Abstract
Keywords
“The DBCD model stresses the importance of early intervention at all chronic disease stages through structured lifestyle changes, with judicious pharmacotherapy/procedures at later stages.”
Introduction
The escalating prevalence of diabetes poses a substantial burden on individuals, healthcare systems, and global economies. The number of adults living with diabetes worldwide has surpassed 800 million in 2022, more than quadrupling since 1990. 1 This significant rise is largely attributed to factors such as increased obesity rates, unhealthy diets, lack of physical activity, and economic hardship. 1 Pharmacological interventions are crucial, but lifestyle modifications are paramount in mitigating chronic disease progression and associated complications. 2 However, implementing and maintaining effective lifestyle changes can be challenging for many individuals, theoretically driven by complex interactions among primary drivers of chronic disease (genetics, behavior, and environment), and pragmatically driven by the networking effects of technological, social, and other infrastructural factors.3-5
Glossary of Artificial Intelligence Tools. a
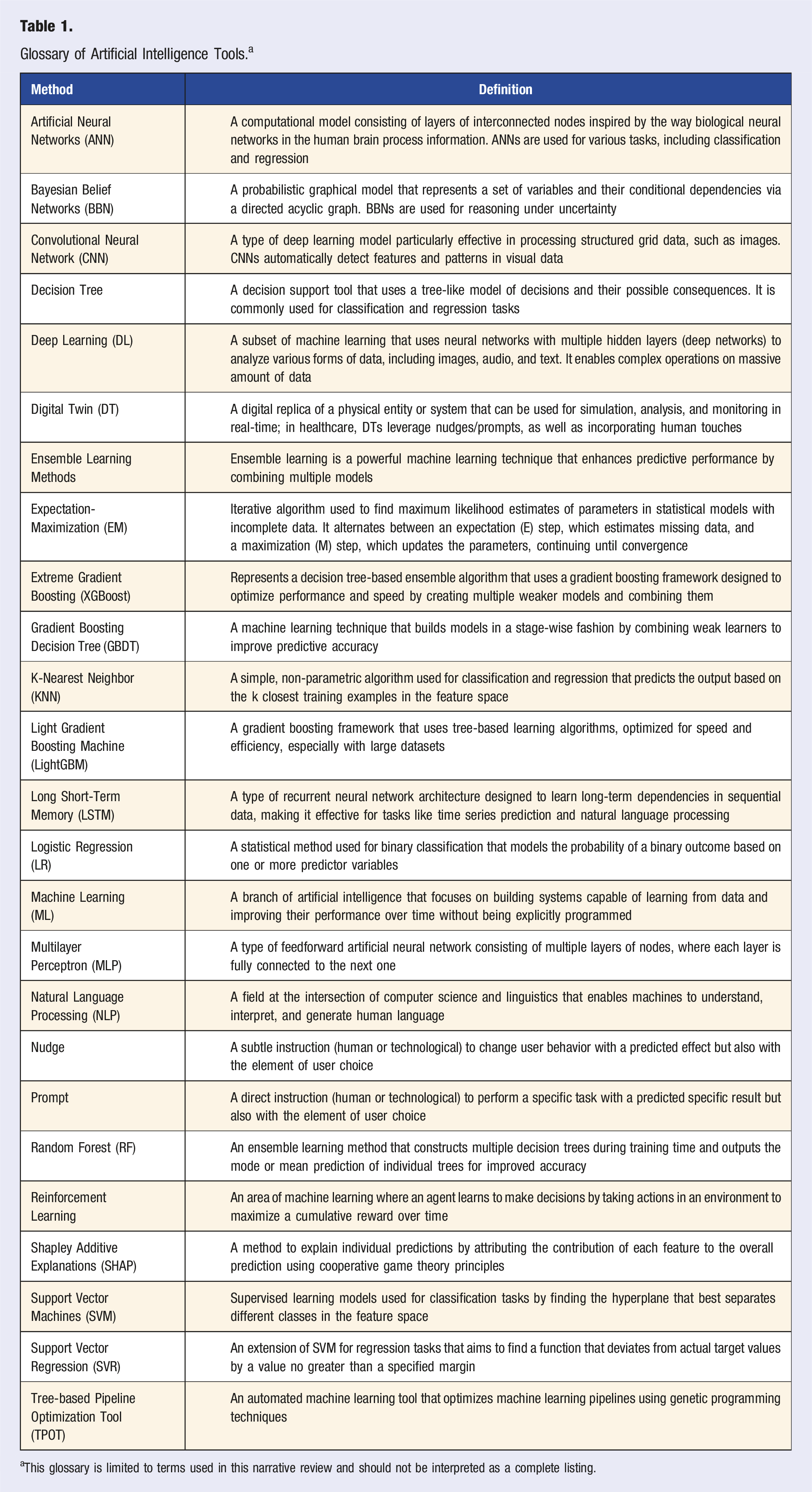
aThis glossary is limited to terms used in this narrative review and should not be interpreted as a complete listing.
The application of AI in diabetes care is particularly compelling due to the ability to address the multifaceted and dynamic aspects of chronic disease. For instance, AI can enhance diabetes care through continuous glucose monitoring (CGM) interpretation and insulin dosing (using insights from multivariate changes over time, such as variations in insulin sensitivity at different times of the day with different meals and varying glycemic indices/loads). 8 Comprehensive AI systems can integrate data on dietary instructions, physical activity, stress levels, and comorbidities to personalize diabetes management strategies. This approach is particularly relevant for type 2 diabetes (T2D) but can also apply to type 1 diabetes (T1D) and gestational diabetes mellitus (GDM). 9 However, despite the burgeoning research, a dedicated review that holistically synthesizes the role and evidence base of diverse AI technologies specifically in empowering lifestyle medicine for comprehensive diabetes management is currently lacking. This narrative review aims to identify and critically evaluate the principal applications of AI in supporting key aspects of lifestyle medicine for diabetes care. Specifically, it examines how AI is utilized in (1) predictive modeling for risk assessment, (2) personalized lifestyle guidance (nutrition and physical activity), (3) remote monitoring and patient self-management, and (4) clinical decision support relevant to lifestyle interventions. The review synthesizes existing scientific evidence on the characteristic features of these AI technologies and their reported effectiveness in improving diabetes-related health outcomes and enhancing patient self-management capabilities. We have organized this review by AI-enabled technologies/methods and their respective scientific evidence.
