sensor data from smartwatches and bands provided similar ranges of data despite some variation (Fuller et al., 2020), we assumed that the raw IMU data from different smartwatch companies would not significantly differ. Among the popular commercial smartwatches, the Galaxy Watch® was selected because it provided direct technical support for IMU sensor data extraction.
The participants were instructed to perform the three task sets in separate recordings to minimize signal drift while ensuring practicality. Sensor calibration was performed before each task set. The participants were instructed to perform each set of tasks as continuously as possible from the first to the last task item, as directed by the therapist who conducted the study. The acceleration and angular velocities were acquired from the smartwatches via the Bluetooth®-linked data logger application at an average sampling rate of 25 Hz with the epoch time recorded in 0.04 sec interval, and processed with an algorithm developed using MATLAB® (Figure 1). Because of sampling through Bluetooth, the sampling rate was inconsistent; therefore, the data calculation was performed on the basis of the timestamp. The sampling rate of 25 Hz may be insufficient to characterize movements; however, one of the main study goals was to use a commercial smartwatch. In a physical activity analysis, it was demonstrated that 25 Hz and 100 Hz accelerometer data were highly correlated (Small et al., 2021). A person is wearing smart watches on both wrists, connected to the tablet installed with the data logger application, via Bluetooth connection. The orthogonal coordination and the gyro sensor axes with corresponding representative anatomical axes are indicated (up). The wearable data logger shows smart watch connection with the real-time acceleration displayed, controlled by a start/stop button (down). Data are stored in the tablet device as a spreadsheet format.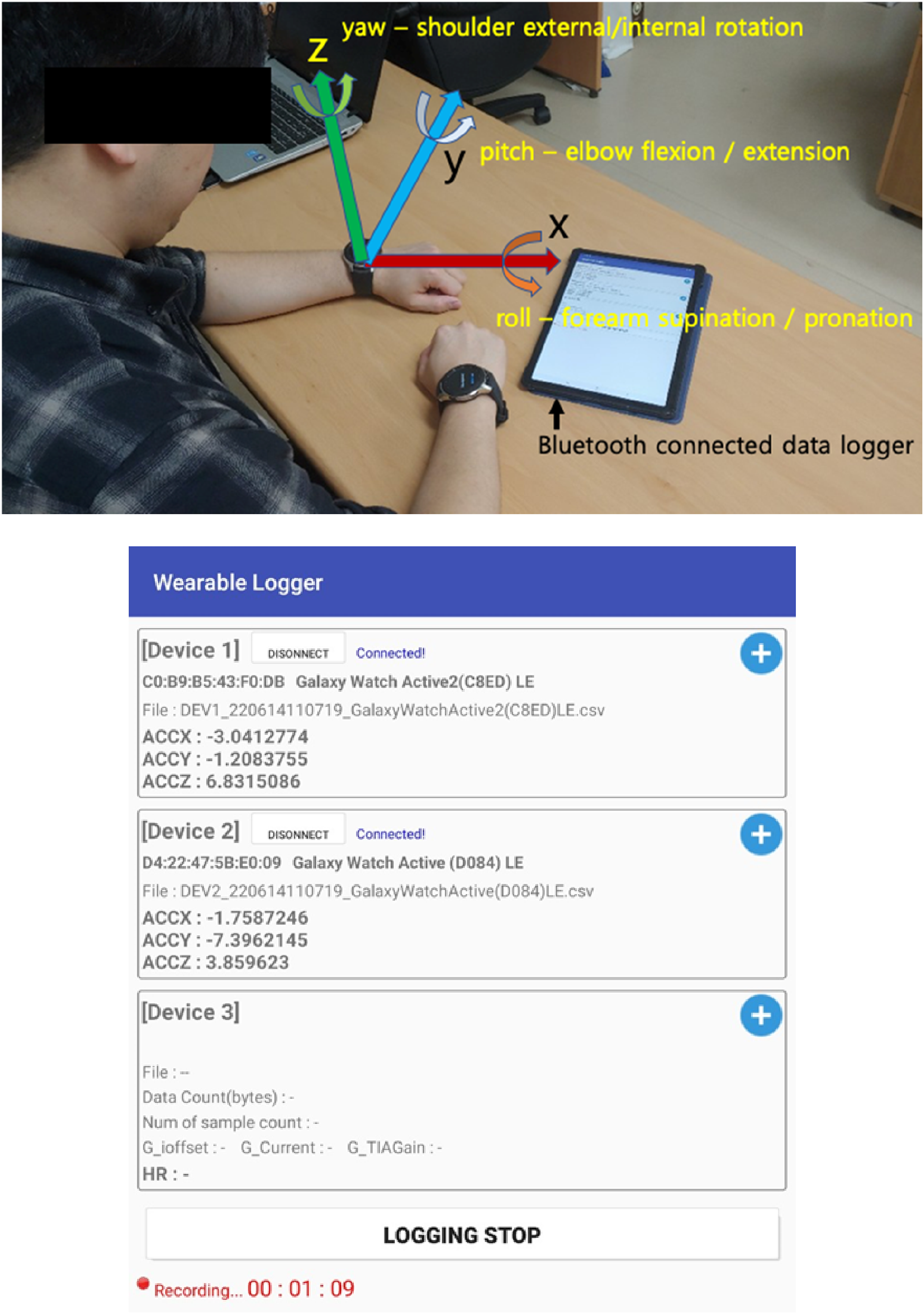
Sensor-based parameters
The orthogonal coordination system is defined as shown in Figure 1, with the x- and y-axes representing the planar direction and the z-axis representing the vertical direction. Based on the literature (Bertomeu-Motos et al., 2018; Cakmak et al., 2022; Shen, 2016) and considering the movement from the calibration position (sitting on a desk and chair with elbow flexed and forearm pronated), the gyro sensor data axes were defined in relation to the anatomical joint movements as follows (Figure 1): (1) yaw — primarily representing the shoulder internal/external rotation angle with partial shoulder adduction/abduction portion, (2) pitch — primarily