Abstract
‘Patients with coronary artery disease have a negative protein balance, and BCAAs exhibit an anabolic effect on myocardial protein metabolism . . .’
Introduction
Heart failure (HF), a complicated syndrome, is accompanied by cardiac ejection fraction impairment and is one of the most important cardiovascular diseases with high incidence, mortality, and disability.1,2 Its prevalence in adults overall is 1% to 3% and is estimated to be 5% to 9% in individuals aged 65 years and older.
3
Cachexia is a term originating from the Greek
Patients with CC show poor prognosis and disability. 6 Inability of the heart to supply blood causes many symptoms such as shortness of breath, dizziness, fatigue, angina, edema, and ascites. 7 These symptoms result in inactivity and a lack of ability to make changes in lifestyle, which affects patients’ quality of life (QoL).2,8 The results of clinical trials have shown that QoL can be considered a marker of the quality of health care and part of the disease treatment plan; hence, its assessment in chronic disease reveals useful information about the health condition of the patient and can be a useful guide to improve the quality of care. 9 In a cross-sectional study, health-related QoL was studied among elderly patients with HF. The results indicated that patients had higher scores in the dimensions of pain, social function, and emotional performance and moderate scores in terms of physical and mental health. Lower scores were related to physical performance assessment, public health perception, and vitality. 10
Fatigue (as a result of muscle wasting) and anorexia are the most commonly reported symptoms by cachectic HF patients. One-third of patients with mild HF and up to 50% of all HF patients are fatigued, which is defined as recurrent fatigue and is a disability in HF patients, making it difficult to perform daily activities. 11 Decreased appetite in patients with HF can increase this disability and malnutrition and affects the prognosis of this medical condition. 12
Melatonin, as a hormone, ameliorates cachexia complications, including malabsorption, inflammation, and autonomic failure, through multiple mechanisms. The positive effects of melatonin have been proposed for damaged cardiomyocytes, clinical improvement, and survival rates in HF patients. 13 Previous studies have also evaluated the effect of melatonin on symptoms such as poor appetite, fatigue, and depression in patients with cancer cachexia. 14
The branched-chain amino acids (BCAAs) consist of leucine, isoleucine, and valine. These amino acids are known to play a number of roles, such as muscle protein synthesis, insulin secretion, and energy production, through their catabolism. 15 In a previous study, it was suggested that BCAAs counteract the effects of anorexia by competing with tryptophan for entry through the blood-brain barrier. 16 BCAAs improve cardiac function in global ischemia of isolated rat hearts. 6 Patients with coronary artery disease have a negative protein balance, and BCAAs exhibit an anabolic effect on myocardial protein metabolism, whereas leucine attenuates myocardial infarction in mice.6,17 These results show that BCAAs may protect the heart from myocardial ischemic injury.
Considering that CC is a multifactorial clinical condition, previous studies suggested that complicated therapeutic protocols are needed to improve features of cachexia such as QoL, fatigue status, and appetite in CC patients. The present study was carried out to investigate the impact of melatonin and BCAA supplementation, separately and together, on QoL, appetite, nutrition risk index (NRI), and fatigue status in CC patients.
Materials and Methods
Participants
In this study, 84 eligible patients referred to the Shahid Madani Medical Research Training Center in Tabriz, Iran, were recruited. The sample size was calculated based on changes in albumin levels with regard to the results of a previous study done by Okabayashi et al, 18 with an error of 5% and power of 90%; the required minimum sample size was calculated as 17 patients per group, and with anticipation of an overall dropout rate of 20%, increased to 21 patients within each group. Eligibility criteria included the following: (1) agreement to participate in the study; (2) CHF patient; (3) age range of 30 to 70 years; and (4) loss of at least 7.5% of dry body weight over the past 6 months or a body mass index (BMI) lower than 20 kg/m2. Exclusion criteria were patients with diabetes; chronic renal failure; hemodialysis; respiratory failure with pulmonary origin; patients receiving immunosuppressive, anti-inflammatory, and corticosteroid drugs; or history of supplementation with vitamins or antioxidants during the previous 6 months. The method of recruitment was through announcement flyers in the Tabriz Shahid Madani Hospital. The criteria for matching were sex, age, and the New York Heart Association (NYHA) Functional Classification.
