Abstract
Keywords
‘Nonetheless, it is well established that being physically active and having a high level of fitness (ie, aerobic capacity and muscular strength) are positively linked to cardiovascular and overall health . . .’
Introduction
According to the World Health Organization, cardiovascular disease (CVD) is the leading cause of death in the United States and globally. 1 People living with HIV (PLWH) experience the same chronic conditions that affect the general population, including CVD. Several studies have shown that CVD risk is significantly higher in PLWH compared with HIV-negative individuals.2-8 As in the general population, CVD among PLWH is related to traditional risk factors, such as age, smoking, male sex, and being overweight.9,10 In addition, the combined effects of the HIV infection and antiretroviral therapy (ART) cause dyslipidemia, accumulation of visceral fat, inflammation, and metabolic alterations,11-13 thus further increasing CVD risk.
The Framingham Risk Score (FRS) is a common profiling tool that is used to estimate the 10-year risk of CVD using traditional factors (ie, age, gender, total cholesterol [T-Chol], high-density lipoprotein cholesterol [HDL-C], smoking status, systolic blood pressure [SBP], diabetes, and use of blood pressure medication), and it has been validated in Caucasians and African Americans in the United States. 14 The FRS is considered the safest of all profiling tools to predict coronary artery disease and major CV events because it uses clinical characteristics to calculate its risk score. 15
Most studies in PLWH using FRS to predict the risk of CVD have only considered the aforementioned clinical variables that are used to score it.16-20 A few studies have found additional relationships between insulin resistance and various indicators of body composition (eg, waist circumference, body mass index [BMI], and visceral adipose tissue) and their contributions to increased CVD risk as per FRS.10,21-23 However, it is unclear how other variables, such as physical fitness and inflammatory markers, may be related to FRS in PLWH. Nonetheless, it is well established that being physically active and having a high level of fitness (ie, aerobic capacity and muscular strength) are positively linked to cardiovascular and overall health and associated with reduced CVD risk.24,25 Our previous work and others have demonstrated that PLWH are much less aerobically fit and weaker than their age- and gender-matched counterparts.26-28 Many studies have shown that hs-C-reactive protein (hs-CRP) is elevated (>3.0 mg/L) among PLWH, and its role as an inflammatory marker in the pathogenesis of CVD is relatively consistent26,29-31; but how hs-CRP relates to FRS has not been established in HIV.
Therefore, the purpose of the study is to determine if physical fitness, as measured by aerobic capacity and upper- and lower-body strength, and inflammation, as measured by hs-CRP, are related to FRS in a mostly minority sample of PLWH. Our analysis may be the first to evaluate if these variables are associated with CVD risk as per FRS in HIV-infected adults. Given that some of the predictors of FRS (eg, smoking status, SBP, and HDL-C), 9 physical fitness, 32 and hs-CRP30,33 are also already known to be different between men and women, we stratified analyses by gender to determine these relationships for each group.
Methods
Healthy Living for Better Days was a 12-month community-based exercise training program aimed at improving the CV health of PLWH of predominantly low socioeconomic status. Recruitment began in March 2013, and enrollment closed in November 2013. The program was approved by the Institutional Review Board for Human Subjects Research at the University of Miami Miller School of Medicine (ID No. 20121029) and was conducted in accordance with the ethical standards of the Declarations of Helsinki. All participants signed informed consent and HIPAA forms prior to commencing the program.
Participants
A total of 89 HIV-positive patients were enrolled in the program after referral from the Adult Special Immunology Clinic at University of Miami Miller School of Medicine/Jackson Health System and other local HIV clinics. Inclusion criteria were as follows: (1) confirmed HIV infection as established by external laboratory reports from the referring physician, (2) men or women ≥18 years of age, and (3) ability to attend supervised weekly exercise sessions at University of Miami’s Medical Wellness Center. Exclusion criteria were (1) any medical condition in which exercise would be contraindicated and (2) pregnancy, for women.
Assessments
Physical characteristics, physical fitness measures, blood sampling, and questionnaires were all obtained by trained investigators using standard techniques. Participants completed a sociodemographics and health history questionnaire that included gender; race/ethnicity; employment status; household income; daily smoking (none, less than a half pack, a half to 1 pack, or more than 1 pack); the use of lipid-lowering, antihypertensive, diabetes, and/or protease inhibitor medications; and duration of HIV infection.
Weight and height were recorded to the nearest 0.1 kg and 0.1 cm, respectively, to calculate BMI. Waist circumference was measured in inches at the narrowest circumference halfway between the lowest rib and the iliac crest, and hip circumference was measured at the widest point of the pelvis. Waist circumference was divided by hip circumference to calculate the waist to hip ratio (WHR). SBP, diastolic blood pressure (DBP), and heart rate (HR) were measured with an automatic oscillometric device (Omron BP monitor). Three readings were made with the participants seated after they had rested for 5 minutes. The average of the second and third readings was used in the analysis. Pulse pressure was calculated by subtracting DBP from SBP.