Methods
This study employs a narrative review methodology to provide a critically evaluative overview of the multifaceted applications of Artificial Intelligence (AI) in lifestyle medicine for diabetes care. This approach was chosen to facilitate an in-depth synthesis of key technological features, evidentiary findings, and overarching trends.
To identify relevant literature, a primary search was conducted in the PubMed database, selected for its comprehensive coverage of peer-reviewed biomedical and clinical research directly pertinent to healthcare applications. We acknowledge that this focus, while prioritizing studies with a certain level of scientific scrutiny, may not capture all engineering-specific publications or gray literature potentially found in specialized AI/computer science databases. The search strategy in PubMed combined MeSH (Medical Subject Headings) terms and free-text keywords using Boolean operators (AND, OR). Core search concepts encompassed AI-related terms (e.g., “Artificial Intelligence,” “Machine Learning,” “Deep Learning,” “Natural Language Processing,” “predictive model*,” “Algorithms,” “Clinical Decision Support”), diabetes-related terms (e.g., “Diabetes Mellitus,” “Type 1 Diabetes,” “Type 2 Diabetes,” “Gestational Diabetes,” “Prediabetes”), and lifestyle medicine-related terms (e.g., “Lifestyle,” “diet,” “nutrition,” “physical activity,” “behavior change,” “Self-Management,” “remote monitoring,” “personalized intervention*”). An exemplary PubMed search combined these categories, such as (AI terms) AND (Diabetes terms) AND (Lifestyle terms). Furthermore, the reference lists of retrieved articles, particularly influential reviews and key primary research studies, were manually scanned to identify additional relevant publications (a process often referred to as snowballing or citation searching) to complement the database search.
Studies were selected for inclusion if they directly investigated, applied, or discussed the use of AI technologies in supporting lifestyle medicine components (including risk prediction informing lifestyle modification, dietary management, physical activity promotion, behavioral support, remote monitoring for self-management, and clinical decision support systems focused on lifestyle aspects) for individuals with any type of diabetes (type 1, type 2, gestational) or prediabetes, and involved human participants. Articles had to be published in the English language. While no strict limitations were placed on study design to capture a broad spectrum of evidence, the primary search focus was on publications from January 2017 to May 2024 to reflect recent advancements, though seminal earlier works providing critical foundational context were also considered.
The initial search results were screened by title and abstract. Full texts of potentially eligible articles were then retrieved and assessed against the inclusion criteria. Given the narrative nature, the selection aimed to identify key themes and representative examples of AI applications. Data on AI technology, characteristic features, target population, key findings, and study limitations were qualitatively extracted and synthesized. The results are presented thematically, organized by the main AI-enabled application areas identified, and further categorized by their characteristic features and key evidentiary findings, as outlined in the review’s aim.
Results
Predictive AI Models
Characteristic Features
Studies Published on Predictive Models and Risk Assessment. a
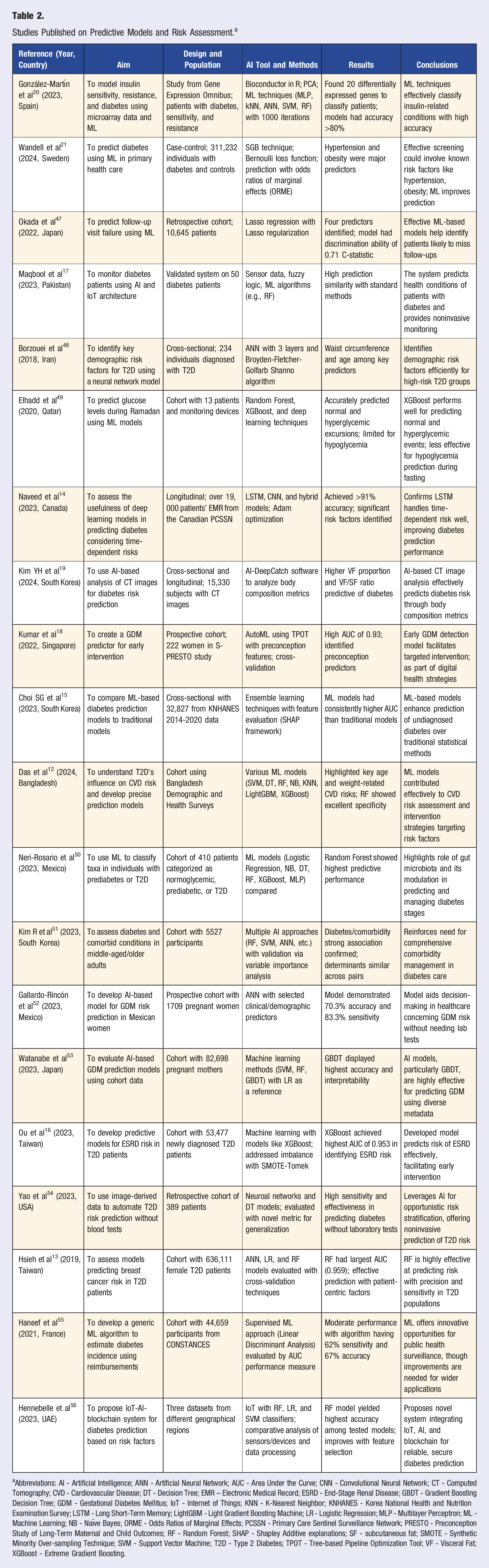
aAbbreviations: AI - Artificial Intelligence; ANN - Artificial Neural Network; AUC - Area Under the Curve; CNN - Convolutional Neural Network; CT - Computed Tomography; CVD - Cardiovascular Disease; DT - Decision Tree; EMR – Electronic Medical Record; ESRD - End-Stage Renal Disease; GBDT - Gradient Boosting Decision Tree; GDM - Gestational Diabetes Mellitus; IoT - Internet of Things; KNN - K-Nearest Neighbor; KNHANES - Korea National Health and Nutrition Examination Survey; LSTM - Long Short-Term Memory; LightGBM - Light Gradient Boosting Machine; LR - Logistic Regression; MLP - Multilayer Perceptron; ML - Machine Learning; NB - Naïve Bayes; ORME - Odds Ratios of Marginal Effects; PCSSN - Primary Care Sentinel Surveillance Network; PRESTO - Preconception Study of Long-Term Maternal and Child Outcomes; RF - Random Forest; SHAP - Shapley Additive explanations; SF - subcutaneous fat; SMOTE - Synthetic Minority Over-sampling Technique; SVM - Support Vector Machine; T2D - Type 2 Diabetes; TPOT - Tree-based Pipeline Optimization Tool; VF - Visceral Fat; XGBoost - Extreme Gradient Boosting.