Randomization and Intervention
This study was a double-blind 4-arm parallel randomized controlled trial. For randomization, eligible participants were randomly assigned to intervention and placebo groups based on the random block procedure developed by random allocation software. The random sequence was administered by an independent third investigator who was not aware of the study clinical process until the outcome data collection was completed. The participants were allocated to 4 groups: (1) melatonin 20 mg/d (1 tablet containing 20 mg melatonin/d); (2) BCAAs contained at a ratio of 2:1:1 leucine, isoleucine, and valine 10 g/d (5 caplets containing 2 g BCAAs/caplet); (3) melatonin 20 mg/d (1 tablet containing 20 mg melatonin/d) and BCAAs 10 g/d (5 caplets containing 2 g BCAAs/caplet) together; and (4) tablet-like melatonin and caplet-like BCAAs filled with cornstarch, as the control group. The participants were asked to take their tablet at night before sleeping and caplets in the evening for 8 consecutive weeks. Compliance was checked by phone calls every week. For ethical reasons, they were permitted to continue routine medications. However, they were not allowed to take antioxidants and/or vitamin supplements during the trial. The participants were allowed to discontinue the trial if they were unwilling to complete the trial or if they experienced any adverse effects during the intervention. A safety assessment was done during the intervention by interviewing participants and evaluating for adverse effects related to the trial.
Measurements of Anthropometric Indices
Anthropometric indices, including weight and height, were measured to the nearest 0.1 kg using a standardized digital column scale and to the nearest 0.1 cm using a portable stadiometer (Seca, Hamburg, Germany), respectively. BMI was calculated by dividing weight in kilograms by height in meters squared.
Quality-of-Life Assessment
The Minnesota Living with Heart Failure questionnaire (MLHFQ), a self-report questionnaire with 21 items that assesses the QoL, was used. The MLHFQ consists of 2 domain scales: physical (physical symptoms such as shortness of breath, fatigue, peripheral edema, and sleep disturbance; 8 items) and emotional (psychological symptoms such as depression and anxiety; 5 items) 19 ; total score of the MLHFQ was calculated by summing the response score of 21 items. Items are scored on a 6-point scale from 0 (best situation) to 5 (worst condition) in terms of severity. The total score is calculated by adding the score of each item. Total scores range between 0 and 105, with a higher score representing lower QoL. The validity and reliability has been established in previous studies. 20 The reported internal consistency of the MLHFQ is very good (Cronbach’s α = .90). 21
Fatigue Status Assessment