Blood was drawn during the morning in at least an 8-hour fasted state. Chemistry and immunoassays were performed by an automated analyzer (Roche Cobas-6000; Roche Diagnostics, Indianapolis, IN) utilizing the manufacturer’s reagents and following manufacturer’s instructions. Serum was used to quantify hs-CRP in a high-sensitivity latex particle–enhanced immunoturbidimetric assay, with a detection limit of 0.1 mg/L with intra-assay and interassay coefficients of variations of 1.1% and 2.2%, respectively. According to the American Heart Association guidelines, participants were categorized at the following levels for risk of developing CVD: (1) low, <1.0 mg/L; (2) average, 1.0 to 3.0 mg/L; and (3) high, >3.0 mg/L. 34 T-Chol and triglycerides (TGs) were determined in serum or plasma by enzymatic, colorimetric assays. Intra-assay and interassay coefficients of variation were 0.7% and 1.8%, respectively, for T-Chol and 0.9% and 2.3%, respectively, for TGs. HDL-C was measured using a third-generation homogeneous enzymatic colorimetric assay, and the intra-assay and interassay coefficients of variations were 0.6% and 1.9%, respectively. Low-density lipoprotein cholesterol (LDL-C) levels were calculated using the Friedewald formula: LDL-C = T-Chol − (HDL-C − 0.20 × TG). Very-low-density lipoprotein cholesterol (VLDL-C) values were calculated as TG/5, unless TG exceeded 400 mg/dL, in which case, it was measured by enzymatic methods (Vitros 750 Analyzer, Johnson & Johnson, New York, NY). Fasting glucose was measured by the hexokinase method with intra-assay and interassay coefficients of variations as 1.9% and 2.7%, respectively. Fasting insulin was measured by laboratory analysis. Homeostatic model assessment for insulin resistance (HOMA-IR) was calculated according to the following formula: Fasting insulin (µU/L) × Fasting glucose (nmol/L)/22.5. 35 Hemoglobin A1C (HbA1C) in whole blood was measured by high-performance liquid chromatography using a fully automated analyzer (Variant II Hemoglobin Testing System, BioRad, Richmond CA), and intra-assay and interassay coefficients of variations were 1.7% and 2.0%, respectively.
Maximum oxygen consumption (VO2max) was estimated with the Rockport One-Mile Fitness Walking Test using American College of Sports Medicine (ACSM) guidelines.32,36 The test was modified for indoor performance with participants walking 1 mile on a treadmill as quickly as possible. Participants could modify the treadmill speed (mph) if necessary. HR was measured for 10 s immediately on completion by palpating the radial artery. Age, gender, body weight, and walk time were used in a regression equation to calculate VO2max. ACSM reference values were then used to categorize participants on VO2max (ie, aerobic capacity) by age and gender into very poor, poor, fair, good, excellent, or superior. 32
Muscular strength was measured using the ACSM protocol for 1 repetition maximum (1-RM) testing. 32 Participants completed a maximum of 4 trials of 10, 8, 6, and 3 repetitions with rest periods between 2 and 4 minutes between trials. The initial weight was selected within the participant’s perceived capacity (50%-70%), and resistance was progressively increased until the participant reached the maximum. The final maximum weight lifted successfully 1 time with proper form for bench press (upper-body strength) and leg press (lower-body strength) was recorded as the 1-RM. ACSM reference values were then used to categorize participants on strength by age and gender into very poor, poor, fair, good, excellent, or superior for the upper body (1-RM on the bench press divided by body weight) and well below average, below average, average, above average, or well above average for the lower body (1-RM on the leg press divided by body weight). 32
The Framingham Risk Score
The 10-year risk of a CVD event was scored via an online calculator—https://www.framinghamheartstudy.org/risk-functions/cardiovascular-disease/—using age, diabetes (yes or no), smoking (yes or no), hypertension (yes or no), SBP, T-Chol, and HDL-C as the predictors. A fasting glucose level ≥126 mg/dL or already undergoing treatment for diabetes was classified as diabetic. Two participants who were younger than 30 years of age (the minimum age required to compute a score) were excluded from the analysis, bringing the final sample size to 87. Each participant was then categorized as having low (<10%), intermediate (≥10%-19.9%), or high (≥20%) risk of a CVD event. 14 The Framingham heart/vascular age was recorded for each participant as part of the information provided by the calculator. The participant’s heart/vascular age was subtracted from the chronological age to get a difference between the 2 values, which was used to create categories for being younger, the same age, or older.
Statistical Analyses
Frequency and descriptive statistics were calculated on all variables. The χ2 test was used to examine the degree of association of categorical variables with gender. All continuous variables were analyzed with the independent-samples
Results
Sociodemographics, FRS, Health History, and Medication Use
A total of 47 women and 40 men were included in this analysis. See Table 1 for the descriptive information of the sample by gender for age, race/ethnicity, employment status, and annual household income. All were nonsignificantly different between the genders, except for race/ethnicity, because men were more likely to be white or Hispanic, and women were more likely to be black (χ2[3] = 8.0;
Sociodemographic Characteristics of the Sample.
