Abstract
Exercise could indirectly affect body weight by exerting changes on various components of appetite control, including nutrient and taste preferences, meal size and frequency, and the drive to eat. This review summarizes the evidence on how exercise affects appetite and eating behavior and in particular answers the question, “Does exercise induce an increase in food intake to compensate for the increase in energy expenditure?” Evidence will be presented to demonstrate that there is no automatic increase in food intake in response to acute exercise and that the response to repeated exercise is variable. The review will also identify areas of further study required to explain the variability. One limitation with studies that assess the efficacy of exercise as a method of weight control is that only mean data are presented—the individual variability tends to be overlooked. Recent evidence highlights the importance of characterizing the individual variability by demonstrating exercise-induced changes in appetite. Individuals who experience lower than theoretically predicted reductions in body weight can be characterized by hedonic (eg, pleasure) and homeostatic (eg, hunger) features.
‘For weight control, exercise is intended to increase energy expenditure with the aim of creating a sustained energy deficit.’
Introduction: Compensatory Responses to Exercise and Weight Loss
“Exercise may stimulate appetite so that persons who exercise increase their eating and do not lose as much weight as expected” and “a person who exercises in the early evening may go to sleep earlier or require more rest in the evening.”
More than 30 years ago, Epstein and Wing suggested that energy balance is under such tight control that the energetic cost of exercise training will be mediated by compensatory adaptations.
1
For weight control, exercise is intended to increase energy expenditure (EE) with the aim of creating a sustained energy deficit. A lower than expected weight loss with exercise or energy restriction may be considered in 2 categories: errors inherent in the calculation of the predicted weight loss and errors associated with metabolic or behavioral adaptations.
2
The current review concerns compensatory adaptations that occur once exercise has been imposed. Any biological or behavioral compensatory responses will attempt to prevent the energy deficit by maintaining energy balance. We acknowledge that there are a range of adaptations that could occur to offset the exercise-induced increase in EE, including reductions in resting metabolic rate and nonexercise activity thermogenesis. Tremblay et al
3
provide a comprehensive review of the role adaptive thermogenesis can play in determining the ability of individuals to lose body weight. However, the focus of this review is compensatory changes in
Exercise and Weight Loss: Individual Variability
It is important to recognize that for a majority of published research on the effects of exercise on weight loss, mean data are reported, which may disguise differential responses between individuals and miss identification of important subgroupings of “responders” and “nonresponders.” The phenomenon of individual variability in response to energy balance perturbations is certainly not new.6-8 However, exercise-induced individual variability has yet to be exploited, in particular, the mediating role of compensatory responses. Recently, there is an emerging interest in characterizing the variability9-16 to better understand the relationship between exercise and weight management.
The interindividual variability in changes in body weight after 12 weeks of supervised exercise intervention can be partially explained by differences in compensatory responses in appetite.10,11,17 The intervention consisted of 5 moderately-high-intensity (70% V
There is a need to understand what accounts for the variability and how that information can be used to develop weight management strategies to facilitate weight loss (and maintenance) in those people who experience a lower than expected weight loss. Furthermore, there is a need to educate people that improvements in health occur even in the absence of expected weight loss. This variability demonstrates that some individuals experience minimal exercise-induced weight loss; however, they still experience other health benefits such as reduced blood pressure and waist circumference. 18
The Interaction Between Exercise, Appetite, and Food Intake: What Does the Evidence Say?