To measure fatigue, we used the Fatigue Symptom Inventory (FSI), a self-report questionnaire with 14 items that assesses the frequency, severity, and daily pattern of fatigue as well as its perceived interference with QoL. 22 The FSI total score is calculated by adding the scores of each subscale, resulting in total scores ranging from 0 to 131. Items are scored on a 10-point scale, with lower scores indicating less fatigue. The FSI has been found to be a valid measure of fatigue with acceptable psychometric properties (Cronbach α = .89). 23
Nutritional Status Assessment
The Simplified Nutritional Appetite Questionnaire (SNAQ) was used to assess appetite. SNAQ is a self-report questionnaire with 4 items. 24 Participants are presented with items related to appetite, are asked to choose 5 statements for each item, and select the best alternative that describes their appetite. The total score is calculated by adding the score of each item. The maximum possible score is 20; a score <14 indicates poor appetite. 25
NRI is a practical tool that identifies nutritional depletion earlier to CC or the clinical manifestation of body wasting in HF patients. NRI is calculated as follows: [1.519 × Serum albumin (g/L)] + 41.7 [Current body weight (kg)/Ideal body weight (kg)]. 26 There is a higher probability of nutritional depletion with a lower NRI. 27
Statistical Analysis
SPSS version 16 (SPSS Inc, Chicago, IL) was used for statistical analysis. Distribution of data was examined using the Kolmogorov-Smirnov test. Results are shown as mean (SD) and median (percentile 25 to 75) for normally and nonnormally distributed quantitative data, respectively, with frequency (percentage) for qualitative data. For nonnormally distributed variables, a log transformation was conducted prior to the analysis. A χ2 test and a 1-way ANOVA test, followed by Tukey’s test, were used to compare the 4 groups for baseline measures of the qualitative and quantitative data, respectively. Comparison of the 4 groups at the end of the study was completed using analysis of covariance followed by Sidak’s test after adjusting for baseline parameters. Comparison between baseline data and final results within each group was made using a paired samples
Results
Of a total of 84 patients, 70 completed the trial (melatonin group, n = 18; BCAA group, n =17; melatonin+BCAA group, n = 18; placebo group, n = 17; Figure 1). Baseline participants’ characteristics in different groups are given in Table 1. No significant differences were observed in age, sex, height, 6-month weight loss, marital status, education status, smoking status, HF duration, family background of HF, NYHA functional class, and NRI and albumin levels among the 4 investigated groups (
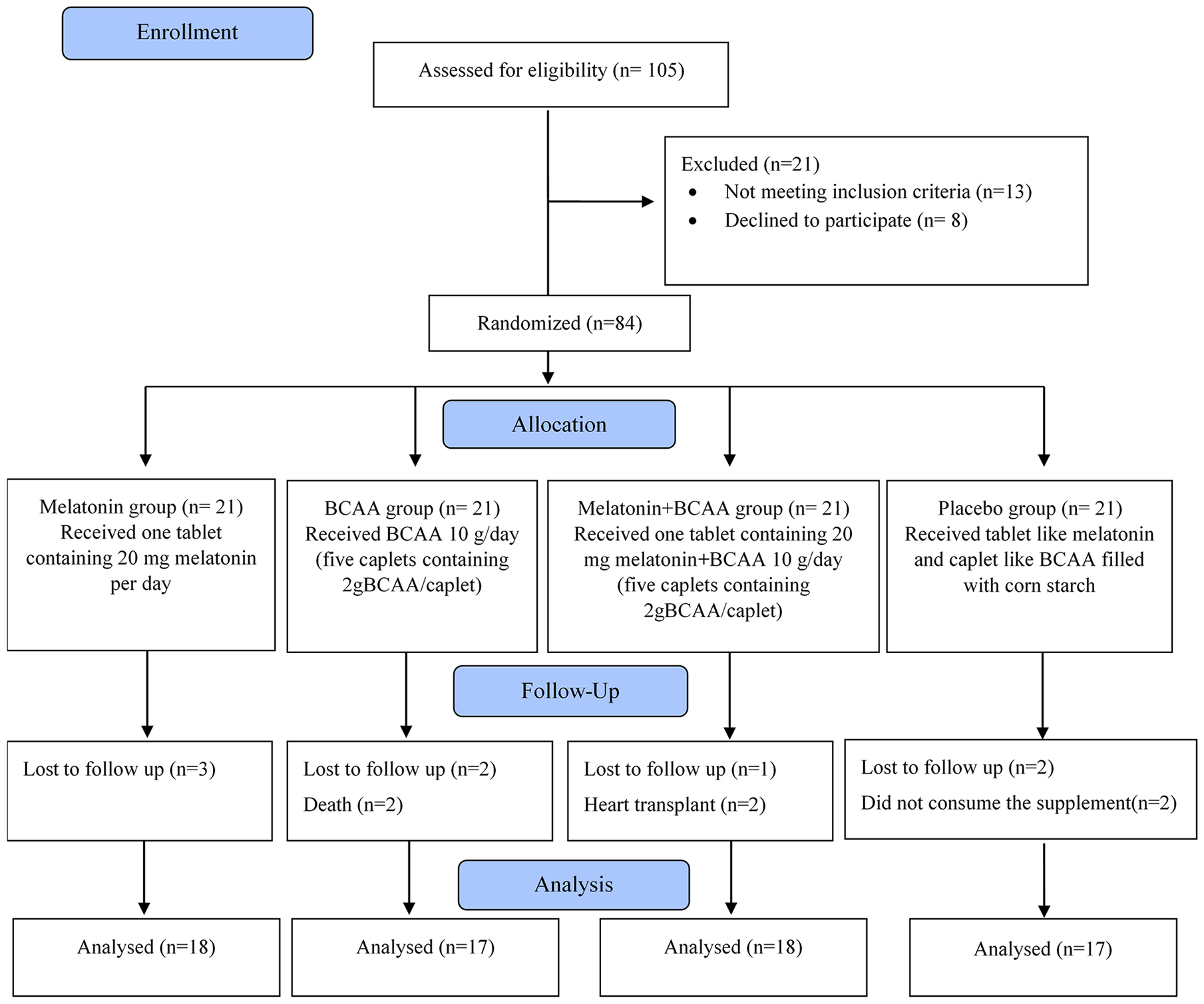
Flow diagram of the study.
