Abstract
The microbiome connects the gut health to the rest of the body’s organs, including the skin. The pathophysiology of intestinal dysbiosis leads to the expression “leaky gut.” Ongoing speculations are aimed at restoring the gut microbiota via modulating lifestyle habits to prevent and potentially reverse autoimmunity. This review finds the connections between gut dysbiosis and skin conditions. It also collects evidence of some lifestyle pillars that influence the gut microbiome including dietary intakes, exercise, sleep, stress, and toxin exposures. It addresses why maintaining a healthy intestinal microbiome is important for the health of all organs in the human host. More people are affected by gut dysbiosis resulting in pro-inflammatory effects on different organs, like the cutaneous tissue, one of the largest epithelial surfaces. It is essential to take care of the gut health because this is where most of the immune system resides. The connection between the intestinal tract with its microbiota and the cutaneous system with its microbiota seems to be mediated by the immune system of the human host. Therefore, this review enhances the understanding of the research on the gut microbiome, its relationship to skin health, and the interplay between the gut and various autoimmune cutaneous conditions.
“Gut dysbiosis results from a depletion of beneficial bacterial species in the presence of over-production of other harmful species.”
Introduction—Background and Purpose
Maintaining healthy skin and gut microbiomes is crucial to achieving healthy epithelial homeostasis and immunity. The lifestyle influences on the microbiome composition are evident for multiple conditions beyond the skin.1,2 Both genetic factors and environmental triggers have impact on the gut microbiome and the immune system response.3,4 This clinical review serves to showcase the importance of keeping a healthy microbiome to protect against harmful pathogens. It should inform the readers of current supporting research on the direct impact of gut health on skin health. Furthermore, its focus is mainly on the environmental triggers manifesting as lifestyle factors that contribute to the health or the disorder of the gut microbiome, which ultimately regulate the balance of the skin microbiome. It seeks to find a current evidence-based understanding of the connections between intestinal dysbiosis and skin conditions, such as psoriasis. There is a vicious cycle between gut inflammation and gut dysbiosis that will be discussed as a bidirectional relationship, in which gut dysbiosis may lead to pro-inflammatory responses in the human host, and inflammation can cause more dysbiotic microflora in the gut. The gut-skin connection is mediated by the host’s immune system, and there are ongoing investigations of the linkage between the gastrointestinal system and comorbid chronic skin disorders.2,4 A concise summary based on evidence from published literature is needed to help clinicians better understand the gut-skin axis and provide science-backed recommendations for the patients.
Epidemiology and Definitions
Before diving in, the terms microbiota and microbiome should be clearly defined. While “microbiome” is an umbrella term that includes the entire variety of microorganisms, from bacteria, viruses, and fungi, along with their metabolites and surrounding environments, the term “microbiota” is less comprehensive as it represents only a subgroup of microorganisms, from symbiotic, commensal, and pathogenic located in an established environment. 1 Therefore, these terms are sometimes used loosely or interchangeably, in the appropriate context. Gut microbiota is also known as gut microflora or gut flora, for short. The microbiome of the gut is the largest endocrine organ that produces more than thirty hormone-like compounds, such as short-chain fatty acids (SCFAs), cortisol, neurotransmitters like gamma-aminobutyric acid (GABA), dopamine, and serotonin. 2 Autoimmune dermatologic conditions, like psoriasis as a prime example, result from dysfunction of the epithelial barrier and an increased in cellular turnover rate.2,5 The immune-mediated connection of dysbiosis of the gut and the skin was well described in a recent study published in 2024 where the authors utilized the bidirectional two-sample Mendelian randomization (TSMR) method to explore this causal relationship between the microbiome of the gut and the following conditions: psoriasis, atopic dermatitis, acne, and lichen planus. 6 This was the first bidirectional TSMR to link evidence of intestinal-cutaneous axis.
