Abstract
This review evaluates physical activity as a candidate for an adjunct treatment, in conjunction with antiretroviral therapy (ART), for people living with HIV (PLWH). Evidence is summarized that chronic, non-resolving inflammation (a principal feature of immune system dysfunction) and a dysfunctional state of the gut environment are key factors in HIV infection that persist despite treatment with ART. In addition, evidence is summarized that regular physical activity may restore normal function of both the immune system and the gut environment and may thereby ameliorate symptoms and non-resolving inflammation-associated comorbidities that burden PLWH. Physicians who care for PLWH could thus consider incorporating physical activity into treatment plans to complement ART. It is also discussed that different types of physical activity can have different effects on the gut environment and immune function, and that future research should establish more specific criteria for the design of exercise regimens tailored to PLWH.
“Incorporating physical activity into treatment plans may ameliorate lingering effects of HIV”
Introduction
Current treatment of infection with human immunodeficiency virus (HIV) focuses on countering the destruction of human immune cells and associated weakening of immune defenses. 1 Such intervention aims to prevent progression to stage 3 HIV, or acquired immunodeficiency syndrome (AIDS).2-4 At the same time, people living with HIV (PLWH) exhibit systemic, low-grade immune system activation, a hallmark of immune system dysfunction, that persists even when viral replication is effectively suppressed 5 by antiretroviral therapy (ART). This prolonged immune system activation is commonly referred to as chronic inflammation, but the term non-resolving inflammation has recently been introduced to emphasize that this persistent immune response primarily constitutes a failure to terminate pro-inflammatory signaling.6,7 Such non-resolving inflammation is a root cause of multiple diseases and disorders that can occur as comorbidities in PLWH.8-13
This review presents an overview of HIV effects that are successfully addressed by ART, as well as effects that persist in ART-treated PLWH. These lingering effects are aspects of immune system dysfunction, and can be linked to a persistent disruption of the gut environment. Since ART-treated PLWH experience such dysfunction, it is necessary and relevant in modern HIV treatment to understand how to restore a functional immune system and gut environment. Because dysfunction of these systems is implicated in chronic illnesses that disproportionately burden PLWH, ameliorating non-resolving inflammation should be a priority for physicians aiming to improve the quality of life of their patients with HIV.
Here, we review the effect of physical activity on immune system function and the gut environment. Attention is given to various types of physical activity and their apparent different impacts on the gut environment. Based on review of the available evidence, a proposal is made that certain types of physical activity may be candidates for adjunct treatment, complementary to ART, that may restitute the gut environment and restore normal immune system function (eliminate non-resolving inflammation) in PLWH. We propose that regular, voluntary (interest-based) physical activity with adequate periods of recovery is most likely to have a restorative effect on the gut environment and immune system function and is therefore an attractive candidate for an additional treatment complementary to ART in PLWH. Incorporating physical activity into treatment plans may ameliorate lingering effects of HIV that are not addressed by ART and enhance the quality of life of PLWH.
HIV Effects and Antiretroviral Therapy
Viruses like HIV interact with the immune system in complex ways; they use components of the immune system for their own replication, and at the same time precipitate a dysfunctional state of the immune system as a whole. The retrovirus HIV enters immune cells (especially CD4+ T cells) and employs their molecular vehicles for its own replication. 14 To do this, HIV activates gene regulators that trigger replication, such as the transcription factor NF-kB.15,16 The latter is a key regulator of the immune response and orchestrates inflammatory signaling.17,18 Constitutive, low-grade activation of NF-kB is associated with non-resolving inflammation in HIV-infected individuals.15,19 Furthermore, the take-over of CD4+ T cells by HIV eventually results in destruction of immune cells via programmed cell death—mainly as triggered by the human host’s immune system in an attempt to eliminate the virus. 5 Doitsh and Greene state that, during HIV infection, “most cells are not dying because of a toxic action of products encoded by HIV. Rather, death occurs as a consequence of a powerful defensive innate immune response launched by the host against the virus leading to a cellular form of suicide rather than virological murder.” 5 This attack by the immune system reverberates system-wide as non-resolving inflammation. The attack on immune cells involves reactive oxygen species (ROS) that function in the programmed cell death of CD4+ T cells 20 and also serve as signals that trigger a system-wide, snow-balling, and continuous mobilization of the immune system, 21 that is, non-resolving inflammation. This non-resolving inflammation involves systemic disruption of redox homeostasis (balance between oxidants and antioxidants) and the gut environment (see next section), and plays a key role in cardiovascular disease, obesity, GI distress, cancer, mental illness and many other conditions.22-27 The recognition of the role of non-resolving inflammation as a root cause for disease has been called “one of the most important scientific discoveries in health research in recent years.”28,29 Notably, PLWH exhibit a higher incidence of many of these and other comorbidities, which emphasizes the critical importance of addressing non-resolving inflammation in this population.