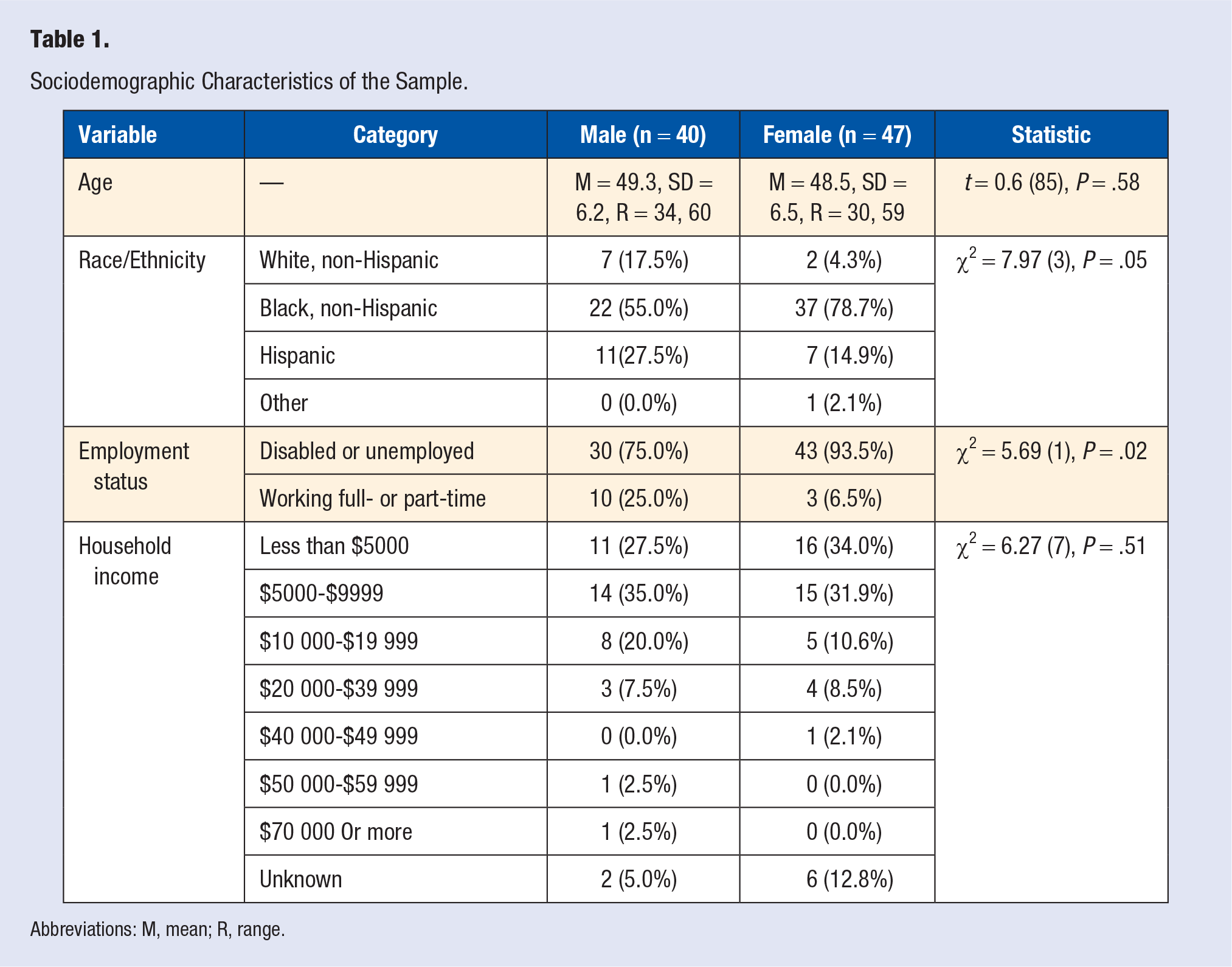
Abbreviations: M, mean; R, range.
See Table 2 for the descriptive information of the sample by gender for FRS, FRS categories, the difference between age and Framingham heart/vascular age, the number of years since HIV diagnosis, smoking status, and the use of lipid-lowering, antihypertensive, diabetes, and protease inhibitor medications. Men had a more than double FRS of a potential CVD incident in the next 10 years compared with women (
Health History Characteristics of the Sample.
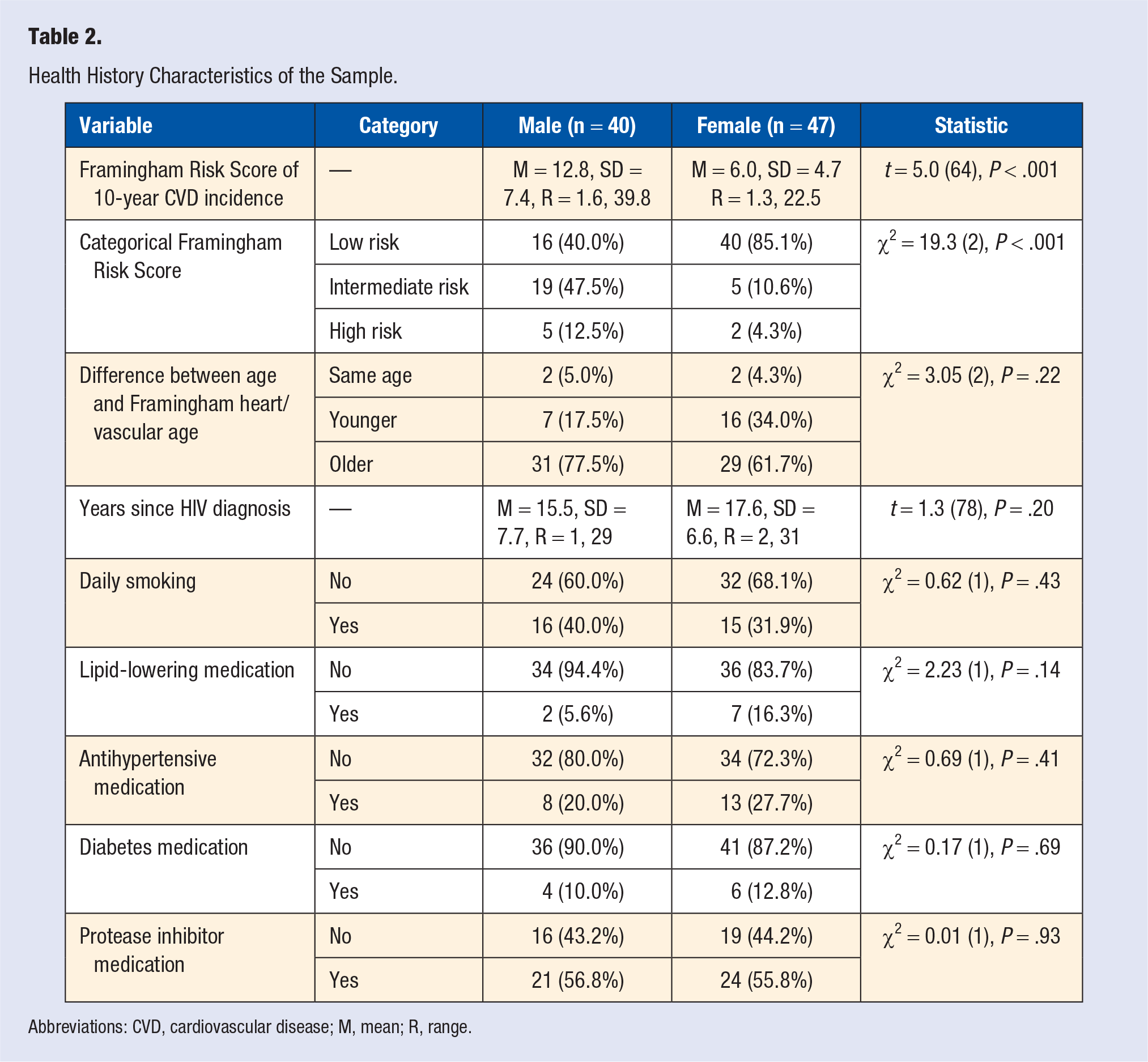
Abbreviations: CVD, cardiovascular disease; M, mean; R, range.
