Abstract
As pregnancy progresses, physical changes may affect physical activity (PA) measurement validity. n = 11 pregnant women (30.1 ± 3.8 years) wore ActiGraph GT3X+ accelerometers on the right hip, right ankle, and non-dominant wrist for 3–7 days during the second and third trimesters (21 and 32 weeks, respectively) and 12 weeks postpartum. Data were downloaded into 60-second epochs from which stepping cadence was calculated; repeated-measures analysis of variance was used to determine significant differences among placements. At all time points, the wrist accelerometer measured significantly more daily steps (9930–10 452 steps/d) and faster average stepping cadence (14.5–14.6 steps/min) than either the hip (4972–5944 steps/d, 7.1–8.6 steps/min) or ankle (7161–8205 steps/d, 10.3–11.9 steps/min) placement, while moderate- to vigorous-intensity activity at the wrist (1.2–1.7 min/d) was significantly less than either hip (3.0–5.9 min/d) or ankle (6.1–7.3 min/d). Steps, cadence, and counts were significantly lower for the hip than the ankle at all time points. Kappa calculated for agreement in intensity classification between the various pairwise comparisons ranged from .06 to .41, with Kappa for hip–ankle agreement (.34–.41) significantly higher than for wrist–ankle (.09–.11) or wrist–hip (.06–.16). These data indicate that wrist accelerometer placement during pregnancy likely results in over counting of PA parameters and should be used with caution.
Comparability of overall physical activity levels across different accelerometer placements
Introduction
There are many well-documented benefits of physical activity (PA) during pregnancy for both mother 1 and her offspring. 2 However, self-report and device-based PA measures suggest that <25% of pregnant women meet the PA guidelines of at least 150 minutes of moderate- to vigorous-intensity PA.3-5 In order to make more precise, outcome-oriented PA recommendations, reliable and valid PA measurement during pregnancy must be both available and simple to use. Device-based measures (pedometers and accelerometers) are becoming increasingly popular for PA surveillance, but there are considerations for their use. Physical and behavioral changes that occur across gestation may affect device validity for PA measurement, which would in turn affect the determination of adherence to PA recommendations as well as measurement of longitudinal changes in PA across pregnancy.
Walking is the most commonly reported form of PA among pregnant women3,4 and seems to be resistant to many of the commonly reported barriers to PA during pregnancy. 6 The simplest way to measure walking is to count daily steps, but there is increasing interest in using walking cadence, measured in steps per minute, as an indicator of walking intensity.7,8 In the general population, a cadence of 100 steps/min has been shown to closely correspond to moderate-intensity PA (ie, 3 METs)9-11 while 130 steps/min is approximately equivalent to vigorous-intensity PA (ie, 6 METs).9,12 Cadence can and has been used as a tool to estimate walking intensity in pregnant women,13,14 but its utility should be examined further in estimation of walking intensity in pregnant women.
The location of wear for step counting devices varies due to design of various monitors as well as personal preference of the wearer. One such study reported challenges with compliance using ankle accelerometry, 15 though ankle-worn ActiGraph steps were more accurate than those worn on the hip in non-pregnant adults who walked overground at both slow and fast walking speeds. 16 Another laboratory-based study found relatively comparable reliability coefficients for hip and ankle accelerometer counts but did not report steps. 17 Further, it is important to note that not all devices are designed to measure steps or related parameters at more than one site. Some devices are designed for a specific site (ie, Fitbit and Apple Watch are designed for wrist wear, while many pedometer algorithms are designed for hip use). Consideration must be taken to use devices as intended, though some devices such as the ActiGraph are unique in that they are designed to be able to be worn at several different body locations.