Baseline Characteristics of the Study Population. a
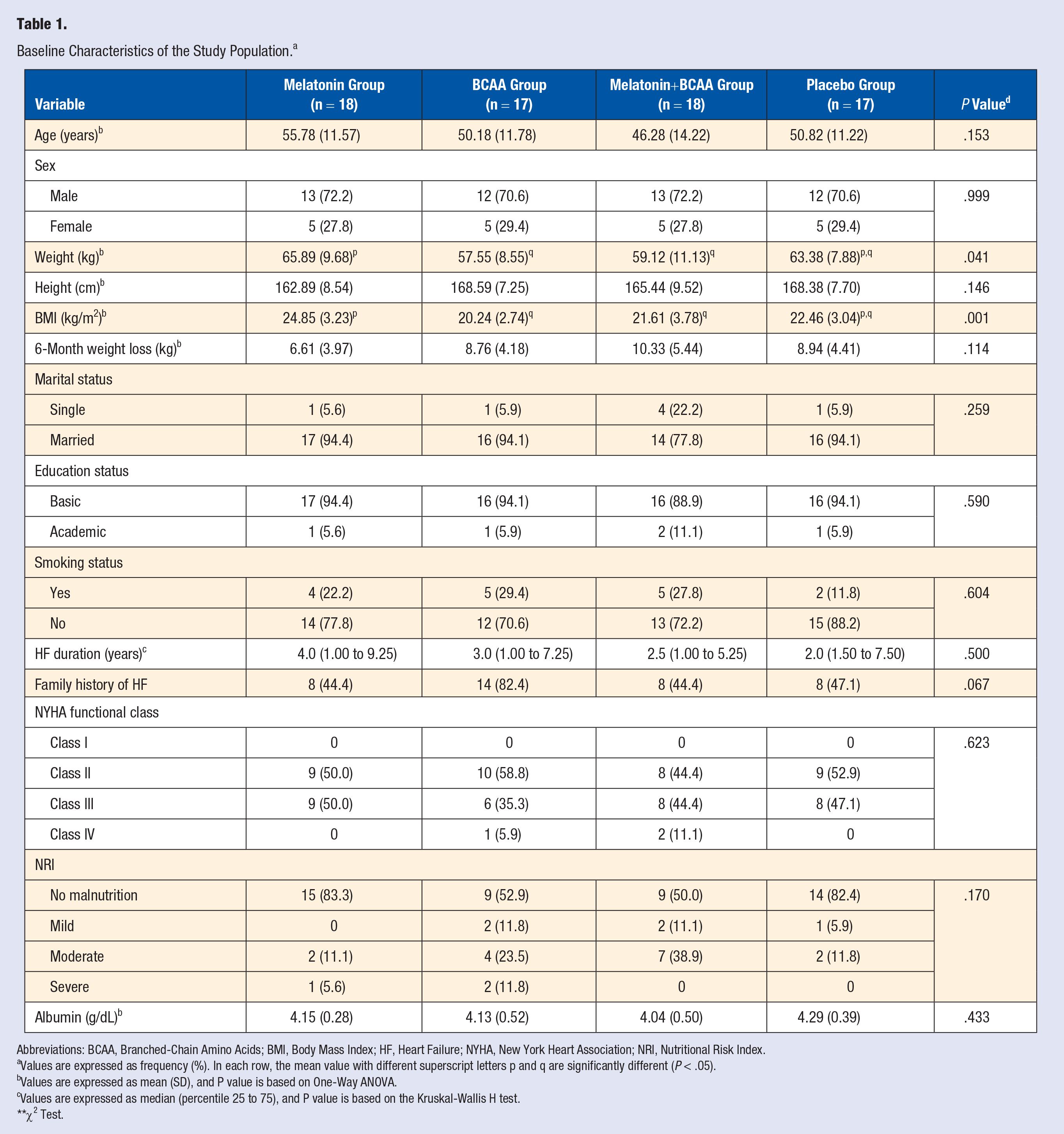
Abbreviations: BCAA, Branched-Chain Amino Acids; BMI, Body Mass Index; HF, Heart Failure; NYHA, New York Heart Association; NRI, Nutritional Risk Index.
Values are expressed as frequency (%). In each row, the mean value with different superscript letters p and q are significantly different (
Values are expressed as mean (SD), and P value is based on One-Way ANOVA.