the elbow flexion/extension and partially including the shoulder flexion/extension, and (3) roll — primarily the forearm supination/pronation axes. An integration matrix, followed by a rotation matrix, was applied to the angular velocity data acquired from the gyro sensor to estimate the roll, pitch, and yaw angles with respect to the initial position and direction. The algorithm was verified by comparing the angle data (roll and pitch) with the results derived from the angle estimation using the gravity vector. Data from both methods showed similarities.
Previously studied sensor-based parameters including the mean MSS for the acceleration (x-, y-, and z-axes; in m/s2) and joint motion angles (roll, pitch, and yaw; in degrees), maximum acceleration (m/s2), and total performance time (s) were extracted from the acquired data (Nam et al., 2022). MSS is defined as the distance (or displacement) of a parameter in the same direction of positional or angular movement changing the direction, as calculated using equation (1) (Nam et al., 2022):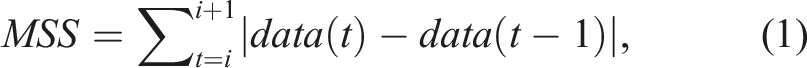
The mean MSS was calculated for all motion segments within the task set that exceeded 10° for angles and 0.05 m/s2 for accelerations.
Statistical analysis
For all sensor-based parameters extracted and calculated from the smartwatch on the affected side, the Pearson’s correlation analyses were performed between the parameters and the clinical outcome measures (WMFT and UEFI scores). All the statistical analyses were performed using the SPSS 28.0 software (IBM, Armonk, NY, USA). p values less than 0.05 were considered statistically significant. As our study aimed to extend the feasibility of the MSS established in the previous study and extracted only potentially relevant parameters are extracted, multiple comparison adjustment was unnecessary; however, the Bonferroni correction method was applied and analyzed, with a p value less than 0.01 statistically significant.
For the parameters showing a significant correlation with the clinical measures, the percentage relative range (PRR = (maximum – minimum)/average × 100%) was calculated to assess clinical feasibility regarding spread of the data across a range of severity. PRR is typically used to assess the measure of spread, and it should be minimized when measuring in the same state repeatedly, whereas when measuring different states, it should give a sufficient value so that it gives significantly different values when the measuring target is in a different state or level (Coelho & Lourenco, 2021; Yetkin & Alotaibi, 2023).
Results
Demographic data
Demographic Data of the Participants.