Physical Characteristics and Fitness
Table 3 shows the descriptive statistics for SBP, DBP, pulse pressure, HR, VO2max, 1-RM upper-body strength, 1-RM lower-body strength, waist circumference, WHR, and BMI by gender. Waist circumference (
Physical Characteristics of the Sample. a
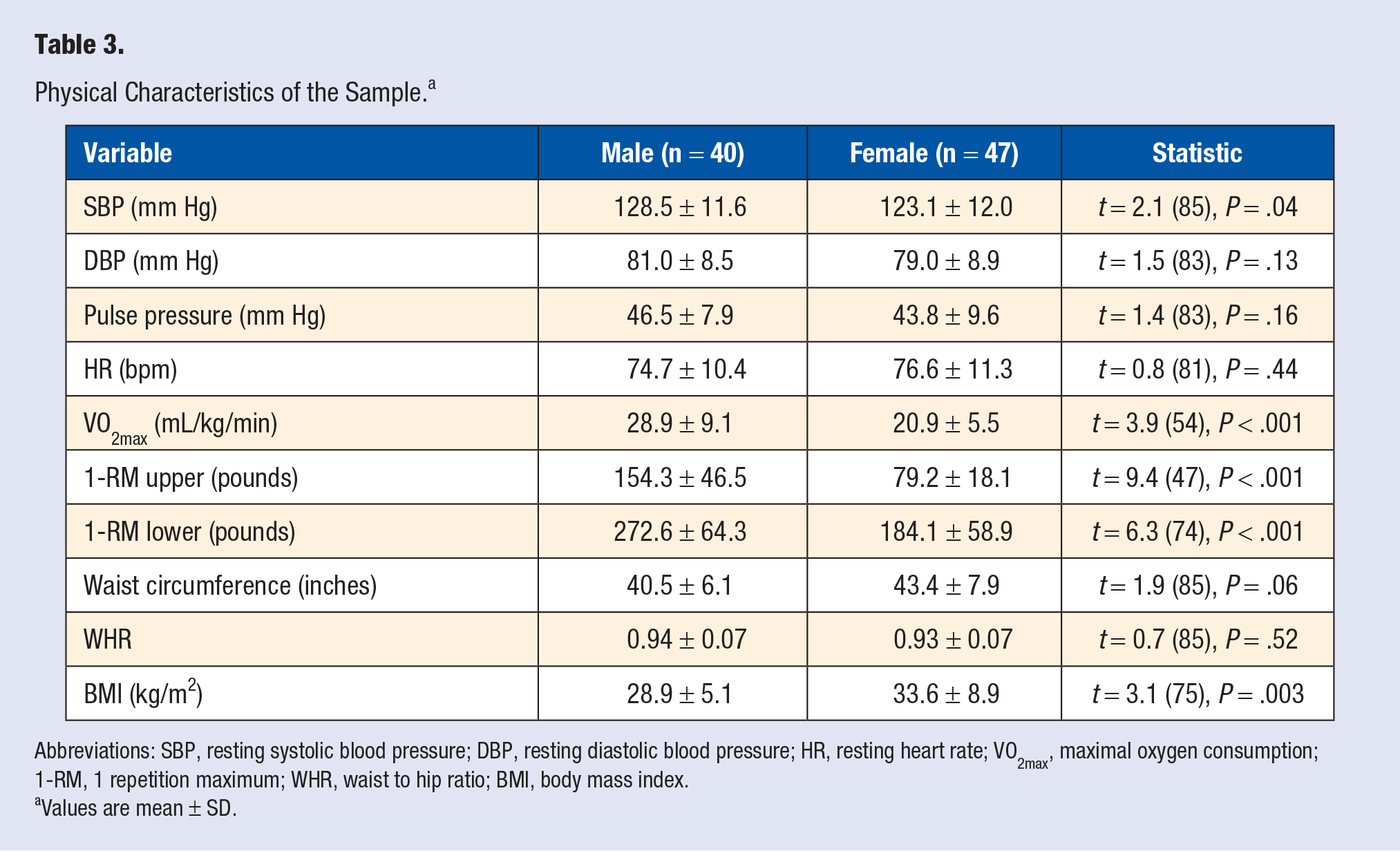
Abbreviations: SBP, resting systolic blood pressure; DBP, resting diastolic blood pressure; HR, resting heart rate; VO2max, maximal oxygen consumption; 1-RM, 1 repetition maximum; WHR, waist to hip ratio; BMI, body mass index.
Values are mean ± SD.
