Abstract
Objective. Well-being and quality of life can vary independently of disease. Instruments measuring well-being and quality of life are commonly used in neurology, but there has been little investigation into the extent in which they accurately measure wellbeing/quality of life or if they merely reflect a diseased state of an individual. Design. Systematic searches, thematic analysis and narrative synthesis were undertaken. Individual items from instruments represented in ≥ 5 publications were categorised independently, without prior training, by five neurologists and one well-being researcher, as relating to ‘disease-effect’ or ‘Well-being’ with a study-created instrument. Items were additionally categorised into well-being domains. Data sources. MEDLINE, EMBASE, EMCARE and PsycINFO from 1990 to 2020 were performed, across the 13 most prevalent neurological diseases. Results. 301 unique instruments were identified. Multiple sclerosis had most unique instruments at 92. SF-36 was used most, in 66 studies. 22 instruments appeared in ≥ 5 publications: 19/22 ‘well-being’ outcome instruments predominantly measured disease effect (Fleiss kappa = .60). Only 1/22 instruments was categorised unanimously as relating to well-being. Instruments predominantly measured mental, physical and activity domains, over social or spiritual. Conclusions. Most neurological well-being or quality-of-life instruments predominantly measure disease effect, rather than disease-independent well-being. Instruments differed widely in well-being domains examined.
Most neurological well-being instruments measure disease-effect, rather than disease-independent well-being, and differ widely in well-being domains examined
Introduction
‘Human well-being is an activity of the soul in accordance with virtue’,1,2 stated Aristotle in the 4th century before the common era (BCE). 3 Even today, the World Health Organization defines health as ‘A state of complete physical, mental and social-well-being, and not merely the absence of disease or infirmity’. 4 While the use of wellbeing scales in medical publications has burgeoned in recent decades, there is little consensus as to what they measure: scholarly discussion on the principles of instrument creation, related dimensions and guidance for use in clinical studies remains sparse. Despite the elegance of the WHO definition of ‘health’, the definition of ‘well-being’ is not agreed upon.1,4 The underlying principles that guide well-being instrument development are often disparate in nature or differ by discipline. Terminology is employed interchangeably and confusingly across related topics with, for example, ‘well-being’, ‘quality of life’ (QOL), and ‘life satisfaction’ all being used, at times synonymously.
Disease-specific quality-of-life instruments may be insensitive to detect well-being improvement if the disease itself is unchanged by the intervention. With neurological diseases ranked globally as the leading cause of disability adjusted life years (DALYs), 2 an intervention that could shift a proportion of the patient population from significantly disease-impacted to flourishing in the presence of – or in spite of – the same degree of ongoing disease would be a major public health advance. Instruments examining well-being independent of the effects of disease are more likely to be sensitive in this context.
A study by Bassi et al (2014) was among the first to examine disease and well-being simultaneously in a cohort of people with MS (PwMS). 5 Despite having high depression and lower quality-of-life scores on the Beck Depression Inventory and SF-36, their ratings on the Satisfaction with Life Scale (SWLS) – which is a disease-independent rating scale – paralleled those of caregivers and healthcare professionals. 5 Having disease-impacted disability did not necessarily lead them to a decrease in perceived well-being. The ‘disability paradox’ described by Albrecht and Devlieger (1999) similarly reports high levels of well-being in individuals despite significant limitations in daily functions. 6 Hence, disease and well-being are not merely differing ends of the same spectrum; they may co-exist and vary independently. For a given degree of disease, it is possible for patients to have vastly differing experiences of well-being. An item within a well-being instrument will typically relate to a specific ‘domain’ or ‘dimension’ of well-being. As defined by Linton et al (2016), domains can be split into mental, social, physical, spiritual, activities and functioning, and personal circumstances. 1 While Linton et al surveyed 99 self-reporting assessments to develop these domains, part of the exclusion criteria were instruments with a specific disease focus, as are common across neurological diseases (e.g. ‘QOL measurements in Epilepsy’ (QOLIE)). It is not clear, therefore, if the categories defined by Linton et al 1 are generalisable to disease-specific instruments.
Previous literature has rarely examined measurement of well-being in the context of disease: particularly whether specific items and overall instruments relate predominantly to well-being vs disease effect. An analysis of the most frequently used well-being measures specifically in neurology, and their domains of focus, has not been previously attempted. Understanding well-being domains would allow health professionals and researchers to choose an instrument to match their focus, the patient’s needs, or a particular study question.
Hypothesis
It is our hypothesis that the well-being and quality-of-life measures commonly employed within neurology are often disease-centric, and focus on the impact of disease, rather than on well-being, independent of disease.
Aims
The aims of the current review were to (1) identify instruments regarded as measuring well-being in neurological diseases, (2) identify if items in neurological instruments relate to disease effect or well-being, (3) describe aspects of an item that make it more likely to relate to disease effect or well-being, (4) describe the regularly sampled domains in the most common neurological instruments, and (5) describe the qualities that may be incorporated into an ideal instrument for measuring well-being in neurological disease. The wider objectives were to provide researchers and health professionals with a framework to identify an appropriate neurological well-being instrument for their needs, and to instruct development of future instruments in the measurement of well-being in the presence of disease.
Methods
Search Strategy and Selection Criteria
The study protocol was agreed prospectively by collaboration of five authors (BZ, RC, WDS, AV and SM). Database searches included MEDLINE, Embase, Emcare and PsychINFO in line with PRISMA guidelines. Similar strategies were used for Embase, Emcare and PsychINFO. Additional searching was undertaken in 2020 using search engines, cross-checked with subject matter experts. All databases were searched with an identical timestamp criterion, with a starting year of 1990 to February 2020. This ensured old and new instruments were captured. Restrictions were set to English language only.
Pre-agreed inclusion criteria were established. Studies were included if they had an instrument that focused on the most prevalent and highest incidence neurological diseases as defined by the Global Burden of Disease. 2 These diseases included headache, migraine, Alzheimer’s disease, dementia, epilepsy, Parkinson’s disease, multiple sclerosis, brain and other central nervous system cancers, motor neuron disease (MND), traumatic brain injury, stroke, encephalitis, meningitis, spinal cord injury and tetanus. Studies with instruments focussing on quality of life, wellness, well-being, health-related quality of life and life satisfaction in the above neurological diseases were included.
