Abstract
Per- and polyfluoroalkyl substances (PFAS) are a class of toxic, ubiquitous, anthropogenic chemicals known to bioaccumulate in humans. Substantial concern exists regarding the human health effects of PFAS, particularly metabolic syndrome (MetS), a precursor to cardiovascular disease, the leading cause of mortality worldwide. This narrative review provides an overview of the PFAS literature on 4 specific components of MetS: insulin resistance/glucose dysregulation, central adiposity, dyslipidemia, and blood pressure. We focus on prospective cohort studies as these provide the best body of evidence compared to other study designs. Available evidence suggests potential associations between some PFAS and type-2 diabetes in adults, dyslipidemia in children and adults, and blood pressure in adults. Additionally, some studies found that sex and physical activity may modify these relationships. Future studies should consider modification by sex and lifestyle factors (e.g., diet and physical activity), as well quantifying the impact of PFAS mixtures on MetS features and related clinical disease. Finally, clinicians can follow recently developed clinical guidance to screen for PFAS exposure in patients, measure PFAS levels, conduct additional clinical care based on PFAS levels, and advise on PFAS exposure reduction.
“In reviewing studies examining PFAS and MetS features, we observed evidence for an association between perfluorooctanoic acid (PFOA) and T2D in adults.”
Introduction
Per- and polyfluoroalkyl substances (PFAS) are a large class of anthropogenic fluorinated chemicals, where some or all hydrogen atoms on an alkane backbone have been replaced by fluorine atoms (except those associated with functional groups).1,2 Because the carbon-fluorine bond is the strongest single bond in organic chemistry, these compounds are environmentally persistent and have been dubbed “forever chemicals.”2,3 PFAS are environmentally ubiquitous and can recirculate in water and the atmosphere. 4 This has resulted in PFAS having been found on all 7 continents, including Antarctica.4,5
PFAS have been detected in drinking water, food, household dust, and numerous consumer products and industrial applications where they are then capable of entering the human body, primarily through ingestion (Figure 1).4,6-11 As a result, PFAS have been found in the blood serum of nearly every human tested on earth in addition to a multitude of other matrices including breastmilk, urine, hair, placental cord blood (serum, whole, and plasma), and amniotic fluid.12-18 To date, only a handful of PFAS have been studied in humans. However, of those that have been studied, most have been found to be both toxic and bioaccumulative, with half-lives ranging from a few months to a few years.19-22 Due to their ubiquitous nature, environmental persistence, toxicity, and bioaccumulative properties, substantial concern exists regarding the human health effects of these compounds. A summary of primary per- and polyfluoroalkyl substances exposure pathways.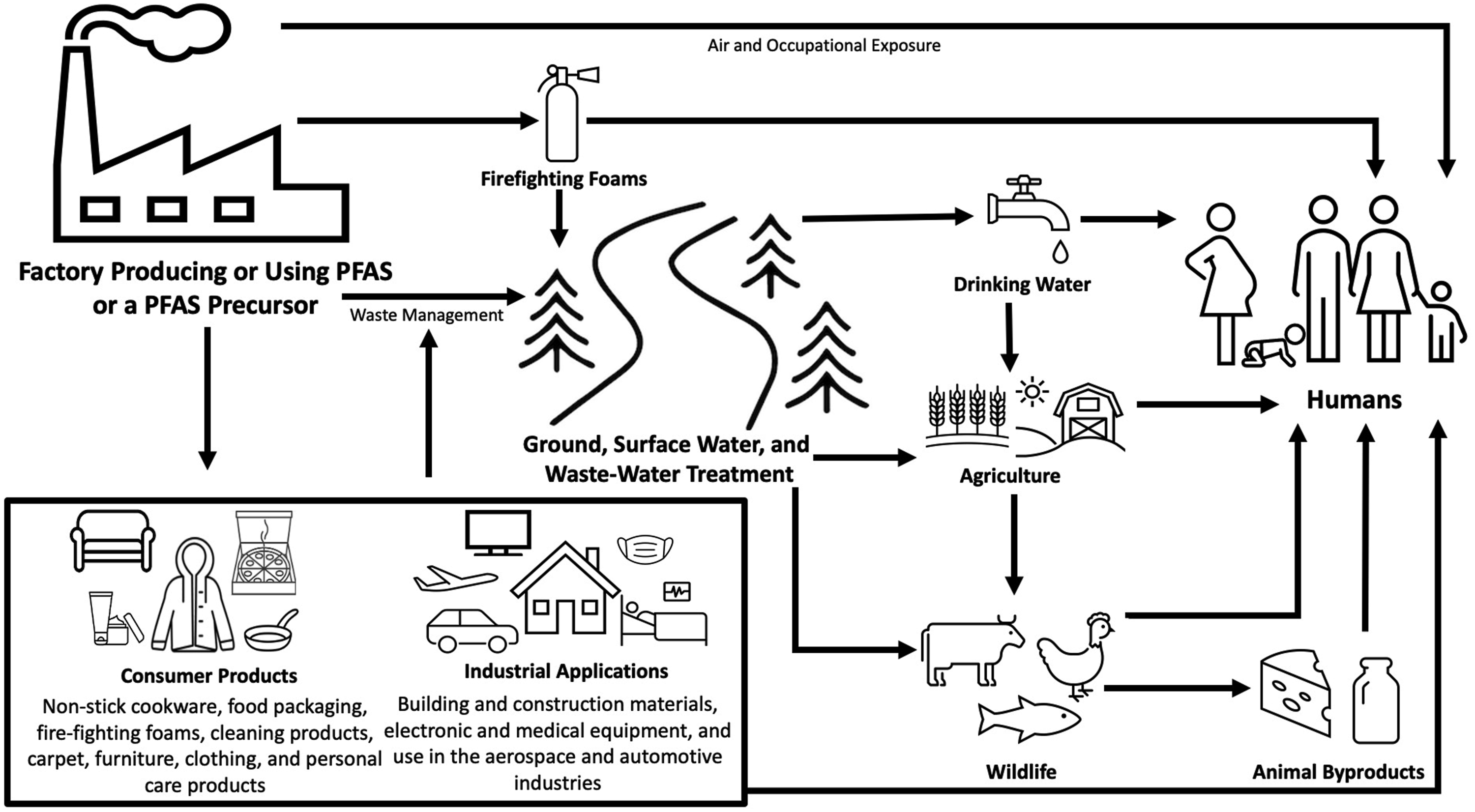