Values are expressed as median (percentile 25 to 75), and P value is based on the Kruskal-Wallis H test.
χ2 Test.
MLHF questionnaire score of study participants at baseline and also at the end of intervention is displayed in Table 2. Assessments of each overall, physical, and emotional dimension of QoL by the MLHF questionnaire, indicated that there was no statisticaly significant difference among study groups at baseline (
Comparison of MLHF Questionnaire Score of Study Participants at Baseline and End of Intervention. a
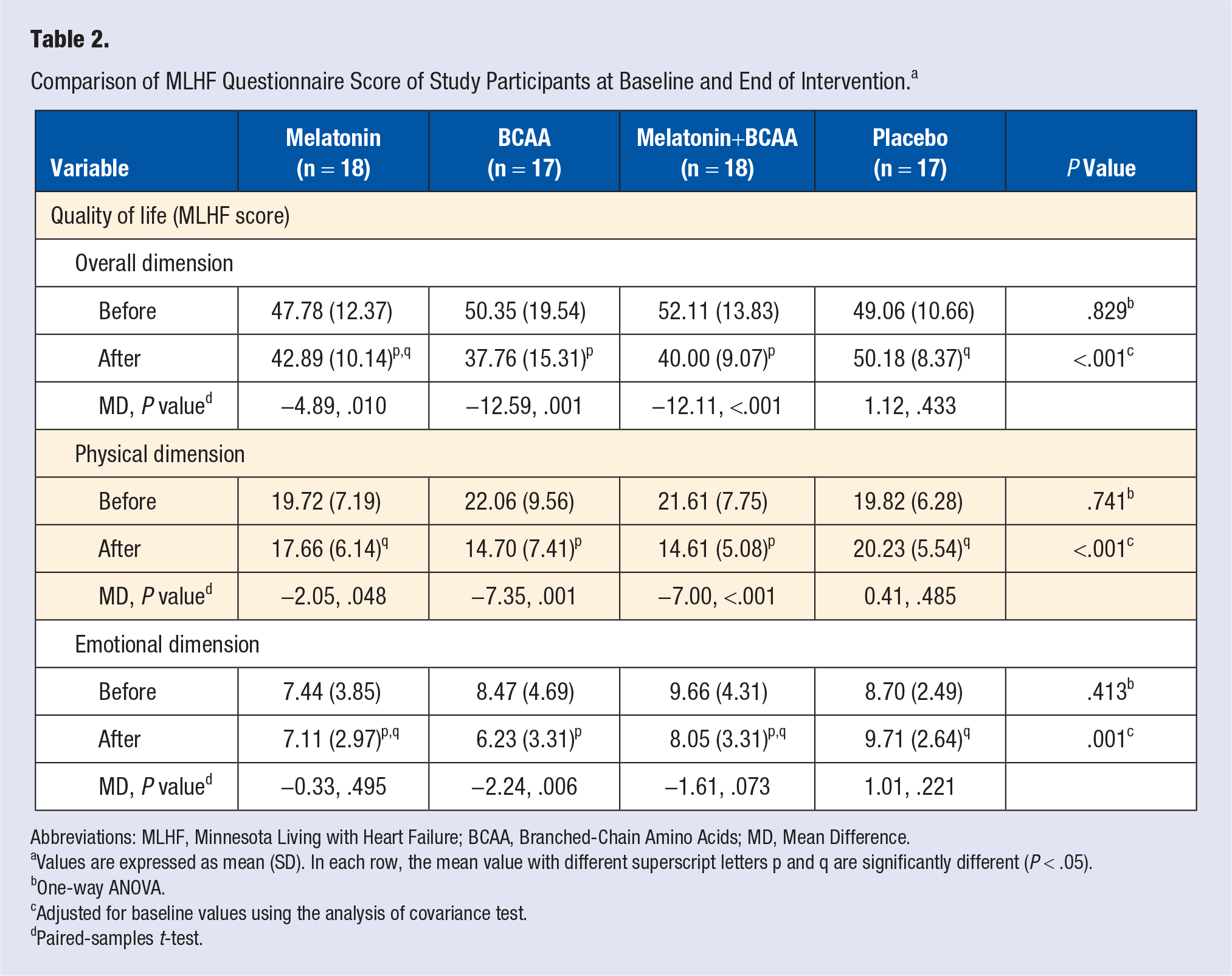
Abbreviations: MLHF, Minnesota Living with Heart Failure; BCAA, Branched-Chain Amino Acids; MD, Mean Difference.