Another key component of the human immune system that interacts with HIV is the interferon system that detects pathogens and triggers defenses. The interferon system exhibits low activity in CD4+ cells 30 and is further suppressed by HIV infection. 31 Such impaired interferon activation in CD4+ cells allows sustained viral replication in these cells. Subsequent activation of interferon in other cells—once HIV infection is established—fails to eradicate the virus and may even promote non-resolving inflammation and further deterioration of host health. 30 As stated above, all aspects of immune-system activation involve ROS production that has the potential to snow-ball into non-resolving inflammation under exacerbating conditions, such as a dysfunctional gut environment (see section below). However, additional research is needed to elucidate the complex interactions between HIV and the interferon system. 32
Current HIV treatment based on ART arrests the replication cycle of HIV and has been shown to increase immune cell numbers in most PLWH.33-35 However, some PLWH are “immunological nonresponders” who do respond to ART with cessation of viral replication, but fail to exhibit restoration of CD4+ cell counts.36-38 More commonly yet, PLWH who receive regular ART treatment that halts viral replication typically show varying levels of persistent non-resolving inflammation.8,39-41 Addressing immune-system dysfunction in the context of HIV infection is thus critical even in the era of ART.
HIV and the Gut-Immune Link
Additional mechanistic insight into the repercussions of non-resolving inflammation induced by HIV focuses on the gastrointestinal tract and the gut microbiome.42-46 Figure 1 depicts the gut environment in its functional state without HIV infection (Figure 1A) as well as in its dysfunctional state during chronic HIV infection (Figure 1B). Schematic depiction of a healthy gut environment 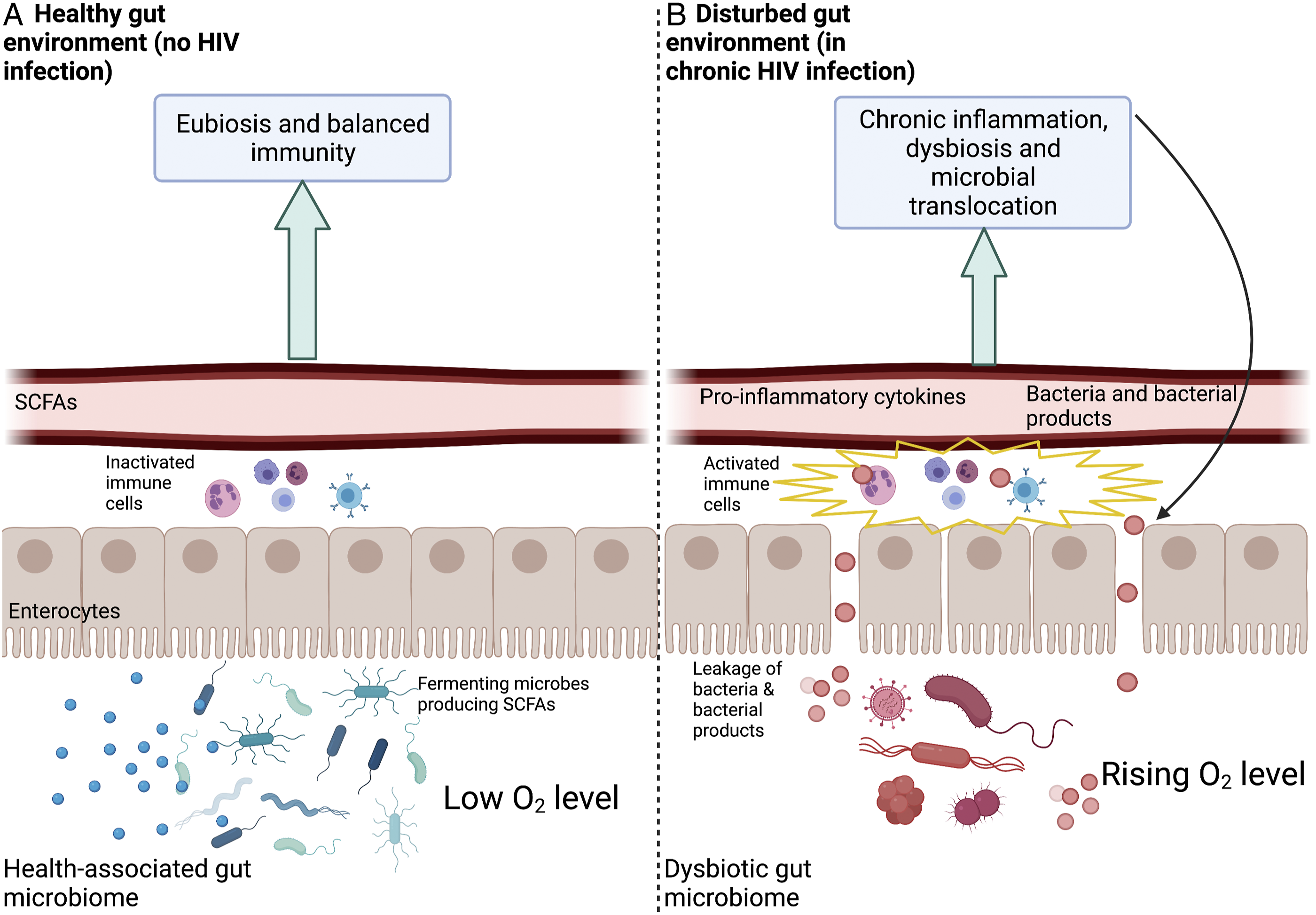
The Gut-Immune Link in Health
Figure 1A depicts a functional, health-promoting (eubiotic) gut microbiome characterized by prominent presence of anaerobic, fermenting bacteria that thrive in low-oxygen environments. 47 These microbes produce short-chain fatty acids (SCFAs) that serve as an energy source for the cells (enterocytes) lining the gut48-50 (for details, see below) and can also diffuse into the bloodstream.51,52 Locally, these SCFAs have essential roles in supporting gut-barrier integrity.53,54 For example, they act as gene activators for the production of proteins that maintain gut-barrier integrity.54-56 An example of a key SCFA is butyrate (produced by fermenting gut microbes) that increases expression of the tight-junction protein Claudin-1 involved in supporting gut-barrier integrity. 57 In addition, butyrate also represses a protein (Claudin-2) 58 that has been suggested to increase barrier permeability. 59 Gut-barrier integrity supports beneficial gut microbes and these gut microbes, in turn, maintain gut-barrier integrity. 47