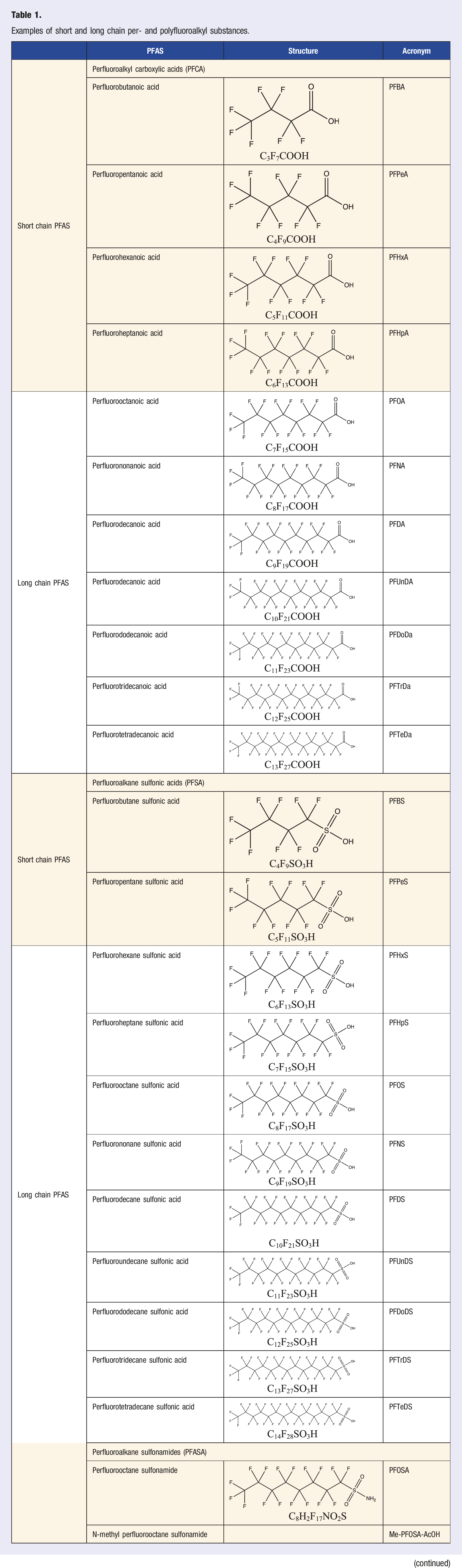
Examples of short and long chain per- and polyfluoroalkyl substances.
Multiple PFAS have been associated with a multitude of adverse health effects including increased preeclampsia risk,30,31 increased risk of miscarriage, 30 preterm birth,29,30 and lower birth weight, 29 decreased vaccine response in children, 32 increased incidence of liver disease, 33 increased risk of kidney-, 34 testicular-, 34 and prostate-cancer, 34 as well as multiple adverse cardiometabolic35-38 and cardiovascular 39 outcomes. Of the adverse health effects associated with PFAS, those associated with metabolic syndrome (MetS) are of particular concern. MetS is an umbrella term referring to a constellation of cardiovascular-related metabolic outcomes.40,41 Examples include insulin resistance, glucose dysregulation, increased central adiposity, dyslipidemia, and increased blood pressure (BP). MetS are of particular concern as these abnormalities are often precursors to cardiovascular disease (CVD), the leading cause of mortality responsible for an estimated 32% of deaths globally. 42 As such, identification and intervention on a modifiable risk factor for MetS could assist in decreasing the incidence of CVDs, thus having a substantial impact on the overall disease burden. 43 Furthermore, gestational and early life exposure is of particular interest as infants or children are potentially more vulnerable to PFAS given that they may have higher PFAS exposure and be more susceptible to environmental exposures than adults due to their rapid development. 44
The aim of this narrative review is to summarize the current evidence on PFAS and MetS. As the purpose of this review is to provide a general overview of PFAS and MetS rather than explore a single specific component of MetS in depth, a narrative review, rather than a systematic review, was conducted. To accomplish this aim, we will first discuss the biological mechanisms of PFAS action and specific populations that may be affected by PFAS differently or exposed to differing PFAS dosages. We will then discuss the reviews to date on PFAS and MetS. Next, we will provide an overview of the literature on PFAS and 4 specific components of MetS: (1) insulin resistance or glucose dysregulation, (2) central adiposity, (3) dyslipidemia, and (4) BP. We will focus this review on prospective cohort studies as these provide the best body of evidence compared to other study designs such as cross-sectional studies. 45 Finally, we will summarize recent recommendations set forth by the National Academies of Science Engineering and Medicine for assessing, reducing, and following up on PFAS exposure in clinical settings.