The human body possesses remarkable innate healing capabilities. Given the right support and conditions, it can often restore balance and health on its own. This underscores the importance of providing the body with the proper nutrients, environment, and lifestyle habits conducive to optimal health and healing. For example, one of the most naturally healing nutrients is derived from the breakdown of fibers consumed by the human host, known as SCFAs, which come in three main types: acetate, propionate, and butyrate.2,7 Particularly, butyrate is a four-carbon SCFA created as a result of intestinal microbial fermentation of indigestible fibers. 7 It transmits signals to receptors in the colon and the liver and has powerful anti-inflammatory properties via suppressing the production of pro-inflammatory cytokines and deactivating NF-kappaB expression. 8
Gut-Skin Axis Description
The gut microbiome and the skin microbiome host a diversified population of species consisting of bacteria, viruses, and fungi. In a healthy environment, they remain symbiotic with the human host. However, when the balance is disrupted and the barrier function is impaired, dysbiosis of either or both microbiomes is the result. Skin homeostasis recovery after disruption through the gut microbiota gives rise to both innate and adaptive immunity.2,9 The immune system connecting the gut and the skin could explain how lifestyle choices can influence the microbiomes of both. Recent research published by Feng F et al, in 2024 concluded that there is a causal relationship between the gut microbiota and immune-mediated skin conditions. 6 Just over 5 years ago, it was reported there was emerging evidence that the gut microbiome may have direct influences on the pathophysiology and immune response of the skin via the metastasis of the gut microbiota and their metabolites to the skin. 9
Dysbiosis Pathophysiology
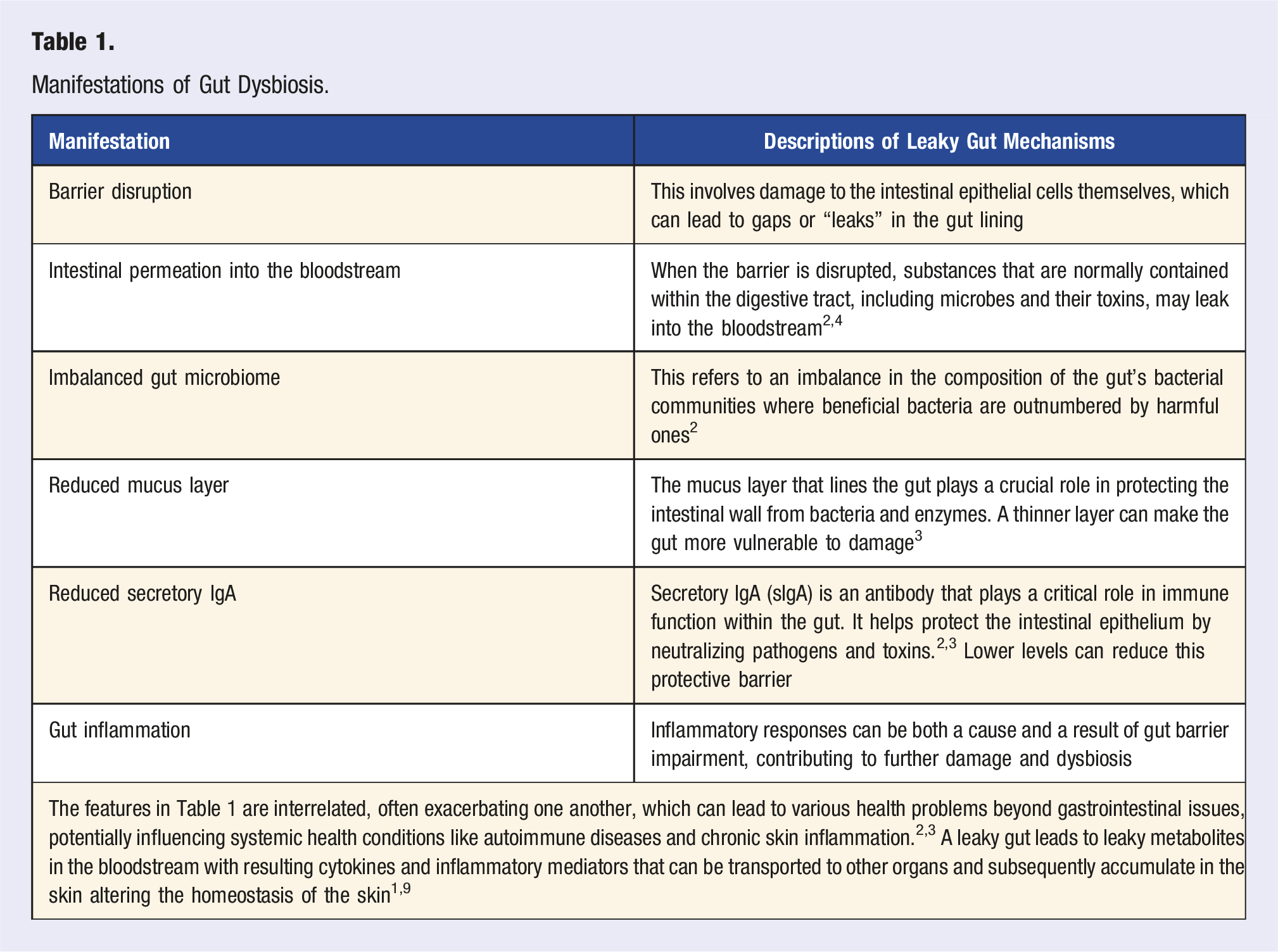
Manifestations of Gut Dysbiosis.
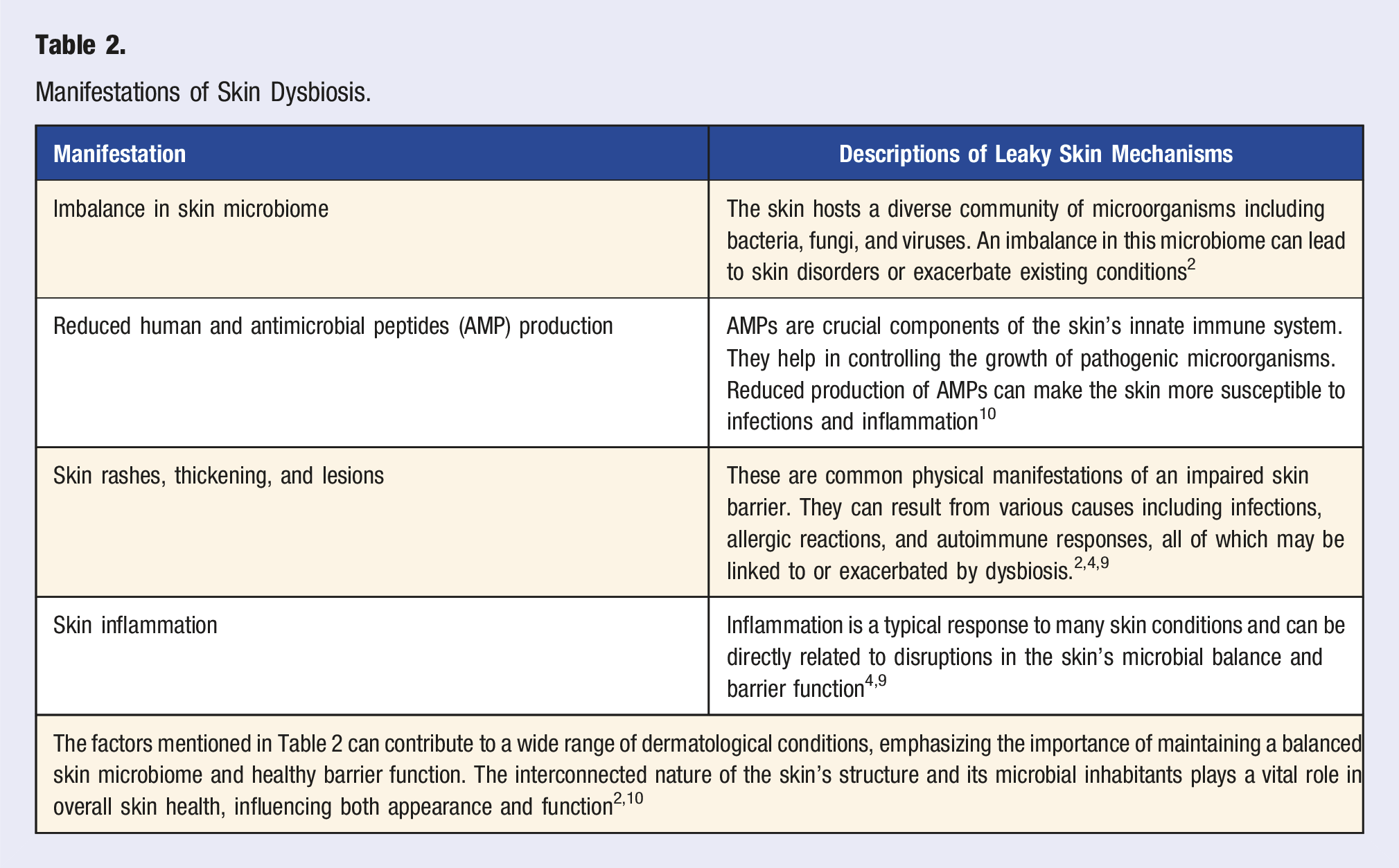
Manifestations of Skin Dysbiosis.
