Abstract
“Lifestyle factors play a critical role in the development, possible progression and growing incidence of many cancers, especially endometrial and ovarian cancer.”
Introduction
Cancer remains the second leading cause of death worldwide with 20 million newly diagnosed cancer cases and 9.7 million deaths from cancer reported in 2022. The incidence of cancer continues to rise and is expected to reach 35 million cases by 2050. 1 This increase is largely driven by population growth and modifiable lifestyle factors such as diet, physical inactivity, smoking, and obesity. Specifically, obesity is a significant contributor to cancer-related mortality. Among women with a body mass index greater than 40, the risk of death from all cancers is 60% higher than that for women with normal BMI.
Obesity is a predominant risk factor in gynecologic cancers, especially in endometrial cancer, influencing both cancer development and prognosis. Despite these known associations, the role of lifestyle interventions, including dietary and physical activity modifications in improving outcomes for gynecologic cancer patients remains underexplored. This review synthesizes current knowledge on the impact of lifestyle interventions on ovarian and endometrial cancer outcomes. We explore mechanistic links, evaluate existing guidelines, and discuss evidence from other cancer types to offer insights into enhancing the care of women with gynecologic cancers through lifestyle intervention.
Mechanistic Links Between Lifestyle Factors and the Development of Gynecologic Cancers
Lifestyle factors influence cancer development and progression through complex biological mechanisms, including inflammation, angiogenesis, hormonal regulation, insulin resistance, and gut microbiome modulation. Understanding these pathways is critical to leveraging lifestyle interventions as an integral part of gynecologic cancer care.
Obesity, Inflammation and Immune Function
Obesity creates a pro-inflammatory state through the secretion of adipose tissue-derived cytokines, such as tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6). 2 These inflammatory mediators contribute to tumor progression by promoting cancer cell proliferation and survival. Chronic inflammation associated with obesity is a well-established driver of endometrial and ovarian cancer. 2 On the other hand, exercise can reduce inflammation and improve immune surveillance. 3 In a study that investigated the effects of exercise on inflammatory markers in patients with ovarian cancer, those who engaged in regular exercise exhibited significantly lower levels of pro-inflammatory cytokines, such as IL-6 and TNF-α. 4 Furthermore, a metanalysis found that cancer patients who adhered to a Mediterranean diet rich in fruits, vegetables, and healthy fats, had significantly lower levels of inflammatory markers IL-6 and TNF-α compared to a standard diet. 5 These factors may create a more favorable environment to inhibit tumor growth and potentially mitigate tumor progression.
A combined exercise and dietary program in breast cancer survivors showed that participants in the intervention group had a 20% increase in natural killer (NK) cell activity and a 15% increase in the CD8+ T cell population, compared to the control group. 3 In another study, participation in a structured exercise and nutrition program correlated with a higher rate of pathologic complete response during neoadjuvant chemotherapy for breast cancer. 6 As such, engaging in exercise has been shown to mitigate the inflammatory effects of obesity and enhance immune function.
Angiogenesis
Diets rich in high fat and sugar elevate the levels of vascular endothelial growth factor (VEGF) that fuels tumor growth. 7 Similarly, an increase adipose tissue leads to the secretion of adipokines and cytokines (e.g., leptin and interleukin-6), which further stimulate angiogenesis and create a tumor-promoting microenvironment.8-10 Excess adipose tissue also serves as a source of aromatase activity, leading to increased conversion of androgens to estrogens, which can promote the proliferation of estrogen-sensitive tissues and enhance angiogenic signaling pathways. 11 This increase in circulating estrogens, along with inflammatory cytokines, increases the susceptibility to gynecologic cancers such as endometrial and ovarian cancers.
On the other hand, a diet rich in fruits, vegetables, and whole grains, which are abundant in anti-inflammatory and anti-angiogenic compounds, can inhibit angiogenesis. For example, polyphenols found in green tea, resveratrol in red wine, and curcumin in turmeric have been identified as potent inhibitors of angiogenesis, exerting their effects by downregulating VEGF and other angiogenic factors.12-14 Regular physical activity can also have a profound impact on the regulation of angiogenic factors. Exercise has been shown to reduce the expression of VEGF and other pro-angiogenic markers in both adipose tissue and systemic circulation in animal models. 15 Therefore, angiogenesis is a key driver of tumor growth under the effects of obesity, while a balanced diet and regular physical activity can provide antiangiogenetic benefits.
