Abstract
Pediatric obesity is complex—in every way. The pathogenesis, which is a result of a multitude of factors combined with social and environmental factors makes it a crisis with overwhelming challenges. It is difficult to treat pediatric obesity; however, guidelines attempt to clarify how to approach treatment. These strategies are mostly interventions in lifestyle because only 1 medication is approved for treating obesity in children. The approach to the treatment of pediatric obesity will need to be a one that relies on the available data, a continued drive for additional research and publication, sharing of practice experience, and continued efforts at prevention.
‘Children may be taking medications that can cause weight gain; if this occurs, a risk versus benefit analysis needs to be performed . . .’
In a world where there are never-ending, myriad medications to treat a multitude of health conditions, childhood obesity only has 1 Food and Drug Administration (FDA)-approved pharmacological possibility (starting at the age of 12)—a stark contrast to what one might expect in response to an epidemic. In 2015-2016, it was estimated that the prevalence of obesity among youth 2 to 19 years old was 18.5%. The incidence was higher in non-Hispanic black and Hispanic youth compared with other races. 1 Health professionals and public health advocates speak to this via a plethora of platforms—scientific journals, the media, TED talks, and documentaries, to name a few. Skinner et al 2 used the NHANES data from 1999-2016 and found that even with the public health initiatives, the prevalence of childhood obesity continues to be on the rise, especially in adolescents and children aged 2 to 5 years. 2 The World Health Organization depicts the current crisis perfectly: “obesity is one of today’s most blatantly visible—yet most neglected—public health problems.” 3
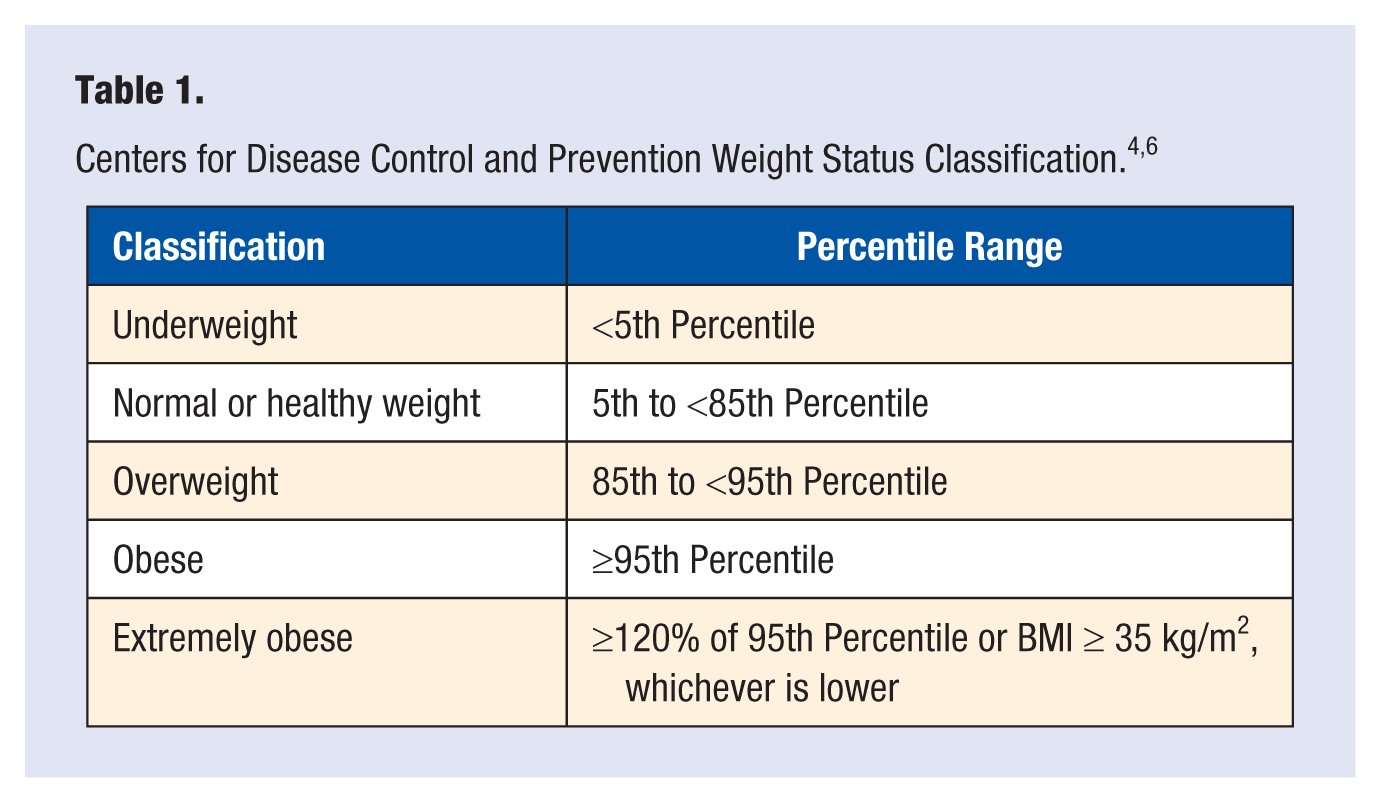
The classification of weight status per the United States Centers for Disease Control and Prevention (CDC) is defined in Table 1. 4 Body mass index (BMI) is calculated in children ≥2 years of age, and the CDC normative BMI percentiles are used to diagnose overweight or obesity. Considering that BMI is an imperfect measurement, clinicians also need to consider that according to race/ethnicity, BMI may correlate risk of comorbidities differently in certain racial/ethnic groups and that BMI may be increased in those with higher muscle mass.4,5
Causes of Obesity
The pathogenesis of obesity is complex and involves multiple factors—the ideology that a calorie is a calorie is no longer foundational when attempting to understand the multiple variables that contribute to obesity. It is an interplay of the following factors: molecular, genetic, developmental, behavioral, and environmental. 