Studies of paediatric and adolescent patients were excluded during the full-text screening stage. Conference abstracts without subsequent publication, letters, opinion papers, and animal studies were excluded.
Ethics Approval
Ethics approval was not required for this study design.
Screening
Search results were compiled using Endnote software. Duplicate references were automatically detected and then individually assessed with subsequent removal if deemed appropriate. One reviewer extracted the relevant instruments for consensus review: each study was screened based on title and abstract, and all full-text articles assessed for inclusion.
Data Extraction and Synthesis
An abstraction datasheet was created using Microsoft Excel (Microsoft Office 365 Pro plus). Instrument characteristics (e.g. instrument name, disease type and authorship) were extracted from the eligible full-text publication.
Tool Creation
Input from neurologists within Australia and America was collaborated to agree prospectively upon 2 category definitions: ‘disease effect’ and ‘well-being’. Disease effect was defined as ‘relates to the disease progression, outcome and/or subsequent co morbidities. Can be in relation to the underlying disease, associated diseases, disease complications, or side effects of treatment’. Well-being was defined as ‘relates to the degree of well-being, not merely the absence of disease: in the spectrum of flourishing’. A third optional category ‘unequivocally well-being’ was used and defined as ‘item has no association with disease effect and excludes all criteria of ‘disease and its predictable associations’.
Domain categories and their subsequent definitions were adopted unchanged from Linton et al, who had described well-being domains across generic well-being instruments – excluding disease-specific instruments – and encouraged further research into instrument use in disease-specific contexts. Domain definitions from Linton et al were supplied unchanged to investigators and the items were categorised according to the definitions.
Instruments that appeared ≥ 5 times across all studies were analysed in this way – a frequency chosen in advance to reflect an established and validated instrument with peer-considered applicability – using the tool created through Microsoft Excel.
Instruments were obtained through online search strategies. Three instruments – EQ-5D-5L, PROMIS and SIP – were unable to be obtained and were therefore omitted. Further consultation with neurology sub-specialty experts was undertaken to ensure no commonly used instruments had been inadvertently missed and to reduce search bias: no additional instruments were identified. A total of 22 unique instruments emerged, and these were examined in detail. Items from all instruments were collated into Microsoft Excel, totalling 734 items. Each item on each instrument was compared with the category definitions by the 6 authors of the study, acting independently and blinded to results of the other authors. These 6–5 neurologists, one from America and 4 from Australia, and one well-being researcher – sorted each item into respective categories and one or more well-being domains.
All neurological diseases investigated – as defined by the Global Burden of Disease study – were represented in the analysis: one or more instruments that appeared in the top 5 most common instruments for that disease were included in the instrument tool for analysis.
Statistical Analysis
The qualitative analysis was undertaken using IBM SPSS Statistics (software for statistical analysis, version 26 [program]: IBM, Australia, 2019). The tool data was tabulated, and an inter-rater reliability was performed using Fleiss kappa for disease effect vs well-being effect. Mean averages were taken from domain results and then converted into ratios for analysis.
Results
Instrument Identification
The database search identified 1906 citations. With duplicates removed, there were a total of 1245 references. After title and abstract screening, 900 records were excluded, and the remaining 345 articles were assessed for eligibility with full-text review. Ultimately, 245 records were included in this review.5,7-250 Reasons for exclusion defined prospectively were categorised as (1) non-neurological focused disease, (2) not well-being or similar related term, (3) conference abstract without publication, (4) language other than English, and (5) adolescent or paediatric population. Search strategy following PRISMA guidelines is presented in Figure 1. Search strategy with excluded studies and reasoning as per PRISMA guidelines.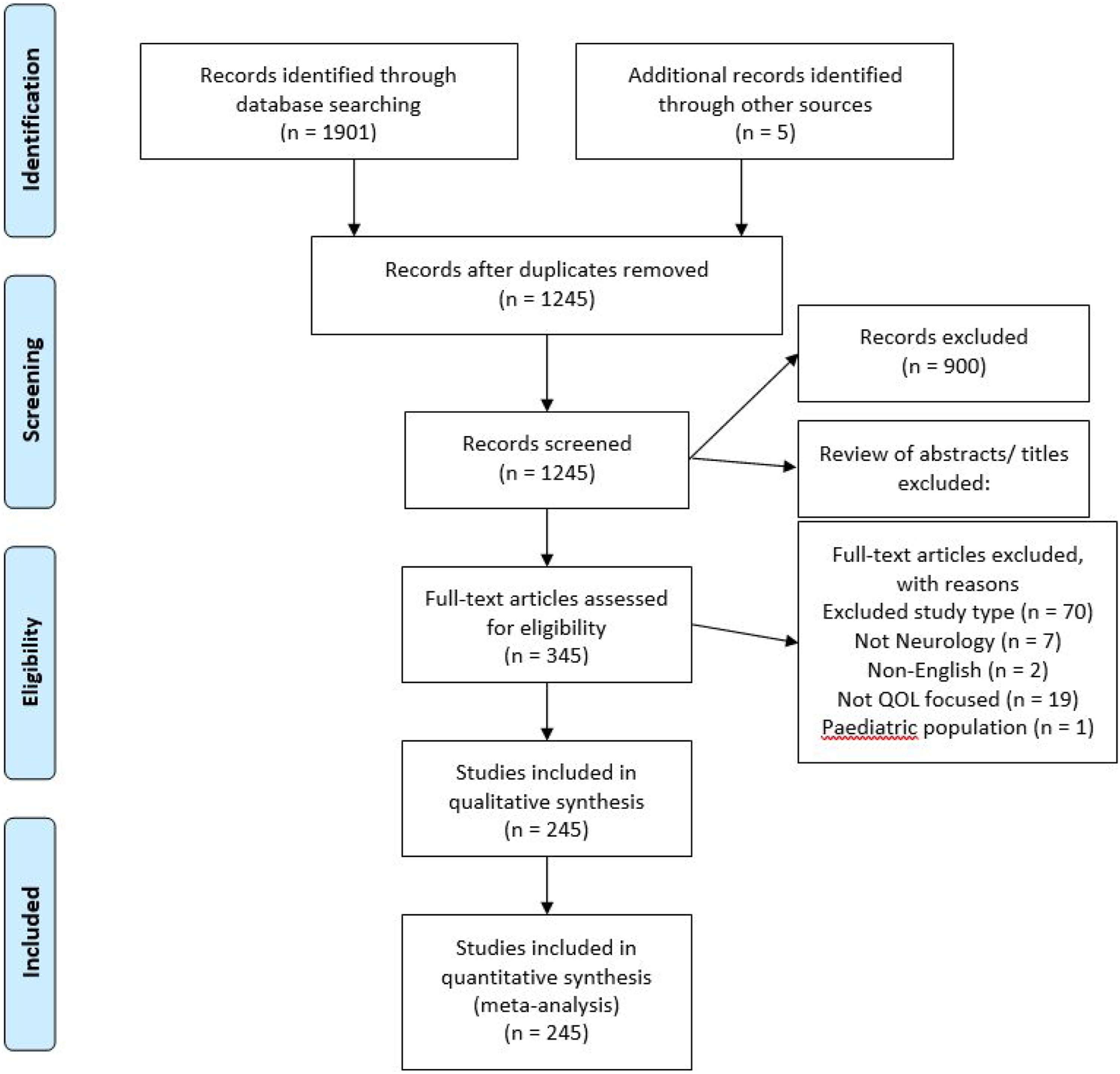
Critical Appraisal
Instrument usage was clearly stated in all publications. Data collection was performed appropriately in 98% of publications.