Sensor-derived parameters showing clinical correlation
Task Set 1 primarily included gross ADL tasks, including overhead and behind-body activities. While performing Task Set 1, the maximum MSS (R = 0.710, p = .032 vs WMFT; R = 0.729, p = .026 vs UEFI) and maximum acceleration (R = 0.689, p = .040 vs WMFT; R = 0.677, p = .045 vs UEFI) in the z-direction (up-down axis) were significantly correlated with the WMFT and UEFI scores, respectively. For gyro sensor-derived parameters, the mean MSS for the pitch (primarily representing elbow flexion/extension) axis showed a significant correlation with the WMFT score (R = 0.784, p = .012), whereas the roll (primarily representing forearm supination/pronation) and yaw (primarily representing shoulder rotation) axes were not significantly correlated. The total performance time was also statistically insignificant.
Task Set 2 consisted of 15 tasks extracted from the WMFT items, mostly consisting of planar movements. For these tasks, the mean MSS of acceleration in the x (R = 0.849, p = .004) and y (R = 0.753, p = .019) directions was significantly correlated with the WMFT scores. Among the gyro sensor-derived parameters, the mean MSS for the roll (forearm supination/pronation, R = 0.815, p = .007) and yaw (shoulder rotation, R = 0.870, p = .002) axes were significantly correlated with the WMFT score.
Smart Watch-Derived Parameters and Its Correlation With WMFT Scores.
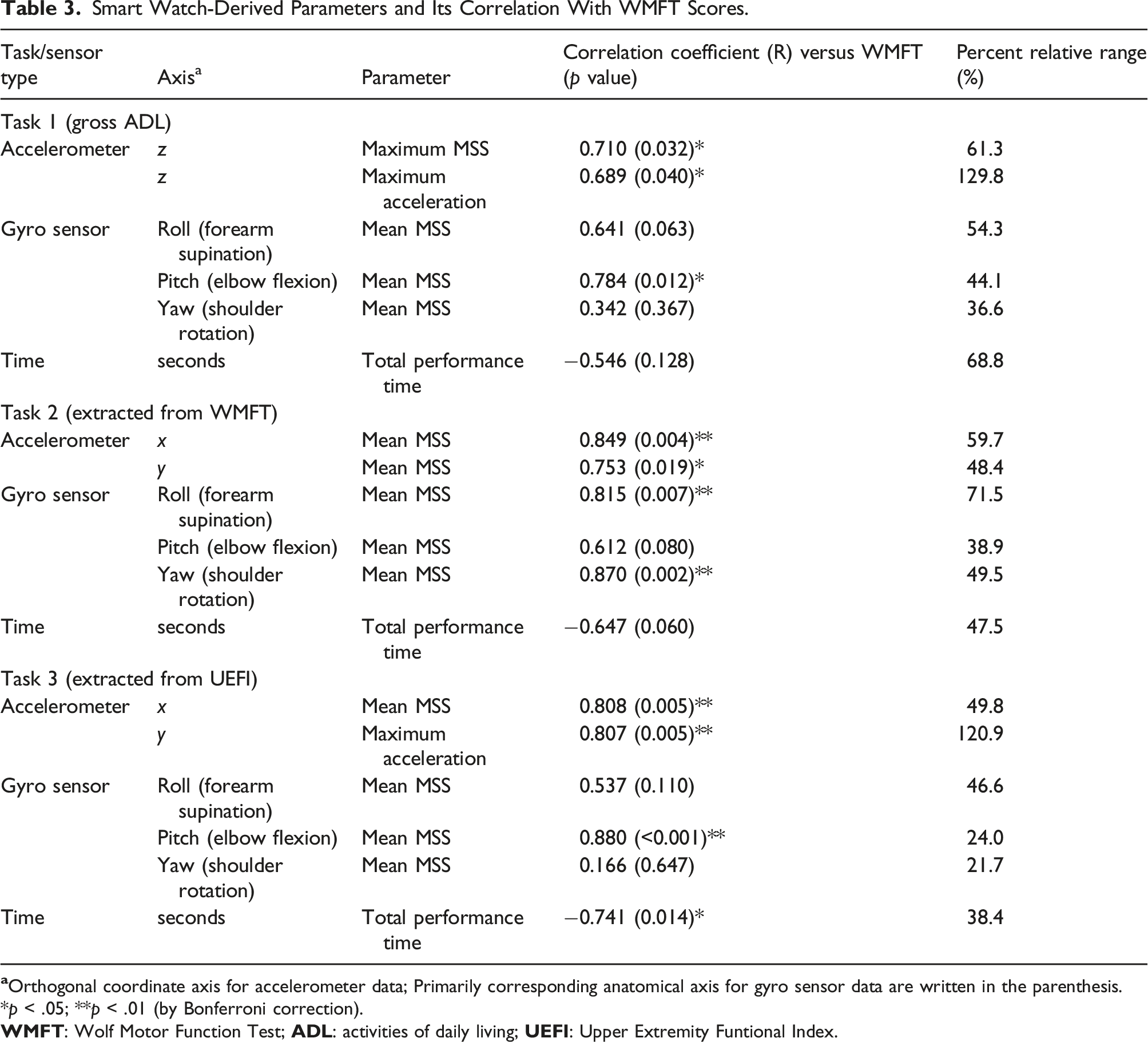
*p < .05; **p < .01 (by Bonferroni correction).