Hormonal Regulation and Insulin Resistance
Obesity-induced hyperestrogenism is a key driver of carcinogenesis in both endometrial cancer and ovarian cancer. 16 Dietary fiber, particularly insoluble fiber, which is found in whole grains and vegetables, binds to estrogens in the intestinal lumen. 16 This binding prevents estrogen reabsorption in the enterohepatic circulation, promoting excretion via feces. 16 As a result, circulating estrogen levels decrease, which may reduce estrogen-driven proliferation of endometrial cancer. A meta-analysis by Bandera et al 17 (2008) 17 reported an inverse association between fiber consumption and endometrial cancer risk, suggesting that a high-fiber diet may mitigate the effects of elevated estrogen levels in obese individuals. Similar findings were noted in ovarian cancer patients suggesting the protective role of dietary fiber in regulating estrogen metabolism. 17
Hyperinsulinemia exacerbates ovarian and endometrial cancer cell growth by activating the PI3K/Akt signaling pathway and promoting systemic inflammation.18,19 Hyperinsulinemia also suppresses the production of sex hormone-binding globulin (SHBG), leading to increased levels of free estrogen, which stimulates endometrial cell proliferation. 20 Alternatively, lifestyle changes, such as rigorous exercise and dietary programs exercise, along with pharmacologic agents, such as metformin can reduce hyperinsulinemia and improve insulin sensitivity, disrupting tumor-promoting pathways. 20 In a breast cancer cohort, reducing insulin-like growth factor 1 (IGF-1) through lifestyle modifications such as engaging in structured aerobic exercise, adhering to a low-glycemic or Mediterranean diet, and incorporating metformin therapy enhanced tumor chemosensitivity, ultimately inhibiting cancer cell proliferation and metastasis. 21
The Gut Microbiome
The gut microbiome, the collection of the microorganisms residing in the intestines, has emerged as a critical factor influencing cancer development and progression. The gut microbiome interacts with the immune system and can influence systemic inflammation, which modulates cancer growth. Additionally, certain gut bacteria can modulate the metabolism of estrogens and other hormones, impacting hormone-driven cancers such as endometrial cancer. 22 A diet high in fiber and low in processed foods promotes a diverse and balanced microbiome, which can produce short-chain fatty acids (SCFAs) which have anti-inflammatory and anti-cancer properties.23,24 Conversely, a diet high in fat and sugar can disrupt the microbiome, leading to dysbiosis and the production of toxic metabolites that can promote inflammation and cancerous proliferation. 25 Modulating the microbiome through diet offers a promising avenue for reducing cancer progression.
Overview of Current Guidelines on Lifestyle Interventions in Patients with Cancer
Leading organizations, including the American Cancer Society (ACS) and the National Comprehensive Cancer Network (NCCN), have emphasized the importance of lifestyle interventions as part of comprehensive cancer care. Their recommendations center on maintaining a healthy weight, engaging in regular physical activity, and adhering to a balanced diet. 26 Lifestyle interventions that include diet and exercise during and after cancer treatment can improve quality of life, reduce chemotherapy toxicity, maintain healthy body weight and composition, and potentially enhance survival.26-28
Dietary Recommendations
Cancer-directed treatments can be associated with muscle wasting and immunosuppression. 29 Thus, adequate protein intake is recommended for maintaining muscle mass and strength during cancer treatment. 30 The ACS guidelines also advise limiting the intake of processed foods and red meats, refined grains, and sugars, which are linked to inflammation and adverse cancer treatment outcomes. 26 Omega-3 fatty acids, found in fatty fish and flaxseeds, provide anti-inflammatory effects, which may help mitigate cancer progression and improve treatment tolerance.30,31 However, the evidence on their benefits during chemotherapy is limited.