Biological Mechanisms of PFAS Action and Exposure in Specific Populations
The biological mechanisms of action of the PFAS-MetS association are likely multifaceted and only beginning to be understood, with most mechanistic studies focused on PFOA or PFOS specifically. Preliminary animal and human studies have demonstrated that PFAS may have the capacity to target peroxisome proliferator-activated receptor-α (PPAR-α) and, to a lesser extent, peroxisome proliferator-activated receptor-γ (PPAR-γ).46-51 Through this mechanism, PFAS could adversely affect lipid and glucose metabolism or promote adipogenesis.46-51 However, studies have found that PPAR-α may respond less to PFOA exposure in humans than in rodents; as such, interpretation of animal studies should be made with care.51-53 To address this, Schlezinger et al. has conducted research on PFOA in humanized PPAR-α mice that were fed an American diet.54,55 This research found increased PFOA may induce liver triacylglycerides, skew serum triacylglycerides toward more saturated fatty acids, and activate humanized PPARα and CAR in the liver at human relevant exposure levels.54,55 However, these effects of PFOA were both independent and dependent on PPARα, suggesting that PFOA may act via multiple biological pathways. 54 Other studies on other nuclear receptors such as constitutive androstane receptor (CAR) and pregnane X receptor (PXR) have also been conducted, but results from these studies have been inconsistent.46,54,56-59
PFAS body burden, measured by serum or plasma concentrations, has been found to vary by sex, with studies finding higher concentrations of PFAS such as PFOS, PFOA, perfluorononanoic acid (PFNA), and perfluorooctane sulfonic acid (PFHxS) in males compared to females.60,61 Potential reasons for this include female reproductive-related characteristics such as menstruation, maternal-fetal transfer during pregnancy, and lactation, as these can be a source of PFAS-elimination.60,62-64 This is further re-enforced by greater disparities in many of these PFAS concentrations in males and females among those of pre-menopausal age compared to those of a post-menopausal age, suggesting characteristics before menopause, such as lactation or menstruation, may contribute to the disparity between males and females. 60
In addition to sex-specific differences, differences in several PFAS concentrations by race and/or ethnicity have also been observed.23,65,66 For example, in the 2017–2018 National Health and Nutrition Examination Survey (NHANES), participants who identified as Hispanic had lower concentrations of PFHxS, PFNA, 2- (N-methylperfluoroctanesulfonamido) acetic acid (Me-PFOSA-AcOH), and PFOS compared to those who identified as non-Hispanic (there were no difference by Hispanic vs non-Hispanic for perfluorodecanoic acid (PFDA) or perfluoroundecanoic acid (PFUA)). Additionally, those who identified as Asian and/or those who identified as non-Hispanic white generally had higher concentrations for PFHxS, PFNA, PFOA, and PFOS compared to those who did not identify as Asian or non-Hispanic white. 23 A study by Nguyen (2020) et al. using 1999-2014 NHANES and another study by Nelson (2012) et al. using 2003-2006 NHANES data evaluated demographic patterns for PFOA, PFOS, PFNA, and PFHxS, with Nelson et al. also looking at Me-PFOSA-AcOH and PFDEA.65,67 These studies found similar patterns for these PFAS, observing that racial disparities are persistent even when controlling for socioeconomic status (SES).65,67 Furthermore, Nguyen et al. also found those who identified as non-Hispanic white had the highest PFOA concentrations, with those identifying as non-Hispanic Black or other/multi-racial having the highest PFDA concentrations even after controlling for SES, suggesting these trends may be PFAS specific. 67 More so, Nelson et al. observed participants from higher income communities had higher PFAS levels for all measured PFAS compared to participants from lower income communities. 65 Although the reason for these disparities are not fully known, they may be the result of cultural differences, dietary differences, and/or behavioral differences such as usage (or degree of usage) of PFAS-containing products. 65
Disparities regarding some PFAS concentrations by age have also been observed.23,68 In the US, older adults have been found to have higher concentrations of several PFAS compared to those who are younger; however, this age-related difference appears to be PFAS specific.23,68 For example, participants 60 years and older from the 2017-2018 NHANES had higher PFNA, PFUA, n-PFOA (a linear isomer of PFOA), and PFOS concentrations compared to participants under 60 years; however, no differences were present for perfluorodecanoic acid (PFDeA), PFHxS, or Me-PFOSA-AcOH by age. 23 Reasons for these specific PFAS concentrations being higher in older individuals compared to those who are younger could be due to multiple reasons. These reasons include bioaccumulative properties of legacy PFAS like PFOA and PFOS who had higher use historically,23,69,70 as well as age-related differences in diet and/or behavior. Conversely, children have been observed to have higher PFAS concentrations compared to adults for PFAS such as PFOA, PFOS, PFNA, and PFHxS.71,72 This difference in these PFAS concentrations between children and adults is thought to be primarily the result of higher body size to surface area ratios, breastfeeding, proximity to the floor which could increase dust inhalation and/or ingestion, increased hand-to-mouth behavior, and/or dietary differences.71,72 Furthermore, some occupations can lead to higher exposure to certain PFAS, with firefighters and those who work in making, processing, or using PFAS or a PFAS precursor having higher serum/plasma levels than the general population.73,74
Reviews on PFAS and Cardiometabolic Outcomes
To our knowledge, only three other review articles have synthesized the literature on PFAS and MetS to date.75,76 The first article was written by Jeddi (2021) et al. and focused on MetS as an overall diagnosis; as such, this review did not incorporate literature that focused on a specific components of MetS. 75 This systematic review found 12 cross-sectional studies (10 in the general population and 2 in an occupational setting), with half of these studies conducted in North America (5 in the US and 1 in Canada). 75 Jeddi et al. did not find any studies investigating the PFAS-MetS relationship in children and only one study, 77 evaluating this relationship in adolescents. 75 The most common PFAS assessed in these 12 cross-sectional studies were PFOA (N = 7), PFOS (N = 7), PFHxS (N = 5), and PFNA (N = 5). 75 As such, this review was not able to draw conclusions on other PFAS or effects in children or adolescence due to the limited number of studies. In their review, the authors conclude there is no evidence of an association between PFAS and MetS, but caution is warranted given that the reviewed studies were cross-sectional and did not include studies of children and adolescents who might be more vulnerable to the effects of PFAS. 75
The second review on PFAS and MetS in adults was conducted by Gaston (2020) et al. 76 This review was restricted to studies published between January 2018 and April 2019 and focused on synthetic chemicals (including PFAS) and cardiometabolic health across the life course, specifically in vulnerable populations. 76 Gaston et al. found 9 studies on PFAS and cardiometabolic health in vulnerable populations (8 studies with children [5 prospective and 3 cross-sectional] and one prospective study with pregnant people). 76 Among these studies, only 1 study investigated sex as a potential effect measure modifier (EMM), finding stronger PFAS-dyslipidemia associations among girls compared to boys.76,78 Additionally, one study investigated EMM by maternal race (White, Black, Hispanic, Asian) and found no consistent patterns for race/ethnicity after a false discovery rate (FDR) correction (q < .05).76,79 Gaston et al. concluded the overall evidence between PFAS and cardiometabolic outcomes in their review was inconclusive, but felt the association between PFAS and cardiometabolic outcomes merited continued investigation. 76
Another review by Rappazzo (2017) et al. focused on PFAS and general health outcomes in children and adolescents. 80 This systematic review found 16 studies specific to cardiometabolic outcomes (9 cohort studies and 7 cross-sectional), with studies conducted in Europe (N = 7; mainly Denmark N = 4), the United States (US; N = 5), and Taiwan (N = 4). 80 Rappazzo et al. observed inconclusive evidence across studies regarding markers of insulin resistance such as glucose77,81 and insulin.77,80-82 Additionally, this review also found limited and inconsistent evidence on measures of central adiposity,82-85 and only a single study on PFAS and BP, 86 which observed no association. 80 Rappazzo et al. reports the strongest evidence between PFAS and a cardiometabolic outcome in children and adolescents was for the PFAS-dyslipidemia association, citing five cross-sectional studies.77,80,87-90 Specifically, Rappazzo et al. details that the majority of included studies found an association between low-density lipoprotein cholesterol (LDL-C)87-90 and total cholesterol87-90 but inconsistent evidence for high-density lipoprotein cholesterol (HDL-C)81,87,88 and triglycerides.80,87-89 This review urges investigators to focus future studies on longitudinal assessments to establish temporality and mixture-based approaches as well as emphasizes the need for more cohesive outcome definitions. 80
Cohort Studies on PFAS and Cardiometabolic Outcomes
In our narrative review, we found 52 cohort studies on PFAS and our 4 cardiometabolic outcomes of interest (19 on insulin resistance/glucose dysregulation,17,91-108 19 on central adiposity,82-85,92,95,106,108-119 12 on dyslipidemia,92,95,108,116,118,131-136 and 17 on BP95,106,111,116,118,122,137-146; numbers do not add to 52 due to overlapping studies). Additionally, 3 well-conducted prospective case-control studies were noted as well.120-122 Details on included studies by MetS subcomponent are available in Supplemental File 1. Most of our studies were identified from previous reviews.38,39,51,75,76,80,123-126 However, some were located from a PubMed search on PFAS and each outcome, by reviewing the reference section of identified articles, and through peer recommendation. As no review articles on central adiposity were found, the following PubMed search strategy was employed: PFAS and (“BMI” OR “weight” OR “obese” OR “overweight” OR “length” OR “waist” OR “fat” OR “height” OR “adiposity” OR “anthropometric”). This search returned 280 studies, of which 15 (79% of included studies) were included in the review. Data was extracted by a data single extractor and all studies extracted were in English. We performed the last search on October 7, 2022.
