Abstract
Background. It is hypothesized that normal weight individuals develop diabetes through different pathophysiological mechanisms and that methods of prevention may differ in the absence of overweight/obesity. In this study, we compared the effect of lifestyle health coaching (LHC) on fasting plasma glucose (FPG) in normal weight, overweight, and obese US adults with prediabetes. Methods. Subjects were 1358 individuals who completed baseline and follow-up evaluations as part of an LHC program (follow-up = ~6 months). Participants were stratified, based on baseline body mass index (BMI), into normal weight (n = 129), overweight (n = 345), and obese (n = 884) cohorts. LHC included counseling, predominantly via telehealth, on exercise and nutrition. Results. BMI decreased (P < .001) in the overweight and obese participants but was unchanged in the normal weight participants. FPG decreased (P < .001) in all 3 cohorts, and the magnitude of decrease did not differ significantly among cohorts. FPG decreased to <5.6 mmol/L in 58.1%, 49.3%, and 41.4% of the normal weight, overweight, and obese participants, respectively. Conclusions. To our knowledge, this study is the first outside of Asia to show that LHC is as effective in managing FPG in normal weight adults with prediabetes versus those who are overweight/obese.
Although overweight/obesity is a major risk factor for prediabetes, recent research suggests that as many as 20% of US adults with prediabetes have a normal body mass index.
Over one-third of US adults are estimated to have prediabetes. 1 In addition to being a precursor for type 2 diabetes, prediabetes is associated with increased hospitalizations and health care costs.1-3 Although overweight/obesity is a major risk factor for prediabetes, recent research suggests that as many as 20% of US adults with prediabetes have a normal body mass index (BMI). 4 Moreover, in a pooled analysis of 5 longitudinal cohort studies, individuals who were normal weight at the time of incident diabetes experienced higher total mortality during follow-up as compared with those who were overweight or obese. 5
Currently recommended approaches to the management of prediabetes and prevention of type 2 diabetes include therapeutic lifestyle changes and, if needed, pharmacotherapy. 6 However, it is hypothesized that normal weight individuals develop type 2 diabetes through different pathophysiological mechanisms and that methods of prevention may differ in the absence of overweight or obesity. 4
While lifestyle intervention is the foundation of diabetes prevention programs, with the exception of studies conducted in Asia where prediabetes is more common at lower BMIs,7-9 scarce data are available on the effectiveness of lifestyle intervention in normal weight adults with prediabetes. Indeed, normal BMI is an exclusion criterion for participation in many diabetes prevention programs, including the US Medicare Diabetes Prevention Program. 10 Therefore, the aim of this study was to evaluate, in a US population, the effectiveness of an evidence-based, technology-enabled, lifestyle health coaching (LHC) program in normal weight versus overweight and obese adults with prediabetes.
Methods
Study Design
A cohort of 1358 consecutive US adults (age = 51.6 ± 11.6 years) who completed both an initial and follow-up assessment as part of their participation in an evidence-based, technology-enabled LHC program comprised the study population. Subjects were either self-referred (typically, after completing a health risk assessment as part of an employer-sponsored health promotion program) or referred by their health care providers. All subjects provided informed consent and the research protocol was approved by the University of the Witwatersrand Human Research Ethics Committee (Medical; Clearance Certificate No. M200844).
Subjects were evaluated at baseline and again after an average of ~6 months of intervention. At baseline, all subjects met the fasting plasma glucose (FPG) criterion for the diagnosis of prediabetes as defined by the American Diabetes Association, namely, FPG = 5.6 to 6.9 mmol/L. 11 Subjects did not have known diabetes mellitus at program entry, were not taking diabetes medication at program entry, and remained off diabetes medication throughout the study. In addition to FPG, weight, height, BMI, waist circumference, resting blood pressure, and fasting serum lipids and lipoproteins were evaluated at baseline and follow-up using verified measurements obtained from employer-sponsored biometric screenings or the participant’s health care provider (not all measurements were obtained for all participants).