A healthy gut is characterized by a steep oxygen gradient between the (hypoxic) gut lumen and the distal zone of the intestinal epithelium.60,61 The enterocytes of the intestinal epithelium require oxygen for their oxidative metabolism that supports their roles in barrier formation,
55
nutrient absorption
62
and immunity.61,63,64 Enterocytes use butyrate as the fuel they burn with oxygen to produce ATP for these various functions.
61
Therefore, butyrate produced by fermenting gut microbes stimulates oxygen uptake by enterocytes and helps maintain a low oxygen environment within the gut.55,65 By requiring oxygen for their own metabolism, enterocytes thus contribute to maintaining hypoxic concentrations within the gut lumen,
61
which supports anaerobic, SCFA-producing fermenting microbes.60,66 In addition to directly supporting gut-barrier integrity, SCFAs play a role in dampening the activity of NF-kB and the inflammatory response.67-71 In the state where gut microbial eubiosis and gut-barrier integrity are maintained, the immune cells associated with the gut lining are not activated to induce systemic effects72,73 (Figure 1A). Any condition that interferes with enterocyte function (eg, as seen during HIV infection) triggers production of messengers that initiate a pro-inflammatory cascade involving pro-inflammatory cytokines.74,75 Such adverse conditions include an insufficient supply of butyrate (as fuel) for enterocytes or of oxygen (to burn butyrate) as can be the case during exhaustive exercise (see Figure 2B in the next major section below). The effects of a regimen of regular moderate physical activity 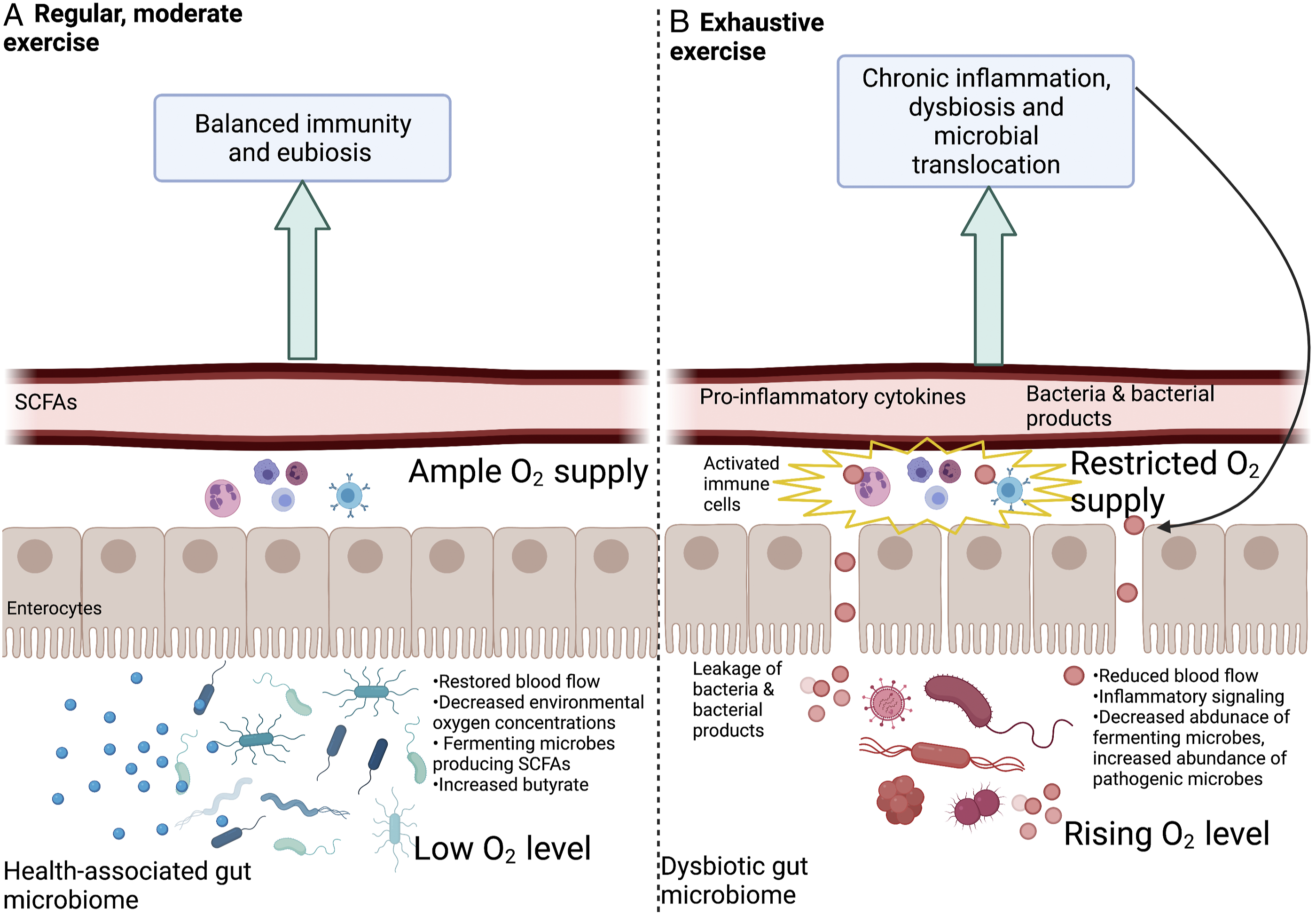
The Gut-Immune Link as Affected by HIV
Figure 1B illustrates a feed-forward loop of loss of gut-barrier integrity and non-resolving inflammatory responses in PLWH. Early HIV infection is characterized by widespread destruction of CD4+ T cells present in gut mucosa tissue. While ART causes some recovery of these cells, this recovery is not always complete and mucosa impairment may persist after individuals receive treatment.76-78 Disruption of T cells in the gut mucosa results in impaired gut-barrier integrity, which allows leakage of gut microbes and microbial products into the bloodstream (Figure 