Exercise Recommendations
Exercise has been shown to alleviate treatment-related side effects such as fatigue, anxiety, and depression, thereby improving the overall quality of life for cancer patients.32,33 According to the ACS guidelines, moderate-intensity aerobic exercise, such as brisk walking, cycling, or swimming, for at least 150 minutes per week, is recommended for cancer patients. 34 Stretching exercises as well as resistance training exercises should be incorporated at least 2 days each week. Resistance training in the form of weightlifting or bodyweight exercises (e.g., squats, lunges, push-ups) can help preserve muscle mass and strength during treatment. 35
Weight Management Recommendations
Weight management is a cornerstone of survivorship and supportive care. Strategies to achieve and maintain healthy weight should be multicomponent, integrating dietary modification, structured physical activity, and behavioral counseling. Intensive behavioral programs, typically involving ≥12 sessions within the first year, achieve clinically meaningful weight loss of 5-10% within 6-12 months, with approximately 40% of participants in gynecologic oncology cohorts attaining ≥5% weight loss at 6 months.36,37 Core behavioral strategies, including self-monitoring, goal-setting, problem solving, and relapse-prevention follow-up, are critical for sustaining results. Weight loss typically plateaus after 6 months due to metabolic adaptation, so ongoing support and maintenance strategies are critical.
Benefits of Lifestyle Interventions in Gynecologic Cancer Care and Current Evidence
Lifestyle interventions provide multiple benefits across the cancer care continuum from prevention to treatment and survivorship, improving physical, psychological, and treatment-related outcome.
Cancer Prevention Benefits
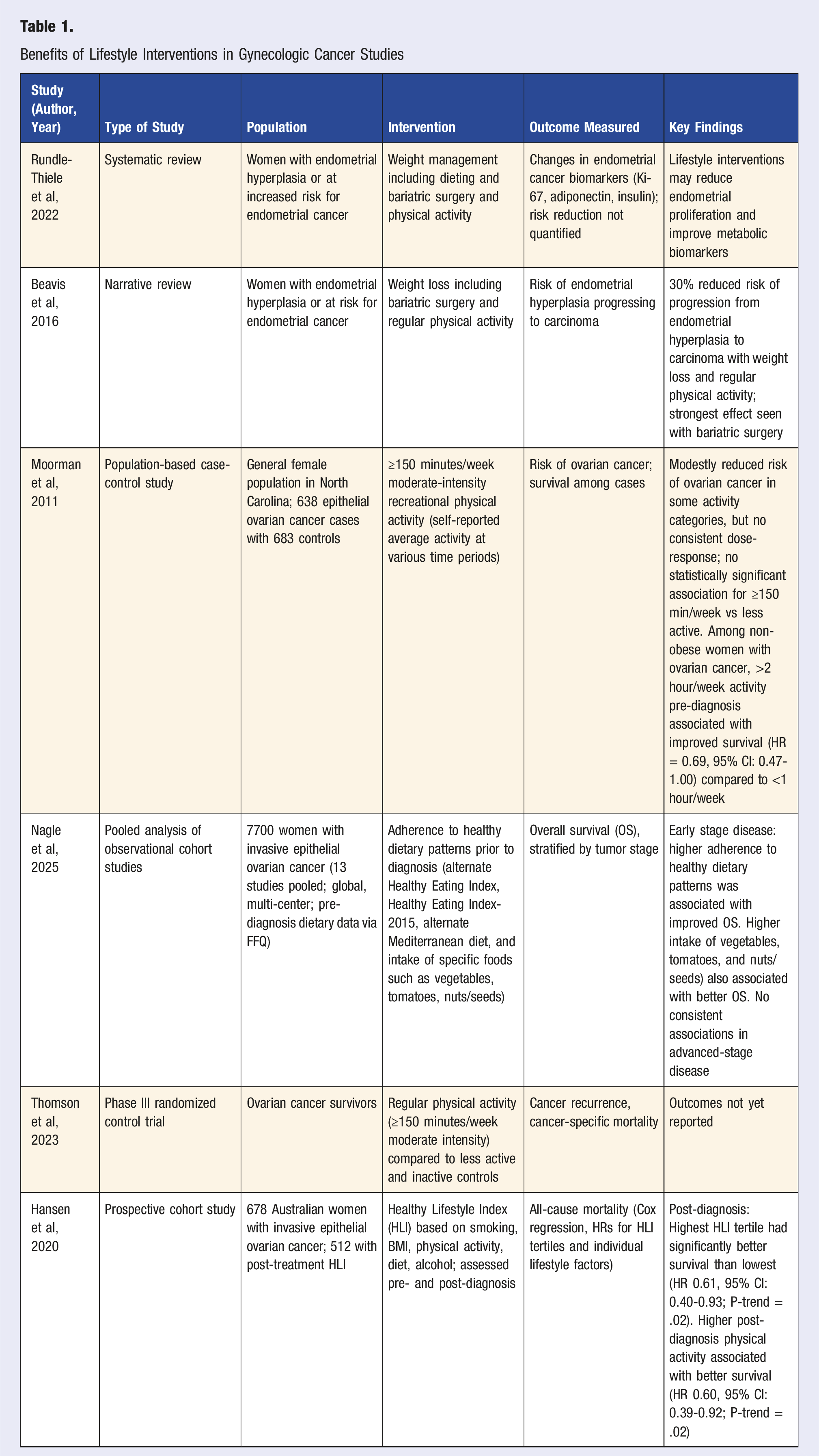
The risk of progression from endometrial hyperplasia to carcinoma is significantly influenced by various lifestyle factors. Interventions such as weight loss and regular physical activity, defined in studies by roughly 30 minutes of moderate-intensity cardiac and musculoskeletal activity 5 days a week, can play a crucial role in mitigating this risk by reducing estrogen levels, decreased chronic inflammation and improving insulin sensitivity. 38 A study by Rundle-Thiele et al, (2022) 39 investigating the impact of lifestyle interventions on the risk of progression from endometrial hyperplasia to carcinoma found that an intervention consisting of diet, physical activity and bariatric surgery was associated with favorable changes in endometrial cancer biomarkers, such as reduced endometrial proliferation (measured by Ki-67), increased adiponectin, and reduced circulating insulin levels. 39 A recent meta-analysis similarly concluded that weight loss and physical activity are effective strategies for reducing the risk of endometrial hyperplasia progression to carcinoma. 40 The authors note that physical activity and weight loss are associated with improved quality of life in endometrial and ovarian cancer survivors, and that small interventional trials show promise for increasing physical activity and weight maintenance. 40 Furthermore, in a population-based cohort study by Mooreman et al, regular physical activity was associated with a reduction in ovarian cancer risk particularly among women engaging in at least 150 minutes of moderate-intensity exercise per week. 41