Intervention
The LHC program, including its evidence-based design, has been described in detail elsewhere.12,13 Briefly, the LHC program is based on multiple programmatic designs for the delivery of lifestyle management and chronic disease risk reduction interventions shown to be effective in randomized clinical trials and is conducted in accordance with published national evidence-based clinical guidelines, where appropriate. Moreover, the effectiveness of the LHC program in terms of improvements in cardiovascular disease risk factors has previously been verified in multiple peer-reviewed publications, including randomized clinical trials.12,13
LHC included counseling on exercise training, nutrition, weight management, stress management, and smoking cessation. Participants were preassigned to receive up to 18 one-on-one live LHC sessions during the first year of program participation, with at least 50% of the sessions generally occurring within the first 3 months of intervention. LHC was provided by specially trained nonphysician health professionals guided by an online participant management system and using behavioral interventions derived from several behavior change models and strategies, primarily, adult learning theory, social learning theory, motivational interviewing, single concept learning theory, and the stages of change model. The LHC program included the following core components: (a) initial assessment; (b) formulation of individualized cardiovascular disease risk factor goals (including for FPG) based on national clinical guidelines; (c) formulation of an individualized action plan for achieving risk factor goals through evidence-based lifestyle intervention and, where appropriate, physician referrals for consideration of medication changes to optimize blood pressure and/or lipid management in accordance with national clinical guidelines; (d) action plan implementation, including one-on-one behaviorally oriented live coaching (face-to-face in community-based settings or, predominantly, via telephone and the Internet); (e) online access to a variety of resources specifically designed to supplement the coaching process, including personalized goals, action plan and progress reports, an individualized series of standardized behaviorally oriented educational modules with accompanying audios, interactive self-monitoring diaries and logs, recipes, and health-related challenges; and (f) reassessment after a mean of ~6 months.12,13
During LHC, counselling on exercise training focused on the achievement of ≥150 minutes of moderate intensity aerobic exercise per week. Counseling on nutrition focused on a healthy eating pattern that, depending on each subject’s preferences, was consistent with the Dietary Approaches to Stop Hypertension (DASH) and/or Mediterranean dietary patterns.14,15 For subjects with a baseline BMI <25.0 kg/m2, weight management focused primarily on the avoidance of weight gain, whereas for those with a baseline BMI ≥25.0 kg/m2, weight management focused on the achievement of ≥5% weight loss and/or a BMI <25.0 kg/m2.
Statistical Analyses
For the purpose of this study, participants were stratified into 1 of 3 study cohorts using BMI data from the initial assessment, as follows: normal weight cohort (BMI 18.5-24.9 kg/m2; n = 129), overweight cohort (BMI 25.0-29.9 kg/m2; n = 345), and obese cohort (BMI ≥30.0 kg/m2; n = 884).
For categorical variables, statistical significance of differences among participants in the 3 BMI cohorts was evaluated at baseline and follow-up using Fisher’s exact test. For continuous variables, statistical significance of baseline differences and changes from baseline among participants in the 3 BMI cohorts was evaluated using one-way analysis of variance, followed by Tukey’s post hoc test when a significant F-ratio was obtained. Statistical significance of within-cohort changes from baseline was evaluated using paired t tests, where appropriate. Tests were 2-sided, and statistical significance was established at P ≤ .05.