While the gut microbiome continues to modify throughout the lifetime of a human host, the skin microbiome stabilizes earlier on in life around the age of three. 11 However, the microbial composition of the skin can still be altered depending on the microbial biodiversity of the gut and skin microbiota, including modifications of the surrounding soils, which can either enhance or degrade certain cutaneous bacterial species. 9 Furthermore, there are commensal microbes within the skin that regulate mucosal-associated invariant T (MAIT) cells which react to T-cell receptor-mediated recognition of metabolites, serving to prevent skin dysbiosis by defending bacterial skin infections and improving healing of skin wounds.11,12 MAIT cells are also associated with regulating the intestinal epithelial barrier and the lung epithelial cells; thus, they interact bidirectionally with the local microbiota, suggesting their involvement in maintaining a balanced microbiome and contributing to overall epithelial health and function.11,12
Why Choose the Connection Between Gut Health and Skin Health?
Clinical Significance
At the time this clinical review is written, there has been a significant amount of new research and published articles released over the past several years on the lifestyle factors and their relationships with gut microbiome and psoriasis, specifically, one of the most well-acknowledged autoimmune types of skin conditions.5,10 Certain cutaneous autoimmune disorders are triggered by the inflammatory process occurring at the gut permeability level, so-called leaky gut.1,2,11 Most of our immune system is located along the wall of the intestine, so identifying the root causes of the leaky gut could prevent autoimmune cascades or pro-inflammatory responses from occurring and recurring in the body. The skin and the gut tissues have a high cellular turnover rate and are extremely responsive to stressors from the environment and lifestyle choices. Increased permeability of both the epidermal skin and intestinal barriers, also known as leaky skin and leaky gut, respectively, can result from various factors including inflammation. When the barrier function of the skin or intestine is compromised, it allows pathogens, allergens, and other potentially harmful substances to interact more readily with inflammatory receptors on immune cells, triggering immune responses and potentially leading to inflammatory conditions and allergic reactions.2,10
Immunologic Connection
There is growing evidence suggesting a connection between gut and skin dysbiosis through the immune system, particularly through the imbalance of T-helper (Th) cells.
2
Th2 skewing is a condition where there is an overactivity of Th2 cells relative to other types of T-helper cells like Th1 and Th17. This imbalance can influence both gut and skin health in the following ways2,10: • • •
Understanding the gut-skin axis and the role of immune imbalance provides insights into potential therapeutic strategies for treating conditions related to both the gut and skin by targeting microbial balances and immune responses.2,5,10
Why is a Leaky Gut Leading to a Leaky Skin Important?