Values are expressed as mean (SD). In each row, the mean value with different superscript letters p and q are significantly different (
One-way ANOVA.
Adjusted for baseline values using the analysis of covariance test.
Paired-samples
Table 3 shows the comparison between appetite, fatigue questionnaire, and NRI score within and between groups. Mean SNAQ, FSI, and NRI scores were not statistically different among the 4 study groups at baseline (
Comparison of Appetite, Fatigue Questionnaire, and Nutrition Risk Index Score of Study Participants at Baseline and End of Intervention. a
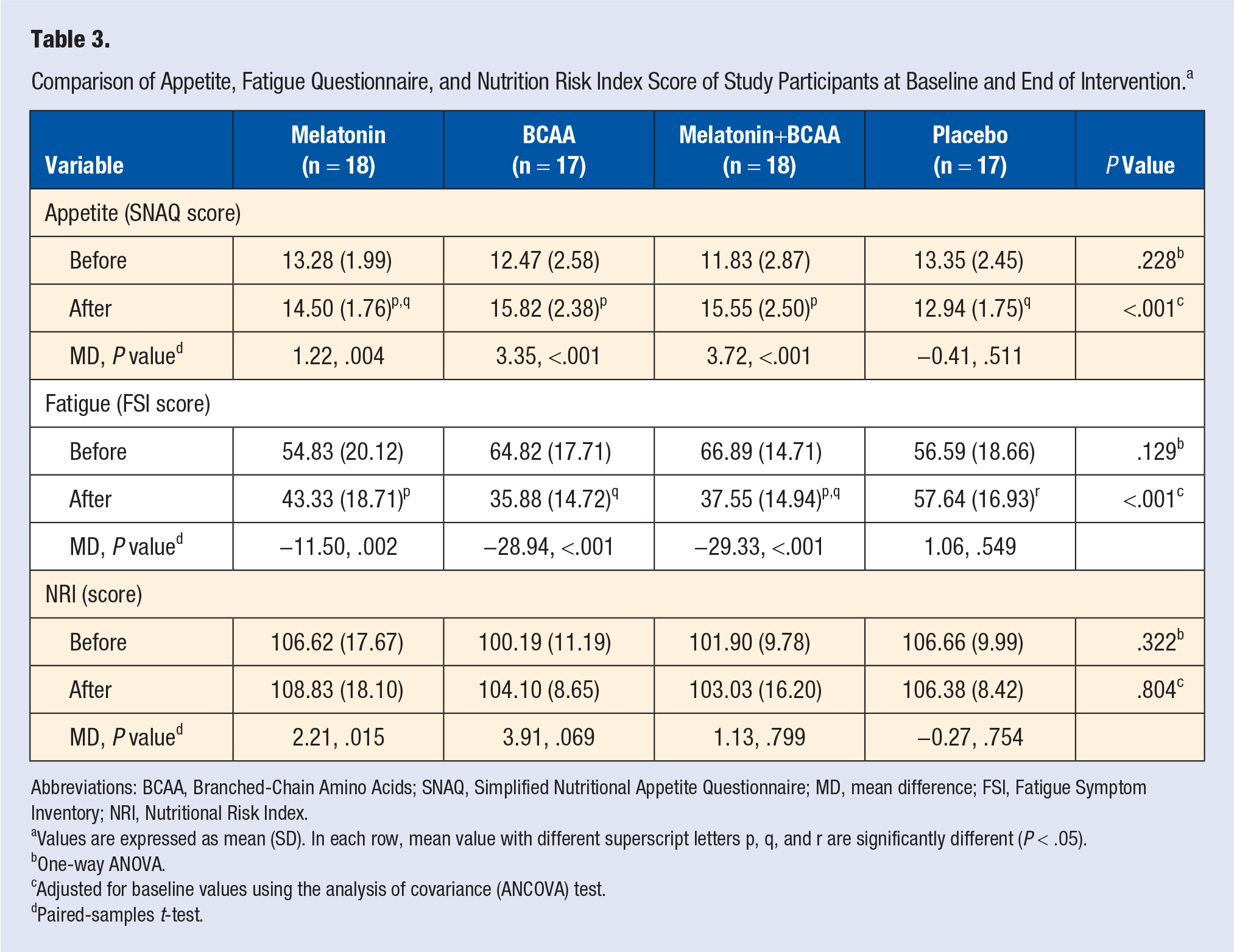
Abbreviations: BCAA, Branched-Chain Amino Acids; SNAQ, Simplified Nutritional Appetite Questionnaire; MD, mean difference; FSI, Fatigue Symptom Inventory; NRI, Nutritional Risk Index.