Results
Baseline Characteristics
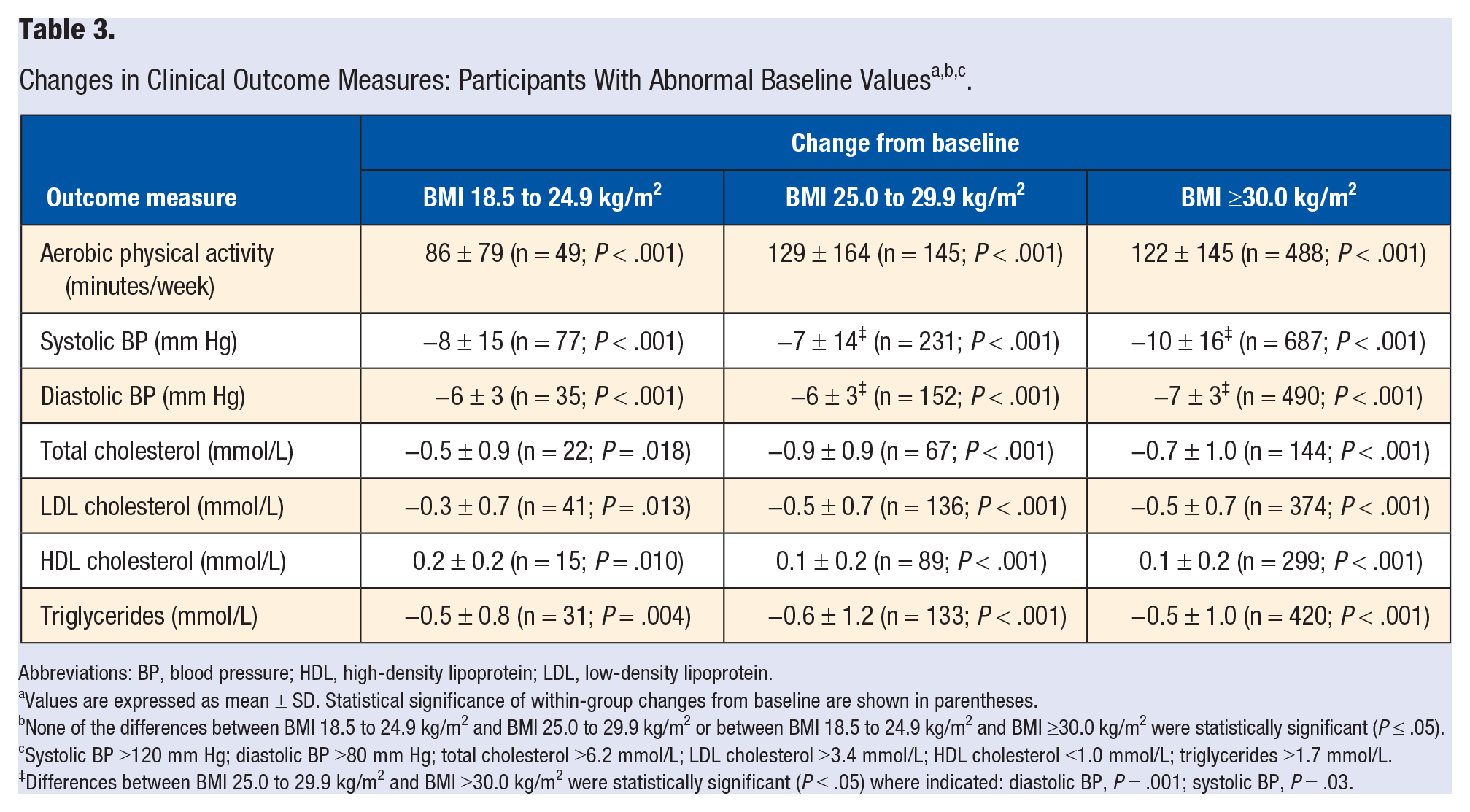
Select baseline demographic and clinical characteristics of the subjects in the 3 study cohorts are shown in Table 1. No statistically significant differences between the normal weight and overweight cohorts were observed for age, self-reported race, cigarette smoking, weekly minutes of aerobic physical activity, systolic blood pressure, total cholesterol, and LDL cholesterol. Compared with subjects in the overweight cohort, subjects in the normal weight cohort included more females and had more favorable mean values for FPG, weight, BMI, waist circumference, diastolic blood pressure, HDL cholesterol, and triglycerides. No statistically significant differences between the normal weight and obese cohorts were observed for gender, cigarette smoking, and total cholesterol. Compared with subjects in the obese cohort, subjects in the normal weight cohort were older, included more Asians and fewer blacks, and had more favorable mean values for FPG, weight, BMI, waist circumference, weekly minutes of aerobic physical activity, systolic and diastolic blood pressure, LDL cholesterol, HDL cholesterol, and triglycerides.
Baseline Demographic and Clinical Characteristics of Study Participants a .
Abbreviations: BMI, body mass index; BP, blood pressure; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Where appropriate, values are expressed as mean ± SD.
Differences between BMI 18.5 to 24.9 kg/m2 and BMI 25.0 to 29.9 kg/m2 were statistically significant (P ≤ .05) where indicated: weight, BMI, waist circumference, diastolic blood pressure, HDL cholesterol, P < .001; gender, P = .001; fasting plasma glucose, P = .003; and triglycerides, P = .004.
Differences between BMI 18.5 to 24.9 kg/m2 and BMI ≥30.0 kg/m2 were statistically significant (P ≤ .05) where indicated: age, Black, Asian, weight, BMI, waist circumference, systolic blood pressure, diastolic blood pressure, HDL cholesterol, triglycerides, P < .001; fasting plasma glucose, P = .001; gender, P = .002; LDL cholesterol, P = .021; and aerobic physical activity, P = .041.
Differences between BMI 25.0 to 29.9 kg/m2 and BMI ≥30.0 kg/m2 were statistically significant (P ≤ .05) where indicated: age, gender, Asian, weight, BMI, waist circumference, aerobic physical activity, systolic blood pressure, diastolic blood pressure, P < .001; Black, P = .001; HDL cholesterol, P = .002.
In addition to having more favorable mean baseline FPG values, a higher percentage of subjects in the normal weight cohort (84.5%) had baseline FPG values between 5.6 mmol/L and 6.1 mmol/L versus those in the overweight (72.2%) and obese (67.6%) cohorts (P = .006 for normal weight vs overweight cohort; P < .001 for normal weight vs obese cohort; and P = .132 for overweight vs obese cohort).
Using the American Heart Association’s harmonized definition for the metabolic syndrome, 1 sufficient data were available at baseline to make a definitive determination of the presence or absence of the metabolic syndrome in 121, 337, and 850 of the normal weight, overweight, and obese subjects, respectively. Of these subjects, 32.2% of the normal weight, 56.7% of the overweight, and 88.4% of the obese individuals met the criteria for the presence of the metabolic syndrome (P < .001 for normal weight vs overweight subjects; P < .001 for normal weight vs obese subjects; and P < .001 for overweight vs obese subjects).
LHC Program Participation
Subjects in the obese cohort (8.9 ± 4.2 LHC sessions) completed more LHC sessions than those in the overweight (7.7 ± 4.3 LHC sessions; P < .001 vs obese cohort) and normal weight (6.7 ± 4 LHC sessions; P < .001 vs obese cohort and P = .069 vs overweight cohort) cohorts.