7 In addition to the various potential abnormalities within the body, weight gain may occur because of other health conditions or medications. Children may be taking medications that can cause weight gain; if this occurs, a risk versus benefit analysis needs to be performed to determine whether the medication causing the weight gain can be discontinued and/or changed to one that does not cause weight gain. Unfortunately, sometimes there are no alternative medications, and hence, the weight gain needs to be addressed with lifestyle interventions. Some of the more common therapeutic classes that may contribute to weight gain include the following: glucocorticoids, which can stimulate appetite and increase the desire to eat more protein and carbohydrates; diabetes medications, including insulin and sulfonylureas; and second-generation antipsychotics (SGA) such as risperidone, olanzapine, and quetiapine. Although these SGAs have fewer motor effects than those previous classed, they may cause weight gain and insulin resistance and increase lipid levels. Additionally, antidepressants such as citalopram and escitalopram may increase BMI and visceral fat as also certain antiepileptic drugs such as valproic acid, gabapentin, pregabalin, and vigabatrin. It is thought that the propensity for an individual to have weight gain secondary to some of these medications is a combination of the actual medication with genetics, neurotransmitters, hormones, and lifestyle. 8
Approach to Treatment
Although there is a good amount of research about different methods of preventing obesity, there is no clear guidance about the treatment strategy to address childhood obesity. In general, it is the health care practitioner’s clinical judgment that is used when conversing with the child and family about dietary and lifestyle habits. Because of numerous factors, including social barriers, early diagnosis and referral for treatment is often delayed, and sometimes, the clinicians who are treating childhood obesity do not have the clinical support or adequate training that is necessary to treat this complex issue. Although there are more pediatric weight management centers and specialists who provide surgery for adolescents, the overall available locations of these specialty practitioners is sparse. The Obesity Medicine Association created the Pediatric Obesity Algorithm in 2016, an “evidence-based roadmap for the diagnosis and management of children with obesity.” 9 It describes evidence-based approaches to the following: initial assessment, physical exam, focused review of systems, diagnostic work-up, management with age-specific guidance, comorbidity management, and medication and surgical approach. 9
The Endocrine Society published evidence-based guidelines in January 2017, discussing the assessment, treatment, and prevention of childhood obesity (updated from the previous 2008 version). These guidelines include recommendations about the following: diagnosis, genetic testing for obesity syndrome, prevention, and treatment, including lifestyle considerations, psychosocial comorbidities, pharmacotherapy, and surgery. 10 Although both the Pediatric Obesity Algorithm and the Endocrine Society guidelines discuss the place of medications in the treatment for pediatric obesity, the actual options for pharmacotherapy are dismal.