Insulin Resistance and Glucose Dysregulation
Outcomes related to insulin resistance or glucose dysregulation include hyperinsulinemia, impaired fasting glucose, impaired glucose tolerance, and type 2 diabetes (T2D). Nineteen cohort studies examined the relationship between PFAS and insulin resistance or glucose dysregulation.17,91-108 Of these, 8 were in non-pregnant adults,17,91-97 8 were in pregnant adults,17,98-104 and 7 were in children or adolescents.17,93,105-108 Additionally, 2 well conducted prospective case-control studies in non-pregnant adults were also noted.120,121
In non-pregnant adults, 2 studies observed that higher PFOS and PFOA concentrations were associated with higher serum insulin levels, 95 higher insulin area under the curve (AUC), 17 and/or higher Homeostatic Model Assessment of Insulin Resistance (HOMA-IR). 95 In 621 overweight and obese participants in Boston, MA and Baton Rouge, LA (POUNDS Lost Study), higher PFNA was associated with higher cross-sectional blood plasma insulin levels; this study also found higher PFNA and PFDA were associated with a greater increase in blood plasma insulin over the course of 6 to 24 months. 95 However, a prospective nested case-control study by Donat-Vargas (2019a) reported that ΣPFAS, PFDA, and perfluoroundecanoic acid (PFUnDA) were inversely associated with HOMA-IR in 124 case-control pairs in a Northern Sweden. 120 Two other studies observed no association between PFAS and insulin, both evaluating PFOS and PFOA and one also evaluating PFHxS, PFNA, and PFDA.17,92 Similarly, the POUNDS Lost Study also observed higher PFNA was associated with higher glucose blood plasma levels 95 ; however, 3 other studies did not find a PFAS-glucose association with regards to fasting blood glucose levels, glucose AUC, or the Oral Glucose Tolerance Test (OGTT).17,92,93
Also in adults, the majority of studies assessing PFAS and T2D (4 of 6) observed that higher PFAS exposure was associated with greater T2D risk, with half of the studies observing an association with PFOA or n-PFOA specifically.91,94,96,97,120,121 For example, a 2022 study by Park et al. using data from the Study of Women’s Health Across the Nation (SWAN) found higher n-PFOA, PFHxS, Me-PFOA-AcOH, and
In children and adolescents, 7 studies on PFAS and insulin resistance or glucose dysregulation were found, with 4 observing an association17,105,106,108 and 3 not observing an association.92,93,107 Of those finding a relationship, associations were observed for the same 2 PFAS: PFOA (N = 3)105,106,108 or PFOS (N = 2).17,108 For example, a 2021 study by Li et al. found higher prenatal PFOA concentrations were related to higher insulin resistance as measured using HOMA-IR at age 12 years. 108 Furthermore, a 2022 study by Braun et al. explored modification of this association by physical activity (determined from a physical activity questionnaire) and found that prenatal PFOA was associated with worse HOMA-IR scores for those with low but not high physical activity scores. 106
Overall, the studies identified in this review provided inconsistent evidence for an association of PFAS with insulin resistance, and glucose dysregulation in children, adolescents, and adults.17,92,93,95,105-108,120 Furthermore, studies assessing gestational diabetes in pregnant people were also inconsistent.98-104 The strongest evidence for a PFAS-glucose/insulin association in adults was found was for some PFAS and T2D diabetes, with 4 of 6 studies observing an association.96,97 Still, the number of identified studies in non-pregnant adults looking at T2D is low and these studies were heterogenous with regards to population- and analysis-related factors. Additionally, 30% (3 of 10) of the studies assessing insulin resistance, glucose regulation, or T2D in adults were conducted in female-only populations, further contributing to the heterogeneity of these studies.17,92-97,120,121 Of the 7 adult studies in both men and women, 4 evaluated modification by sex, with 2 finding no association and 2 studies finding modification with PFAS-insulin/glucose associations among females but not males.17,92,93,120 This was consistent with studies in pregnant adults but inconsistent with studies in children and adolescents that generally observed no associations.17,93,100,105,107,108 However, these null results could be reflective of a small sample size to assess statistical interaction. More so, the 2 studies evaluating modification by physical activity and diet (1 in adults and 1 in children)91,106 found that both physical activity and/or diet may help to mitigate these associations. As such, future studies should consider evaluating physical activity and diet as a potential modifier.