Instruments Across Diseases
There was a total of 301 unique instruments found across all studies (Figure 2). 92 (30.5%) instruments were found for use in multiple sclerosis, 69 (23%) for spinal cord injury/traumatic brain injury, 64 (21.3%) for stroke, 36 (12%) for epilepsy, 31 (10.3%) for Parkinson’s disease, 28 (9.3%) for dementia/Alzheimer’s disease, 25 (8.3%) for headache/migraine, 16 (5%) for neoplasms, and 13 (4.3%) for MND (Figure 2). Encephalitis, meningitis and tetanus had no unique well-being instruments found. Total number of unique instruments per neurological disease.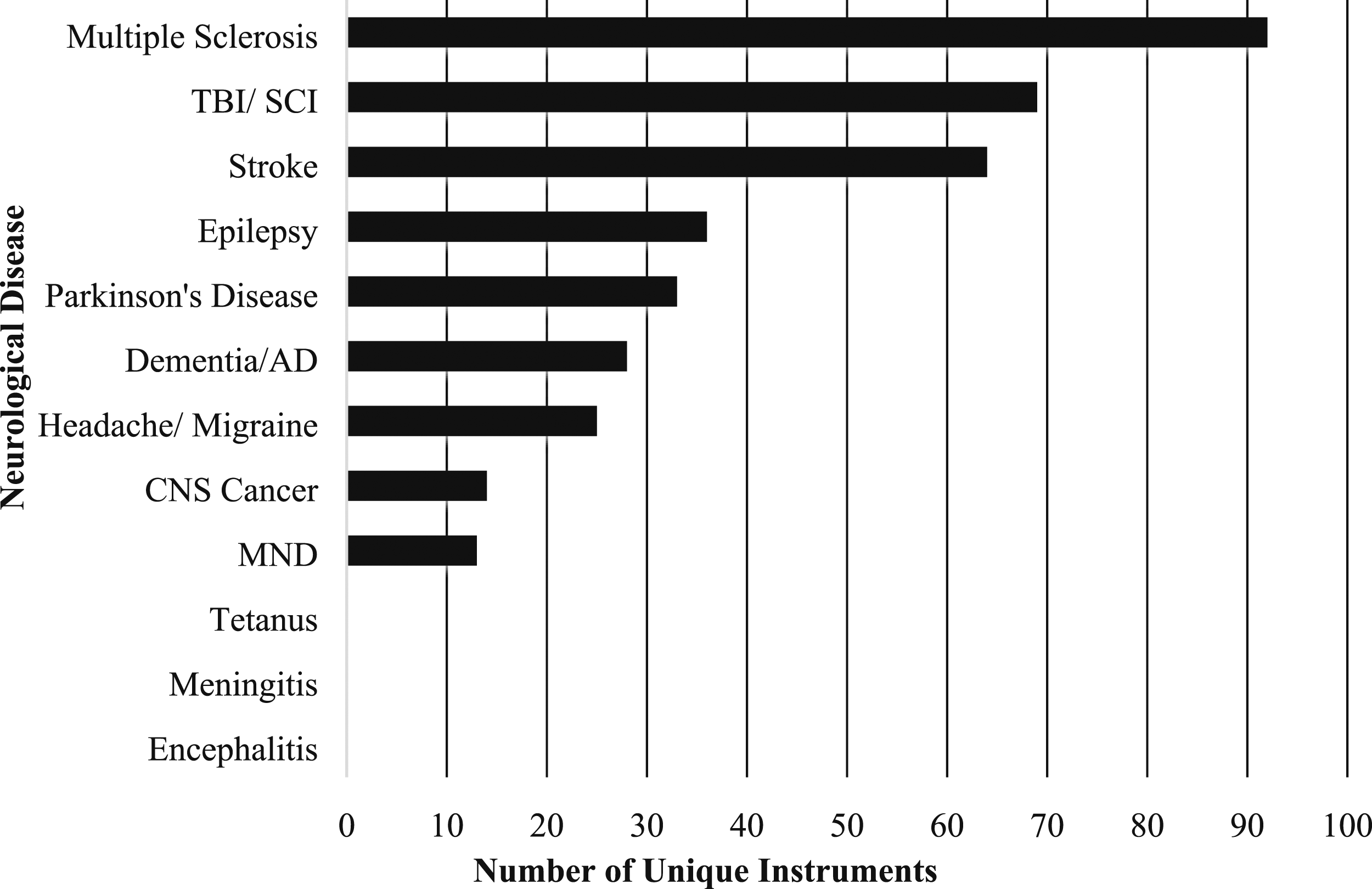
Assays Across Studies
Studies often used more than one assay to measure well-being; the 301 unique assays obtained were used a total of 692 times across the studies in various combinations. The Short-Form 36 (SF-36) was much the most consistently used assay across 66 studies (Figure 3). The EuroQol five-dimensional scale (EQ-5D), Parkinson’s Disease Questionnaire 39 (PDQ-39), Sickness Impact Profile (SIP), the Short-Form 12 (SF-12), the Health Utilities Index 3 (HUI3), and the Nottingham Health Profile (NHP) made up the next top 5 most frequent assays with 29, 24, 15, 13, 13 and 13 studies using these assays, respectively (Figure 3). Quantity of assay use across all studies.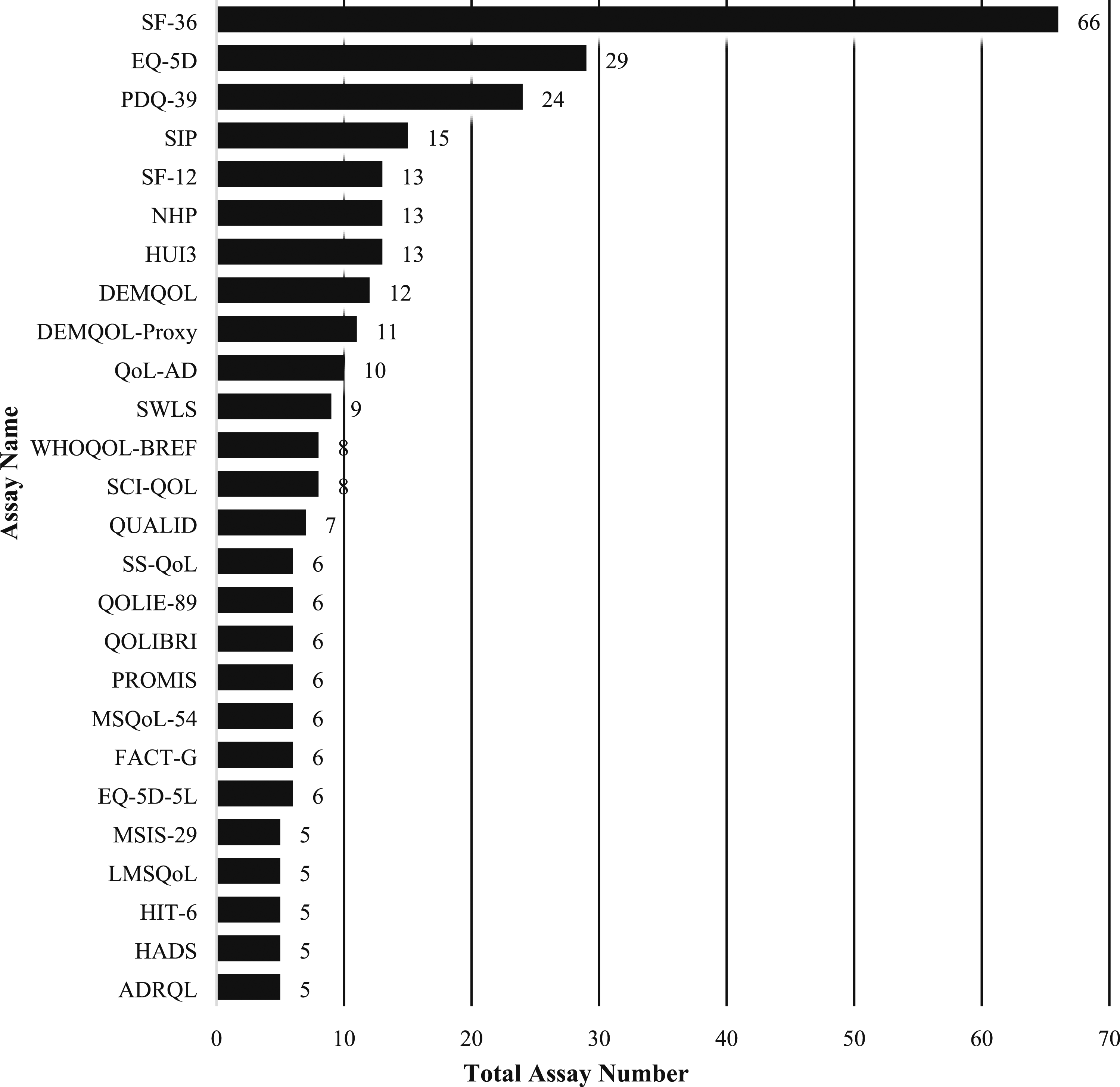
Instrument Focus Across Studies
There was a mixture of disease-specific and disease non-specific ‘generic’ instruments in the top 25 as given in Figure 4. Disease-specific instruments included spinal cord injury quality of life measurement system (SCI-QOL), stroke specific quality of life scale (SS-QoL), the quality of life in late-stage dementia (QUALID) scale, the quality of life scale in epilepsy 89 (QOLIE-89), quality of life after brain injury (QOLIBRI), Quality of Life-Alzheimer’s Disease (QoL-AD); Parkinson’s Disease Questionnaire 39 (PDQ-39), multiple sclerosis quality of life 54 (MSQOL-54), Multiple Sclerosis Impact Scale 29 (MSIS-29), Leeds multiple sclerosis quality of