Serum Biomarkers
Table 4 shows the descriptive statistics for T-Chol, LDL-C, HDL-C, VLDL-C, non–HDL-C, T-Chol:HDL-C, TG, hs-CRP, glucose, HbA1C, insulin, and HOMA-IR for both genders. HDL-C (
Serum Biomarkers of the Sample. a
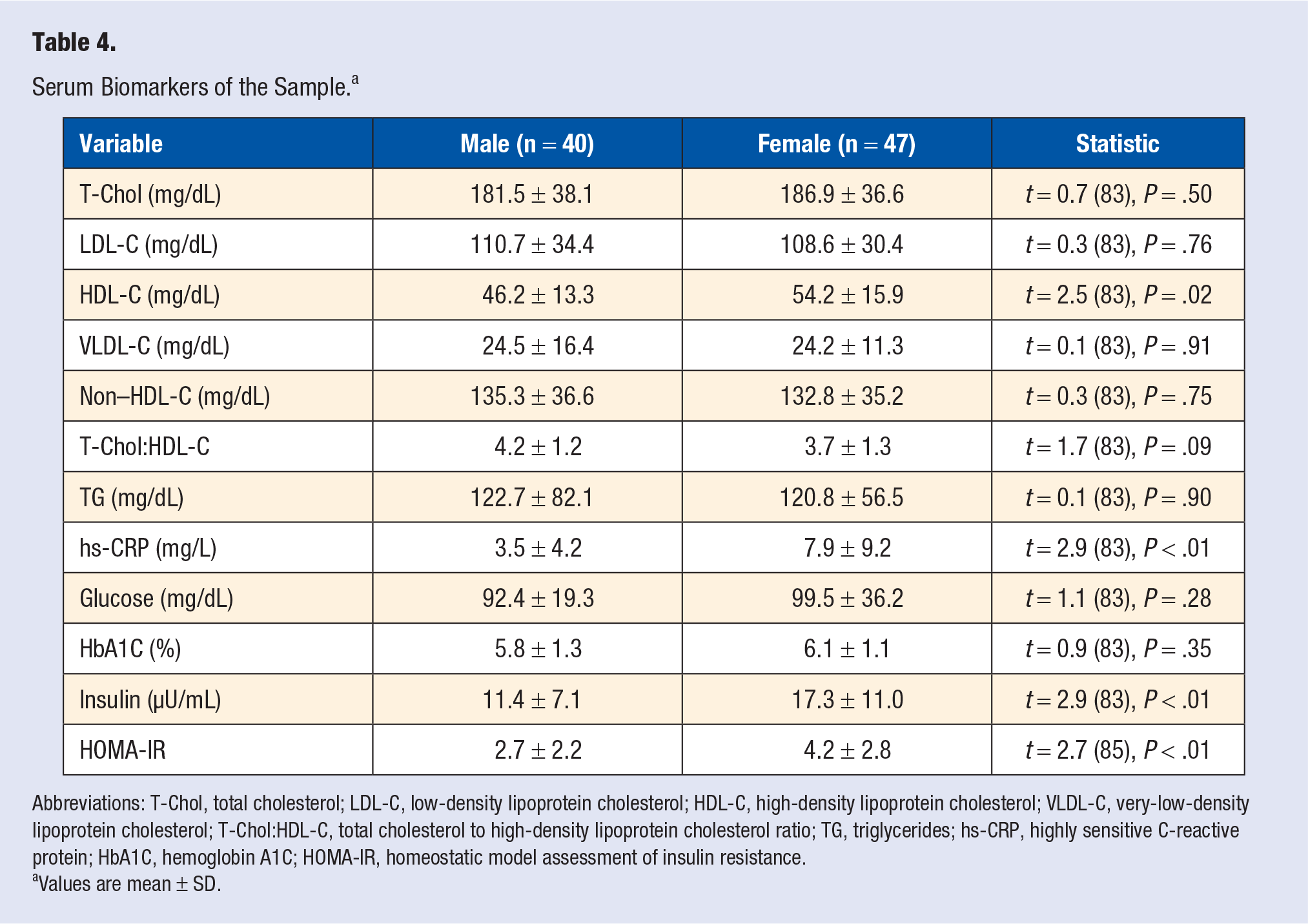
Abbreviations: T-Chol, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; VLDL-C, very-low-density lipoprotein cholesterol; T-Chol:HDL-C, total cholesterol to high-density lipoprotein cholesterol ratio; TG, triglycerides; hs-CRP, highly sensitive C-reactive protein; HbA1C, hemoglobin A1C; HOMA-IR, homeostatic model assessment of insulin resistance.
Values are mean ± SD.