Dermatoses, which encompass a wide range of skin disorders and conditions, pose a significant burden worldwide. In fact, they are recognized as the fourth most common non-fatal burden globally. 1 The role of the gut microbiome is crucial to combat dysbiosis that leads to skin conditions such as psoriasis, eczema, acne, alopecia areata, and more. There is an immense impact on the quality of life of individuals suffering from chronic skin diseases that stem from disruption of the gut microbiome. The negative psychosocial and emotional implications are often even more damaging to those affected by these skin conditions than even the physical discomfort themselves. Skin disorders can vary widely in their severity and impact on well-being, ranging from mild conditions like acne and eczema to more serious diseases like plaque psoriasis and skin cancer. They may affect self-esteem, body image, and interpersonal relationships, leading to emotional distress and reduced quality of life for affected individuals. Therefore, efforts to address the burden of dermatoses through a root-cause analysis include targeting the gut microbiome with advancing research into the prevention and treatment strategies, for example, through lifestyle modifications and the use of probiotics.
How are Disruption and Maintenance of the Healthy and Balanced Microbiomes Occurred?
An unhealthy microbiome can result from not only a lack of diversity in gut bacteria but also various factors like poor dietary choices, inadequate physical activity, low-quality sleep, high stress, occupational exposures, risky substance use, and antibiotic use. This imbalance not only leads to weakened immunity and digestive or skin issues but also contributes to conditions like obesity and autoimmune diseases. For example, both inflammatory bowel disease (IBD) and psoriasis are classically linked via the immune system and are characterized by dysbiosis in the microbiome composition. 2 Balancing the gut microbiome through lifestyle changes and the use of probiotics can help restore and maintain the health of both the gut and the skin.9,10,13 By addressing factors such as diet, exercise, sleep, stress, and toxin exposure, individuals can potentially mitigate the impact of genetic risk factors on their health outcomes.1,13 This approach highlights the significance of personalized care and empowers individuals to take proactive steps toward optimizing their health.
Nutrition
As mentioned earlier, gut dysbiosis refers to an imbalance in the gut microbiota composition, which can significantly impact the way the body processes and responds to dietary interventions. The altered microbial community may affect how nutrients are absorbed, metabolized, and utilized by the body, thereby influencing the effectiveness of dietary interventions aimed at improving health outcomes. This underscores the importance of addressing gut health as part of dietary interventions and personalized health care approaches. The gut microbiota could be influenced by not only genetic predisposition but also dietary changes or a direct outcome of immune system disorders. 4 For example, in the majority of patients with IBD, cases have been linked with gut dysbiosis. 14 Dietary awareness is crucial to the management of IBD because diet plays a critical role in shaping the gut microbial composition. Bacterial diversity is markedly different in humans on a fiber-rich diet compared to a meat-based diet without fiber. In a study in 2020, a low-fiber diet was shown to disrupt the homeostasis associated with health and start the path toward disease. 15
SCFAs are derived from a fiber-rich diet and can be absorbed by the gut epithelial cells as a nutrient supply. This helps improve leaky gut via promoting better tight junctions, increasing mucin and goblet cells, and leading to a reduction in pro-inflammatory cytokines cascading effects. 8 Ten percent of daily caloric needs are met using SCFAs as energy sources. 7 The cells that line the colon are known as colonocytes, and they consume SCFAs in the form of butyrate, which helps the tight junction proteins to be repaired, in the case of a leaky gut. Remember that leaky gut is an expression adopted simply to describe intestinal hyperpermeability that ultimately affects the proper functioning of the intestinal lining, leading to inflammatory downstream effects. 7 Therefore, targeting dysbiosis with the consumption of SCFAs may represent a root-cause approach to a variety of inflammatory conditions, including immune-mediated skin conditions. The more diversity of plants a human host consumes, the more SCFA-producing good gut microbes grow, including a species known as Faecalibacterium prausnitzii, which provides a variety of benefits to the host. 9 For example, a study showed psoriatic patients have a depleted number of the protective F prausnitzii species compared to healthy controls. 16 Another beneficial bacterial strain that is highly abundant in a healthy gut is called Akkermansia muciniphila. An imbalance in the gut microbiota favors pro-inflammatory processes because of a lower abundance in F prausnitzii and A muciniphila.1,13 F prausnitzii produces butyrate with anti-inflammatory properties to help maintain the integrity of the gut barrier. Further research is still needed to define the effects of diet on specific cutaneous diseases, especially randomized controlled trials. 17 Nonetheless, there is a promising future for targeted microbiome strains as part of a nutritional therapeutic approach to prevent and reverse immune-mediated or inflammatory conditions. 17
Physical Activity
Research is still limited on the effects of an active lifestyle on the gut microbiome. However, several studies have shown that aerobic exercises significantly change the composition of the gut microflora while comparing elite vs non-elite athletes. 14 Regular physical exercise positively impacts the gut microbiota and decreases gut inflammation, thereby strengthening the gut lining to prevent leaky gut. 18 Furthermore, exercise not only enhances the diversity of gut microbiota but also modulates its function. 19 The more active an individual is, the higher the abundance of beneficial bacteria and the lower the number of harmful species. 18 An important contribution of exercise to gut health is the reduction of transit time for food going through the GI tract. As a result, this reduces the exposure of potential pathogens to the gut lining, which indirectly affects the bloodstream and reduces the circulation of harmful species that may end up causing an inflammatory effect on the skin.