life scale (LMS-QOL), Headache Impact Test 6 (HIT-6), functional assessment of cancer therapy – general (FACT-G), DEMQOL-Proxy, DEMQOL and the Alzheimer’s Disease-Related Quality of Life (ADRQL). Disease non-specific ‘generic’ instruments included Satisfaction with Life Scale (SWLS), Sickness Impact Profile (SIP), Short-Form 36 (SF-36), Short-Form 12 (SF-12), Nottingham Health Profile (NHP), Health Utilities Index 3 (HUI3), Hospital Anxiety and Depression Scale (HADS), EQ-5D-5L and EQ-5D. Disease-specific instruments vs disease non-specific ‘generic’ instruments.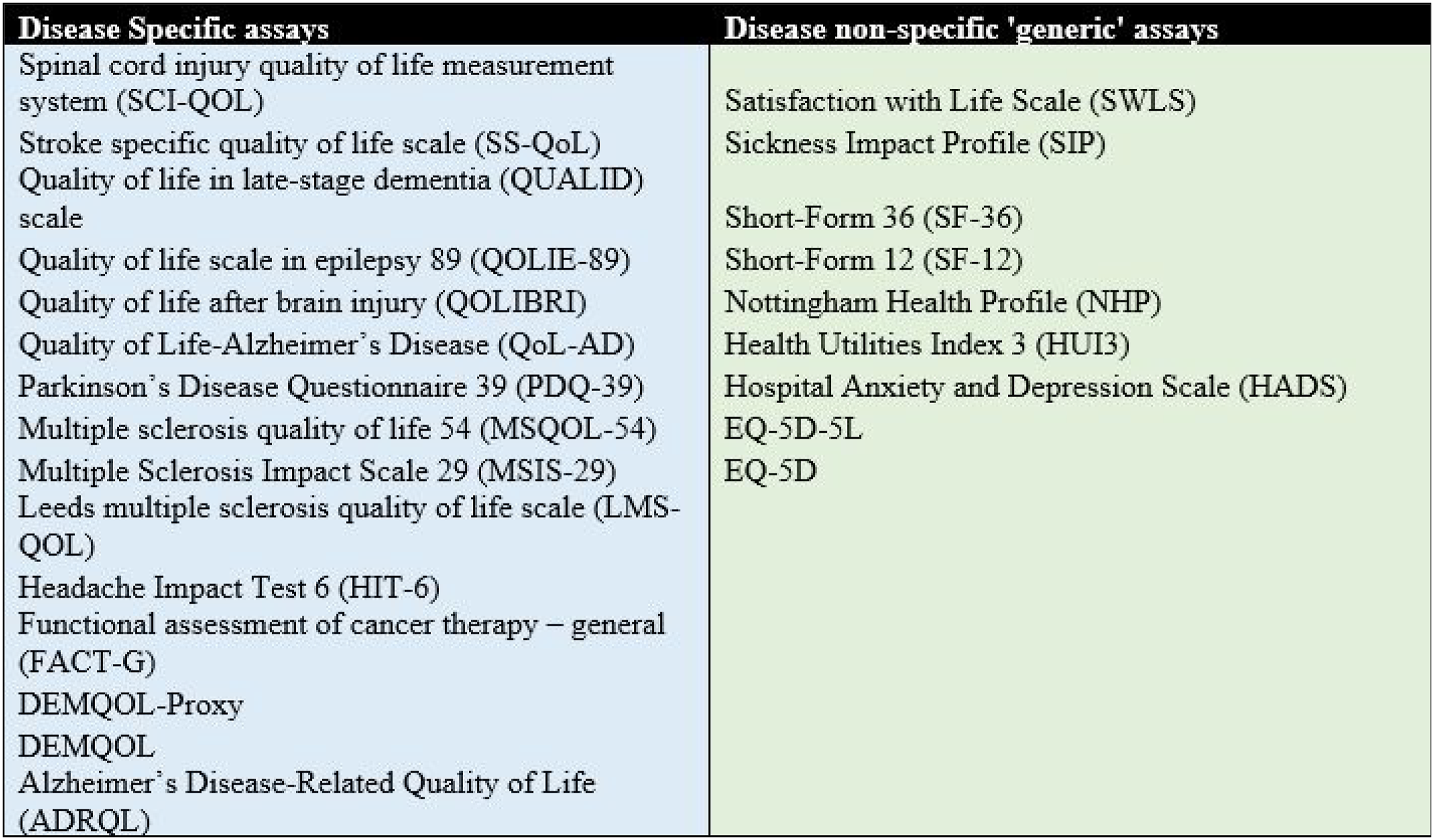
Well-Being vs Disease Effect
The mean percentage ratios of well-being vs disease-effect items as ascertained by application of the tool are given in Figure 5. Among the instruments used, only the SWLS was rated as measuring well-being effect unanimously across all items (k = 1). Conversely, the instruments MSIS-29, SS-QoL, HIT-6 and the PDQ-39 were rated with 100% disease effect measurement across all items. Of the remaining 17 instruments, the Quality of Life-Alzheimer’s Disease (QoL-AD) and the Spinal Cord Injury Quality of Life measurement system (SCI-QoL) measured > 50% well-being effect with a ratio of 74:26 and 61:39, respectively. Ratio of disease-effect vs well-being effect in the top 22 instruments by tool use.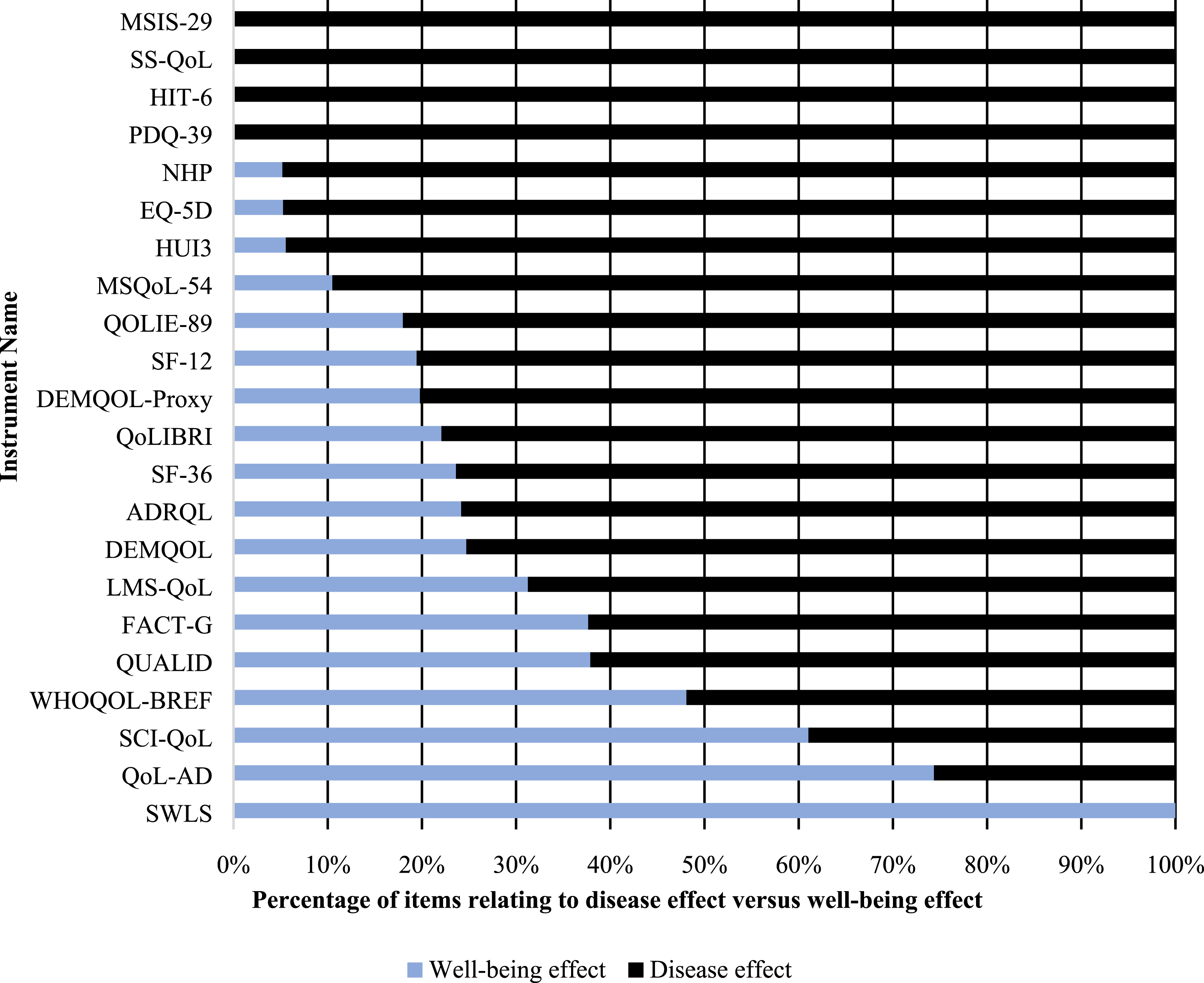
There was a total of 14/22 instruments that were aligned to a specific disease. The disease aligned instruments demonstrated a trend towards examining disease effect. A total of 12 (85.7%) of the 14 disease-specific instruments trended towards disease-effect measures. Of the 8-disease non-specific ‘generic’ instruments, 7 (87.5%) trended towards measuring disease effect (Figure 5).
The average Fleiss inter-rater reliability kappa across all instruments was .6, demonstrating moderate agreement between investigators. The instruments that had well-being effect greater than 40%, QoL-AD, SCI QoL and WHOQOL-BREF, had low kappa values at .005, .28 and .266, respectively.