In the general population, an increasingly popular device placement site is the wrist. Acceptability was significantly higher for wrist vs hip wear in a study of both pregnant and non-pregnant adults, 18 and in a study of pregnant women who wore both hip and wrist accelerometers, compliance was significantly higher for wrist- vs hip-worn devices. 19 Though compliance seems to be higher with wrist wear, no evaluation of accuracy for step counting and cadence has been reported in pregnant women. In the general population, wrist-worn ActiGraph monitors have been shown to undercount steps compared to hip-mounted ActiGraphs during treadmill and overground walking but overcount during activities of daily living, 20 suggesting use of the hands during some activities may impact step counting. In pregnant women, hip-worn ActiGraphs have been shown to have good accuracy for fast walking and poorer accuracy during slow walking, 21 while other work has demonstrated that the hip-mounted ActiGraph significantly undercounts during free-living walking compared to a criterion measure. 22 Unfortunately, there are no algorithms designed specifically for step counting via ActiGraph GT3X+ devices in pregnant women for any device placement, which further complicates comparisons of both sites and various devices.
Despite decades of research reporting PA outcomes in pregnant women, 23 more work is needed to establish valid ways of capturing pregnancy PA. Given the aforementioned lack of research comparing accelerometer placement sites and cadence- and counts-based outcomes longitudinally across pregnancy, our study’s purpose was to compare daily steps, stepping cadences, and accelerometer count measurements when worn at the hip, wrist, and ankle.
Methods
Participants
Pregnant women were recruited prior to 20 weeks gestation from local obstetrics clinics, health clubs, and via word of mouth to participate in a longitudinal study of PA monitor validity and reliability. A total of n = 11 (age = 30.1 ± 3.8 years) pregnant women agreed to wear accelerometers at three wear sites (wrist, hip, and ankle) for 7 days at three time points. Prior to beginning the study, all participants signed informed consent forms, and all study procedures were approved by the university Institutional Review Board.
Procedures
Participants reported to the laboratory at approximately 21 and 32 weeks gestation and 12 weeks postpartum. The gestational weeks were selected because they occur at approximately the mid-point of the second and third trimesters, respectively, and 12 weeks postpartum was chosen as a surrogate measure for the pre-pregnancy state, as is often used in other pregnancy work.14,17 At each time point, participants were fitted with ActiGraph GT3X+ accelerometers on the non-dominant wrist (dorsal side), right hip (anterior axillary line), and right ankle (lateral malleolus). All accelerometers were initialized to record triaxial data at 30 Hz and were time-synchronized to the same computer. At the initial laboratory visit, participants were instructed on proper device placement and were asked to wear the devices during all waking hours (except when bathing or swimming) for seven consecutive days while maintaining their normal activities.
Statistical Analysis
Upon device return, accelerometer data were downloaded as vector magnitude activity counts (with low-frequency extension disabled) and steps in 60-second epochs within the ActiLife software (version 6.13.4, ActiGraph Corp., Pensacola, FL). Once downloaded, non-wear was determined, separately for each monitor, as ≥60 consecutive minutes of 0 counts using Troiano default algorithm within ActiLife. The Cole–Kripke and ActiGraph proprietary algorithms were used to determine sleep and wake periods for each monitor. 24 All time recorded as non-wear or sleep by any of the monitors was removed from the datasets for all three monitors, and the remaining data were time-aligned and coded as wear/awake time by all three monitors. A valid day was considered as any day with ≥480 minutes of wear time. To be included in the analysis, participants needed to have at least one valid day of data at each of the three time points, and all valid days at each time point were included for each participant.25,26 Since the focus of this analysis was on comparability of overall PA levels across different accelerometer placements and not on characterizing normal activity patterns, data were analyzed with each valid day serving as a data point (no averaging across the week). Thus, participants could contribute up to seven data points per time point (second trimester, third trimester, and 12 weeks postpartum) for a possible total of 21 data points per person. A subanalysis using weekly averages for each participant was also conducted; point estimates were similar to the daily analysis but with lower power due to smaller sample size when analyzing weekly data (n = 11 at each time point) compared to daily data (n = 52-66, see Results). Therefore, only the daily data are presented in the Results.