Values are expressed as mean (SD). In each row, mean value with different superscript letters p, q, and r are significantly different (
One-way ANOVA.
Adjusted for baseline values using the analysis of covariance (ANCOVA) test.
Paired-samples
Discussion
To the best of our knowledge, this is the first study conducted to investigate the effects of BCAAs plus melatonin on QoL, fatigue, and nutritional status in HF patients with CC. HF patients frequently experience fatigue and appetite loss, which has a negative effect on clinical outcomes.28,29 HF still causes major incapacity in these patients despite many treatment methods. Earlier researches have emphasized that diet and some supplementations, as adjunct conventional therapies, could improve some HF symptoms.30,31 Therefore, identification of the effects of diet and cosupplement therapy of melatonin and/or BCAAs on QoL will possibly facilitate the improvement of new methods for increasing QoL and also secondary prevention in patients with HF.
We have established that the BCAAs/melatonin combination improves QoL, fatigue, and appetite status to a greater extent in comparison with melatonin alone. No effect was observed for melatonin plus BCAA, in terms of NRI.
Self-reported measures of the MLHFQ questionnaire are regularly applied to assess the intervention plan outcomes in HF patients. This questionnaire is a precise instrument for assessment of QoL in HF patients. In these patients, QoL could be reduced along with increasing level of NYHA functional class.19,32 The results of this study indicate that supplementation with BCAAs and melatonin, and a combination of these 2 interventions, could significantly improve overall dimensions of the MLHFQ questionnaire and also emotional and physical subscale scores. Earlier studies have demonstrated the effects of BCAAs or melatonin supplementation on QoL score in different clinical settings18,33; however, the combined effects of BCAAs plus melatonin supplementation still have not been studied in these patients. Mean MLHFQ score after 8 weeks showed a significant reduction in the BCAAs group when compared with the placebo group, suggesting an improving in QoL. The study findings are in agreement with the results of Rondanelli et al, 34 who reported an improvement in QoL score after supplementation of the aged patients with 4 gr essential amino acids twice a day for 8 weeks. Similar findings were also observed after taking 5 mg of melatonin for 2 months in breast cancer patients. 35 In contrast to these findings, some studies did not find any positive effect of BCAAs or melatonin supplementation on QoL. For instance, in a study by Amstrup et al, 36 melatonin did not exhibit any effect on QoL or sleep in elderly adults after 12 months.
Previous reports recommended that patients with HF have protein energy malnutrition (PEM), which in turn would result in a decrease in muscle mass, albumin, and immunity.30,37 Therefore, PEM clearly affects QoL and the prognosis of patients with HF. Recent clinical data support the favorable effects of BCAAs on protein synthesis and appetite in HF.34,38 Therefore, supplementation with BCAAs might improve the QoL in HF patients. To the best of our knowledge, this research is the first to address this question using MLHFQ questionnaires. With respect to that, it has been established that BCAAs and melatonin improve the nutritional status and thereby decrease PEM and modulate inflammatory response, and these mechanisms might be the reasons for the positive effects of cosupplementation on QoL.6,39 The QoL score improved in our study participants mainly because of enhanced appetite score and reduced fatigue score during the study intervention.
We found that appetite score was higher in the melatonin, BCAAs, and cosupplementation groups compared with the placebo at the end of the study period. We have also found that adding BCAAs to melatonin could augment the appetite levels much more in comparison with any of the 2 supplementations given alone. Although the exact mechanism of melatonin action is not known yet, there is accumulating evidence that melatonin supplementation could stimulate appetite in cachectic patients.13,40 Much attention has been paid to supplementation in HF patients, which would improve their nutritional intake. Moreover, BCAAs have anti-inflammatory and antioxidant effects and also the ability to modulate immune function, which in turn would result in improving the muscle strength.16,41,42 Also, clinical data from HF patient groups recommend that BCAAs supplementation could also increase appetite and caloric intake.34,43 BCAAs plasma levels increase following BCAAs consumption and impede tryptophan entrance into the brain, consequently leading to serotonin level reduction, and along with that appetite increases.43,44 On the other hand, the anti-inflammatory and antioxidant activity of melatonin could yield favorable effects on appetite, 40 which appear to be decreased in HF patients. Altogether, these results show that BCAAs/melatonin cosupplementation would improve appetite score, which in turn could increase appetite and muscle mass.