Instrument Domains
Instruments had a large variance in the domains they measured (Figure 6). Instruments that primarily measured ‘mental’ domains included DEMQOL (48.2%), QUALID (45.7%), LMS-QoL (40.7%), ADRQL (43.3%), HDI (41.3%), DEMQOL-PROXY (36.2%), FACT-G (32.7%), QOLIE-89 (33.9%), QOLIBRI (32.4%), SWLS (26.7%), QoL-AD (23.1%) and SCI-QoL (21.7%) (Figure 6). No instruments had ‘social’ as their primary domain; however, 24.9% of items in ADRQL had a social domain focus. Instruments that focused predominantly on ‘physical’ domains included EQ-5D (38.2%), HIT-6 (34.8%), MSQoL-54 (31.9%), MSIS-29 (35%), SF-36 (33.4%), NHP (33.4%) and PDQ-39 (32.5%) (Figure 6). No instruments had ‘spiritual’ as their primary domain; however, 13.3% of items in the SWLS had a spiritual domain focus. Instruments that focused on ‘activities and functioning’ as their primary domain included HUI3 (36.8%), SF-12 (32.8%), MIDAS (37%), SS-QoL (32.2%), MSQ (31.5%) and WHOQOL-BREF (21.7%) (Figure 6). No instruments had ‘personal circumstances’ as their primary domain; however, 18.8% of items in SCI-QoL had a personal circumstances focus. Proportion of domains per instrument from tool use.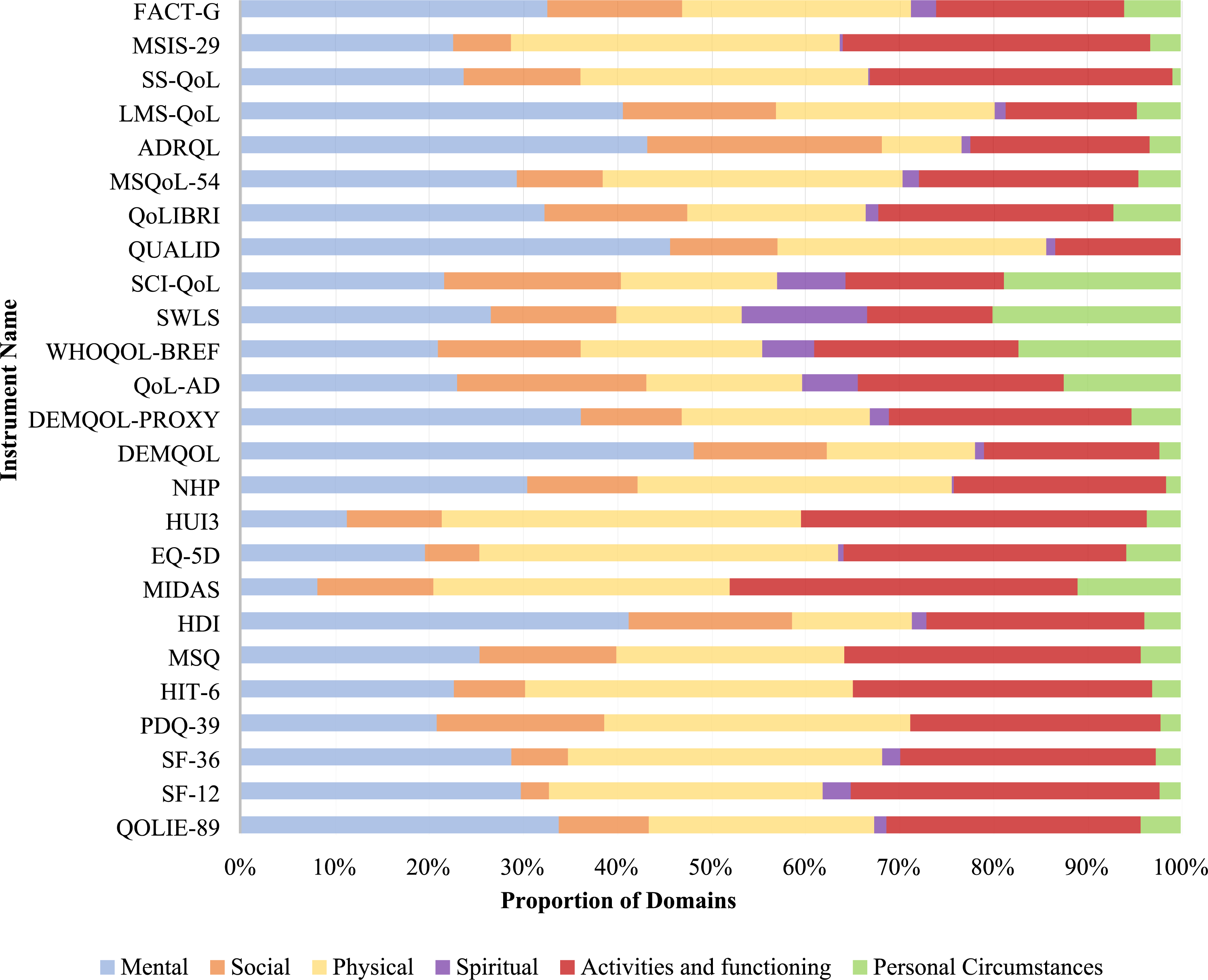
Discussion
In a census of quality-of-life and wellbeing instruments used across the range of neurological diseases, most available instruments predominantly measure disease effect, rather than disease-independent well-being: we have demonstrated this through development of a tool that can be used to address this question reproducibly and with good inter-rater agreement. Concerningly, this focus on disease effect rather than disease-independent wellbeing includes many measures which have become the standard ‘quality of life’ or ‘well-being’ instruments used in several neurological diseases, for example, PDQ-39, QOLIE-39 and SF-36. In addition, the specific well-being domains examined varied widely across the available instruments. This study provides a snapshot of the currently used instruments and their focus, and will help guide instrument selection and further instrument development in measuring well-being in neurological disease.