Outcomes
For the entire cohort, baseline FPG (5.9 ± 0.3 mmol/L) decreased by 0.3 ± 0.6 mmol/L (P < .001) during the study. Based on the American Diabetes Association FPG criteria, 11 611 (45.0%) subjects normalized their FPG (ie, achievement of FPG <5.6 mmol/L; P < .001) whereas 55 (4.1%) subjects developed a value compatible with a diagnosis of type 2 diabetes mellitus (ie, FPG ≥7.0 mmol/L; P < .001) during the study.
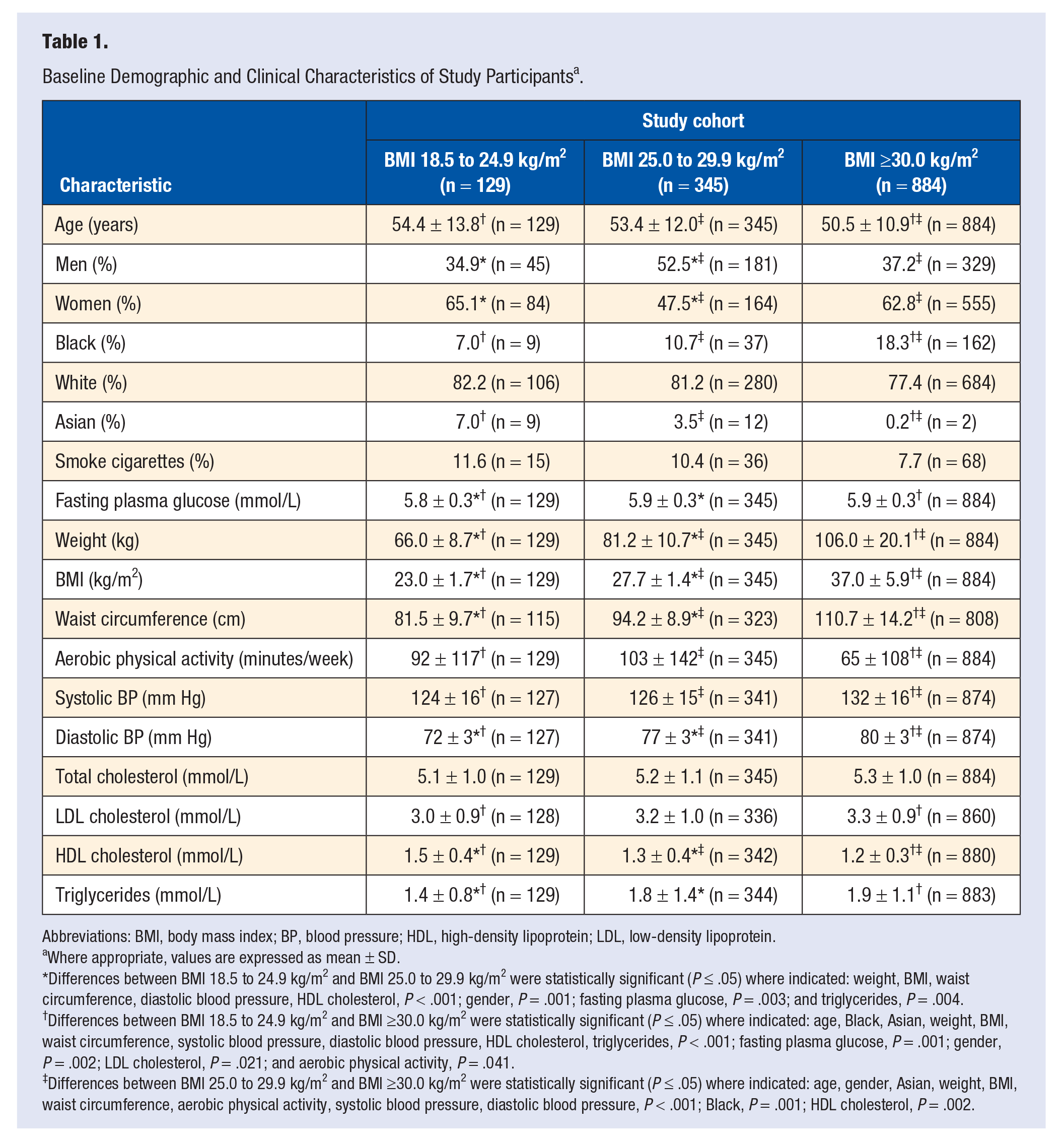
The effects of LHC on key clinical outcome measures in participants from the 3 cohorts are summarized in Table 2. FPG decreased (normal weight = −0.4 ± 0.6 mmol/L decrease; overweight = −0.3 ± 0.7 mmol/L decrease; and obese = −0.3 ± 0.6 mmol/L decrease) significantly (P < .001) in all 3 cohorts and the magnitude of decrease did not differ significantly among the cohorts (P = .142 for difference among the 3 cohorts). In contrast, weight, BMI, and waist circumference decreased significantly (P < .001) in both the obese and, to a lesser degree, overweight cohorts but remained essentially unchanged in the normal weight cohort. Although weekly aerobic exercise increased significantly (P < .001) in all 3 cohorts, the magnitude of increase was significantly greater (P = .035) in the obese versus the normal weight cohort.