Central Adiposity
For our review, 19 cohort studies on central adiposity were identified, with most studies in children and adolescents (14 studies in children/adolescents; 6 studies in adults).82-85,92,95,106,108-119 All but one study assessed central adiposity using waist circumference (WC), which has been found to be a good predictor of central adiposity and subsequent T2D diabetes and CVD.82-85,92,95,108-119,127 As such, we emphasize this outcome in the discussion below. Additionally, other central adiposity measurements utilized in these studies included waist-to height ratio,85,116 waist-to-hip ratio, 117 hip circumference 117 or girth, 109 visceral fat via DXA106,108 or CT 95 scan, trunk-, android-, or gynoid-fat percentage, 115 and subscapular to triceps skinfold thickness. 117
Six studies on PFAS and central adiposity in adults were identified.84,92,95,109-111 In 3 studies, higher concentrations of PFOS were associated with increased WC92,95,110 (the other 3 studies found no association84,109,111). Ding (2021a) et al. evaluated 10 PFAS in relation to WC in 1381 women (around 50 years old) in SWAN. 110 This study observed that women in the highest PFOS, Et-PFOSA-AcOH, and Me-PFOSA-AcOH tertiles had annual increases in WC of 12% (P = .002), 15% (P = .0006), and 10% (P = .02), respectively, compared to women in the lowest tertile. 110 Halldorsson (2012) et al. evaluated the association between prenatal PFAS concentrations and WC at 20 years of age using 665 mother–child pairs from the Danish National Birth Cohort. 84 This study observed that females born to women PFOA concentrations in the 4th quartile had a greater risk of having an elevated WC (>88 cm in females and >102 cm in males) 128 compared to those in the first quartile; no associations were observed for males. 84
Among 8 studies observing PFAS-WC association in children,82,92,112-115,117-119 half observed an inverse association.114,118,119 Bloom (2022) et al. found that higher prenatal blood plasma perfluoroundecanoic acid (PFUnDA) concentrations were associated with increased WC in 803 4- to 8-year-old multiethnic US children in the Fetal Growth Study. 112 Additionally, a 2021 study by Papadopoulou et al. observed a positive association between pre- and post-natal PFAS mixture and WC in 1101 mother–child pairs in a study of 6 multinational European studies. This study was inconsistent with the 2 other PFAS mixture studies,108,119 one finding no association 108 and the other finding an inverse association for boys and a positive association in girls. 119 Additionally, modification of the PFAS-central adiposity relationship by child sex was evaluated in 12 of the 14 studies, with 7 not observing modification,82,83,92,108,112,115,116 3 observing stronger effects in boys and no effects in girls,117-119 and 2 observing stronger effects in girls and no effects in boys.85,113
Overall, the association between PFAS and WC was inconsistent in adults, children, and adolescents.82,84,92,95,109-115,117-119 All 6 studies in adults that evaluated relations of PFOS and PFOA with central adiposity reported similar median concentrations of these compounds.84,92,95,109-111 As such, differences between studies on the PFOS- or PFOA-WC relationship in adults are likely not the result of differences in dose. On the other hand, studies in children appeared to differ by dose; however, no clear patterns in relation to median serum concentrations were observed. Furthermore, although the majority of studies in children did not observe modification by child sex, half (N = 6) had samples sizes under 500 total participants, with only 2 studies having sample sizes over 1000 children.82,85,92,108,113,115,117-119 As a result, many of these studies may be underpowered to detect subtle sex-specific differences. Therefore, null results in relation to EMM of the PFAS-central adiposity association in children should be interpreted with caution. Furthermore, only two studies from the same cohort evaluated modification by diet, finding no association.82,108 However, as both of these studies contained less than 225 mother-child pairs, more and larger longitudinal studies are needed to make inferences about whether diet modifies the PFAS-central adiposity relationship.
Dyslipidemia
Dyslipidemia refers to an imbalance in lipids such as triglycerides and lipoproteins. 129 Our review found 12 PFAS-dyslipidemia cohort studies, with 8 in adults92,95,130-135 and 5 in children or adolescents.92,108,116,118,136 Additionally, a prospective nested case-control study was noted as well. 122
The association of PFAS with dyslipidemia was fairly consistent in the adult literature, with most studies finding an association for triglycerides (N = 5/7),92,95,122,130,131,133,134 LDL-C (N = 4/5),95,130,131,133,134 HDL-C (N = 3/5),95,130,131,133,134 and hypercholesterolemia (N = 2/2),132,135 but not total cholesterol (N = 2/5)95,122,130,131,134 or hypertriglyceridemia (N = 0/1). 132 However, not all associations in these studies were in the expected directions. For example, a study by Donat-Vargas (2019b) et al. observed PFAS was inversely related to triglycerides in 187 participants in the Vasterbotten Intervention Programme (VIP) Study in Northern Sweden. 122 Similarly, 2 of 3 studies found a positive relationship between PFOA and HDL-C, with Liu (2018) et al. being the only study to observe an inverse relationship. 95 Additionally, a study by Dunder (2022) et al. observed associations between a 10-year change in 6 of 8 PFAS evaluated with changes in total cholesterol, triglycerides, LDL-C, and/or HDL-C in 404 70-year old participants from the Prospective Investigation of Vasculature in Uppsala Seniors (PIVUS) study. 130 For example, this study observed that decreases in perfluoroheptanoic acid (PFHpA), PFHxS, PFOA, and PFNA concentrations were associated with greater decreases in total cholesterol, triglyceride, and HDL-cholesterol levels. 130 These findings were similar to a study by Fitz-Simmons (2013) et al. that also evaluated within-person change in PFAS. 131 They found that declines in PFOA and PFOS concentrations were associated with decreases in LDL cholesterol over a 4.4 year period in 560 adults in the Ohio Valley using the C8 Short-Term Follow-up Study. 131 The change-in-change approach used by both studies is robust to confounding from fixed within-person factors, thus enhancing the strength of the association between some PFAS and dyslipidemia.
Only 5 cohort studies evaluated PFAS and dyslipidemia in children and adolescents.92,108,116,118,136 These studies were primarily in Europe (N = 4), with all studies mostly evaluating the same 4 PFAS (PFOS [N = 5], PFOA [N = 5], PFHxS [N = 4], and PFNA [N = 4]), and one study also assessing PFDA.92,108,116,118,136 Of these studies, 3 observed an association between PFAS and triglycerides, with 2 studies specifically finding a PFHxS-triglyceride association.92,108,116,118,136 The majority of studies assessing HDL-C and LDL-C observed an association (HDL-C: 3 of 4 studies; LDL-C: 2 of 3 studies), but a PFAS-total cholesterol association was only observed in 1 of 2 studies.92,108,116,118,136 Bloomberg (2021) et al. found that higher PFNA exposure at 5 years was associated with higher total cholesterol at age 9 in 490 mother–child pairs in the Faroe Islands. 136 This paper also observed several cross-sectional associations at age 9, including positive associations of several PFAS concentrations with higher total cholesterol, HDL-C, or LDL-C levels. 136 Additionally, Papadopoulou et al. used the HELIX cohort to assess a combination of pre- and post-natal PFAS mixture concentrations in 1101 mother-child pairs. 118 This study found PFAS was positively associated with HDL-C but negatively associated with LDL-C and triglycerides; these negative associations were the only negative associations observed in this literature for PFAS and dyslipidemia (including HDL-C associations). 118
The strongest evidence for an association between PFAS and a component of MetS in our review was found for dyslipidemia, specifically in relation to higher PFAS and higher LDL-C. However, all but one observed a positive association for PFAS and HDL-C, which is unexpected as one would hypothesize an inverse association (i.e. higher PFAS would lead to lower HDL-C). Reasons for this unexpected directionality are unclear and could be truly biologic but also may be reflective of confounding by positive behaviors associated with better health outcomes that also lead to increased PFAS exposure (examples could include drinking more water or eating more fish). Additionally, while all of these studies were conducted longitudinally, many of the observed associations were cross-sectional. Therefore, the inability to establish temporality for these cross-sectional associations must also be considered. Furthermore, only one study evaluated PFAS mixture on individual dyslipidemia outcomes, finding that the PFAS mixture was positively associated with HDL-C and negatively associated with LDL-C and triglycerides. 118 As such, the potential for joint effects should not be dismissed and future studies should consider mixture analyses in addition to assessing PFAS in single-pollutant models.