Steps/day were calculated for each day, both as the raw number recorded from each accelerometer as well as steps/day scaled to a 16-waking-hour day to normalize data for potentially differing wear times; this approach has been used previously in order to provide data indicative of a “typical” day with 16 hours awake and 8 hours of sleep. 14 Total daily (and scaled to 16-waking-hour day) moderate-intensity activity was determined by counting the number of minutes with a stepping cadence of 100–129 steps/min, vigorous-intensity activity as the number of minutes with a stepping cadence of ≥130 steps/min, and moderate- to vigorous-intensity activity (MVPA) as the number of minutes with a stepping cadence of ≥100 steps/min.12,14 Also, mean, median, maximum (highest recorded cadence in a single minute), and peak cadence (the average of the highest 30 daily step cadence minutes), as well as the number of minutes spent in cadence ranges by units of 10 (0-9, 10-19, 20-29, etc.) as well as <100 steps/min were calculated for each day.13,14 It is worth noting that cadence rates were developed by analyzing walking cadence via hip-worn monitors. 27 In addition to steps data, total daily vector magnitude (and scaled to a 16-waking-hour day) were calculated for each accelerometer.
Agreement statistics (percent (%) agreement and Kappa) for characterizing activity intensity (sedentary/light as <100 steps/min, moderate as 100–129 steps/min, and vigorous as ≥130 steps/min) were calculated for each pairwise accelerometer comparison (hip–wrist, hip–ankle, and wrist–ankle). Finally, correlations were calculated to compare daily steps and counts data for each pairwise comparison.
Each of these aforementioned variables were compared among the hip, wrist, and ankle accelerometers, separately for each time point, using repeated-measures analysis of variance with an alpha level of P < .05 used to denote statistical significance. In the event of a significant overall test statistic, post hoc pairwise comparisons were made using a Bonferroni correction to account for multiple comparisons. These analyses were conducted in SPSS version 24.0 (IBM Corp., Armonk, NY).