The acute effects of exercise on appetite are consistent and relatively well understood. The majority of research demonstrates that acute exercise does not increase the drive to eat (ie, hunger) or EI.19-23 An exercise-induced increase in EE of 4.6 MJ/d over 2 days failed to stimulate an automatic increase in EI. 24 However, when the amount of EE is increased to 1.5 MJ and 3 MJ/d over 14 days, there is evidence of partial (~30%) compensation.25-27 Exercise has also been demonstrated to improve the sensitivity of appetite control and that regular exercisers are better at detecting manipulated differences in energy content between meals compared with their sedentary counterparts.28,29
Most of the evidence from long-term studies (4-18 months) suggests no significant change in EI across the intervention.30-32 It is important to note that in most long-term studies there is a tendency for the exercise sessions to be unsupervised. When exercise
Explaining the Variability: Mechanisms Associated With the Compensatory Changes in Appetite and Food Intake
Appetite Peptides
Peripheral signals including long-term signals such as leptin and insulin and short-term signals released by the gastrointestinal tract play an important role in appetite control and may help explain changes in appetite with exercise (see Martins et al 5 for a comprehensive review). Long-term signals appear to respond primarily to changes in body composition and energy availability and similar to other weight loss strategies have been shown to decrease in response to chronic exercise.5,38,39 These changes would be expected to stimulate appetite.
Short-term satiety signals—in particular appetite peptides—have been targeted as potential mediating mechanisms involved in the effects that exercise exerts on EI and, subsequently, on body weight and are the focus of this section. Ghrelin is an orexigenic peptide secreted from the stomach, and increased ghrelin suppression is associated with reduced appetite.40,41 The majority of studies indicate that acute exercise has no influence on total ghrelin concentrations42-47; however, one study showed that total ghrelin levels remain suppressed for 1 hour after exercise. 48 The consensus is that acute exercise appears not to influence total ghrelin levels independent of mode, intensity, or metabolic state in lean and overweight/obese adults. 5 Sixty minutes of running at ~75% VO2max induced a suppression of acylated ghrelin (AG), which was associated with a simultaneous suppression of appetite during and immediately after exercise in lean, well-trained males. 49 This evidence suggests that total ghrelin and AG respond differently to exercise. No changes in total ghrelin and a decrease in AG were reported after intense exercise in obese and lean individuals.50,51
Cholecystokinin (CCK), polypeptide YY (PYY), and glucagon-like peptide-1 (GLP-1) are anorexigenic peptides released in response to intestinal nutrients. 40 Fifty to 60 minutes of cycling (65% HR max) induced a significant increase in PYY, GLP-1, and pancreatic polypeptide (PP), but did not induce any change in total ghrelin. GLP-1 remained elevated into the postexercise period, whereas the increase in PYY was short-lived.52,53
It is worth noting that in chronic exercise studies, if weight loss occurs, changes in peptides could be associated with changes in weight loss and/or body composition—rather than exclusively due to exercise per se. A significant increase in fasting total ghrelin plasma levels has been reported after exercise-induced weight loss in lean 54 and obese adults 55 and in obese children.38,55 However, no change in AG fasting plasma levels after long-term exercise was reported in overweight children and adolescents.38,55 We have recently shown a significant increase in fasting AG plasma levels and an intensified postprandial suppression of AG in obese adults after 12 weeks of exercise-induced 3.5 kg reduction in body weight. 39
The increase in total ghrelin and/or AG plasma levels observed in exercise studies in which weight loss occurs could be a counterregulatory mechanism to restore energy balance. Furthermore, studies that demonstrate a return to baseline of total ghrelin during sustained weight loss support the hypothesis that total ghrelin is involved in the regulation of acute, but not chronic, changes in EB.55,56 It remains to be seen if the same is true for AG, since changes in AG do not necessarily parallel changes in total ghrelin.
The evidence regarding the effects of chronic exercise on the release of anorexigenic gut peptides is relatively scarce. Fasting CCK levels do not change in response to chronic exercise, 57 whereas fasting and postprandial levels of PP increase in sedentary males, 58 and fasting PYY levels increase in overweight adolescents. 59 Exercise-induced weight loss appears to exert differential effects on fasting (decrease) and postprandial (increase delay) levels of PYY and GLP-1 in obese adults. 39 Further support comes from a 5-day exercise intervention showing no change in fasting GLP-1 and an increase in postprandial GLP-1 release in lean and overweight individuals. 60 There is a need for further studies to systematically examine the interactions among gastric emptying, gut peptides, and appetite in response to acute or chronic exercise.