1B). Specifically, lipopolysaccharide (LPS), a cell surface component of gram-negative bacteria, is often identified as a marker of such microbial translocation and increased levels of LPS are observed during chronic HIV infection.79,80 Translocation of LPS activates the immune cells associated with the gut lining (Figure 2B) in a process mediated, for example, by NF-kB.81,82 Continuous activation of NF-kB contributes to a cascade of non-resolving immune system activation and may explain the persistent inflammatory state observed in PLWH.83-85
Furthermore, the disrupted gut barrier in chronic HIV infection (Figure 1B) results in increased oxygen concentrations within the gut, which shifts the gut microbiome to a disrupted state (dysbiosis) with specific losses in anaerobic fermenters and SCFA production. Increased concentrations of oxygen in the gut as a result of gut-barrier impairment also favor increases in gut bacteria that are facultative anaerobes capable of switching to aerobic (oxygen-dependent) cellular respiration and fast growth.86-88 These fast-growing facultative anaerobes can become pathogenic when undergoing such rapid growth.26,88,89 In addition to accelerating the cycle of leaky gut and non-resolving inflammation in PLWH shown in Figure 1B, this shift in microbiome composition is associated with GI distress and other symptoms.86,90
Since non-resolving inflammation and a dysfunctional gut microbiome are implicated in several conditions that disproportionately burden both untreated and treated PLWH, including cardiovascular disease, chronic kidney disease, cancer, and depression, restoration of the gut environment and normal immune function must be a priority for improving the quality of life of PLWH. The next section examines the potential of regular, moderate physical activity, used as a therapy complementary to ART, to reduce non-resolving inflammation and restore gut barrier integrity.
Physical Activity and the Gut-Immune Link
Figure 2 summarizes effects of physical activity on the gut microbiome, gut-barrier integrity, and inflammation that have emerged from recent studies in various human populations (Figures 2A and 2B).
Intensity, duration, and frequency of exercise, as well as age and training status of individual participants, can all influence the effect of exercise on health.91-95 More research is needed to elucidate the threshold of the transition between beneficial and detrimental exercise. A key consideration may be the effect of physical activity on blood flow to the gut, and the fact that acclimation (habituation) to a regular exercise regimen can restore this blood supply.