Results
Comparison of Steps and Activity Counts Among Hip, Wrist, and Ankle Accelerometer Placements by Trimester.
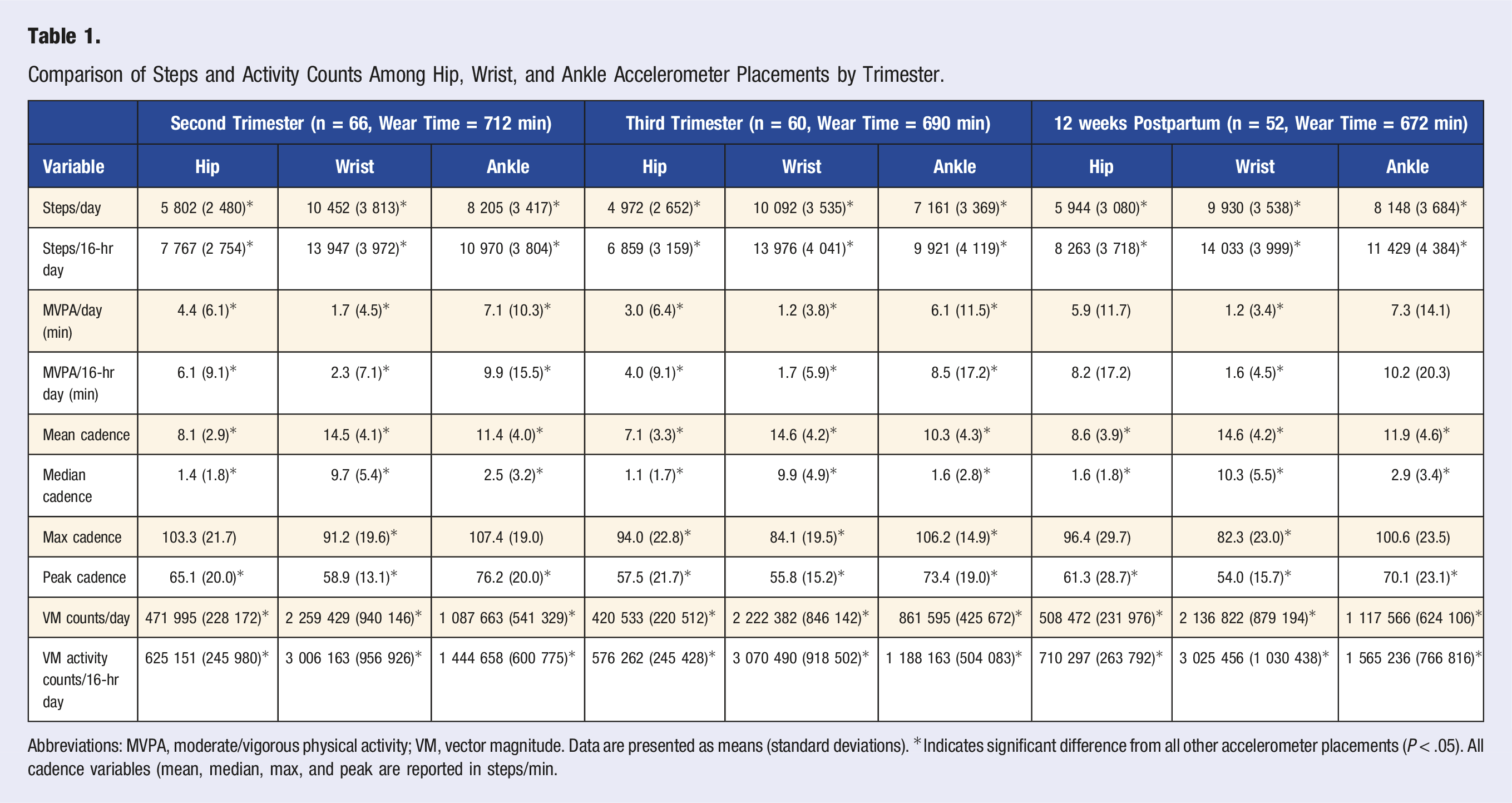
Abbreviations: MVPA, moderate/vigorous physical activity; VM, vector magnitude. Data are presented as means (standard deviations). *Indicates significant difference from all other accelerometer placements (P < .05). All cadence variables (mean, median, max, and peak are reported in steps/min.
Comparison of Time Spent in Step Cadence Ranges by Accelerometer Placement and Trimester.
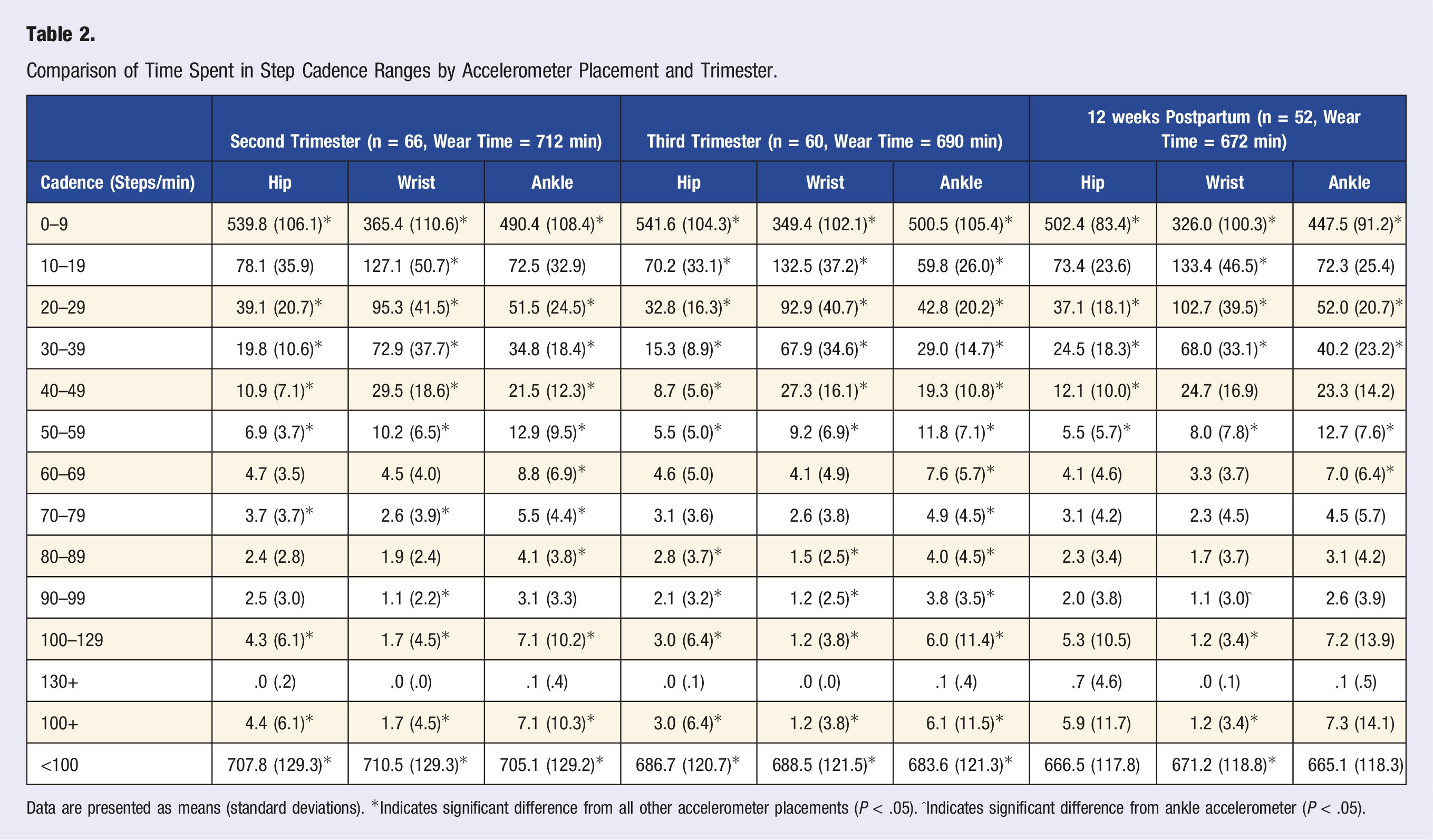
Data are presented as means (standard deviations). *Indicates significant difference from all other accelerometer placements (P < .05). ^Indicates significant difference from ankle accelerometer (P < .05).
Agreement Statistics Among Hip, Wrist, and Ankle Accelerometers by Trimester.