Gastric Emptying
Given persuasive evidence that gastric emptying influences appetite and EI,61,62 it is possible that the gut and its related hormonal activity also moderate the responses to acute or chronic exercise. As food fills the stomach and subsequently empties, a variety of factors including gastric distension, nutrient stimulation of intestinal mechanoreceptors and chemoreceptors and several gut peptides released from the gastrointestinal tract, contribute to satiation (control of meal size) and satiety (postmeal inhibition of eating). As satiation and satiety are important processes in appetite regulation, interindividual differences in gut physiology and the strength of these signals could contribute to compensatory responses in food intake and, consequently, variability in exercise-induced weight loss.
Surprisingly, very few studies have directly examined the effects of exercise on gastric emptying and appetite. One study recently reported perceived hunger feelings were negatively correlated with total stomach volume following fluid ingestion after acute exercise. 63 However, most studies conducted on exercise and gastric emptying have focused on the efficacy of providing optimal rates of carbohydrate and fluid replenishment as ergogenic aids in sport. Overall, the evidence points to a delay in gastric emptying with strenuous exercise,64-72 whereas mild to moderate exercise stimulates (ie, accelerates) gastric emptying.69,70,73-75 There have also been reports of no change in gastric emptying with moderate-intensity exercise.67,76 Mechanisms proposed that could contribute to exercise-induced alterations in gastric emptying include changes in contraction frequencies, antral area 77 and gastric myoelectrical activity,78,79 hormonal71,74,80 and neural factors (mainly vagal in origin),73,81 gut blood flow, 69 and the mechanical effects of “jostling of the gut”69,71,74,82 during exercise.
In terms of adaptation to regular exercise, very few studies have examined the impact of chronic exercise exclusively on gastric emptying at rest or during exercise. Figure 1 shows data from a study indicating that basal rate of gastric emptying is faster in marathon runners compared with sedentary controls. 83 Training-induced enhanced parasympathetic tone was proposed as one possible explanation.
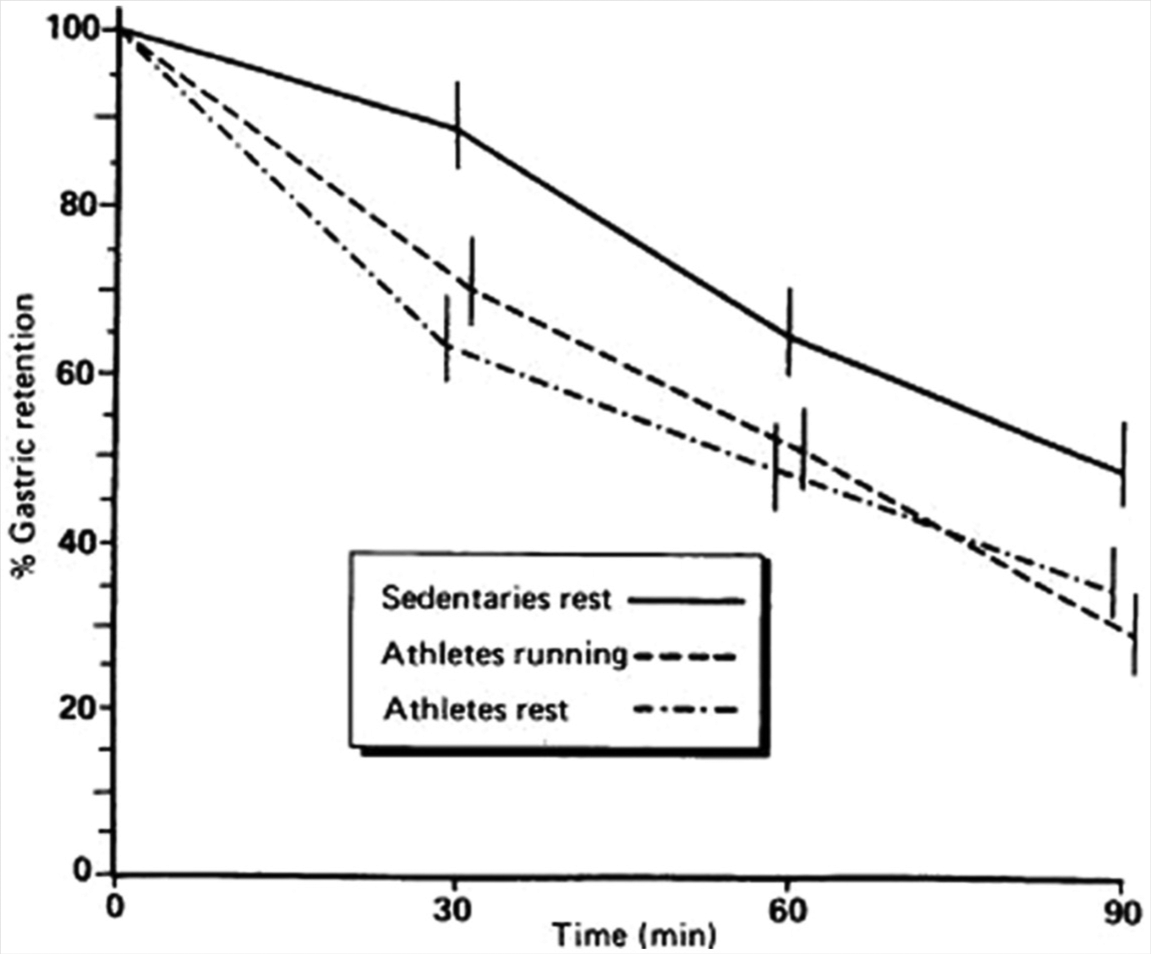
A Comparison of Mean (± Standard Deviation) % Gastric Retention Between Sedentary Individuals (At Rest Only) and Marathon Runners (At Rest and
Of course, physical activity and total EE might not be the only factors to vary between inactive and active people. Dietary habits, including total EI, frequency of eating, and macronutrient intake, could also vary. Collectively, these dietary factors could affect gastric motility, via the quantity, frequency, and quality of nutrients that pass through the gut and small intestine.
Harris et al 84 reported rapid orocecal transit time in chronically active individuals with concomitant high EIs, and they concluded that the high EI associated with chronic exercise may be associated with significant gastrointestinal adaptations. However, the causal nature of this association is not possible to determine from this cross-sectional study.