Fatigue is also seen in more than half of the patients with HF, although the exact mechanism of these symptoms still remains unidentified.45,46 This study discovered that single supplementation or their combination has an effect on FSI score. There are inadequate data for the effects of BCAAs and melatonin on fatigue. The study results are in agreement with some earlier data. In rats with type 2 diabetes, melatonin supplementation in combination with exercise for 5 weeks possibly influenced fatigue through improvement in antioxidative and anti-inflammatory activities. 47 Van Heukelom et al 48 reported that a 3-month oral administration of 5-mg melatonin was an effective treatment for patients with chronic fatigue syndrome. In this study, reduction in fatigue in the melatonin plus BCAAs group was much more in comparison with other groups. There are no general standard guidelines for fatigue treatment and screening in HF patients. Consequently, specialists may fail to improve this condition in most patients. These results definitely suggest that both BCAAs and melatonin supplementation contribute to the beneficial effect on fatigue. One possible mechanism of benefit from melatonin supplementation is the reduction in oxidative stress and inflammation. Accumulated data from different cachectic patients indicated that BCAAs supplementation might reduce catabolism and weight loss, which may have beneficial effects on fatigue levels.6,16 Generally, this evidence recommends that melatonin+BCAAs cosupplementation might contribute to lower morbidity and fatigue through improvement in protein metabolism, which leads to gain in muscle mass.
PEM with poor prognosis is prevalent in HF patients. 49 The NRI is an objective tool that can appraise nutritional status in this pathological condition. 50 An important finding of this study is that nutrition risk score was improved by melatonin supplementation. In contrast to a previous study, this study established that BCAAs supplementation and/or combination with melatonin did not have any beneficial effects on NRI score. A human study also found that BCAAs supplementation for 4 weeks affected the NRI in those patients with neurological disorders. 51 Earlier studies recommended that melatonin might contribute to the nutritional status increase through decrease in inflammatory responses in HF patients.41,52
Along with the therapeutic usefulness of melatonin seen in numerous clinical trials, very low toxicity of melatonin was reported over an extensive range of doses. 53 Furthermore, melatonin safety has been established in doses that noticeably exceed those that were used in cardiovascular diseases. 54 In the previous studies, an improvement was observed clinically in cancer cachexia with 18 or 20 mg melatonin supplementation per day. As a result, we used a dose of 20 mg/d melatonin in this study.14,55 In addition, different BCAA doses ranging from 8 to 14.4 g/d were given in order to improve body composition, QoL, and appetite in degenerative diseases.34,56-58 Therefore, this study was conducted with 10 g/d of BCAA.
Lack of serum BCAAs and melatonin assessment as well as other oxidative stress indices and short intervention times were some of this study’s limitations. Also, other essential prognostic factors, including cardiac function and exercise capacity, were not assessed in this study. Regardless of these limitations, this is the first double-blind study that investigated the effect of BCAAs plus melatonin on MLHFQ, SNAQ, and FSI questionnaire as well as NRI score in patients with HF. Further studies are required to confirm the anticachectic properties of BCAAs plus melatonin.
Conclusion
The findings of this study indicate that cosupplementation of BCAAs and melatonin for 8 weeks improved the CC outcomes such as QoL, fatigue status, and appetite in cachectic HF patients, but it could not affect NRI. Although separate supplementation with melatonin and BCAAs had positive effects on QoL, fatigue, and appetite of study participants, this study demonstrated that adding BCAAs to melatonin augmented these effects more than either of the 2 other supplements given alone.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is supported financially by Tabriz University of Medical Sciences as a thesis for the PhD degree of the first author.
Ethical Approval
The study protocol was approved by Medical Ethics Committee of TBZMED (No: IR.TBZMED.REC.1396.767).
Informed Consent
All participants were informed of the study and then asked to sign a written informed consent form.
Trial Registration
The trial was registered in the Iranian Registry of Clinical Trials (Registration No. IRCT20121028011288N14) on February 1, 2018.