Effect of Regular Physical Activity With Replete Blood Flow to the Gut
It is well established that regular physical activity can reduce inflammation in various populations.96-98 In principle, physical activity has the potential to divert blood flow from the gut to the working muscles. 99 However, during the establishment of a regular regimen of physical activity, a process of acclimation (habituation) takes place that restores blood supply to the gut during physical activity. 75 Such regular physical activity is associated with fully functional enterocytes and a high abundance of fermenting, butyrate-producing microbes75,100-102 (Figure 2A). While some gut microbes produce butyrate from sugars derived from fiber or complex sugars,103,104 other microbes convert lactate produced by working muscles to SCFAs like butyrate.105,106 The butyrate produced serves to strengthen the gut barrier through the above-described support for enterocytes and, for example, stimulated expression of proteins involved in barrier tightening and downregulation of proteins involved in barrier loosening. A strengthened barrier and associated microbiome eubiosis decrease leakage of microbial products into the bloodstream, and decrease activation of immune system regulators such as NF-kB and the resulting non-resolving inflammation107-113 (Figure 2A). Suppression of NF-kB by physical activity was associated with reduced muscle loss in a mouse model 114 and this effect should also be assessed in humans. Exercise-associated decreases in muscle loss could offer important relief for PLWH since PLWH are often burdened by loss of muscle mass and function. 115
Effect of Physical Activity Associated With Continuous Low Blood Flow to the Gut
The type of exercise that increases the risk for non-resolving inflammation may be identified by its effect on the gut—as exercise that continuously exceeds the capacity for acclimatory restoration of blood flow to the gut at a level sufficient to prevent hypoxia in enterocytes. As far as impacts on the gut environment are concerned, a regimen of continuously exhaustive exercise has similar effects to those seen during the initial transition phase from inactivity to an established regimen of regular, moderate physical activity.74,75 Both activities draw blood away from the gut—either continuously or only during a transition phase of acclimation. Whereas acclimation to regular physical activity can restore blood flow to GI cells, 75 this may not be the case for a regimen of continuously exhaustive exercise. 116 Insufficient oxygen supply to the enterocytes is as detrimental as hypoxia within the gut lumen is beneficial. The ability to maintain sufficient blood flow to the gut during exercise is critical to supporting gut barrier integrity and gut microbiome eubiosis74,75,117,118 (Figure 2A). As detailed above, insufficient blood flow to enterocytes interferes with their functions and triggers production of messengers such as pro-inflammatory cytokines74,75 (Figure 2B).
In summary, exercise that continuously exceeds the capacity to maintain sufficient blood flow to the gut results in enterocyte hypoxia, which impairs gut-barrier integrity, 119 increases oxygen concentrations in the gut lumen, and decreases the abundance of anaerobic fermenting microbes120,121 (Figure 2B). Such microbiome dysbiosis and impaired barrier parallel changes observed during chronic HIV infection and promote microbial translocation, chronic activation of gut mucosa-associated immune cells, upward-spiraling immune system activation and further impairment of the gut barrier (Figure 2B).
Additional Effects of Physical Activity on the Gut and Immune System
Additional mechanisms may also contribute to the effects of physical activity on the immune system. For example, adipose tissue around the waist (visceral fat) is metabolically active and releases inflammatory hormones. 122 Excess visceral fat thereby leads to chronic NF-kB activation and non-resolving inflammation.123,124 Exercise can contribute to preventing excess adipose tissue as one avenue to reduce non-resolving inflammation.96,125-127
Moreover, regular physical activity may also restore immune system balance by supporting redox homeostasis. 128 All exercise generates oxidants (reactive oxygen species, ROS) in working muscles which, in turn, stimulate production of endogenous antioxidant enzymes that help keep ROS in check.129,130 Moderate amounts of ROS are essential to induce these important antioxidant enzymes. 131 Redox homeostasis supports immune function, that is, stimulates immunity against infection without inducing excessive self-attack or non-resolving inflammation. 132 Regular non-exhaustive physical activity presumably stimulates ROS and antioxidant production in a balanced ratio that maintains redox homeostasis. Antioxidants are needed to keep ROS from triggering excessive NF-kB activation and programmed cell death.133,134 Continuously exhaustive exercise may produce ROS at a level that exceeds the capacity for antioxidant enzyme production and results in chronic redox imbalance. It should also be noted that most of the antioxidant enzymes induced by physical activity require dietary mineral cofactors, such as zinc, selenium, copper, and manganese, and also cooperate with dietary antioxidant vitamins, such as vitamins C and E. 135
The Transition Between Beneficial and Detrimental Exercise
Intensity and Habituation
There is interest in identifying a quantifiable transition point between beneficial and detrimental physical activity. The authors 136 of a recent review of the relevant evidence proposed that “vigorous endurance training with ≥60 min at ≥70% of VO 2max increases the intestinal permeability, with an enhanced effect observed in hot environments, at high altitude, and under dehydration.” Exercise at 70-80% of VO2max (maximal aerobic capacity, or maximal oxygen consumption) was reported to be the point at which blood flow to the gut decreased by other authors.137,138 It should be noted that this recommendation ties the threshold to individual work capacity as a feature of individual fitness level that is, in turn, presumably associated with acclimation/habituation to a particular exercise regimen and varies with individual differences in training status as well as other personal factors.