Cancer Treatment Benefits
Chemotherapy-Related Adverse Events and Toxicities
Physical activity can alleviate chemotherapy-induced fatigue, nausea, and neuropathy.42,43 Mustian et al found that exercise interventions produced a mean weighted effect size (WES) of 0.30 (95% CI, 0.25-0.36) for reducing cancer-related fatigue, which is a moderate effect. Breast, GI, colon, and multiple myeloma cancer patients demonstrated chemotherapy-induced peripheral neuropathy (CIPN), and reported that a structured exercise program led to modest reductions in CIPN symptoms (e.g., a mean reduction of 0.42-0.46 points on a 0-10 scale for numbness/tingling and hot/coldness, respectively). 44 Hence, exercise and dietary interventions can help reduce the toxicity and side effects associated with chemotherapy.
Improvement in Nutrition
Nutritional support can prevent weight loss and malnutrition, which are common side effects of chemotherapy, thus improving patients’ ability to complete their treatment regimens. 45 The prevalence of malnutrition in cancer patients at the time of first medical oncology visit is 51%, with 9% overtly malnourished and 43% at risk for malnutrition. This study also found that 64% of patients had experienced weight loss (between 1-10 kg) in the previous 6 months and over 40% reported anorexia. The severity of malnutrition correlated with advanced cancer stage.45,46 Therefore, integration of dieticians within oncology teams to develop personalized nutrition plans that address the specific needs of cancer patients, is highly beneficial.
Psychological and Quality of Life Benefits
Regular physical activity provides ample mental health benefits. A review by Wanjau et al 42 (2023) found that individuals with high physical activity levels had a 17% lower risk of incident depression and a 26% lower odds of developing anxiety) compared to those with low activity. 42 Furthermore, a study published in Gynecologic Oncology found that patients undergoing treatment for endometrial cancer who participated in a structured exercise program experienced less fatigue and improved physical functioning compared to those who did not exercise. 43 Therefore, exercise plays a key role in alleviating anxiety and depression in patients undergoing cancer treatment.
Cancer Survivorship Benefits
Benefits of Lifestyle Interventions in Gynecologic Cancer Studies
Challenges and Future Directions: The Tenets and Feasibility of Lifestyle Interventions
While lifestyle interventions offer a broad spectrum of benefits in managing symptoms and improving outcomes in gynecologic cancer care, notable gaps in knowledge remain. 51 The mechanisms by which lifestyle factors influence specific tumor microenvironments and recurrence rates require further investigation.52,53 Addressing these gaps through robust clinical trials and longitudinal studies will help refine evidence-based guidelines and maximize the therapeutic potential of lifestyle interventions in gynecologic oncology.