Sleep
Sleep is a biological necessity that takes up about 30% of every 24 hours of life. Needless to say, it is essential for the health and well-being of every organ in the body. In modern society, sleep deprivation unfortunately has become more common, and subsequently, detrimental consequences are faced. Recent studies trended the changes in the gut microbiota because of sleep deprivation leading to harmful effects on other body systems, due to the interconnectedness of the gut microbiome and other organs, including the skin.1,8 Depending on the depletion of the numbers of various beneficial bacteria in a sleep-deprived individual, certain pathological changes can be initiated or exacerbated, such as autoimmune diseases. Sleep deprivation-induced immune impairments are a result of the inflammatory cascades of events that occur in the host, most importantly, at the level of an impaired intestinal gut barrier. There is an interesting area of research that connects sleep, melatonin supplementation, and changes in the gut microbiota. Melatonin, often called the “sleep hormone,” not only regulates sleep cycles but also plays a role in modulating the gut microbiome. 8 This evidence supports the notion that melatonin could potentially be used as a therapeutic agent not just for correcting sleep disorders but also for maintaining or restoring gut microbiota balance, particularly in conditions induced by or associated with sleep deprivation. Further studies in humans are needed to validate these findings from animal models and to explore the potential therapeutic implications of melatonin on gut health and beyond. 8
Stress Resilience
During periods of excessive stress, the body’s stress response can impact the immune system and, in turn, affect the integrity of the gut barrier and the balance of the gut microbiota. 20 Stress-induced activation of the immune system can lead to increased inflammation, which may compromise the tightness of the intestinal barrier, resulting in a leaky gut. This altered barrier function can allow harmful substances to pass through more easily, potentially disrupting the balance of the gut microbiota and leading to various extra-intestinal issues, including leaky skin. Gut inflammation has been linked to the risk of development of mental health disorders. 18 Chronic stress can lead to an imbalance in the body’s antioxidant defenses, resulting in increased oxidative stress. This oxidative stress can damage cells and disrupt various physiological processes, contributing to further inflammation in the brain and throughout the rest of the body. 20 Consequently, inflammation can impact neurotransmitter function and disrupt neural circuits involved in mood regulation, ultimately resulting in the development or exacerbation of depression. Understanding the interplay between oxidative stress, inflammation, and depression is crucial for developing effective therapeutic interventions targeting these pathways via a root-cause approach. Studies suggest that certain probiotic strains can help modulate neurotransmitter levels, reduce inflammation, and improve stress response, all of which may contribute to improved mental health outcomes.1,20 As mentioned earlier in this review, suffering from a chronic skin condition is already mentally straining and this stressor has the potential to beget more stress, leading to further suppressed immune function as the vicious cycle continues, unless carefully interjected. While more research is needed to fully understand the mechanisms and efficacy of psychobiotics, which are probiotic bacteria with potential mental health benefits, the findings so far are promising and highlight the potential of using probiotics as adjunctive therapy for mental health conditions like depression and anxiety.18,20
Avoidance of Risky Exposures like Smoking
Particulate matter exposure has been shown to increase leaky gut. 21 Other toxic environmental factors, including cigarette smoking, also promote gut dysbiosis. Once ingested into the gastrointestinal tract, the gut microbiota is altered, leading to a disrupted immune response and interrupted antimicrobial regulatory activity. 22 Due to the connectedness of immune-induced gut and skin diseases, as in the cases of IBD and psoriasis, one could associate the pathogenesis of IBD with the induction of psoriatic flares as a result of smoking, with the model of smoke-driven microbiota alterations. 22 Similarly, the microbiome disturbances due to antibiotic use can profoundly lead to immune system impairment. The phrase “antibiotic-driven gut microbial dysbiosis” was used to describe how adverse effects could occur on the human host after the use of antibiotics, resulting in improper interactions of the immune system with extra-intestinal organs, including the skin. 23