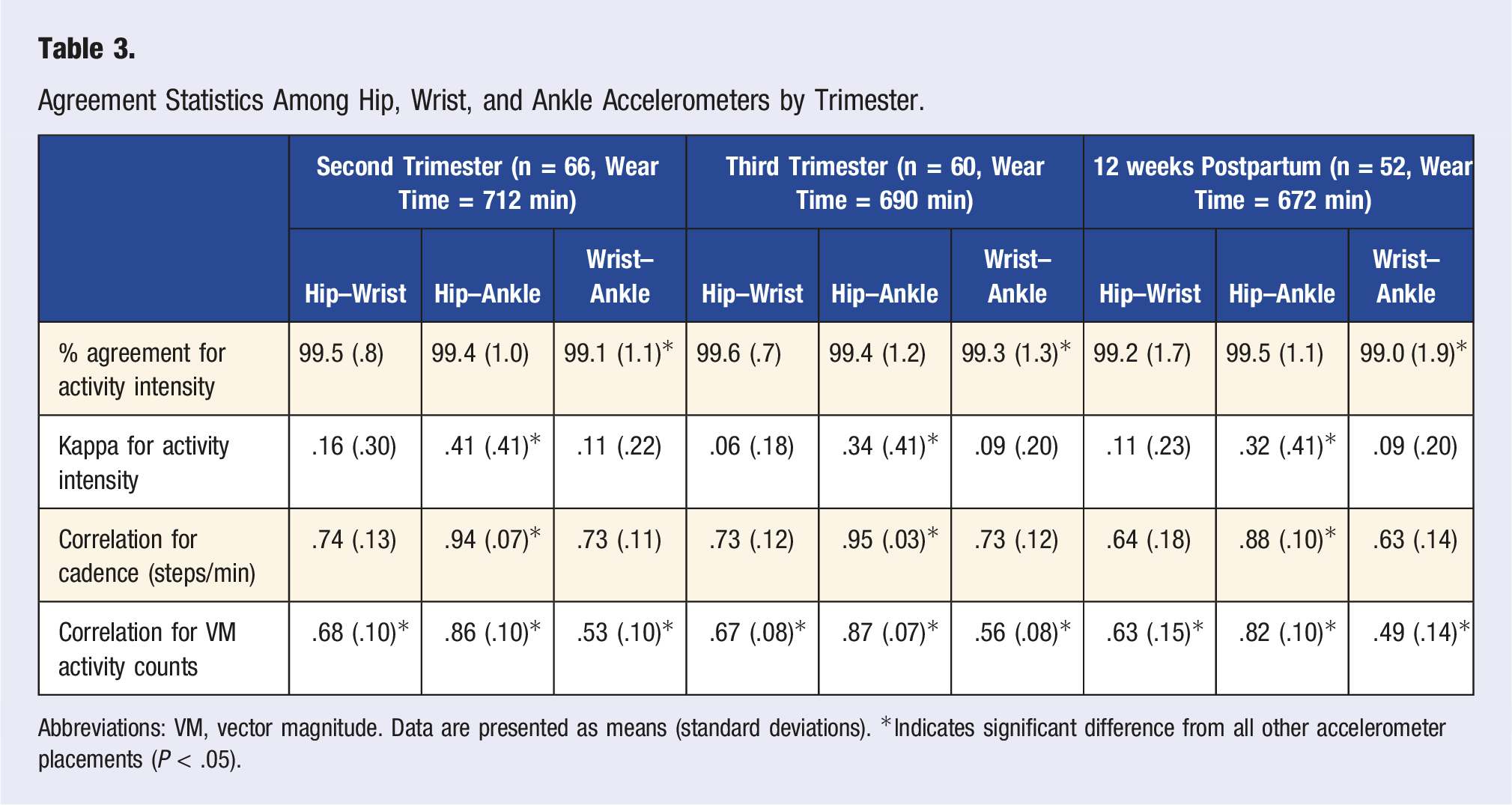
Abbreviations: VM, vector magnitude. Data are presented as means (standard deviations). *Indicates significant difference from all other accelerometer placements (P < .05).