For a given degree of disease, patient’s experiences can be vastly different. Disease and perceived well-being exist on differing spectra and can vary seemingly independently. Nevertheless, when we set out to measure well-being in the setting of neurological diseases, often the ‘quality of life’ or ‘well-being’ instruments used primarily assess the degree or impact of disease, rather than disease-independent well-being.
The consequences of this are several. Firstly, an intervention focussing on well-being – that is potentially disease-independent – is unlikely to be meaningfully measured on a disease-specific instrument: a different instrument should be selected to measure this. Measuring flourishing independent of neurological disease becomes particularly important in studying wellbeing where disease-modifying treatments are few, such as in ALS/MND or stroke. Secondly, the converse is also important: if we have an intervention that is disease-modifying, in addition to measuring the disease impact, we may also want to measure flourishing independently, and use 2 appropriate scales.
Disease-Effect vs Well-Being
We defined disease effect as ‘relates to the underlying disease, associated diseases, disease complications, or side effects of treatment’. This was inclusive of mental health and its associations, as mental health effects are a common complication of neurological diseases. We adopted the WHO approach to health to define well-being as ‘relates to the degree of well-being, not merely the absence of disease: in the spectrum of flourishing’. 4
We obtained good agreement as to which items were related to disease, which to well-being. The points of difference are informative. There was unanimous consensus (k = 1) that MSIS-29, SS-QOL, HIT-6 and PDQ-39 all exclusively measured disease effect. All items included in PDQ-39 were preceded by the sentence: ‘Due to Parkinson’s disease, how