Physical Fitness and Inflammation by FRS
The categorical breakdowns are shown for aerobic capacity, upper- and lower-body strength, and hs-CRP level by FRS category for men (see Table 5) and women (see Table 6). Most men and women were categorized as very poor or poor on aerobic capacity, very poor or poor on upper-body strength, and well below average or below average on lower-body strength. However, these frequencies were statistically unrelated to FRS categories. For men, a statistical trend was noted for the relationship between hs-CRP level of CVD risk and FRS category (χ2 = 7.72[4];
Physical Fitness and Inflammation by Framingham Risk Score for Men. a
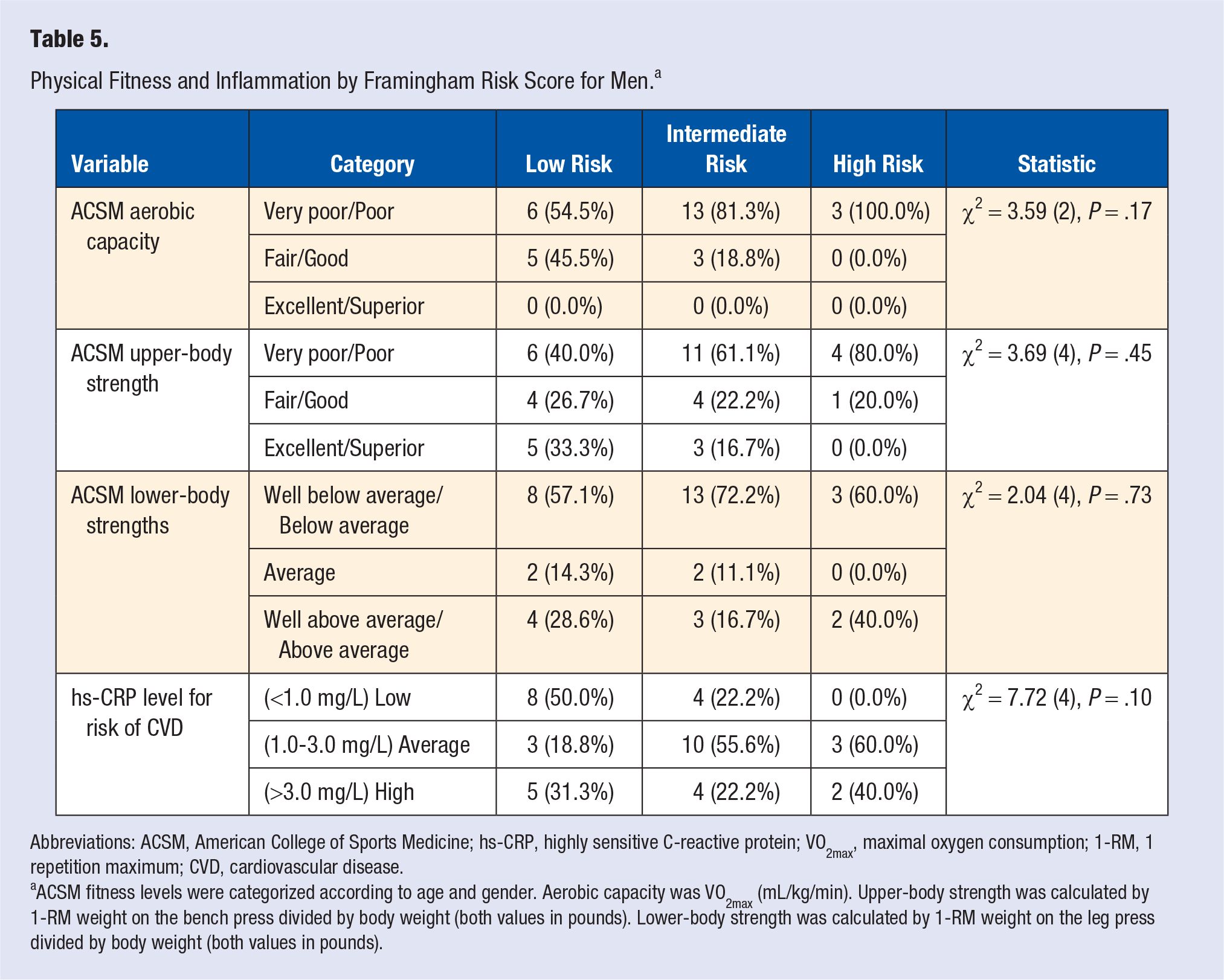
Abbreviations: ACSM, American College of Sports Medicine; hs-CRP, highly sensitive C-reactive protein; VO2max, maximal oxygen consumption; 1-RM, 1 repetition maximum; CVD, cardiovascular disease.
ACSM fitness levels were categorized according to age and gender. Aerobic capacity was VO2max (mL/kg/min). Upper-body strength was calculated by 1-RM weight on the bench press divided by body weight (both values in pounds). Lower-body strength was calculated by 1-RM weight on the leg press divided by body weight (both values in pounds).
Physical Fitness and Inflammation by Framingham Risk Score for Women. a
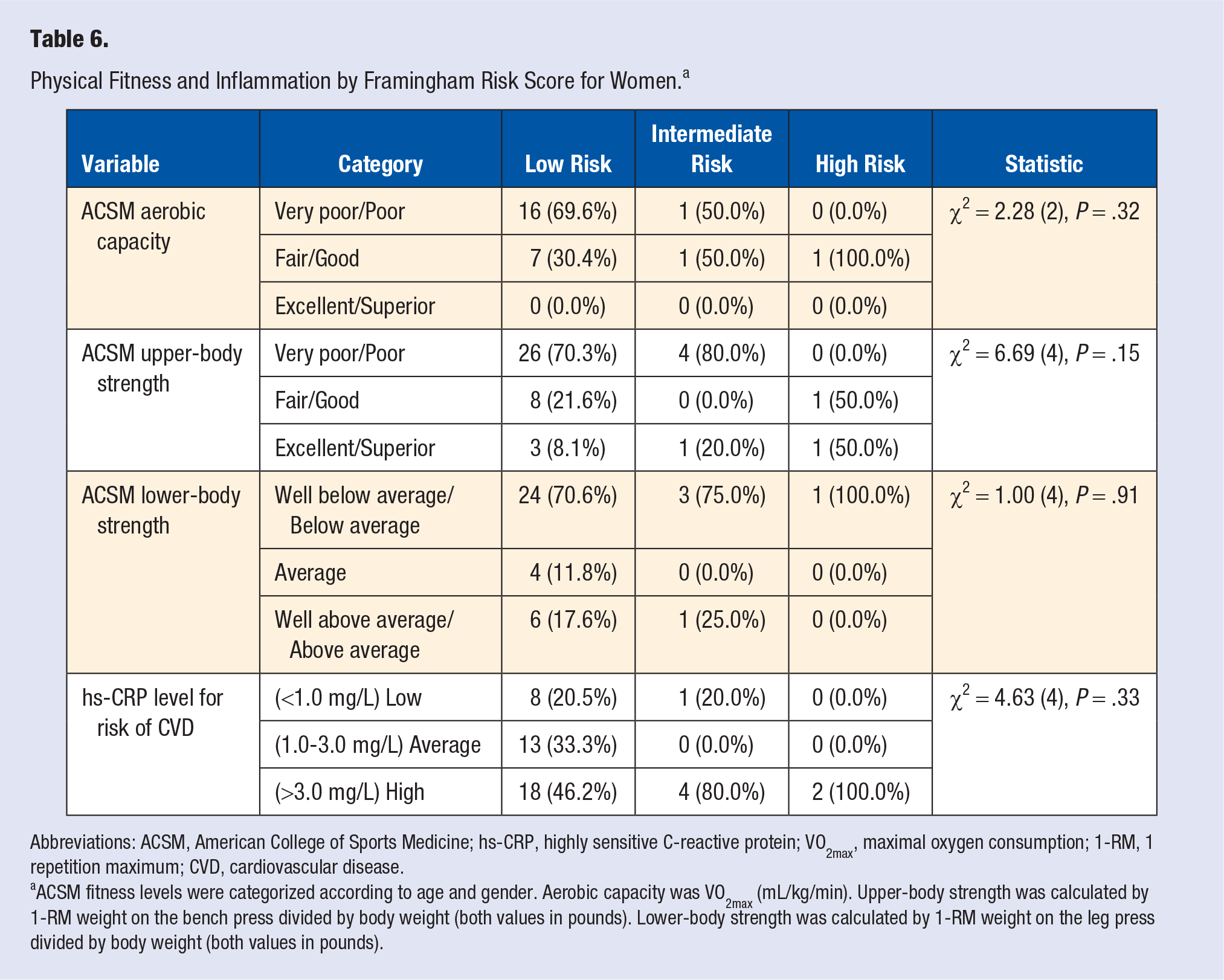
Abbreviations: ACSM, American College of Sports Medicine; hs-CRP, highly sensitive C-reactive protein; VO2max, maximal oxygen consumption; 1-RM, 1 repetition maximum; CVD, cardiovascular disease.
ACSM fitness levels were categorized according to age and gender. Aerobic capacity was VO2max (mL/kg/min). Upper-body strength was calculated by 1-RM weight on the bench press divided by body weight (both values in pounds). Lower-body strength was calculated by 1-RM weight on the leg press divided by body weight (both values in pounds).
Correlation Analysis and Linear Regression Model for Men
The FRS was correlated with physical characteristics and fitness variables and serum biomarkers that are not used in its scoring. Statistically significant relationships were found with VO2max (
Correlation Analysis and Linear Regression Model for Women
The FRS was correlated with the same set of assessments as for the men. Statistically significant relationships were found with T-Chol:HDL-C (
Discussion
Although FRS underpredicts the incidence of CVD in PLWH,37,38 by as much as 50% according to the Veterans Aging Cohort Study, 7 it has still been commonly used in HIV studies because of its relationships to clinical characteristics in predicting the 10-year risk of CVD events.10,23,39,40 The exact relationships between Framingham risk factors and mechanisms of action that explain the increased risk of CVD among PLWH, such as inflammation 29 and endothelial dysfunction, 41 are still debatable, but the heightened risk is thought to be a combination of the effects of viral load, ART, and comorbid disease. 7 In the current study, we showed no association between the FRS and selected variables that could be expected to lead to increased CVD risk, such as duration of HIV infection, use of protease inhibitors, hs-CRP, and LDL-C. Nonetheless, we did show a significantly greater FRS in male participants whose 10-year calculated risk was more than double that of female participants (12.8 vs 6.0). Additionally, according to FRS categories, men were more likely to be at intermediate or high risk of a 10-year event of CVD (60%), whereas most women were at low risk (85%). These findings are consistent with prior work showing that HIV-positive men have greater CVD risk than their female counterparts. 9 Of note, the only FRS predictors statistically different between men and women in the current study were SBP (higher in men) and HDL-C (higher in women), whereas the other variables were similar. These same SBP and HDL-C gender differences were also noted in a previous study. 9