Evidence for Effects of Physical Activity on Inflammatory Markers in PLWH
Summary of Selected Studies Published Over the Past Ten Years on the Effects of Interventional Physical Activity Programs on Inflammatory Markers and/or CD4+ Cell Counts in PWLH. Significant Effects (P<.05) Are Noted in Relation to the Duration and Type of the Physical Activity Completed. Longitudinal Study Assesses Variables Over an Extended Period of Time.
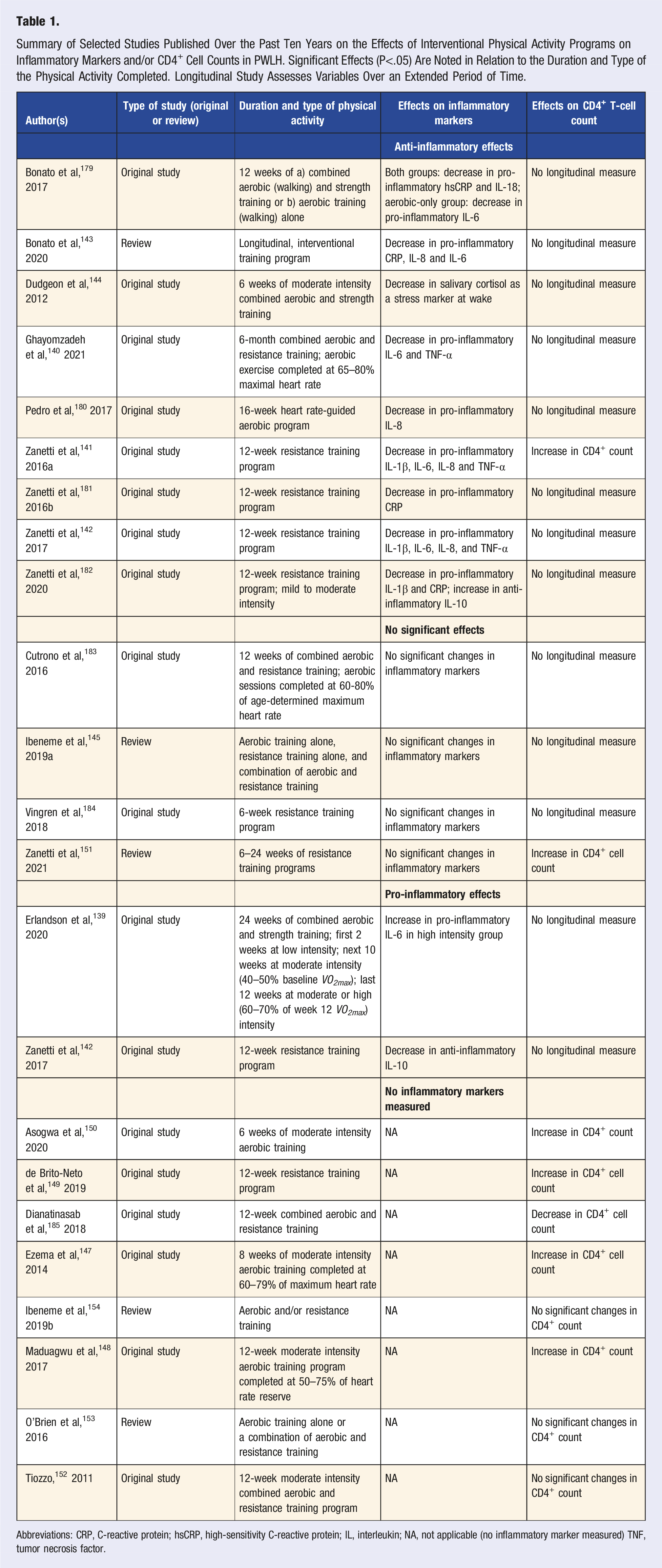
Abbreviations: CRP, C-reactive protein; hsCRP, high-sensitivity C-reactive protein; IL, interleukin; NA, not applicable (no inflammatory marker measured) TNF, tumor necrosis factor.
A recent clinical trial consisting of combined aerobic and resistance training over six months, with aerobic exercise performed at 65–80% maximal heart rate, resulted in statistically significant decreases in percent body fat and decreased levels of the pro-inflammatory cytokines IL-6 and TNF-α. 140 A 12-week resistance training program also significant decreased subcutaneous body fat, IL-6 and TNF-α in sedentary PLWH 141 as well as in PLWH with metabolic syndrome. 142 A recent review 143 concluded that physical activity is associated with positive changes in the inflammatory environment of PLWH. However, other trials found a mix of decreases in some inflammatory markers and no change in others (eg, Dudgeon et al, 2012). 144 A meta-analysis 145 of a limited number of studies using diverse training regimens concluded that physical activity does not significantly reduce inflammation in PLWH.