Blood Pressure
A total of 17 cohort studies have evaluated PFAS and BP.95,106,111,116,118,122,137-146 Of these, the majority (N = 9) were in pregnant people,138-146 4 were in adults,95,111,122,137 and 4 were in children.106,108,116,118
Of the 4 studies in adults, all observed associations between PFAS and BP were positive, with increasing exposure leading to an increasing outcome.95,111,122,137 All of these studies also observed some PFAS-BP association, with all studies evaluating systolic blood pressure (SBP; N = 3) and hypertension (N = 2) and most studies evaluating DBP (2 of 3 studies) finding a positive association.95,111,122,137 Additionally, all studies observed an association with PFOS specifically.95,111,122,137 For example, a 2022 study by Ding et al. found higher sm-PFOS and Et-PFOSA-AcOH were related to hypertension, higher SBP, and higher DBP in 1058 women in the SWAN study. 137 Furthermore, this study observed higher PFOS was associated with greater hypertension risk and higher SBP, higher n-PFOS was associated with greater hypertension risk, and higher Me-PFOSA-AcOH was associated with higher SBP and DBP. 137 Liu (2018) et al. was the only other study to observe a PFAS-BP association in a PFAS other than PFOS, finding higher PFNA was associated with higher DBP and SBP, and higher PFOS, PFOA, and PFHxS were associated with higher DBP in 621 participants in the POUNDS Lost study. 95
In pregnant people, the majority of PFAS-BP studies evaluated preeclampsia (N = 6 of 9 studies) and pregnancy-induced hypertension (N = 6 of 9 studies), finding inconsistent results.138-146 Regarding BP, PFAS was consistently associated with SBP and DBP, with PFOS-DBP associations and PFHxS-SBP associations observed in 3 of 4 studies.138,139,142,146 However, while all observed PFOS-DBP associations were positive, the directionality of the PFHxS-SBP associations were inconsistent, with the majority of studies observing an inverse relationship.138,139,142,146 A 2021 study by Birukov observed an inverse PFHxS-SBP association and a positive PFOS-DBP association but no associations for PFOA, PFNA, or PFDA in 1283 pregnant people in Southern Denmark. 138 Borghese (2021) et al. found higher PFHxS was weakly associated with higher odds of preeclampsia. 139 Furthermore, this study observed positive PFOA-SBP, PFOA-DBP, PFHxS-SBP, and PFOS-DBP associations. 139 Preston (2022) et al found positive associations between several PFAS and gestational hypertension, DBP, and SBP in 1558 pregnant people in Boston, MA. 142 Yang (2022) et al. observed positive associations between PFOS, PFDA, PFUnDA, and PFDoDA and risk of gestational hypertension and lower SBP. 146 Additionally, this study observed inverse associations between PFHxS and perfluorotridecanoic acid (PFTrDA) and SBP as well as PFHxS and DBP. 146
To date, only 4 cohort studies have evaluated PFAS-BP associations in children, all assessing the same 4 PFAS: PFOS, PFOA, PFNA, and PFHxS.106,116,118 Li et al. evaluated pre- and post-natal PFAS on the mean of the DBP and SBP z-score in 221 12-year old children in the HOME study finding no association. 108 Also using the HOME study, Braun (2022) et al. assessed the impact of prenatal PFAS on SBP in 166 12-year old children in the HOME study observing no association. 106 Similarly, Manzano-Salgado (2017) et al. evaluated the impact of prenatal PFAS on DBP and SBP in 1083 children at ages 5 and 7 years in Spain, also observing no associations. 116 Conversely, Papadopoulou et al. found that prenatal PFAS mixture was associated with children’s SBP but not DBP at ages 7–11 years in 1101 mother-child pairs in a multi-country European. 118
PFAS was consistently associated with both DBP and SBP in the only 4 studies assessing BP in non-pregnant adults, emphasizing a potential association between some PFAS and BP.138,139,142,146 Therefore, more studies are needed to evaluate this association. Additionally positive associations between PFAS and BP or hypertension risk were mostly consistent across studies, with PFOS-BP associations specifically noted in all studies in non-pregnant adults.95,111,122,137 However, inferences are largely limited by the number of studies in non-pregnant adults (with only 4 adult studies having been performed).95,111,137 As a result, more studies are needed assessing PFAS in adults. Furthermore, 2 of the 4 studies on PFAS and BP in adults were conducted in female-only populations.111,137 Therefore, future studies would ideally consider evaluating PFAS and BP in populations with both men and women. In pregnant people, most studies assessed preeclampsia and hypertension, finding inconsistent results with regards to an association.138-146 However, as preeclampsia and hypertension are fairly rare, the number of cases in each study were relatively small, thus, analyses may have been underpowered to detect an association.138-146 Therefore, larger prospective studies are needed, specifically ensuring large enough sample sizes of preeclampsia or hypertension cases. In children, as only 4 PFAS-BP studies were conducted, inferences should be made with care.106,116,118 Two of 4 studies did not observe any PFAS-BP associations106,116; however, all but Li et al. were conducted using only prenatal PFAS measurements.106,116,118 As such, future studies should additionally consider collecting information on postnatal PFAS concentrations to evaluate how these may related to MetS outcomes during childhood and adolescents.