often during the last month have you…’. This prefacing, and the inclusion of the disease name, was felt unanimously to place the focus on disease. HIT-6 adopts a similar approach and includes the word ‘headache’ in all their items, for example, ‘when you have headaches, how often is the pain severe?’ MSIS-29 and SS-QOL adopted similar approaches but used exclusively negative verbs in all items, for example, ‘how much have you been
As the instruments became weighted towards well-being, the consensus among investigators decreased, SWLS excluded QoL-AD, SCI QoL and WHOQOL-BREF had significant disagreement. There were 3 themes that likely lead to this disagreement; firstly, the mixture of positive and negative language used in items significantly impacted investigators categorisation, for example, the combination of using both ‘satisfied and ‘worries’ caused significant disagreement in the item ‘how satisfied are you with the amount of worries in your life?’ from SCI-QOL. Secondly, items that omitted introspective thought – and had a purely physical focus – had significant disagreement. When an item related purely to actions, for example, ‘how do you feel about your ability to do things like chores around the house?’ (QoL-AD), the consensus was low. Finally, questions relating to areas such as ‘sleep’, ‘fatigue’ and ‘sexual function’ had significant disagreement, as to whether mainly impacted by disease or reflecting well-being.
Domains of Well-Being
We also examined these frequently used instruments as to the specific domains of well-being. We now have data, not previously available, that catalogues the priorities of the currently available instruments. We hope this may be relevant when selecting an instrument for wellbeing that is most appropriate. For example, whether the intervention is primarily disease-specific (such as an antidepressant) or potentially disease-independent (such as yoga or mindfulness), an instrument enriched for items in the ‘mental’ well-being domain would be ideal in this setting.