Collectively, the limited evidence suggests chronic exercisers have a faster gastric emptying. Further studies are needed to determine the temporal patterns of changes in gastric emptying with exercise and with exercise-induced weight loss. The associated implications of exercise-induced changes in gastric emptying for meal size, frequency, and the ability to compensate for prior EI also remains to be established.
Exercise-Induced Changes in Substrate Oxidation: Carbohydrate Balance
Consistent with a psychobiological approach to appetite control, in which physiological mediators can drive behavior, 85 exercise-induced changes in substrate metabolism may influence appetite and food intake. Substrate metabolism has long been implicated in the energostatic control of EI, with postmeal satiety and EI thought to be mediated by vagal afferent nerve activity stemming from changes in fatty acid oxidation and the hepatocellular ATP/ADP ratio.86,87 Furthermore, aerobic exercise is known to alter substrate utilization and availability during and following exercise. This is of importance as it has been suggested that short-term feeding is designed to maintain the body’s stored glycogen levels at a set point, for example, the glycogenostatic theory.88-90 Due to its limited capacity for storage, challenges to glycogen availability via diet or exercise may elicit compensatory changes in EI designed to restore carbohydrate balance. 88
The evidence to support a direct link between substrate metabolism during exercise and compensatory eating is limited and contradictory.90,91 However, Burton et al
92
recently reported that a positive CHO balance at the end of a 6-hour “high energy turnover” condition (involving 60 minutes of walking at 50% V
Where CHO balance has been measured over longer periods (1-3 days) following dietary and/or exercise manipulation of glycogen availability, a negative CHO balance has been shown to be predictive of greater ad libitum EI over subsequent days.92,94-97 While such findings have not always been consistent,98-100 a negative CHO balance has also been shown to be predictive of long-term weight gain. 101 Eckel et al 101 measured energy balance in 39 participants following either a 15-day isocaloric high fat (50% fat) or high CHO diet (55% CHO), and subsequently tracked body composition over a 4-year period in these individuals. There was a significant inverse relationship between CHO balance following the high CHO diet and the change in fat mass over the follow-up period. Individuals who had the highest positive 24-hour CHO balance (indicative of CHO storage) following the high CHO diet gained less body mass, fat mass, and percentage body fat over the follow-up period. As such, while further research is needed, there is preliminary evidence that the need to maintain CHO balance might play a role in the control of food intake and body weight.