One of the subject’s watch data was partially not analyzable and calculable owing to severe signal noise, so the parameters using these data were not included in the correlation analysis.
Parameters showing potential clinical feasibility
The new sensor-based parameter must show a sufficient but not excessively wide range of distribution such that regular variations in measurements and calculations do not result in misrepresentations for practical use as a clinical parameter representing upper limb functions. The PRR values for the significant sensor-based parameters are listed in Table 3. We have compared the PRR values with that of the WMFT scores for the study participants as a reference value, which was 58.5%. The mean MSS of the parameters that showed high correlation with the WMFT and similar PRR to the reference value were the mean MSS for elbow flexion (44.1%) during Task 1, mean MSS for forearm supination (71.5%) during Task Set 2, and the mean MSS in the x-direction (49.8%) during Task Set 3. The total performance time for Task Set 3 had a PRR of 38.4%. The selected parameters showing sufficient PRR are shown in Figure 2. Selected sensor-derived parameters showing high correlation (R values indicated in the graph) with Wolf Motor Function Test (WMFT: total range 0–75) scores and with sufficient percent relative range (PRR) are shown (PRR values – A: 44.1%, B: 71.5%, C: 49.8%, D: 38.4%). 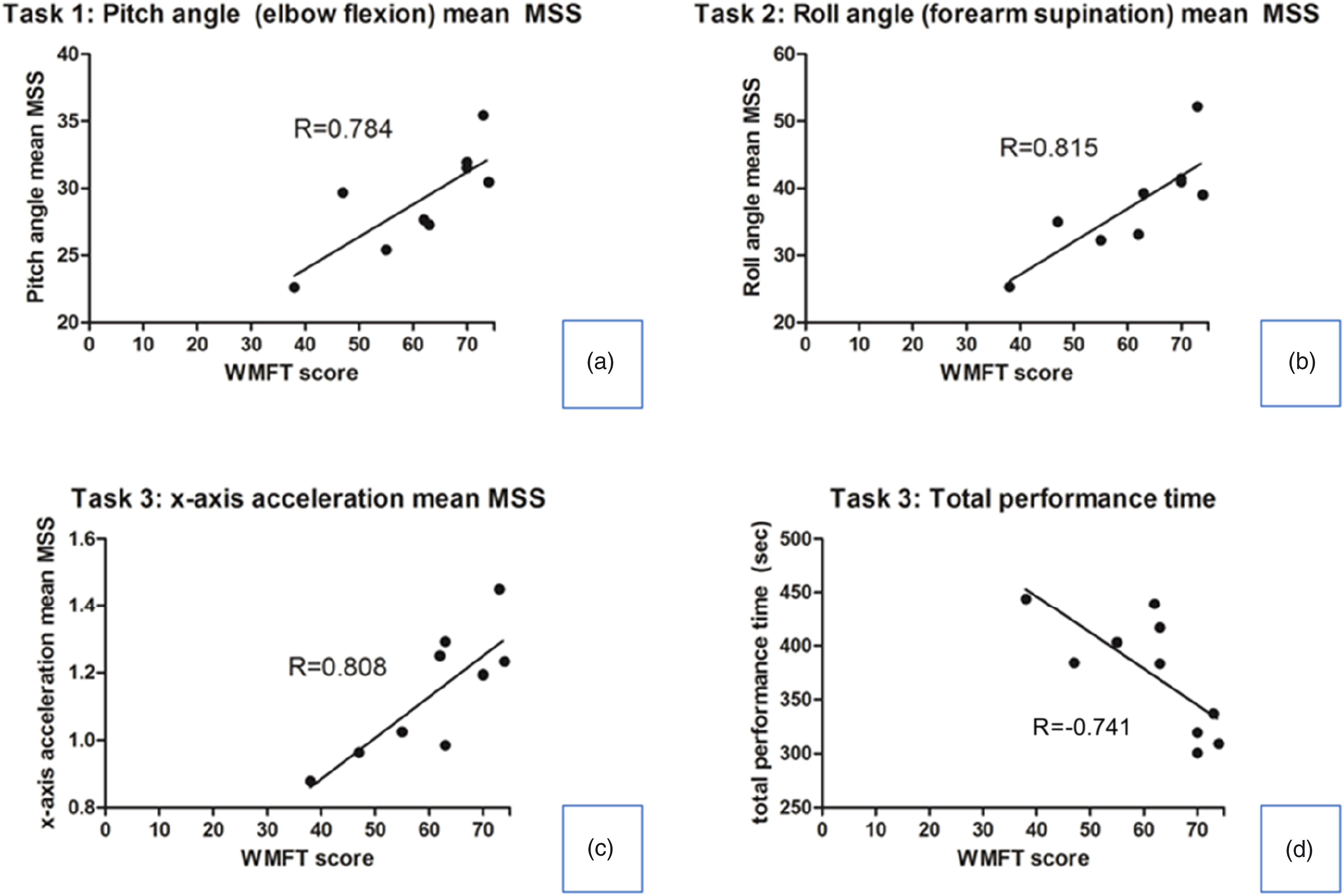
Discussion
The present study is an extension of our previous study that aimed to determine the reproducibility and expandability of its application in other disease categories involving upper extremity impairments. The major difference between the findings of the present and previous studies was the use of a single commercial wrist smartwatch instead of a multi-sensor IMU system, respectively, to acquire data from the upper extremity. Although the upper extremity movements are complex and certain gyro sensor axis movement does not correspond to a single anatomical joint movement, the most relevant anatomical axis corresponding to each sensor axis were designated for intuitive analysis of the data, and the results of the present study are consistent and promising for the generalization of the newly proposed parameter.