Pharmacotherapy
In 2007, a review article about the pharmacological agents available began with the story of dexfenfluramine, a medication that was touted as “weight-reducing efficacy and good tolerability . . . [with side effects] generally mild and transient” 11 . This medication was found to be correlated to valvular heart disease and primary pulmonary hypertension in some patients, and it was taken off the market 18 months after its approval. When the article was published, it discussed orlistat, sibutramine, metformin, and rimonabant. 11 Rimonabant, a selective CB1 endocannabinoid receptor antagonist, which had been approved in Europe, failed to receive FDA approval and, subsequently, was also removed from the market worldwide because of psychiatric side effects, including an increased risk for suicidal ideation. Then in 2010, sibutramine, which reduced hunger and increased satiety by acting centrally to inhibit reuptake of serotonin, norepinephrine, and dopamine, was also taken off the market because of an increased cardiovascular risk. 12
There are no FDA-approved medications to treat obesity in children younger than 12 years, and there is only 1 medication approved for this indication in adolescents between 12 and 16 years old. Although there have been medications that have come to market to treat obesity in adults, this has not been the case in children. Additionally, there is a sparsity in guidance on clinical trial design for pharmacotherapy for pediatric obesity. 13
Orlistat
Orlistat is a gastrointestinal tract lipase inhibitor. It works by decreasing intestinal fat absorption by up to 30%. Orlistat is FDA approved in adolescents 12 to 16 years old based on evidence from a 54-week efficacy and safety study and a 21-day mineral balance study in obese adolescent patients of that age range. The efficacy and safety trial was a randomized, double-blind study that included 539 obese adolescents with a BMI ≥2 units above the 95th percentile. The participants were either given orlistat 120 mg 3 times a day (n = 357) or placebo (n = 182), with a mildly hypocaloric diet (30% fat calories), exercise, and behavioral therapy. There was a mean reduction in BMI of 0.55 kg/m2 compared with an average increase of 0.31 kg/m2 in placebo-treated patients (P = .001). Additionally, secondary measures found a decrease in fat mass and waist reduction in the orlistat group. The adverse effects were as seen in the adult trials—fatty/oily stool, oily spotting, and oily evacuation. Approximately 9% to 50% had mild-moderate gastrointestinal adverse effects in the orlistat group versus 1% to 13% in the placebo group. It is imperative that a fat-soluble vitamin be taken daily, dosed apart from the orlistat. 14
A Brief Discussion About Other Medications That Treat Obesity
There are other medications used in the adult population that treat obesity and have been studied in children. In 2013, a review was published that critically evaluated the limited data available via a PubMed search for the following keywords: children or adolescents, obesity, appetite or satiety, drug or pharmacotherapy, and clinical trial or meta-analysis. A total of 1296 articles were found in the primary search; 74 studies were included in the analysis for the following drugs: phentermine, chlorphentermine, diethylpropion, mazindol, dextroamphetamine, fenfluramine, sibutramine, amantadine, topiramate, recombinant leptin, orlistat, metformin, octreotide, growth hormone, triiodothyronine, and caffeine/ephedrine. It is important to note that most of these medications are not FDA approved to treat obesity in any population; some may have had prior approval but have since been voluntarily withdrawn from the market for safety concerns, and some have an off-label use or are being evaluated because of the potential their mechanism of action has on weight loss. There are very limited studies in children younger than 5 years, and most are <6 months in duration. 15
Metformin has been studied for pediatric obesity but currently only has FDA approval for treating type 2 diabetes in people with diabetes older than 10 years. In addition to diabetes, metformin is approved to be used in polycystic ovary syndrome and prediabetes. Ameer and Weintraub 8 showed a modest reduction in body weight and/or BMI; however, because this was not the primary end point, the data need to be interpreted cautiously. Additionally, phentermine is approved in adolescents/adults >16 years old for short-term weight control, and the combination of topiramate/phentermine is approved in adults only for obesity. 4 Topiramate alone is used on label as an antiepileptic for children ≥2 years old. Finally, glucagon-like peptide receptor agonists or mimetics (GLP-1 RAs) have been approved for the management of type 2 diabetes in adults. Most recently, in July 2019, one GLP-1 RA medication, liraglutide (Victoza), has been approved to treat type 2 diabetes in children ≥10 years of age.16,17 Of note, liraglutide is the same medication that, at a higher dose, has been approved to treat obesity in adults.