Changes in Clinical Outcome Measures: All Participants a .
Abbreviations: BMI, body mass index; BP, blood pressure; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Values are expressed as mean ± SD. Statistical significance of within-group changes from baseline are shown in parentheses.
Differences between BMI 18.5 to 24.9 kg/m2 and BMI 25.0 to 29.9 kg/m2 were statistically significant (P ≤ .05) where indicated: BMI, P = .002; diastolic blood pressure, P = .004; total cholesterol, P = .015; LDL cholesterol, P = .028; and weight, P = .05.
Differences between BMI 18.5 to 24.9 kg/m2 and BMI ≥30.0 kg/m2 were statistically significant (P ≤ .05) where indicated: weight, BMI, waist circumference, diastolic BP, total cholesterol, P < .001; LDL cholesterol, P = .001; and aerobic physical activity, P = .035.
Differences between BMI 25.0 to 29.9 kg/m2 and BMI ≥30.0 kg/m2 were statistically significant (P ≤ .05) where indicated: weight, BMI, waist circumference, systolic blood pressure, diastolic blood pressure, P < .001.
As can be seen in Figure 1, FPG decreased (P < .001) to <5.6 mmol/L in 58.1%, 49.3%, and 41.4% of the normal weight, overweight, and obese cohorts, respectively (P < .001 for normal weight vs obese cohort and P = .015 for overweight vs obese cohort). FPG increased to ≥7.0 mmol/L in 3.1% (P = .122), 3.5% (P < .001), and 4.4% (P < .001) of the normal weight, overweight, and obese participants, respectively (differences among cohorts were not statistically significant). Similarly, although not shown in Figure 1, when performing additional FPG-related statistical analyses that specifically excluded the 23 Asian study subjects, FPG decreased (P < .001) to <5.6 mmol/L in 58.3%, 49.5%, and 41.4% of the normal weight (n = 120), overweight (n = 333), and obese (n = 881) participants, respectively (P < .001 for normal weight vs obese participants and P = .012 for overweight vs obese participants), and increased to ≥7.0 mmol/L in 2.5% (P = .247), 3.3% (P < .001), and 4.4% (P < .001) of the normal weight, overweight, and obese participants, respectively (differences among cohorts were not statistically significant).
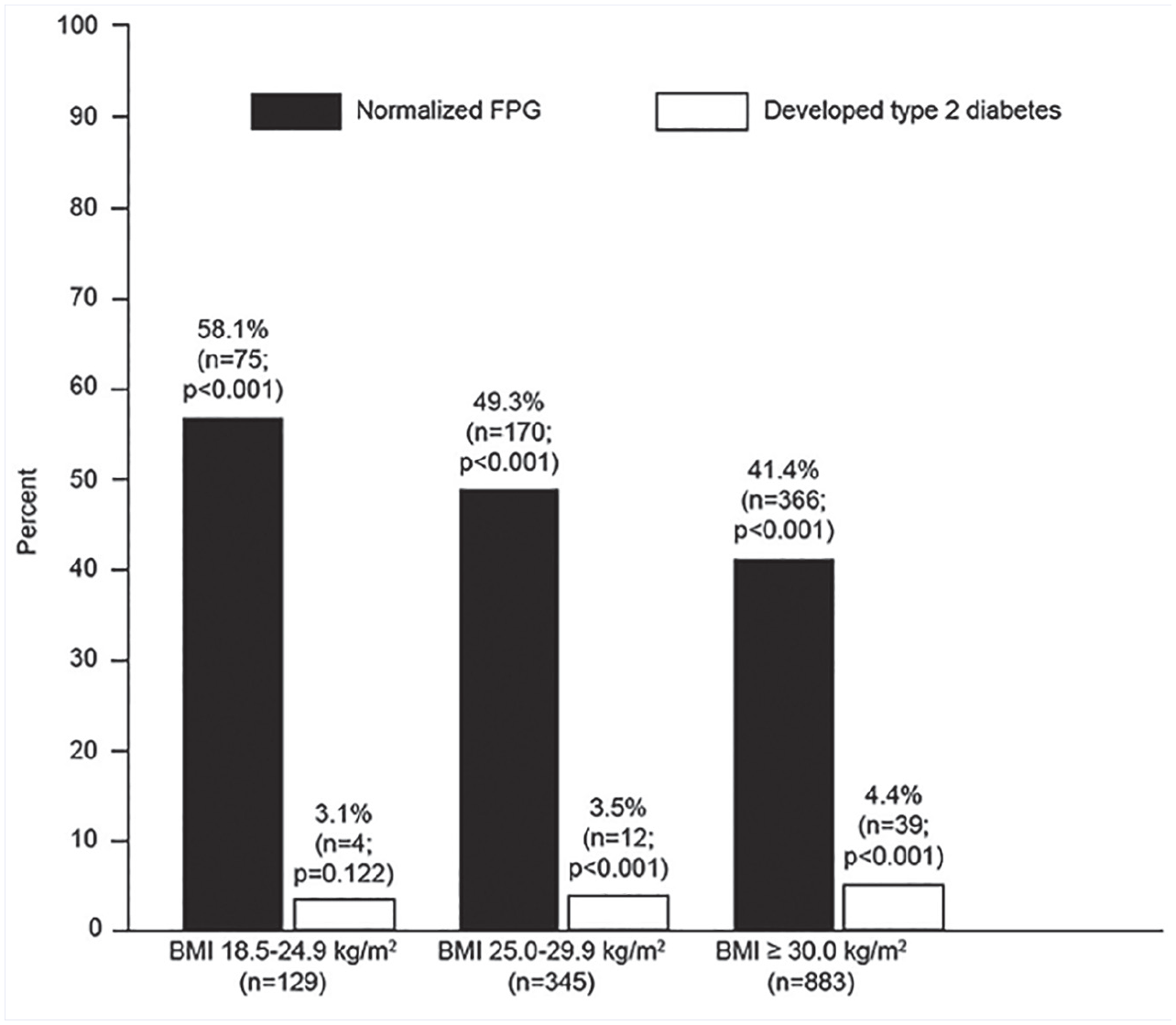