Evidentiary studies spanned diverse geographical regions (Bangladesh, Canada, France, Iran, Japan, Mexico, Pakistan, Qatar, Singapore, South Korea, Spain, Sweden, Taiwan, United Arab Emirates, and the U.S.) from 2018 to 2024. These studies focused on developing predictive models for T2D, gestational diabetes mellitus (GDM), and associated complications such as cardiovascular disease (CVD) and end-stage renal disease (ESRD). The studies employed a variety of AI and ML techniques, including random forest (RF), support vector machines (SVM), artificial neural networks (ANN), long short-term memory (LSTM), and ensemble learning methods. The data sources included electronic medical records (EMR), gene expression data, demographic surveys, and sensor data from Internet of Things (IoT) devices.
There are four main categories of AI/ML techniques used in predictive models in diabetes care. Random forest is widely used for high accuracy and interpretability.12,13 Deep learning techniques such as bidirectional LSTM and convolutional neural network (CNN) were employed for time-dependent risk prediction. 14 Ensemble learning methods such as XGBoost and LightGBM were effective in improving prediction accuracy.15,16 Lastly, IoT Integration studies13,14 combined IoT with AI for noninvasive monitoring and prediction. 17
Key Findings
Many studies reported high accuracy (>80%) in predicting T2D and related conditions. For example, Naveed et al 14 achieved >91% accuracy using LSTM models, and Kumar et al 18 reported an area under the curve (AUC) of 0.93 for GDM prediction, demonstrated by a receiver-operating characteristic (ROC) curve indicating high sensitivity and specificity of the LSTM model for GDM prediction. The significance of such high predictive accuracy lies in its potential to identify individuals who would most benefit from intensive, personalized lifestyle modification programs. For instance, some studies leveraged noninvasive methods, such as computed tomography (CT) image analysis 19 and IoT-based monitoring, 17 to predict diabetes risk without blood tests.
The successful application of AI tools in predictive modeling and risk assessment holds significant implications for proactive lifestyle medicine. Predictive models enable early identification of high-risk individuals, facilitating timely interventions.18,20 This approach moves beyond reactive care, offering a pathway to integrate AI-driven risk stratification into primary care to promote preventative lifestyle changes, as suggested by Wandell et al21, who highlighted the role of known lifestyle-related risk factors (hypertension, obesity) in ML-enhanced screening. Ultimately, the “role” of predictive AI in this context is to make lifestyle medicine more targeted, timely, and impactful.
Personalized Lifestyle Guidance
Characteristic Features