Summary of the Clinical Practice Guidelines by the Endocrine Society to Assess, Treat, and Prevent Pediatric Obesity
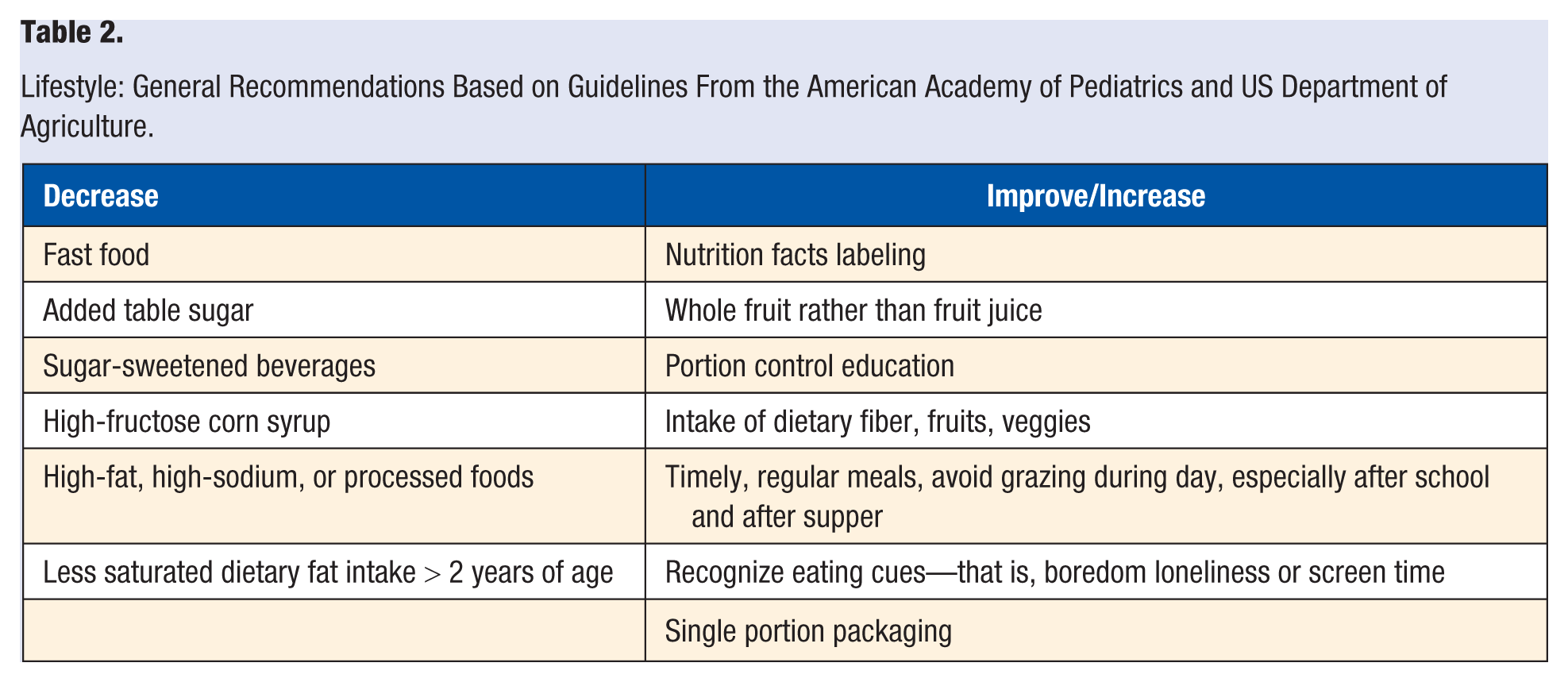
The guidelines offer detailed, evidence-based recommendations about assessing, treating, and preventing pediatric obesity. This brief summary focuses on the approach to treatment. Pharmacotherapy is recommended only after formal intensive lifestyle modifications have failed to limit weight gain or ameliorate comorbidities. They recommend avoiding pharmacotherapy in clinical practice for those <16 years old who are overweight but not obese. Only FDA-approved medications should be used by those with clinical experience, and this should only occur in conjunction with concomitant lifestyle modification program of the highest intensity available. If after 12 weeks at an appropriate dose the patient does not achieve a >4% BMI reduction, the medication should be discontinued. 10 Other articles in AJLM and other journals as well the American Academy of Pediatrics include fundamental recommendations. Lifestyle interventions and primary prevention serve as the foundation for weight management in children and adolescents. 15 Table 2 describes a summary of these recommendations.
Lifestyle: General Recommendations Based on Guidelines From the American Academy of Pediatrics and US Department of Agriculture.
Conclusion: A Nonmedication Prescription
In the pharmacy world, almost every recommendation for a medical problem combines nonpharmacological and pharmacological options—in the case of pediatric obesity, the prescription for nonpharmacological modalities has more evidence and is much more substantive. Including direct interventions at high-risk adult populations to also influence adolescent family member health behaviors has also been found to be related to positive outcomes. 18 Pharmacotherapy may have a role in helping promote weight loss in obese individuals; however, the data in the pediatric population are lacking. Further research needs to occur to better understand the efficacy and safety of pharmacotherapy. Clinical guidelines, an interdisciplinary approach, and allowing for each individual pediatric patient to be evaluated as such will allow a better opportunity to combat pediatric obesity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.