Clinical Considerations
In response to the ubiquitous human PFAS exposure and concerns over the adverse health effects of PFAS, the National Academies of Science Engineering and Medicine (NESEM) convened a panel to develop clinical guidance for PFAS exposure, testing, and clinical follow-up.
147
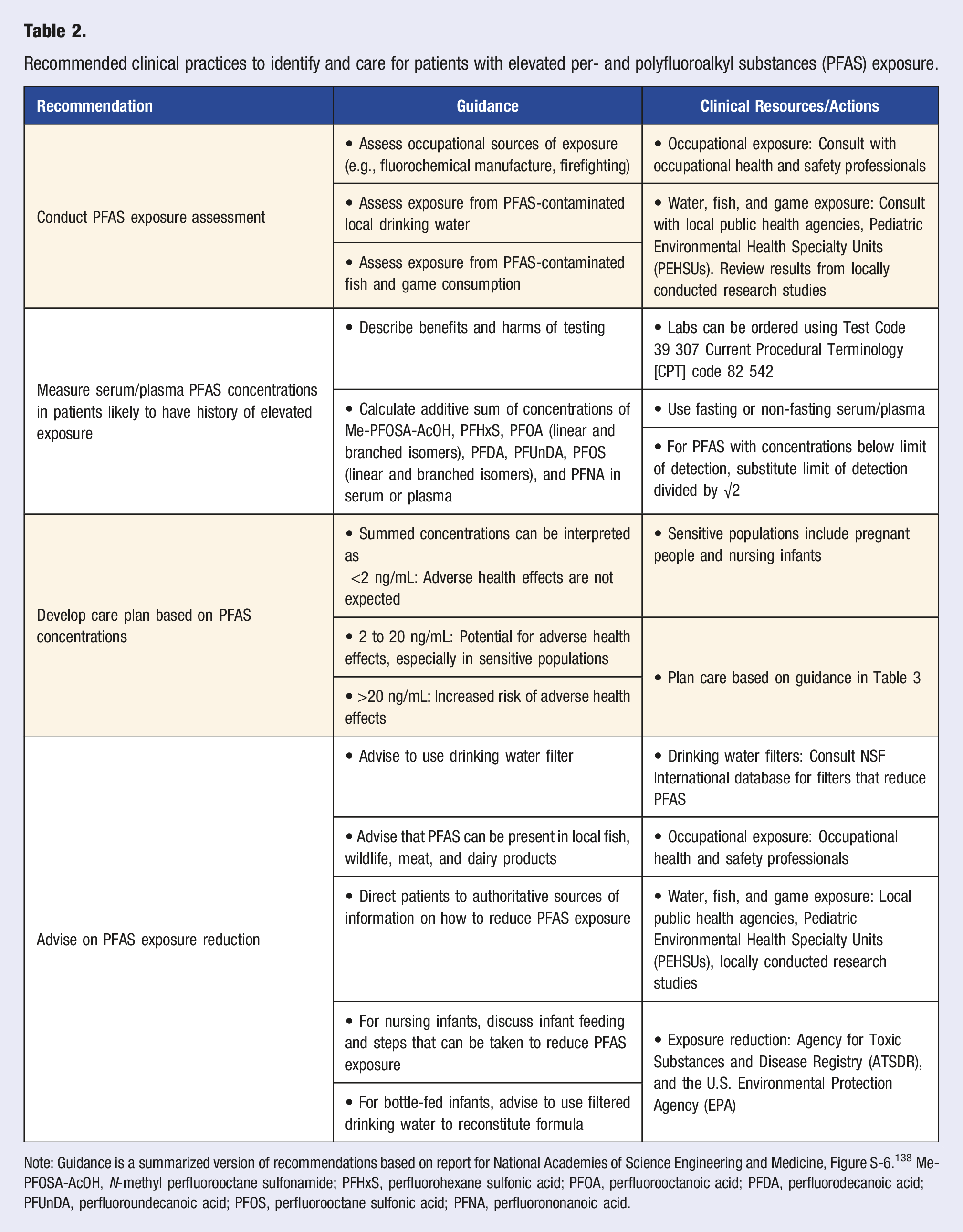
There is precedence for this as exposure controls and/or effects of exposure to some environmental agents are managed clinically. For example, the current recommendations for blood lead testing and lead poisoning treatment in pediatric populations are informed by epidemiological research of lead exposure and its health effects in children. Although there is uncertainty about the risks and benefits of integrating these guidelines into clinical practice, they provide a standard of care that could be adopted, particularly in communities where PFAS contamination is present. Below we highlight practices that the panel recommended for integration into clinical care and describe them in depth in Table 2: 1. Determine if patients are exposed to PFAS and what exposures they are interested in reducing (e.g., working with fluorochemicals, drinking water contamination, etc.). 2. Use shared, informed decision making, offer patients serum/plasma PFAS testing if the patient is likely to have a history of elevated exposure. 3. Use serum PFAS concentrations to guide additional clinical screening (Table 3). 4. Advise patients on ways to reduce PFAS exposure (e.g., drinking water filtration). Recommended clinical practices to identify and care for patients with elevated per- and polyfluoroalkyl substances (PFAS) exposure. Note: Guidance is a summarized version of recommendations based on report for National Academies of Science Engineering and Medicine, Figure S-6.
138
Me-PFOSA-AcOH, N-methyl perfluorooctane sulfonamide; PFHxS, perfluorohexane sulfonic acid; PFOA, perfluorooctanoic acid; PFDA, perfluorodecanoic acid; PFUnDA, perfluoroundecanoic acid; PFOS, perfluorooctane sulfonic acid; PFNA, perfluorononanoic acid. Interpretation of serum/plasma per- and polyfluoroalkyl substances (PFAS) concentrations and clinical guidance for follow-up with patients after PFAS testing.
a
aGuidance is a summarized version of recommendations based on report for National Academies of Science Engineering and Medicine, Figure S-6.
138
bAdditive sum of N-methyl perfluorooctane sulfonamide (Me-PFOSA-AcOH), perfluorohexane sulfonic acid (PFHxS), perfluorooctanoic acid (PFOA; linear and branched isomers), perfluorodecanoic acid (PFDA), perfluoroundecanoic acid (PFUnDA), perfluorooctane sulfonic acid (PFOS; linear and branched isomers), and perfluorononanoic acid (PFNA).