Studies Published on Personalized Lifestyle Guidance. a
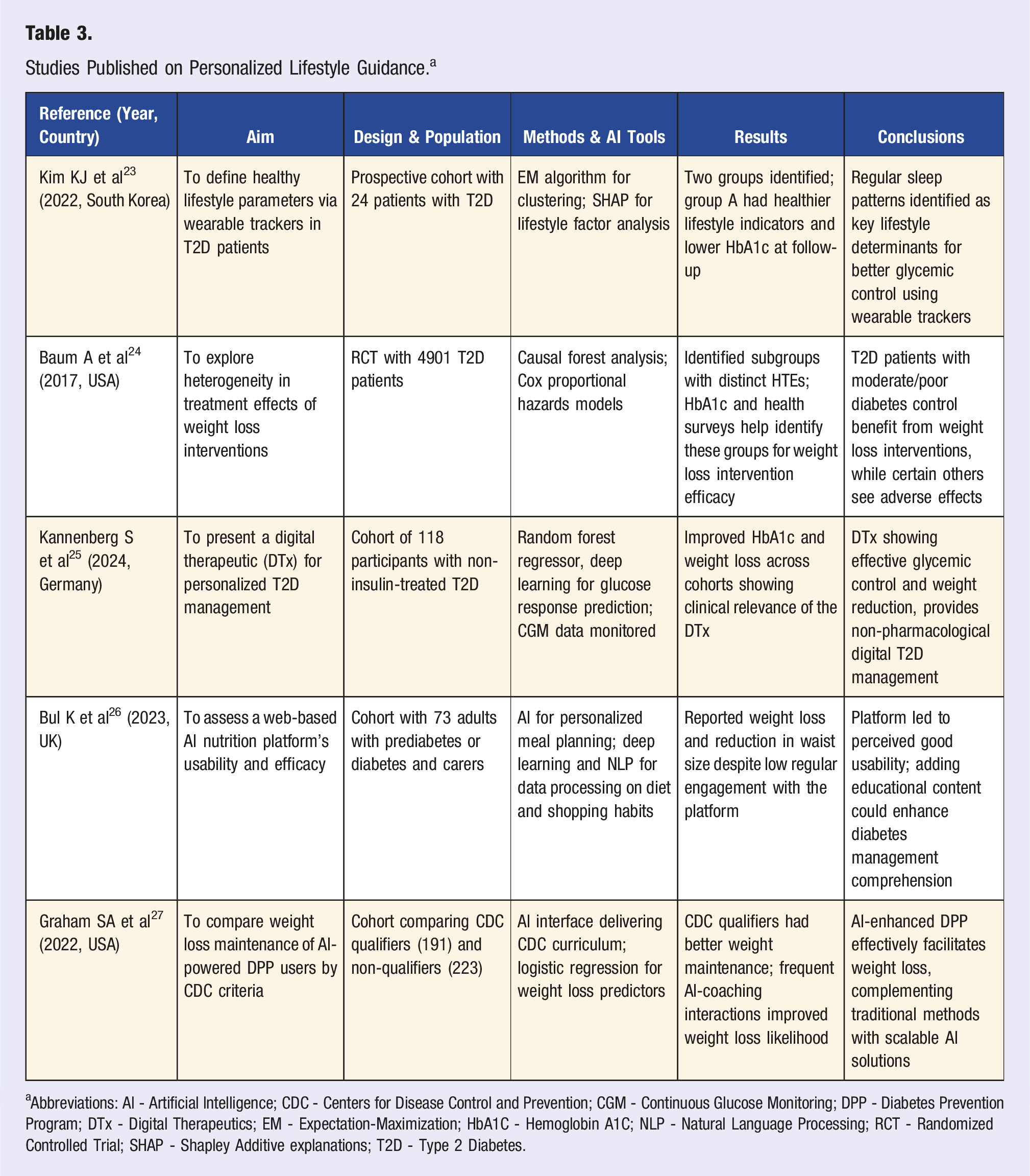
aAbbreviations: AI - Artificial Intelligence; CDC - Centers for Disease Control and Prevention; CGM - Continuous Glucose Monitoring; DPP - Diabetes Prevention Program; DTx - Digital Therapeutics; EM - Expectation-Maximization; HbA1C - Hemoglobin A1C; NLP - Natural Language Processing; RCT - Randomized Controlled Trial; SHAP - Shapley Additive explanations; T2D - Type 2 Diabetes.
Evidentiary studies spanned diverse geographical regions, including Germany, South Korea, the U.S., and the United Kingdom (U.K.) from 2017 to 2024. These studies focused on leveraging AI to provide personalized lifestyle interventions for individuals with T2D or prediabetes. The research designs included prospective cohorts, randomized controlled trials (RCTs), and digital therapeutics (DTx) evaluations, with sample sizes ranging from 24 to 4901 participants. The AI tools and methods employed in these studies include clustering algorithms, causal forest analysis, RF regressors, DL, and NLP. Data sources encompass wearable trackers, CGM data, dietary logs, and health surveys.
There are five main categories of AI/ML techniques used for lifestyle guidance. Clustering algorithms 23 use the expectation-maximization (EM) algorithm to cluster patients based on lifestyle factors, identifying key determinants for glycemic control. Causal forest analysis 24 explores heterogeneity in treatment effects of weight loss interventions, identifying subgroups that benefit most from specific interventions. Kannenberg et al 25 utilized RF regressors and DL models to predict glucose responses and evaluate the efficacy of a DTx for T2D management. In addition, Bul et al 26 applied NLP and DL to process dietary and shopping habit data, enabling personalized meal planning for patients with diabetes, including T1D and T2D. Lastly, AI-powered coaching 27 implements an AI interface to deliver the Centers for Disease Control and Prevention (CDC’s) Diabetes Prevention Program curriculum using logistic regression to identify predictors of weight loss maintenance.
Key Findings
Using AI-enabled wearable devices, Kim et al 23 found that regular sleep patterns were associated with improved glycemic control, while Baum et al 24 found that weight loss interventions were associated with improved glycemic control in those with moderate or poor control. Kannenberg et al 25 demonstrated improved HbA1c and weight loss through AI and CGM data. Bul et al 26 reported weight loss via an AI nutrition platform, even with low engagement, and Graham et al 27 showed that AI-powered coaching enhanced weight loss maintenance, particularly for CDC-qualified participants. These findings underscore AI’s potential for tailored, scalable lifestyle interventions in diabetes management.