Outlook on Probiotics Use
Whether in primary care or specialty care, clinicians must understand the connection between the gut microbiome and its connection to skin health. Even though there is a tremendous amount of published and ongoing research on this connection, there are still frequently missed opportunities to address gut health as it relates to skin health concerns. The connection is not only direct, like an axis, but also indirect through the immunologic responses.2,6,10 A paper published in 2022 by Mahmud R, MD et al clearly defined leaky gut and the interplay among the factors that influence the diversity of the gut microbiome. The authors emphasized the complexity of the factors at play, which include
10
: • • •
The interplay among these factors contributes to the integrity of the gut barrier, with disruptions potentially leading to leaky gut syndrome. Understanding this interplay is crucial for developing strategies to maintain or restore gut health and prevent or treat conditions associated with a compromised intestinal barrier. These strategies include the use of probiotics, prebiotics, dietary intakes, and other consumed substances that affect the skin through the gut microbiome. 10 Probiotics are not limited to supplements; they can be derived directly from plant-based foods. Prioritizing a food-first approach with dietary, fiber-rich options can help restore a leaky gut or a leaky skin by supporting a healthier microbiome. 24 Furthermore, research on psychobiotics has been growing. These beneficial bacteria, when consumed in appropriate amounts, can influence the gut-brain axis, a bidirectional communication network between the gut and the brain. 20
Conclusion and Future Directions
Gut dysbiosis results from a depletion of beneficial bacterial species in the presence of over-production of other harmful species. There are still many unknowns around microbiome-immunity interactions whether in disease or health.25,26 In gut dysbiosis, harmful intestinal microbes, and their metabolites breach a hyperpermeable gut barrier and enter the systemic circulation, targeting distant organs.1,2 Specifically with regards to the gut-skin axis, future research directions will likely dive deeper into lifestyle modifications to gain a fuller understanding of microbiome changes in both the gut and the skin. More studies are needed to explore the specific mechanisms by which gut-skin microbiota and each lifestyle pillar are linked. The question remains: Do changes to the microbiome occur first and then lead to dysbiosis of the gut and skin, or did the changes in the microbiome occur because the disorders of the gut or skin led to alteration of the mucosal immunity that favor certain microbes? Further clinical research is warranted to identify the resolution of specific skin conditions or relapses of immune-mediated skin diseases as the gut microbiome heals or impairs, respectively. Healthy dietary and lifestyle choices working together plus the intervention of supplemental probiotics could improve gut microbial health and ultimately yield beneficial long-term effects on the skin microbiome and overall well-being. Finally, we may speculate that the gut-skin microbiota can be modulated via the use of probiotic strains, including naturally occurring foods from fermented products and well-developed supplemental strains, which may help in the prevention and treatment of immune-mediated skin disorders. There is still more research to be done to showcase how lifestyle influences and the use of probiotics impact the gut microbiome and ultimately the skin microbiome as a result. This review is not meant to expand beyond the axis of the gut and the skin. Nonetheless, there are more opportunities to see the different axes of gut-skin-brain-lung connections to give additional ideas for exploration down the road.
Footnotes
Acknowledgments
The author especially thanks A.T. Still University Doctor of Medical Science Program and its professors for providing the opportunity to research articles to write and publish this review.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article. This Clinical Review received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