Discussion
These data demonstrate that accelerometers placed on the wrist calculate the highest number of steps, counts, and fastest mean walking cadence but the lowest daily minutes of MVPA of the three device placements studied (hip, ankle, and wrist). Similarly, the Kappa statistic showed a significantly greater inter-device reliability estimate between hip and ankle for classification of activity intensity compared to the wrist with the hip or wrist with ankle accelerometer sites, which indicates that the wrist measurements deviate considerably from the other two sites. This may be due to inherent problems with wrist-worn accelerometers in general where movement of the arms does not correspond well to stepping (eg, walking when texting or pushing shopping cart/stroller, activities involving arm movement but no stepping) or due to alterations in typical arm swing during pregnancy, which to our knowledge has not been studied. Other studies in non-pregnant populations have found superior accuracy of hip and ankle placements for step counting compared to the wrist. In community-dwelling older adults, hip-worn PA monitors (Fitbit One, Misfit Shine, and New-Lifestyles NL-1000) were more valid measures of steps taken during overground walking compared to wrist-worn devices (Garmin Vivofit 2, Jawbone UP2, and Fitbit Charge). 28 In a study designed to evaluate the effectiveness of ankle vs wrist accelerometer placement to differentiate among broad categories of movement using a triaxial device called a Wocket placed in various body locations, ankle monitoring was accurate (compared to human observer) 95% of the time, while wrist placement correctly identified the activity type only 84% of the time. 29 Still another study found that estimates of energy expenditure and sedentary time were more accurate when derived from hip vs wrist accelerometers.21,30 In a study comparing various devices and placement sites in pregnant women, the wrist-mounted Fitbit Flex recorded steps had the highest error in step counting compared to numerous other hip- and ankle-worn devices during overground 22 and treadmill 21 walking. As expected from similar research in non-pregnant populations, the wrist placement likely over counts steps and step cadence in pregnant and postpartum women and may be a less accurate placement site than the hip or ankle.
Due to lack of a free-living criterion measure for stepping used in the present study, determination of the most accurate accelerometer placement cannot be discerned from the current study. Few other studies in pregnant or non-pregnant populations have examined the agreement of a single device brand and model at the same three wear locations (hip, ankle, and wrist), so comparisons are difficult. One study of older adults (mean age 84.2 years) examined agreement of ActiGraph GT1M devices on hip, ankle, and wrist compared to hand-counted steps in a laboratory setting, and only the ankle device step counts had positive agreement to hand-counted steps. 31 In a similar study that evaluated estimates of energy expenditure from accelerometers placed on the hip, ankle, and wrist to indirect calorimetry, the ankle placement had the strongest correlation (r = .86) to gold standard energy expenditure measurement. 32 During activities of daily living, reliability for these three placement sites, as measured by the ActiGraph GT3X+, was high, with correlation coefficients ranging from .864 to .989, suggesting that reliability is not problematic for any placement. 33 However, validity was not assessed, making conclusions to which the current study can be compared a challenge.
Strengths of the current study include high compliance to wear the devices, with all participants exceeding minimum inclusion criteria of one day of wear. In addition, the data are longitudinal, with the same women wearing the devices at all three time points. Finally, data were collected in participants’ true free-living environments rather than a laboratory setting; free-living PA is seldom studied in this population, so our study provides new insight into accelerometer comparability during activities that more closely represent typical behaviors of pregnant and postpartum women.
Limitations of this study should also be considered. We used no gold standard for comparison of daily steps or stepping cadence, so only convergent validity measures could be assessed. Future research should utilize other devices, such as a validated pedometer or the StepWatch device (Modus Health, Edmonds, WA, USA) as a criterion measure. Other limitations include the use of convenience sampling, which may not truly represent the behavioral patterns of all pregnant and postpartum women, and lack of demographic characteristics collected. Because the study was not intended to describe characteristics that predict or influence PA behavior, demographic variables such as parity, income, and education were not collected and, thus, cannot be considered in interpreting our findings. Power analysis was not conducted based on the outcome of comparing three sites.
In conclusion, our data indicate that the measurement of daily steps, stepping cadence, and time spent in MVPA based upon accelerometer-collected data depends heavily upon where the device is worn by the pregnant or postpartum woman. In particular, the wrist site overestimates steps and stepping cadence and underestimates MVPA compared to hip and ankle accelerometer placements, likely due to the devices measuring low-cadence stepping during activities movement of the hands (typing, preparing food, etc.) without corresponding whole-body movement. Further research is needed to compare hip, ankle, and wrist accelerometer data to a gold standard in a free-living setting to determine which placement site provides most accurate step counts, cadence measurement, and time spent in MVPA and if the most accurate placement is affected by trimester of pregnancy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval Statement
This study was approved by the Michigan State University Institutional Review Board prior to commencement of data collection.