Furthermore, clarifications regarding the optimal exercise and diet regimens, the frequency, intensity, and type of exercise for specific cancer types, including ovarian and endometrial cancers, are needed. 28 This lack of clarity hampers the ability to provide evidence-based, individualized recommendations for patients at this time. A critical barrier to advancing the field is the lack of large, well-controlled clinical trials investigating lifestyle interventions as adjunctive cancer therapies. Many studies to date have relied on observational data, which are inherently limited by confounding variables such as socioeconomic status and access to health care. 54 Thus, prospective randomized controlled trials are needed to determine causal relationships, establish evidence-based guidelines, and address variability in individual responses to diet and exercise interventions. 55
Lifestyle Intervention through the Lens of Behavioral Adherence Model
A significant barrier to implementing lifestyle interventions in cancer care is behavioral resistance, as modifying established habits and adopting new behaviors can be particularly challenging for patients facing life-altering diagnoses. The behavioral adherence models provide a framework for understanding and promoting lifestyle interventions among patients, particularly those dealing with chronic conditions like cancer (Figure 1). Key tenets of these models include self-efficacy, goal-setting, social support, and the Transtheoretical Model of behavior change.
56
Self-efficacy, the belief in one’s ability to execute specific behaviors, is crucial for initiating and maintaining lifestyle changes.57,58 Goal-setting helps patients create clear, attainable objectives, which can increase motivation and adherence. Social support from family, friends, and health care providers plays a significant role in encouraging and sustaining healthy behaviors.56,59 Behavioral adherence model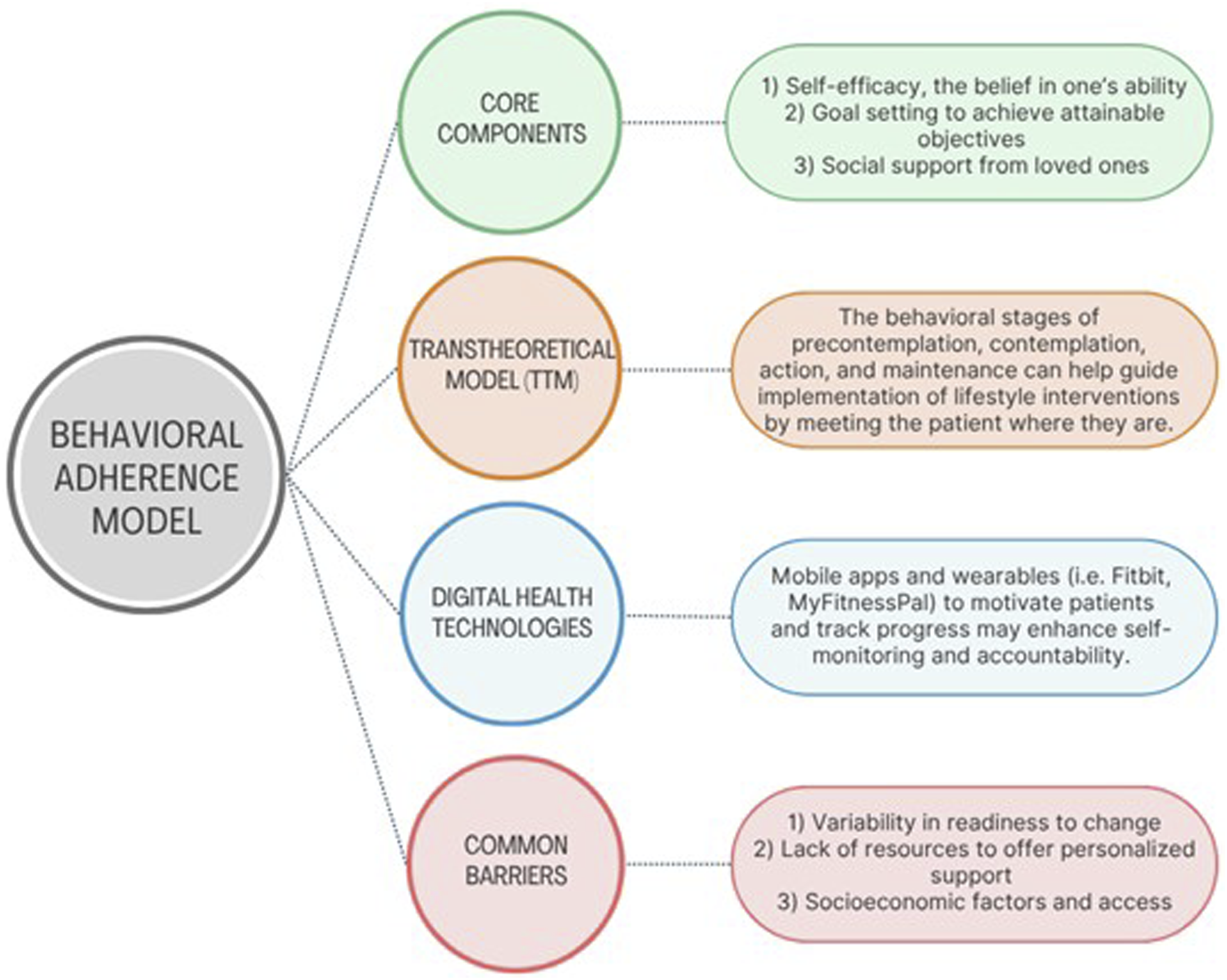
The Transtheoretical Model (TTM) which categorizes behavior change into stages—precontemplation, contemplation, preparation, action, and maintenance—can guide the implementation of lifestyle interventions. 56 By identifying the patient’s current stage, interventions can be tailored to meet their specific needs, enhancing their feasibility and effectiveness. For instance, patients in the contemplation stage may benefit from educational resources and motivational interviewing to increase their readiness for change, while those in the action stage may need more practical support, such as exercise plans or dietary counseling. 60
Example components supported by the literature include: (1) a 10-20 minute motivational interviewing visit focused on ambivalence and readiness; (2) weekly stage-matched modules or counseling sessions; (3) daily feedback from wearable devices to support self-monitoring; (4) SMS prompts for meal or activity logging; and (5) monthly relapse-prevention calls during maintenance. These strategies are consistent with the processes of change and have been shown to facilitate movement through the TTM stages, improve behavioral outcomes, and support maintenance of healthy behaviors in oncology and other clinical populations. 61
The American Society of Clinical Oncology and the American Cancer Society advise offering intensive, multicomponent behavioral programs, typically ≥12 sessions in the first year, targeting 5-10% weight loss over 6-12 months and maintenance thereafter.62-64