One of the primary aims of the current study was to determine if physical fitness was related to the FRS in these HIV+ patients. We showed that greater aerobic capacity was strongly associated with a lower FRS for the men, whereas the same relationship did not exist for the women. Also, neither measure of muscular strength was associated with FRS for men or women. In the general population, it is well established that being physically active and having a moderate to high level of cardiovascular fitness (CVF) are associated with many health benefits, including the reduced risk of CVD and overall decreased morbidity and mortality.42-47 Additionally, CVF (measured by the internationally recognized standard of VO2max) is superior to predicting the risk of CVD compared with self-reported physical activity. 48 Thus, our finding that CVF was related to FRS in HIV+ men may be the first of its kind, providing support for an effect of aerobic capacity on the risk of CVD in HIV+ men that is comparable to that in the general population. We categorized men and women on aerobic capacity based on the ACSM norms for age and gender and showed that most of both genders scored as either very poor or poor based on their VO2max values. Our findings are consistent with a meta-analysis and previous results in our own lab showing that PLWH generally have poor CVF.28,49,50
Although muscular strength was not related to FRS in the current study, it is important to note that we assessed upper- and lower-body strength in male and female HIV+ patients, which is not commonly done in this population. As with aerobic capacity, we showed that most men and women rated as either very poor or poor for upper-body strength and well below average or below average for lower-body strength when categorized by ACSM standardized norms for age and gender. These findings are consistent with a meta-analysis and the previous work from our own group showing that PLWH are weaker than the general noninfected population.27,28 This trend continues despite the fact that scientists have been recently calling for a greater emphasis on strength training as part of a comprehensive approach to exercise from a public health perspective because of its impact on many health- and disease-related risk factors across populations. 51 It was also previously noted that ART can cause peripheral neuropathy and limited ability to recruit motor nerves in the upper-body musculature, which may account for the poor upper-body strength of those in the current study while simultaneously arguing for an emphasis on upper-body training in the HIV+ population.28,52
The other primary aim of the current study was to determine if hs-CRP was related to FRS in PLWH. To our knowledge, no other study has correlated hs-CRP with FRS in PLWH, although one other study utilized hs-CRP >3.0 mg/L as part of a group of aggravating factors to recalculate FRS for PLWH who had low or moderate 10-year risk. 53 Additionally, our results were inconsistent with that of others who showed that elevated hs-CRP and HIV are related to an increased risk of acute myocardial infarction. 29 Beyond that study, the information independently linking hs-CRP as an inflammatory marker to CVD events is sparse in the HIV population, but high hs-CRP has been found to be related to increased HIV disease progression in men 54 and death in women. 55 Although a high level of hs-CRP has been linked to greater predictive accuracy of CV events, such as the risk of incident coronary disease, and adds to the predictive capability of other, established risk factors, such as age, T-Chol, and smoking, in the general population,31,34 its effects in HIV are still not entirely certain. This is likely a result of the effects of ART and the virus itself, which are inherently inflammatory themselves, rendering hs-CRP less capable of predicting outcomes such as atherosclerosis. 56
We also found that hs-CRP was more than twice as high among women compared to men, which is consistent with one study showing that hs-CRP became significantly higher in HIV+ women compared with HIV+ men after 96 weeks of efavirenz-based ART. 30 Interestingly, the change in hs-CRP was unrelated to changes in insulin resistance or fasting lipids. However, another study showed that men without coinfection of hepatitis C had higher hs-CRP than women. 33 Thus, the findings in hs-CRP within the HIV population are inconsistent, at best, regarding its impact on CVD risk or outcomes and the difference between genders.