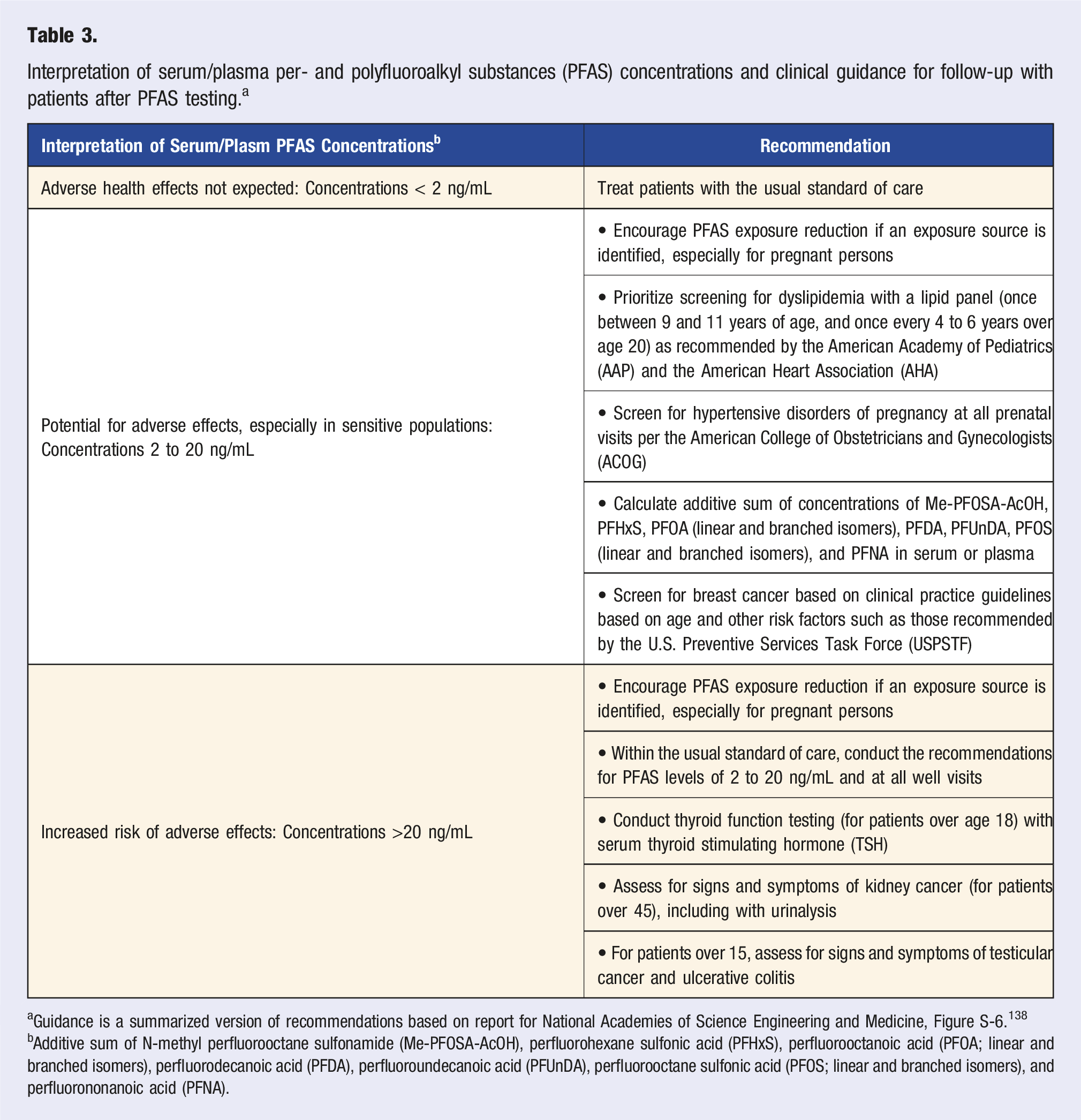
The National Academies panel developed disease screening guidelines based on serum/plasma PFAS concentrations (Table 3). 147 Simple additive sums of Me-PFOSA-AcOH, PFHxS, PFOA (linear and branched isomers), PFDA, PFUnDA, PFOS (linear and branched isomers), and PFNA concentrations can be used to categorize patients into three groups: adverse health effects not expected (<2 ng/mL), potential for adverse effects (especially in sensitive populations; 2–20 ng/mL), and increased risk of adverse effects (>20 ng/mL) for these assessments.
Clinical care and follow-up was developed for health outcomes that the NASEM committee deemed had sufficient, suggestive, or limited evidence of being caused by PFAS exposure. 147 In addition, the panel carefully considered that decisions on PFAS testing and related clinical care are made with uncertainty. As such, the development of these recommendations was made based on the principals of proportionality (risk-benefit assessment), justice, autonomy, feasibility, and adaptability. Ultimately, the panel selected six health outcomes that should be screened for within the standard of care or at all well visits (Table 3).
Clinicians can advise patients on ways to reduce exposure. The primary sources that should be considered include occupational exposures, consumption of PFAS-contaminated drinking water, and consumption of PFAS-contaminated fish, wildlife, meat, or dairy products. Additionally, nursing and bottle-fed infants may be a vulnerable to excess PFAS exposure due to consumption of PFAS-contaminated breastmilk or formula that has been reconstituted with PFAS-contaminated drinking water. 148 While there is still uncertainty about the risks of consuming PFAS-contaminated breastmilk, breastmilk is the optimal source of nutrition for infants with few exceptions. Thus, clinicians will need to engage in informed decision making with nursing mothers to reduce their PFAS exposure and continue breastfeeding. Additional research is needed to understand the routes and contributions of various sources of PFAS, particularly in vulnerable populations like pregnant people and infants.
Conclusions
In reviewing studies examining PFAS and MetS features, we observed evidence for an association between perfluorooctanoic acid (PFOA) and T2D in adults. Additionally, there was also evidence that some PFAS was associated with dyslipidemia in children and adults and possibly blood pressure in adults. While there was emerging evidence suggesting that the relation between PFAS and MetS was modified physical activity and sex, future research should clarify this as it might provide an avenue to prevent exposure-related health effects and identify susceptible subpopulations. Moreover, future studies should incorporate mixture-based analyses in addition to single pollutant assessments to better characterize the effects of aggregate PFAS exposure. Finally, clinicians can integrate PFAS exposure testing and clinical follow-up into their practice to identify patients who might be at increased risk of PFAS exposure-related disease.
Footnotes
Declaration Conflict of Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Braun was compensated for serving as an expert witness on behalf of plaintiffs in litigation related to PFAS-contaminated drinking water.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIEHS grants R01 ES032836 and R01 ES033252.