Percentage (and number) of participants who normalized their fasting plasma glucose (FPG) or developed a value compatible with a diagnosis of type 2 diabetes during study participation.
In the normal weight cohort, the percentage weight loss from baseline did not differ significantly among the participants who normalized their FPG (−0.5 ± 3.1%; n = 75), remained in the prediabetes range (−0.2 ± 2.9%; n = 50), or developed an FPG value compatible with type 2 diabetes (−1.0 ± 5.2%; n = 4) during study participation (P = .804 for difference among groups). Similarly, in the overweight cohort, the percentage weight loss from baseline did not differ significantly among the participants who normalized their FPG (−2.1 ± 4.3%; n = 170), remained in the prediabetes range (−1.3 ± 3.8%; n = 163), or developed an FPG value compatible with type 2 diabetes (−1.8 ± 3.3%; n = 12) during study participation (P = .197 for difference among groups). In contrast, for the obese cohort, the percentage weight loss from baseline was significantly greater in the participants who normalized their FPG (−4.1 ± 4.8%; n = 366) versus those who remained in the prediabetes range (−3.1 ± 4.8%; n = 478) or developed an FPG value compatible with type 2 diabetes (−1.5 ± 3.9%; n = 39) during study participation (P = .007 and P = .004 for the difference between those who normalized their FPG vs those who remained in the prediabetes range and those who developed an FPG value compatible with type 2 diabetes, respectively).
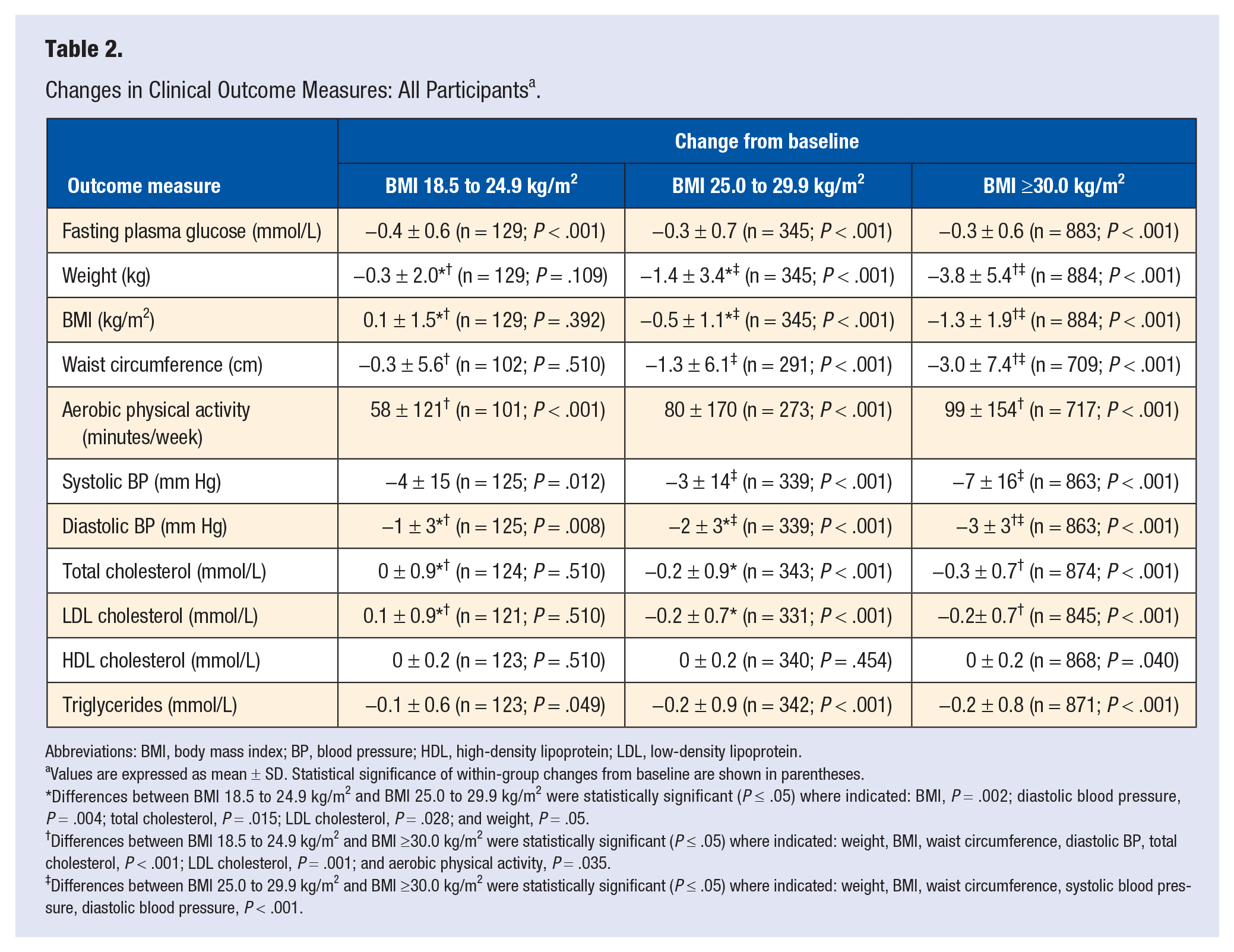
Table 3 shows changes in other select clinical outcome variables for participants in the 3 cohorts with abnormal baseline values. Participants in all 3 cohorts experienced statistically significant mean improvements in blood pressure and serum lipids and lipoproteins. For these variables, the magnitude of improvement for the normal weight cohort did not differ significantly versus the magnitude of improvement for the overweight and obese cohorts.
Abbreviations: BP, blood pressure; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Values are expressed as mean ± SD. Statistical significance of within-group changes from baseline are shown in parentheses.