Changes in Macronutrient and Food Preferences
Compensatory responses in EI to exercise may be partly explained by changes in macronutrient and food preferences. 102 In a recent review, Elder and Roberts 4 identified 12 studies investigating the acute effect of exercise on food palatability and taste perception.103-114 Since their review we have identified a further 6 studies that contribute to this topic.115-120 However, the findings are not consistent and demonstrate both increases and decreases in acuity of taste perception and rated preference for tastes after acute exercise. This between-study variation may be partially explained by differences in the exercise protocols adopted. For example, those studies employing longer and higher intensity exercise sessions (120-180 minutes) tended to detect an effect of exercise,107,114 whereas shorter, lower intensity exercise (10-30 minutes) studies tend not to report significant changes.107,109 This could be partly related to differences in the volume (ie, net increase in EE) of the exercise.
Interestingly, effects were more frequently reported for the perception and preference of salty than sweet or bitter tastes. This suggests any effect of exercise on taste perception and preference is likely to be subtle or subject to a threshold of physical exertion or EE. To date, no studies have independently examined the roles of exertion and expenditure on taste perception and taste preference after exercise. Other studies have tested the effect of exercise on the palatability of complete meals or individual food items categorized by a sensory (eg, sweet) or nutrient (eg, fat) characteristic.106,110 For example, King et al 106 found increased palatability ratings after high-fat and low-fat test meals, whereas Lluch et al 110 found increased ratings for low-fat foods only. Finlayson et al 121 measured hedonic response to images of food immediately before and after acute exercise at baseline and following 12 weeks of daily moderate-intensity exercise. The authors reported an overall decrease in palatability ratings over the 12 weeks; however, on examination of individual variability in net energy balance following the intervention (ie, fat loss accounting for changes in fat free mass), “onresponders” who experienced lower than expected fat loss also demonstrated acute increases in food preference after each exercise session. Figure 2 shows the hedonic response to food (categorized by fat content and taste) after a single exercise bout before and after the intervention.
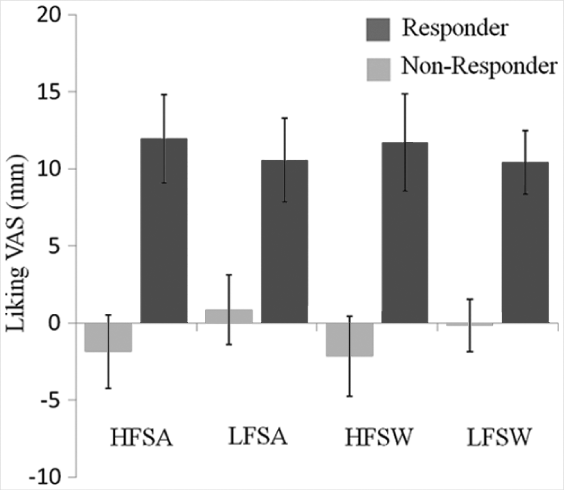
Acute Pre–Post Changes in Liking for Different Foods After a Single Bout of Exercise in Responders (Light Bars) and Nonresponders (Dark Bars) After a 12-Week Exercise Intervention.
In a different study in lean females, Finlayson et al 116 assessed acute exercise-induced changes in implicit hedonic reactions to food. This was achieved by comparing taste responses to an array of food items varying in energy density (manipulated by fat content) and taste before and after 50-minute moderate-intensity cycling (compared to no exercise). The authors observed faster reaction time responses after exercise among those participants who increased their EI compared with participants whose EI did not change. These data help inform findings that individual differences in the hedonic response to food after exercise could be associated with the compensatory EI response to exercise and a poorer weight loss.