The integration of AI into personalized lifestyle guidance has significant implications for diabetes care and prevention. For instance, AI-enabled technologies improve the delivery of personalized recommendations, adherence, and outcomes based on individual data.23,26 Also, DTx and AI-powered platforms offer scalable, cost-effective alternatives to traditional interventions, particularly in resource-limited settings.25,27 Wearable devices and AI-enabled coaching enhance patient engagement by providing real-time feedback and actionable insights.23,27 Identifying subgroups that benefit most from specific interventions allows healthcare systems to allocate resources more effectively. 24
Remote Monitoring and Self-Management
Characteristic Features
Studies Published on Remote Monitoring and Self-Management. a
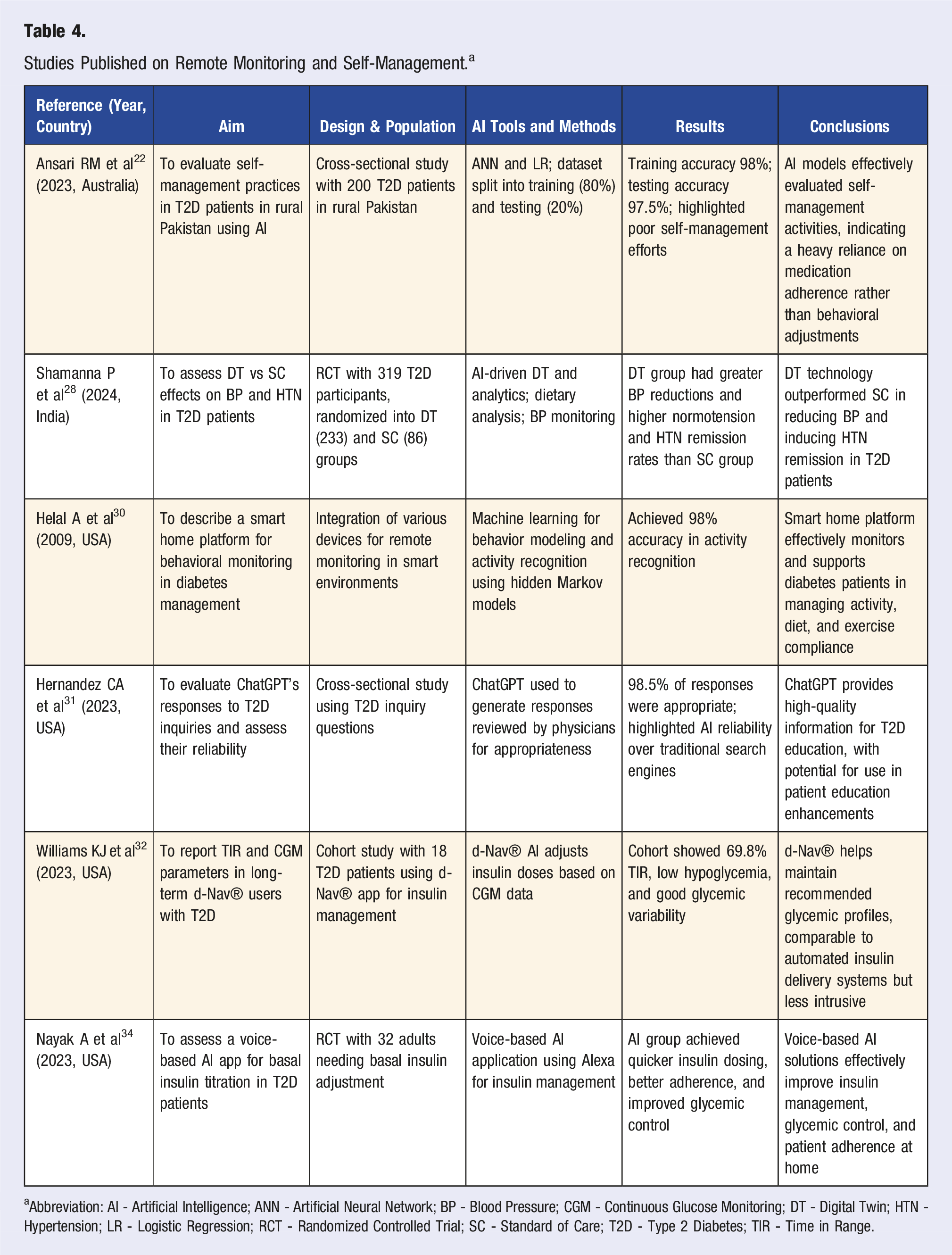
aAbbreviation: AI - Artificial Intelligence; ANN - Artificial Neural Network; BP - Blood Pressure; CGM - Continuous Glucose Monitoring; DT - Digital Twin; HTN - Hypertension; LR - Logistic Regression; RCT - Randomized Controlled Trial; SC - Standard of Care; T2D - Type 2 Diabetes; TIR - Time in Range.
Evidentiary studies spanned various geographical regions, including Australia, India, and the U.S from 2009 to 2024. These studies focused on using AI to improve self-management practices, blood pressure control, and patient education for T2D. The research designs included cross-sectional studies, RCTs, and cohort studies, with sample sizes ranging from 18 to 319 participants. The AI tools and methods employed in these studies included ANN, digital twin (DT) technology, ML for behavior modeling, NLP, and voice-based AI applications. Data sources encompassed CGM data, blood pressure monitoring, smart home devices, and patient inquiries.
There are six main categories of AI/ML techniques used for Remote Monitoring and Self-Management. Ansari et al 22 used ANN and logistic regression methods to evaluate self-management practices in rural Pakistan, achieving high accuracy in identifying poor self-management efforts. Shamanna et al 28 employed AI-driven Digital Twin (DT) technology and analytics to optimize blood pressure control and increase hypertension remission rates in patients with T2D, demonstrating its superiority over standard care. Additionally, Joshi et al 29 demonstrated superior efficacy of DT-enabled personalized nutrition compared with standard of care to improve markers of metabolic dysfunction-associated fatty liver disease. Behavior modeling can be achieved with ML and hidden Markov models to develop a smart home platform (98% accuracy in activity recognition). 30 Conversational AI and education tools, such as ChatGPT’s responses to T2D inquiries, highlighted the reliability and potential for patient education. 31 AI-driven insulin management was part of the d-Nav® app, which adjusts insulin doses based on CGM data, showing equivalency to automated delivery systems in maintaining glycemic control in patients with T2D, 32 similar to what was performed by Nimri et al, 33 in T1D management. Lastly, voice-based AI applications have been implemented using Alexa® for basal insulin titration in T2D. 34
Key Findings
AI-driven remote monitoring tools and self-management applications are highly effective in supporting diabetes management. For instance, DT technology 25 led to greater blood pressure reductions compared to standard care, indicating the value of advanced personalized analytics. 28 Other AI-based applications, such as d-Nav® and the voice-based assistant developed by Nayak et al 34 demonstrated significant improvements in insulin management and glycemic control, offering scalable solutions for home-based care. Furthermore, the Smart Home Platform reported by Helal et al 30 illustrated the practicality of continuous monitoring, and Hernandez et al 31 highlighted the educational potential of AI in healthcare communication.