Comprehensive lifestyle modification interventions that combine dietary change, increased physical activity, and behavioral counseling have been studied are recommended for weight management in gynecologic oncology. Trials from von Gruenigen’s group established feasibility, adherence, and patient-centered benefits of multicomponent lifestyle programs in obese endometrial cancer survivors. In a randomized trial, participants in the intervention arm achieved sustained weight loss with improved diet and activity; quality-of-life gains were mediated by behavioral change constructs (self-efficacy), confirming a plausible mechanism of benefit. Subsequent reports from the same program (including SUCCEED) documented high adherence, durable behavior change, and clinically meaningful weight reduction over 6-12 months, supporting integration into routine follow-up.65,66 Complementing this, a recent real-world implementation by Morris and colleagues showed that a remotely delivered, group-based lifestyle modification intervention embedded in gynecologic oncology practice is feasible across urban/rural settings and yields statistically and clinically significant weight loss of 4.2 kg with 40% of patients attaining ≥5% weight loss at 6 months. 67
Despite these benefits, current evidence does not demonstrate improvements in overall survival or recurrence, largely due to limited sample sizes and short follow-up.68-70 However, interventions are safe, improve cardiometabolic risk factors, and address cardiovascular disease—a major contributor to mortality in this population. Effective programs typically include a 500-750 kcal/day energy deficit, individualized dietary patterns, ≥150 minutes/week of moderate aerobic activity, and behavioral strategies such as self-monitoring and goal-setting. Barriers include limited provider training and patient support needs, highlighting the importance of multidisciplinary care and tailored interventions.68,69
The Role of Digital Health Technologies
Studies have shown that the Transtheoretical Model is particularly effective in promoting dietary changes and physical activity among cancer survivors. 56 Roberts et al reported that multiple studies have found that the Transtheoretical Model-based interventions moderately improved exercise adherence and dietary habits among breast cancer patients. 70 These interventions, which included personalized feedback, stage-matched counseling, and follow-up support, helped patients progress through the stages of change, leading to sustained behavior modifications. 70
Multicomponent interventions, including self-monitoring, goal-setting, and social support, yield the best results for exercise and diet adherence, especially when combined with digital tools like mobile apps and wearables.71-73 A systematic review found that interventions incorporating self-monitoring, goal-setting, and social support were most effective in promoting long-term adherence to physical activity and dietary changes. 74 Programs that used technology, such as mobile apps and wearable devices for tracking progress, also showed promising results in enhancing self-monitoring and accountability. 75 Overall, behavioral adherence models that emphasize personalized, stage-specific strategies, and incorporate elements of self-efficacy, social support, and technology-enhanced self-monitoring appear to be most effective in promoting and maintaining healthy lifestyle changes among patients.71,72
Barriers to Implementation
The variability in individual motivation and readiness to change is a significant barrier in implementing lifestyle interventions. Patients at different stages of the TTM require tailored interventions, yet health care systems often lack the resources to provide such personalized support. 56 Additionally, socioeconomic factors, such as limited access to healthy foods and safe environments for physical activity, can hinder the adoption and maintenance of healthy behaviors.76,77 As a result, individuals from lower socioeconomic backgrounds may struggle to progress through the Transtheoretical Model stages, potentially leading to poorer health outcomes and increased cancer risk. Addressing these disparities by providing tailored interventions, reducing financial and structural barriers, and increasing accessibility to resources is essential for improving outcomes for patients with gynecologic cancers. 78 While technology-enhanced interventions show promise, they may not be accessible to all patients, particularly older adults or those from lower socioeconomic backgrounds. 77