Other than the relationships that we showed between FRS and WHR in men and FRS and HbA1C and T-Chol:HDL-C in women, we also showed that pulse pressure was predictive of FRS in both genders. Although pulse pressure is not as commonly assessed and evaluated for CVD risk as SBP and DBP, another study showed that pulse pressure was higher in HIV+ adults than a noninfected comparison group, and it was related to arterial stiffness. 57 Although the mean pulse pressures of men and women in the current study are considered normal (<60 mm Hg), it is important to note that others have shown that high pulse pressure is independently related to an increased risk of CVD death, 58 and it is a functional marker of organ damage, which is predictive of CV events in hypertensive patients. 59 Thus, our finding that pulse pressure is related to FRS in both HIV+ men and women may be a useful clinical criterion for physicians when evaluating their patients’ risk of CVD.
Limitations
This study showed several interesting and novel findings, but some limitations should be noted. HIV+ patients on ART are a challenging population to study. Many factors are present that could influence CVD risk, including the virus itself, polypharmacy, and the high likelihood of multiple comorbidities. Confounding from multiple medications, comorbid complications, HIV disease, and/or opportunistic infections cannot be entirely ruled out. Additionally, many potentially important variables that could be related to CVD risk were not available—for example, CD4+ cell count, history of opportunistic infections, oral health, injection drug and/or cocaine use, and overall stress. Thus, it is unknown how these variables could have affected our results. Although hs-CRP level was unrelated to FRS, our data are from a sample of mostly black, non-Hispanic patients, which is unique, because most studies that have determined normative values for hs-CRP have been conducted in white, non-Hispanic individuals. 34 The FRS has primarily been validated in non-Hispanic black and Caucasian Americans, so it is unclear if FRS is accurate for the 20% of our sample who are Hispanic. 60 An additional limitation of this study is that FRS tends to underestimate CVD risk in PLWH according to others,7,37,38 but in one study using aggravating risk factors—for example, hs-CRP >3.0 mg/L, presence of metabolic syndrome, and family history of CVD—FRS was thought to overestimate the 10-year risk of CV events. 53 Moreover, one threat to the validity of the data is the lack of a comparison group to a non–HIV-infected group of individuals. The next step in the determination of the usefulness of FRS in predicting the risk of CVD would be to replicate the current analysis in a larger sample size and expand the additional contextual factors to those mentioned that were not collected in the current study.
Conclusions
HIV+ men and women experience CVD risk factors differently, which has been shown in other studies of PLWH and in the general population. In summary, we showed several significant and unique findings, including the following: (1) men had a more than double the 10-year risk of CVD compared with women; (2) aerobic capacity was significantly related to FRS in men but not in women; (3) upper- and lower-body strength were unrelated to FRS for men or women; (4) most of these individuals had low levels of physical fitness compared with age- and gender-matched normative individuals; (5) hs-CRP was more than twice as high in women as in men, (6) hs-CRP was unrelated to FRS, and (7) pulse pressure was related to FRS for both men and women. Despite the interesting results, predicting the risk of CVD is not simple for the HIV population because of the complexities of HIV infection, ART, and other medications that modify the health status of the individual. Nonetheless, considering aerobic capacity and pulse pressure along with other traditional risk factors and lifestyle variables may help clinicians make better treatment decisions for ultimately reducing CVD events, although additional research is warranted to better understand why PLWH generally have poor physical fitness—for example, mitochondrial toxicity caused by ART use. 61 Additionally, proper exercise recommendations based on the standard ACSM prescription for frequency, volume, intensity, and mode of exercise should be utilized by physicians and other clinicians to address the typically very poor fitness levels found in PLWH. 62
Footnotes
Acknowledgements
We are thankful to all the volunteers who participated in this study. This intervention was supported by AstraZeneca HealthCare Foundation’s Connections for Cardiovascular HealthSM (CCH) program. The CCH program funds charitable work, not research, that addresses cardiovascular health issues within the United States and its territories. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and have not been reviewed for approval by the AstraZeneca HealthCare Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the Institutional Review Board for Human Subjects Research at the University of Miami Miller School of Medicine (ID No. 20121029) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all participants in this study.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