Table 1 also includes a summary of recent studies on the effect of exercise on CD4+ T cell counts in PLWH. Non-resolving inflammation and dysbiosis are linked to diminished CD4+ cell counts 146 and low CD4+ counts persist in immunological non-responders (see above). Restoring CD4+ cells is a priority of HIV treatment, and elimination of non-resolving inflammation may facilitate recovery of CD4+ T cell counts in PLWH. Exercise increased CD4+ cell counts in some trials on exercise in PLWH141,147-151 but not in others.152-154 The potential of physical activity to increase CD4+ counts thus warrants further study as a potential avenue to strengthen immunity in PLWH.
Resolving Inflammation and Recovery Time
It should be noted that classification of inflammation regulators into pro- vs anti-inflammatory molecules does not capture the context-dependent roles of these molecules, which applies to both cytokine hormones 155 and another key class of inflammation regulators, the fatty-acid-based eicosanoids.156,157 Moreover, increased acute inflammation following physical activity is not problematic and instead plays a critical role in triggering the synthesis of antioxidant enzymes 131 and thus maintaining immune balance.129,132,158 In fact, when oxidative signals are eliminated by high-dose antioxidant vitamins taken during athletic training, synthesis of endogenous antioxidant enzymes as well as muscle building is prevented. 131 Similarly, anti-inflammatory treatments (eg, NSAIDS) targeting exercise-associated injury can actually prevent healing by blocking the pro-resolution “stop signals” involved in the inflammatory response. 159 Physical activity can thereby acutely increase inflammatory markers while also strengthening pro-resolution pathways. 160 Taken together, these results suggest that short-term increases in markers of inflammation following physical activity may boost, rather than weaken, the anti-inflammatory resolution of inflammation.
The recovery time available for the resolution of acute inflammation may play a role. In other words, exercise performed at an excessive duration, 161 without sufficient recovery time between sessions, 162 may be what is most detrimental. 163 For example, what was described as “vigorous” exercise by the authors164,165 increased the levels of the stress hormone cortisol, whereas “moderate” exercise had no such effect. Likewise, “forced” treadmill running in mice promoted significantly greater increases in cortisol than “voluntary” wheel running. 166 For such exhaustive exercise, constitutive activation of NF-kB and subsequent tissue degeneration, oxidative stress, gut barrier breakdown, gut microbiome dysbiosis and non-resolving inflammation have been demonstrated.167-171 At this point, it can thus not be excluded that detrimental exercise modalities described as “high-intensity,” “vigorous,” or “forced,” may also be lacking in adequate time for recovery and resolution of inflammation between sessions. A notable study by Schlabe et al (2017) 172 suggests that even marathon training, performed as “moderate endurance training” over a period of 12 months, can be safe for PLWH.