Sustainability and Scalability
Promoting lifestyle interventions through behavioral adherence models necessitates personalized, scalable, and sustainable solutions. Ensuring continued engagement and adherence requires ongoing support and reinforcement, which can be resource intensive. 79 A meta-analysis by Stecher et al 80 (2023) demonstrated that mobile health interventions significantly improve physical activity and dietary behaviors, with adherence enhanced by features like reminders and gamification. 80
Digital health technologies offer significant potential in this regard. Mobile applications like MyFitnessPal and CancerFit facilitate dietary tracking and exercise regimens tailored to cancer patients, enhancing adherence and improving health outcomes. 81 Wearable devices, such as Fitbit, provide real-time activity feedback and have been shown to increase physical activity among cancer survivors. 82
Furthermore, telehealth platforms enable remote consultations for nutritional counseling and behavioral coaching, with systematic reviews indicating their effectiveness in improving dietary behaviors. 83 Integrating behavioral adherence models, such as the Transtheoretical Model, into these technologies can tailor interventions based on a patient’s readiness to change. Community-based digital programs like MyFoodDiary foster social support, which enhances adherence to lifestyle changes.84,85 Together, these innovative approaches empower cancer patients to maintain healthy behaviors and improve treatment outcomes through continuous, personalized support. While the integration of lifestyle interventions into routine cancer care is still evolving, there is a need for more standardized protocols and training for health care providers to effectively support behavior change.
Conclusions
Lifestyle factors play a critical role in the development, possible progression and growing incidence of many cancers, especially endometrial and ovarian cancer. The evidence shown in this review underscores the significant benefits of regular physical activity in reducing the risk of recurrence, improving quality of life and potentially improving survival rates. Despite this evidence, lifestyle interventions are often a neglected component in patient counseling and survivorship, and the implementation of these interventions is often nonuniform and limited in scope. Our findings highlight the need for increased investment in incorporating lifestyle interventions into routine gynecologic cancer care and investment in randomized controlled trials that investigate the cancer-specific benefits of these interventions.
Future directions in promoting lifestyle interventions through behavioral adherence models should focus on personalized, scalable, and sustainable solutions such as digital health technologies. Finally, greater value and time should be placed in creating preventative measures to improve outcomes and decrease risks allowing patients the best chance at a good quality of life. More in-depth research in endometrial cancer and other understudied gynecologic cancers should be emphasized to create more informed and personalized health and lifestyle improvements for patients.
Addressing modifiable lifestyle factors by integrating targeted exercise and dietary modifications into patient care, represents a pivotal frontier in enhancing outcomes for women battling gynecologic cancers. Empowering patients with tools to optimize their health and motivating compliance with a healthy lifestyle during and after treatment can potentially augment therapeutic outcomes and redefine standards of survivorship in gynecologic oncology.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