The integration of AI into remote monitoring and self-management holds transformative potential for diabetes care. By delivering real-time feedback and personalized support, AI tools enhance patient adherence and clinical outcomes.22,34 Innovations such as DTx and smart home platforms present efficient and accessible solutions, offering a viable alternative to conventional care models. Other AI-powered platforms, such as ChatGPT, enhance patient education and engagement, providing reliable information for better decision-making. 31 In short, AI enables precise monitoring and intervention, leading to better resource allocation and health outcomes.28,32
Clinical Decision Support
Characteristic Features
Studies Published on Clinical Decision Support. a
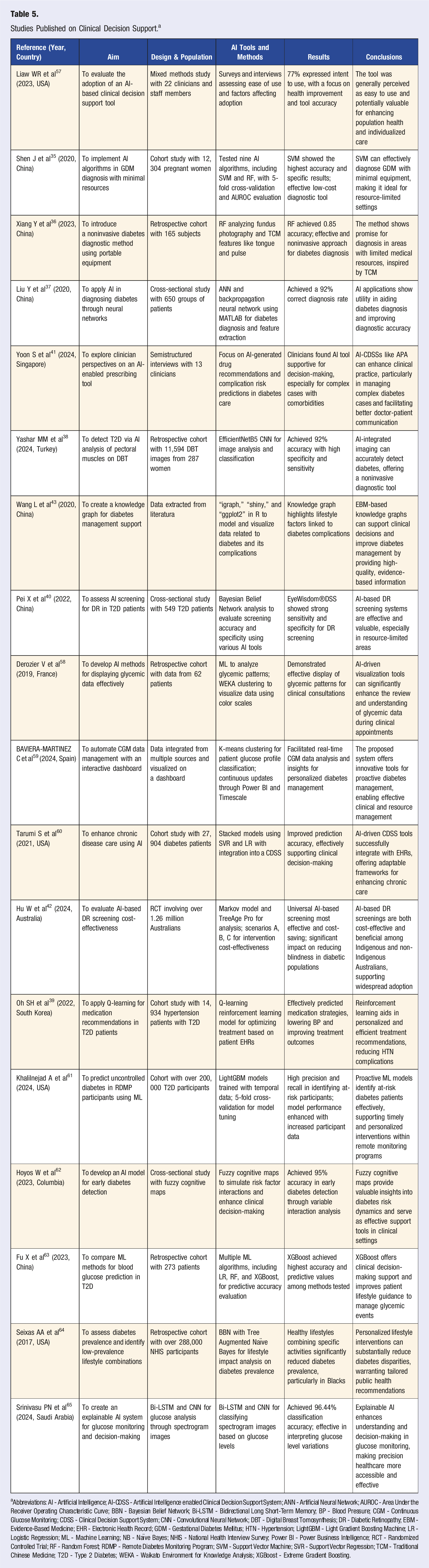
aAbbreviations: AI - Artificial Intelligence; AI-CDSS - Artificial Intelligence enabled Clinical Decision Support System; ANN - Artificial Neural Network; AUROC - Area Under the Receiver Operating Characteristic Curve; BBN - Bayesian Belief Network; Bi-LSTM - Bidirectional Long Short-Term Memory; BP - Blood Pressure; CGM - Continuous Glucose Monitoring; CDSS - Clinical Decision Support System; CNN - Convolutional Neural Network; DBT - Digital Breast Tomosynthesis; DR - Diabetic Retinopathy; EBM - Evidence-Based Medicine; EHR - Electronic Health Record; GDM - Gestational Diabetes Mellitus; HTN - Hypertension; LightGBM - Light Gradient Boosting Machine; LR - Logistic Regression; ML - Machine Learning; NB - Naïve Bayes; NHIS - National Health Interview Survey; Power BI - Power Business Intelligence; RCT - Randomized Controlled Trial; RF - Random Forest; RDMP - Remote Diabetes Monitoring Program; SVM - Support Vector Machine; SVR - Support Vector Regression; TCM - Traditional Chinese Medicine; T2D - Type 2 Diabetes; WEKA - Waikato Environment for Knowledge Analysis; XGBoost - Extreme Gradient Boosting.
Evidentiary studies spanned Australia, China, Colombia, France, Saudi Arabia, Singapore, South Korea, Spain, Turkey, and the U.S. from 2017 to 2024. These studies focused on using AI to improve diabetes diagnosis, treatment recommendations, and management strategies. The research designs included RCTs, mixed methods studies, prospective cohort studies, and cross-sectional studies, with sample sizes ranging from 62 to over 1.26 million participants. The AI tools and methods included SVM, RF, ANN, CNN, reinforcement learning, and Bayesian belief networks. Data sources encompassed EHRs, imaging data, patient histories, and literature.