During gross movements, such as overhead and behind-body tasks, the parameters representing smoothness (represented by MSS in the present study) of the acceleration in the z-axis (up–down direction) and pitch angles (primarily representing elbow flexion/extension) were significantly correlated with clinical functional scores. This finding agrees with that of a study in which similar tasks, such as perineal care, hand-to-back-pocket, and putting the box off the shelf, involved the greatest ROM in humeral elevation (80–110°) and the elbow flexion axis (100–120°) (Gates et al., 2016). In a study which investigated the angles of elbow flexion/extension, shoulder flexion/extension, and forearm supination/pronation in grasping and pinching, these motion axes were significantly involved and changed in performing the motions (Nam et al., 2019a), which are also the motion axes in our study in which the MSS was significantly correlated with arm function.
Previously, motion smoothness was primarily assessed by calculating the jerk of the acceleration data (Carpinella et al., 2014; Rohrer et al., 2002). Recent studies using the jerk methods also suggested that the motion smoothness correlates well with the hemiplegia severity (Datta et al., 2021). Mounting evidence suggests that motion smoothness is more representative of the upper extremity function than the ROM capacity is. Similar concepts including spectral arc derived from the Fourier magnitude spectrum (Balasubramanian et al., 2012) and the number of movement units derived from the camera motion capture system (Alt Murphy et al., 2013), have already been proven to reflect smoothness with a clinical correlation. In the current study, MSS also showed a potential to represent smoothness although more research and evidence are necessary.