None of the differences between BMI 18.5 to 24.9 kg/m2 and BMI 25.0 to 29.9 kg/m2 or between BMI 18.5 to 24.9 kg/m2 and BMI ≥30.0 kg/m2 were statistically significant (P ≤ .05).
Systolic BP ≥120 mm Hg; diastolic BP ≥80 mm Hg; total cholesterol ≥6.2 mmol/L; LDL cholesterol ≥3.4 mmol/L; HDL cholesterol ≤1.0 mmol/L; triglycerides ≥1.7 mmol/L.
Differences between BMI 25.0 to 29.9 kg/m2 and BMI ≥30.0 kg/m2 were statistically significant (P ≤ .05) where indicated: diastolic BP, P = .001; systolic BP, P = .03.
Discussion
Data from seminal randomized clinical trials, namely, the Finnish Diabetes Prevention Study, 16 Diabetes Prevention Program, 17 and China Da Qing Diabetes Prevention Study, 18 have demonstrated the effectiveness of lifestyle intervention in lowering FPG and delaying the onset of type 2 diabetes in individuals with prediabetes. However, the Finnish Diabetes Prevention Study, 16 the Diabetes Prevention Program, 17 and the many other subsequent studies conducted outside of Asia 19 either excluded normal weight individuals, utilized lifestyle interventions designed to produce weight loss in all subjects, and/or did not examine outcomes based on normal, overweight, and obese baseline weight classes. To our knowledge, the present study is the first to show in a US population that evidence-based, technology-enabled, LHC is at least as effective in managing FPG in normal weight adults with prediabetes when compared with those who are overweight or obese.
The relationship between body weight and the prevalence of type 2 diabetes is complex and appears to be modified by race/ethnicity. 7 At similar BMIs, type 2 diabetes prevalence is higher in Asians compared with whites. 7 This finding may be partly explained by differences in body fat distribution in that Asians have a greater propensity to develop visceral versus peripheral adiposity, which is more closely associated with insulin resistance than overall adiposity. 7 Additionally, at any given BMI level Asians have a higher percentage of body fat compared with Whites. 7 Thus, studies on the effect of lifestyle intervention in Asian populations with a BMI <25.0 kg/m2 cannot necessarily be extrapolated to non-Asian populations.