There are six main categories of AI/ML techniques used for clinical decision support. Shen et al 35 used SVM learning to diagnose GDM with high accuracy, making it ideal for resource-limited settings. Random forest methods can be applied to analyze fundus photography and traditional Chinese medicine (TCM) features for noninvasive diabetes diagnosis. 36 Notably, Liu et al 37 utilized ANN for diabetes diagnosis, achieving a 92% correct diagnosis rate, and Yashar et al 38 employed EfficientNetB5 CNN for diabetes detection using digital breast tomosynthesis images with incidental analysis of pectoral muscle. Of further interest, Oh et al 39 implemented reinforcement learning (Q-learning) for personalized medication recommendations in patients with T2D. Lastly, Bayesian belief networks have been applied for diabetic retinopathy screening and lifestyle impact analysis. 40
Key Findings
AI-driven CDSSs contribute to lifestyle-integrated diabetes care in several key ways. Firstly, enhancing early detection and diagnosis is a critical precursor to initiating lifestyle interventions. Studies like Shen et al 35 (GDM diagnosis using SVM), and Yashar et al 38 (T2D detection from DBT images) and Hoyos W et al 62 (early diabetes detection with fuzzy cognitive maps) demonstrate AI’s capacity for accurate and often noninvasive diagnosis, enabling earlier engagement in lifestyle modification programs. Similarly, AI-based screening for complications, such as diabetic retinopathy (Pei X et al 40 ; Fu X et al 64 ), can underscore the urgency for improved lifestyle management.
Secondly, some CDSSs directly support or analyze lifestyle factors. For example, Wang L et al. 43 developed a knowledge graph highlighting lifestyle factors linked to diabetes complications, directly informing clinical advice. Seixas AA et al 64 utilized BBN to explicitly analyze the impact of lifestyle combinations on diabetes prevalence, providing evidence for personalized lifestyle recommendations. Fu X et al 63 reported that XGBoost models for blood glucose prediction could improve patient lifestyle guidance to manage glycemic events.
Thirdly, AI tools that enhance the visualization and interpretation of patient data can empower both clinicians and patients to make informed lifestyle choices. Derozier V et al. 58 demonstrated effective glycemic pattern display for clinical consultations, while BAVIERA-MARTINEZ C et al. 59 developed an interactive dashboard for real-time CGM data analysis, both facilitating a better understanding that can drive lifestyle adjustments. Finally, even CDSSs with broader aims can be relevant to lifestyle medicine. Tools predicting uncontrolled diabetes (Khalilnejad A et al 61 ) can help clinicians identify patients needing intensified lifestyle support. Clinician-facing tools (Liaw WR et al. 57 ; Yoon S et al. 41 ) can improve decision-making; their value for lifestyle medicine increases if they incorporate lifestyle data or prompt discussions about non-pharmacological options. Even medication optimization tools (Oh SH et al 39 ) could indirectly support lifestyle medicine by stabilizing a patient or freeing up clinician time for lifestyle counseling.
Discussion
The integration of AI into comprehensive diabetes care heralds a paradigm shift with substantial implications. Our synthesis indicates that AI is being applied across the continuum of lifestyle management, from predicting risk to personalize preventative lifestyle advice (e.g., Naveed et al, 14 Kumar et al 18 ), to delivering tailored guidance on nutrition and physical activity (e.g., Kannenberg et al, 25 Bul et al 26 ), enabling sophisticated remote monitoring and self-management support (e.g., Shamanna et al, 28 Helal et al 30 ), and informing clinical decisions pertinent to lifestyle interventions (e.g., Wang L et al, 43 Seixas AA et al 64 ). A key theme emerging is AI’s capacity to process diverse, complex datasets—from CGM and wearables to EHRs—to offer personalized and timely interventions that were previously unachievable at scale. For instance, the ability of AI to identify subtle patterns in glycemic responses to specific foods or activities (as seen in personalized guidance studies) offers a clear advantage over generic lifestyle advice.
AI’s capabilities align well with the novel dysglycemia-based chronic disease (DBCD) model,
44
which is pathophysiologically based and emphasizes a continuum of preventive care from insulin resistance (stage 1) to prediabetes (stage 2) to T2D (stage 3) to vascular complications (stage 4). The DBCD model stresses the importance of early intervention at all chronic disease stages through structured lifestyle changes, with judicious pharmacotherapy/procedures at later stages. AI can enhance these efforts by providing precise risk assessments and facilitating early and personalized interventions (Figure 1). Other works in the literature also underscore AI’s potential in improving outcomes through its ability to interpret complex data sets,
45
thus offering insights into the management of T2D and the prevention of micro- and macrovascular complications, particularly cardiovascular events.