Exercise-induced changes in food reward could also be an important consideration in the capacity for exercise to reduce overweight. In particular, an enhanced motivational drive or wanting for food after exercise may help explain why some people overcompensate when given access to food shortly after exercise. 116 The evidence on macronutrient and food preferences following exercise suggests that some individuals appear to compensate for exercise-induced EE as a result of physiologically or psychologically modulated changes in food hedonics.
Psychological Factors: Eating Behavior Traits
Most of the previous discussion about mechanisms underpinning the exercise-induced compensatory changes in eating behavior focuses on physiological issues. Recently, there is emerging evidence to support the role of psychological factors, in particular eating behavior traits. The evidence suggests that eating behavior traits—typically measured using the Three Factor Eating Questionnaire 122 —exert an influence on food intake and that they also play a role in weight loss interventions. 123 The factors of Disinhibition and Restraint, in particular, have been identified as important eating behavior traits that influence weight gain, weight loss, and weight maintenance. These have been labeled as psychological markers of appetite regulation. There are data to suggest that individuals with a high level of Disinhibition are more susceptible to overcompensate for the energy expended during exercise. 124 Conversely, for individuals susceptible to opportunistic eating (the tendency to overeat whenever external food cues are present in the environment), exercise can exert a positive influence on appetite control. For example, in lean women with high trait Disinhibition, an acute bout of exercise has been found to reduce motivation to eat and increase preference for low-fat foods. 125 Similarly, in lean women with high Restraint, an acute bout of exercise increased the perceived pleasantness of low-fat food and reduced the motivation to eat. 110 In concordance with this, lean and overweight males with high Restraint did not show a counterregulatory eating response (an overeating response initiated by the breakdown of cognitive Restraint) following a bout of moderate-intensity exercise. 126 Therefore, the influence of an acute bout of exercise appears to be beneficial, at least in the short term, for men and women who exhibit a high Restraint score.
Evidence from longer-term interventions demonstrates that successful weight loss is associated with a decrease in Disinhibition and Hunger and an increase in Restraint.127-131 Independent of the type of energy balance perturbation, individuals who are successful in losing weight respond typically by increasing their control over eating (ie, restraint) and reducing their opportunistic eating behavior. More specifically, Butryn et al 132 found that individuals who showed a larger reduction in their level of Internal Disinhibition (eg, eating in response to negative affect) experienced the greatest weight loss. 132 Two studies have examined the influence of exercise over a longer term on psychological aspects of appetite regulation. Keim et al 133 found that following a 4-month exercise intervention (aerobic exercise or resistance training 5 days/week), reduced-obese women could be separated into over- and under-compensators. Those who overate where characterized by a high Disinhibition and Hunger scores on the TFEQ, whereas the undereaters showed a decrease in Disinhibition and Hunger scores and an increase in Restraint score during the intervention. In line with this, a recent 3-month exercise intervention, designed to expend 1.25 MJ/d,10,11 demonstrated that overweight and obese individuals with a higher baseline Disinhibition experienced a greater weight loss. In addition, those individuals who experienced a decrease in their level of Disinhibition and an increase in Restraint had a higher weight loss. 134 While it is not possible to establish whether or not these changes occurred in response to exercise or weight loss per se, these findings indicate exercise can be used to modify eating behavior traits that are associated with susceptibility to weight gain.
Conclusion
Resistance to exercise-induced weight loss is partially explained by changes in eating behavior, among other compensatory responses. This review has identified the various behavioral, physiological, and psychological processes potentially mediating changes in eating behavior with exercise. An increased understanding of the effects of exercise on these processes may help explain the individual variability in weight loss response to exercise and could ultimately facilitate the more effective use of exercise in weight management, by tailoring strategies to suit individuals.
Footnotes
Acknowledgements
This work was supported by the Biotechnology and Biological Sciences Research Council (BBS/B/05079) in the United Kingdom.