Improvements in FPG in the normal weight subjects in our study occurred in the context of an intervention focusing on physical activity, a healthy dietary pattern, and the prevention of weight gain and in the absence of significant decreases in body weight, BMI, or waist circumference. Our findings, from a US population with prediabetes, are consistent with those from studies conducted in Asia. In the China Da Qing Diabetes Prevention Study,8,18 577 Chinese adults with impaired glucose tolerance were randomly assigned to a control group or 1 of 3 lifestyle intervention groups, namely, diet, exercise, or diet plus exercise. Participants were not excluded based on baseline BMI and, like our study, weight loss was only encouraged in participants with a baseline BMI ≥25.0 kg/m2. During 30 years of follow-up, compared with the control group, the combined intervention group had a median delay in diabetes onset of 3.96 years, fewer cardiovascular disease events, a lower incidence of microvascular complications, fewer cardiovascular disease deaths, and fewer all-cause deaths, leading to an increase of 4.82 years in median survival and a mean increase of 1.44 years in life expectancy. 8 The interventions were accompanied by only small changes in body weight and the investigators hypothesized that the delay in diabetes onset in the combined intervention group versus the control group was likely to be mainly attributable to factors such as changes in dietary composition and increased physical activity rather than weight loss. 8 Similar observations and conclusions were reported from the Indian Diabetes Prevention Programme, where lifestyle intervention focused on physical activity and dietary composition rather than weight loss in normal weight participants and was equally effective in preventing incident diabetes in Asian-Indian participants with impaired glucose tolerance and a baseline BMI <25.0 kg/m2 versus those with a baseline BMI ≥25.0 kg/m2. 9
The present findings confirm in a US population that evidence-based, technology-enabled, LHC delivered virtually via the telephone and internet, can be an effective tool to help manage prediabetes in normal weight, overweight, and obese participants, with 45% of the subjects normalizing their FPG after a mean of ~6 months of intervention. Moreover, in our study the benefits of the LHC program extended beyond FPG management and included improvements in multiple cardiovascular disease risk factors in the normal weight, overweight, and obese participants. This observation is of significant potential clinical importance, especially in view of the high baseline prevalence of the metabolic syndrome in our subjects, including even those in the normal weight cohort. However, unlike improvements in FPG, which occurred without the use of diabetes medication, the observed improvements in blood pressure and serum lipids and lipoproteins cannot be attributed to lifestyle intervention alone in our study because the LHC program specifically included referral of participants to their physicians for consideration of medication changes to optimize blood pressure and lipid management in accordance with national clinical guidelines. 12 Additional research is needed to clarify the effect of diabetes prevention programs on multiple cardiovascular disease risk factors, especially in normal weight participants with prediabetes who have been underrepresented in prior studies and including the potential role that such programs can play in the identification and referral of participants in need of evidence-based cardioprotective medication management.
The major strength of the present study is the involvement of a large number of free-living US men and women who participated in an already established LHC program without interacting with a team of researchers (ie, an “effectiveness” rather than “efficacy” study). Potential limitations include the relatively short duration of follow-up, nonrandomized study design, absence of a no-intervention control group, and regression to the mean. Additionally, the observed differences in FPG goal attainment among the normal weight, overweight, and obese cohorts in our study can be attributed, at least, in part, to differences in the distribution of baseline FPG values. Finally, we did not have sufficient data to include analyses on energy intake and the specific types and intensity of exercise actually performed, which are also considered to be important lifestyle-related determinants of changes in FPG.20,21
Conclusions
Although further research is warranted, our observation in a US population that LHC is at least as effective in managing FPG in normal weight adults with prediabetes versus those who are overweight or obese is consistent with observations from studies conducted in Asia.8,9 Considering the high prevalence of prediabetes, 1 the finding that almost 20% of US adults with prediabetes are not overweight or obese, 4 and the well-documented adverse clinical sequelae and related health care costs of prediabetes,2,3,6 our results have potentially important clinical and public health implications. In particular, BMI-based eligibility criteria for participation in reimbursable diabetes prevention programs should be reevaluated together with current outcomes-based incentive payments to providers of diabetes prevention programs that focus primarily on weight loss.10,22
Footnotes
Acknowledgements
The authors acknowledge the data analytics and statistical assistance of Dr Richard Salmon, INTERVENT International, LLC.
Authors’ Note
This article contains a subset of data from an abstract presented at the American Diabetes Association’s 80th Scientific Sessions, June 2020.
Data are available on reasonable request. Requests for access to the study data should be submitted to Dr Neil Gordon at
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MKS is a non-salaried research associate of a population health management company (INTERVENT International, LLC). NFG, KSR, BSW, and GCF are members of INTERVENT International, LLC. TLK is an employee of INTERVENT International, LLC. MKS and TLK have family members who are members of INTERVENT International, LLC. DC has no potential conflicts of interest relevant to this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The research protocol was approved by the University of the Witwatersrand Human Research Ethics Committee (Medical; Clearance Certificate No. M200844).
Informed Consent
All subjects provided informed consent.
Trial Registration
Not applicable, because this article does not contain any clinical trials.