46
Examples of Artificial Intelligence Tools for Integration in each State of Dysglycemia-Based Chronic Disease Model.* * The DBCD model is comprised of four stages as depicted to emphasize preventive care. Abbreviations: DBCD - dysglycemia-based chronic disease.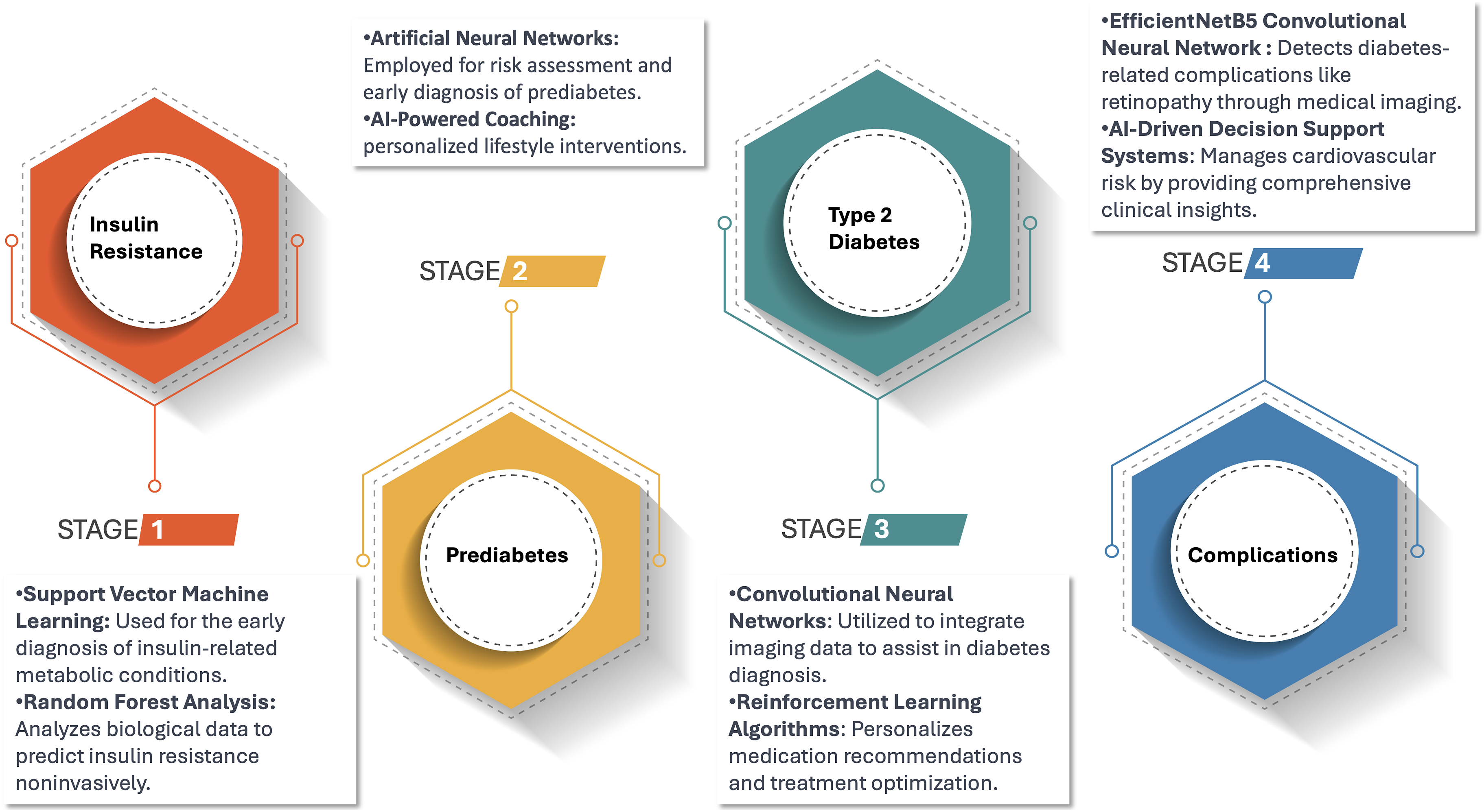
While promising, the evidence base reviewed herein has several limitations that temper immediate widespread application. Many studies rely on region-specific or small datasets, limiting the generalizability of their findings (e.g., some studies in Tables 3 and 4 with smaller cohorts), involved region-specific or relatively small datasets, raising questions about their generalizability across diverse populations and healthcare settings with varying technological infrastructures and socioeconomic realities. The “black-box” nature of some AI models, particularly DL algorithms, poses challenges for interpretability, training, and clinical adoption. Healthcare professionals (HCPs) may hesitate to trust AI recommendations without transparent explanations of how decisions are made. The integration of AI tools into existing healthcare systems faces technical, ethical, and regulatory hurdles, including data privacy concerns and the need for robust validation frameworks. Fourth, the complexity of AI models and the requirement for extensive computational resources can limit scalability, particularly in resource-constrained settings. Fifth, ethical concerns regarding data privacy and patient consent when using AI-driven systems need to be addressed. Finally, while AI models often achieve high accuracy in controlled research settings, their long-term efficacy and real-world performance in diverse clinical environments remain understudied.
Future research on AI in diabetes care will need to address these limitations. Larger, multi-site trials with diverse populations are needed to validate AI tools and establish their generalizability and long-term effectiveness in real-world settings for lifestyle medicine. It will be essential to develop standardized protocols for data collection and AI model validation to ensure consistency and accuracy. Innovations in AI technology should focus on improving user interface design to enhance the accessibility and usability of AI tools for both patients and HCPs. Collaborative efforts between technologists, HCPs, and policymakers will be needed to effectively integrate AI solutions into existing healthcare frameworks. Furthermore, emphasis on ethical AI deployment, including transparent data practices and patient-centered approaches, will be critical to fostering trust and promoting widespread adoption.
This narrative review has several limitations. Firstly, the search was primarily confined to the PubMed database, which, while comprehensive for biomedical literature, may have omitted relevant studies published in dedicated AI/computer science conference proceedings or journals not indexed in PubMed. Secondly, as a narrative review, it does not employ a systematic, reproducible protocol for study selection and data, potentially introducing selection bias. The qualitative synthesis, while aiming for a broad overview, is inherently subjective to some extent. Finally, the focus on English-language publications might have excluded relevant research from other regions. These limitations mean that while this review provides a broad overview of AI in lifestyle medicine for diabetes, it is not an exhaustive account of all existing literature or a quantitative summary of effects.
In conclusion, AI holds considerable promise for revolutionizing lifestyle medicine within diabetes care. This review highlights AI’s emerging capacity to enhance risk prediction for proactive lifestyle interventions, deliver highly personalized guidance for diet and physical activity, empower patients through sophisticated remote self-management tools, and inform clinical decisions pertinent to lifestyle modification. To fully realize this potential, future efforts must focus on robust validation in diverse real-world settings, ensuring equitable access and algorithmic fairness, and seamlessly integrating these powerful tools into patient-centered lifestyle management pathways. Addressing these challenges will be key to harnessing AI to meaningfully improve outcomes for the millions affected by diabetes through enhanced lifestyle medicine.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JM reports receiving honoraria for lectures from Abbott Nutrition and Merck and serves on the advisory boards of Abbott Nutrition and Twin Health. JPGR and AS do not have conflicts to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