We adopted unchanged domain definitions in our tool that were reported in the Linton et al 1 paper on well-being instruments, extracted through their qualitative analysis of disease-independent well-being instruments. The application of items to domains in this way was relatively straightforward and supports Linton et al’s contention that the extracted domains should be applicable in the setting of disease-specific instruments.
The domain results revealed wide variance among instruments as to which domains were more prominent, reflecting that instrument choice should be tailored. Social, spiritual and personal circumstances currently have no focused instrument. Given these are typically introspective domains, and the majority of instruments measure disease-effect over well-being effect, it is not surprising that these are the least represented domains. This likely also reflects the process of disease-specific instrument design, which often begins with a list of disease symptoms and builds an instrument around these and may then neglect these more conceptual or emotional aspects of wellbeing.
Qualities of an Ideal Instrument
The process has also allowed us to describe and highlight the potential and pitfalls in ideal instrument development for assessing well-being in the presence of neurological disease. We used an optional third categorisation in the tool, ‘unequivocally well-being’ and defined this as ‘item has no association with a disease effect and excludes all criteria of “disease and its predictable associations”’. This aimed to give insight into how an ideal well-being item might be structured. Among the unanimously agreed ‘unequivocally well-being’ items, language played the greatest role. If a single negative verb was included in the item, it either had little consensus or was unanimously omitted from the ‘unequivocally well-being’ category.
Overall, in designing an instrument, question design depends on the aim of measurement: if aiming to measure flourishing, then the use of neutral or positive language, avoidance of negative language and the avoidance of disease terminology is important, given pooled experience with application of our review tool. Given our results, the ideal instrument would include (1) a stated aim of examining wellbeing independent of the presence of disease and clearly-voiced definitions; (2) the omission of disease-specific items from the instrument, which requires a reduction in disease to register as improved, and thus diluting the capacity of the instrument to measure disease-independent well-being; (3) the use of neutral or positive language, and avoidance of negative language; (4) the use of a response scale that extends to positivity rather than simply to neutral, to allow an individual to rate items as being impactful and burdensome through to flourishing and absent of negative aspects if required; (5) the use of appropriate vocabulary in items to elicit introspective thought from a patient, allowing assessment of more subjective elements of well-being; and (6) an understanding of what the patient values in order to cater items to specific domains of importance in their subjective assessment of well-being or quality of life, even in the presence of disease.
SF-36 was the most commonly used assay in this census, featuring more than twice as commonly as the next most frequent. This likely reflects its generic nature, and – in this review – possibility to be used across multiple neurological diseases rather than being restricted to single disease states like some other assays, for example, QOLIE, PDQ-39 or SCI-QOL. SF-36 assesses components of life that are almost universal, that is, ‘bathing or dressing yourself’ and ‘did you feel tired?’ However, it does not measure very well against many of our recommendations for an ideal scale. The SF-36 aims to assess ‘views about your health’, rather than wellbeing independent of disease. Negative language is used throughout – ‘does your health now limit you in’, ‘had difficulty in’ and ‘have you had any of the following problems with…’ – which suggests at best neutrality rather than extending into realms of positivity. Relatively few items allow the patient to assess their performance in domains of most importance to them, rather than those nominated by the instrument. Such factors likely explain the relatively poor assessment by our raters in measuring well-being. Whilst this study did not seek to explore why SF-36 is in such widespread use in neurology, it does highlight concerns that an assay that is commonly used and popularly understood to be measuring wellbeing across neurological disease is heavily skewed towards measuring disease effect.
SWLS was the only assay to unanimously measure well-being effect. We believe it aligns with all the above recommendations. In particular, with reference to our recommendations, the SWLS (1) states the intended purpose as being ‘designed to measure global cognitive judgement of one’s life satisfaction’; (2) does not include disease-specific items; (3) uses neutral or positive language – ‘satisfied’ and ‘the conditions of my life are excellent’ – and avoids negative terminology; (4) uses a scale of response extending into positivity ratings ‘strongly agree’, ‘agree’ and ‘slightly agree’; (5) allows introspective subjective thought – ‘so far I have gotten the important things I want in life’; and (6) by being general in questioning, allows an individual to consider their most important domains and answer accordingly. Perhaps the shortcoming might be that – as such a brief scale – it does not allow the clinician an understanding of the particular areas of importance to the patient or, particularly if poorly rated, any understanding of specific concerns of the patient that might be improved. Thus, SWLS gives an excellent assessment of global wellbeing, and could potentially be improved by extension to yield more detailed information around which domains the patient holds as important, and how these are rated.
Strengths and Limitations of the Study Design
This was the first study to analyse neurology-specific QOL and wellbeing disease instruments and their subsequent items. This study also included disease non-specific instruments in an attempt to capture how well-being is currently measured within neurological diseases. The number of reviewers was high, at 6, with moderate agreement achieved between all assessors (k = .6). Criteria for analysis were defined by consensus and prospectively, rigorously and transparently, to allow for future replication.
There were several limitations to the study. A self-designed tool was used to measure well-being vs disease effect. Three instruments were excluded from analysis in the tool: EQ-5D-5L, PROMIS and SIP. Significant attempts were made to find these 3 instruments, without success: this seems unlikely to bias the outcome, and rather argues that these instruments are not in common usage and therefore not entirely relevant to the study outcome. Sorting an item from an instrument into disease effect vs well-being effect required a background knowledge of the disease condition, in order to determine if the downstream consequences of that disease might relate to the disease itself, or stand alone in the measurement of well-being. While 5 of the 6 investigators were neurologists with significant background knowledge on all conditions, one was not. This, however, did not skew the agreement consensus. Whilst this study did not investigate assays used in other fields of medicine, we suspect findings may be similar. It is likely that the generic – disease non-specific – assays would be assessed identically: this would be a rich topic for further research.
Conclusion
Many ‘wellbeing’ or ‘quality of life’ instruments are employed across the breadth of neurological diseases. Most neurological well-being instruments measure disease-effect, rather than disease-independent well-being, and differ widely in well-being domains examined. This recognition is of particular importance in gauging contribution of interventions that may improve perceived wellbeing in spite of minimal or no disease impact. Attention to overall measurement aims and specific domains of interest should guide instrument selection/development in measuring well-being in neurological disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