Future research is needed that includes assessment of time allowed for recovery and resolution of inflammation, and uses consistent standards to quantify work capacity (such as VO2max), what percent of this capacity was reached and for how long during the physical activity, as well as blood flow to the gut across a range of different physical activities. Furthermore, individualized physical activity regimens likely need to be customized for different needs. As stated above, acclimation to a regimen of physical activity can restore blood supply to the gut during exercise. Such acclimation presumably raises the ceiling of work capacity. However, there likely is an upper limit of this acclimation effect that may be exceeded by certain types of exhaustive exercise. The current understanding thus suggests that physical activity recommendations should be expanded to comprehensively emphasize movement that is interest-based, voluntary, completed at an enjoyable intensity, performed regularly, and allows adequate time for recovery and resolution of inflammation.75,173-178
Proposed Benefits of Physical Activity for PLWH
The evidence reviewed above supports a proposal that regular physical activity—allowing sufficient acclimation and recovery that restore blood flow to the gut—may facilitate the resolution of inflammation by ameliorating the leaky gut present in many treated and untreated PLWH (Figure 3). As stated above (Figure 1B), many PLWH experience a dysbiotic gut microbiome, characterized by gut-barrier disruption and microbial translocation that persist despite ART treatment (Figures 1B and 3A). This microbial translocation activates the immune system and leads to release of pro-inflammatory cytokines, microbes, and microbial products into the bloodstream, further triggering widespread immune activation and gut barrier breakdown (see above; Figures 1B and 3A). The non-resolving inflammation experienced by PLWH (Figure 3B) may thus be reduced by adoption of a regimen of regular physical activity that allows sufficient blood flow to the GI tract, increases abundance of fermenting microbes and SCFAs, and strengthens the gut barrier integrity. This type of physical activity may protect against various comorbidities in both treated and untreated PLWH and serve as a complement to traditional ART (Figure 3B). Schematic depiction of proposed changes in the gut of PLWH induced by regular, moderate intensity, interest-driven physical activity. (A) Dysbiosis and impaired barrier present during chronic HIV infection (B) Restored blood flow, eubiosis and reduction of inflammation due to regular physical activity. Created with BioRender.com.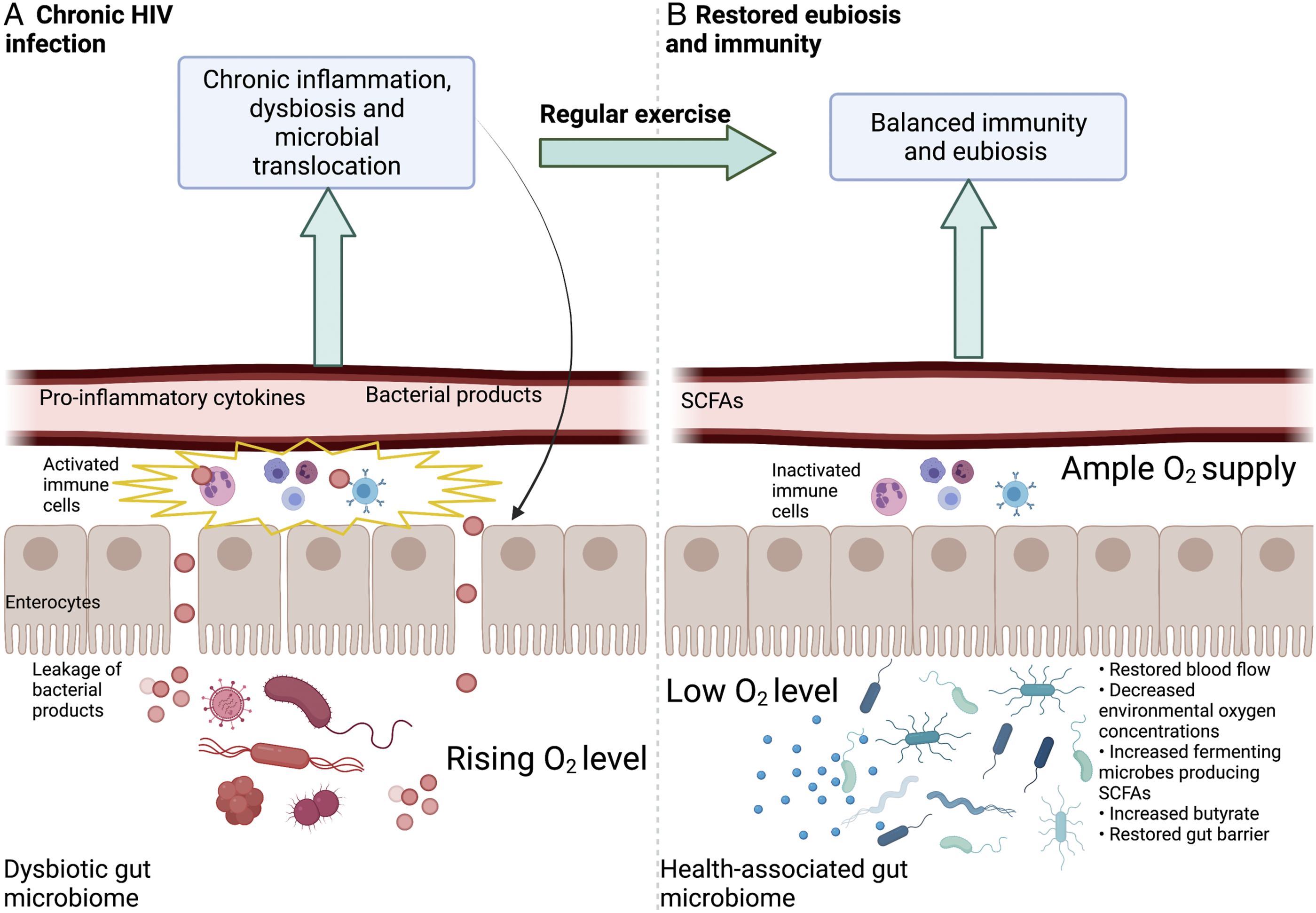
At this time, there is some evidence that exercise can reduce markers of inflammation and improve body composition in PLWH (see above; Table 1).141,143,179 However, more research is needed to establish this because findings are presently not consistent. 145 It is clear, however, that physical activity interacts with the same molecular players that are affected by HIV, such as NF-kB. Whereas an acute bout of exercise was associated with acute activation of NF-kB, acclimation to regular exercise suppressed NF-kB in mice models and prevented non-resolving inflammation. 114
Physical activity thus offers great promise as a potential lifestyle modification for reducing non-resolving inflammation and inflammation-associated comorbidities in PLWH. Physicians and other healthcare providers should be aware of the specific benefits physical activity may offer to PLWH and consider incorporating physical activity into treatment plans as a complement to ART. Additional research is needed to design individualized training plans customized to match individual fitness levels and interest.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