As per the definition of the MSS, when a person performs a movement in slow motion identically to that in normal-speed motion, the MSS would be the same. In reality, slower movements would not be performed smoothly owing to pauses or hesitation so it may result in lower average MSS value, however, this may act as a limitation of the MSS and require additional information or parameter to better represent and distinguish the upper extremity function. In the current study, the total performance time of forearm and fine motor-involving tasks (Task Set 3) showed a significant correlation with the WMFT score. Therefore, it can serve as supporting data to distinguish between fast and slow movements when the average MSS is similar but the speed is significantly different. It is also shown in literature that the people with musculoskeletal pain tend to perform tasks slowly (Seo et al., 2011).
For the clinically relevant and practical application of a specific parameter, the PRR across the patients’ functional levels is very important in addition to the correlation itself. To our knowledge, no well-defined consensus regarding the PRR currently exists in the clinical field. As the PRR of the WMFT in the study participants was 58.5%, the mean MSS for elbow flexion during Task 1, the mean MSS for forearm supination during Task Set 2 and the mean MSS in the x-direction during Task Set 3, which showed a range of 44.1–71.5% appear the most feasible to apply in the clinical setting. These parameters had significantly high correlation with the WMFT and also adequate range of PRR. However, the optimal PRR of a new parameter needs more investigation on a large number of subjects.
Therefore, the results of the current study suggest that the mean MSS in the orthogonal (for acceleration sensors) or anatomical (for gyro sensors) axis, which requires the most movement during the task, shows a high correlation with the performance or functional capacity of the patients in such tasks, with an adequate range of the values across the impairment severity. Furthermore, once the proposed evaluation method is well established, it would be most useful for frequent serial follow-ups of a patient’s rehabilitation progress to assess the improvement trend, rather than to accurately assess or estimate the clinical score. Other similar studies suggest a future direction for wearable technology in this field that utilizes daily feedback and tracking (Held, Luft, & Veerbeek, 2018; Seo et al., 2020).
Another substantial feature of the present study is that the concepts and parameters that were initially proposed and studied in the stroke population (Nam et al., 2022), were also applicable to various patient population with different medical etiologies, including musculoskeletal trauma and brachial plexus injury. The WMFT was originally developed for assessing impairments of neurological origin, mostly those involving the central nervous system (Wolf et al., 2001), whereas the UEFI was intended for assessing musculoskeletal origin impairments (Chesworth et al., 2014). The present study showed that the MSS significantly correlates with both functional evaluation methods, which supports the potential universal feasibility of the proposed sensor-based evaluation of any complex medical condition causing upper extremity impairments.
This study had some limitations. First, as this was a pilot study, the number of participants was small, limiting the significance of its findings. However, the results were consistent with the study that proposed the MSS concept (Nam et al., 2022) that used multiple sensors, providing a basis for further investigations of a larger number of participants to gather evidence and to ensure study reproducibility and universality. Second, the pain levels were not considered in the data collection and analyses. In a high proportion of the musculoskeletal origin impairments, the limb function is significantly affected by pain, along with ROM limitations. In this study, we assumed that the limb dysfunction was a result of various factors, including motor power weakness, structural injury, and pain. However, further investigations should be performed to identify the effects of each neuromuscular aspect and pain or sensory aspect to ensure its universal and precise application. Third, calculating well-established motion smoothness parameters, such as jerk, for the data in this study may have been more informative for comparisons with the MSS. Fourth, investigation between subsets of WMFT scores may have demonstrated more selective correlation with each task set. This shall be performed in further studies involving greater number of subjects. Finally, only one type of commercial smartwatch was used. Although we assumed that raw data from the IMU sensors in a smartwatch would be similar across different manufacturers, inter-device validation needs to be performed in the future step.
Fourth, investigation between subsets of WMFT scores may have demonstrated more selective correlation with each task set. This shall be performed in further studies involving greater number of subjects.
Conclusion
Accelerometer and gyro sensor data acquired and processed by a single commercial smartwatch demonstrated significant correlations with the clinical outcome measures for upper extremity impairments. Additionally, the mean MSS for the major axes and directions showed consistent potential for clinical applications in functional assessments. However, the reliability and consistency of the suggested parameters and their clinical feasibility require further investigation.
Footnotes
Acknowledgements
Galaxy Watch® hardware and the Wearable Logger software development were supported by Samsung Electronics Co., Ltd., Suwon, Korea.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the Korea Workers’ Compensation and Welfare Service.
