Abstract
The majority of US adults are overweight or obese, which is a primary risk factor for type 2 diabetes and other chronic diseases. Recent advances in behavioral treatment of obesity have produced significant short- and long-term weight losses that reduce the risk of type 2 diabetes and cardiovascular disease. This article reviews key components of effective behavioral treatment interventions, including diet, exercise, and behavioral and psychosocial strategies. The authors review newer treatment modalities that may enhance dissemination (Internet, smartphone) and discuss applications to clinical practice. Practitioners face multiple barriers to effectively delivering lifestyle interventions in today’s health care setting but, nonetheless, remain powerful motivators in helping patients initiate and maintain weight loss efforts that reduce the risk of type 2 diabetes and other chronic diseases.
Introduction
Obesity and type 2 diabetes are common, chronic disorders with major public health implications. Obesity affects approximately 33% of the adult US population1-3 and is a primary risk factor for cardiovascular disease (CVD), hypertension, insulin resistance, and dyslipidemia (high triglycerides, high LDL cholesterol, and low HDL cholesterol).4-6 Type 2 diabetes is a similarly prevalent disorder, affecting an estimated 25 million American adults (6.8% of population) and another 79 million who have prediabetes, or impaired fasting glucose. 7 Type 2 diabetes is a known risk factor for CVD, stroke, and all-cause mortality as well as lower-limb amputations, kidney failure, and cognitive dysfunction and dementia. 8
Lifestyle intervention treatment “packages” have been evaluated in several clinical trials and have shown positive long-term results on health outcomes.
It is well accepted that obesity is related to the development of type 2 diabetes.1,9-11 Obese adults, relative to those of normal weight, have a 3 times higher prevalence of type 2 diabetes.3,12 Morbidly obese adults (obesity class 3; BMI >40 kg/m2), compared with normal weight adults, fair much worse and have up to a 7 times higher prevalence of type 2 diabetes.3,12 Gradual weight gain leading to obesity also increases the risk for developing type 2 diabetes 5 because for each 1 kilogram increase in weight there is an approximate 9% increase in the relative risk for diabetes. 13
Overweight and obese individuals with diabetes who attempt weight reduction face considerable challenges. However, new advances in behavioral treatment of obesity have enabled significant short- and long-term weight losses that appear to lower the risk of developing type 2 diabetes and can result in improved glycemic control and CVD risk factors in individuals with diabetes.14-16 This article reviews the key components of effective behavioral weight control interventions that may reduce diabetes risk factors and improve complications of type 2 diabetes. We review the components of successful short- and long-term intervention approaches that target diet, exercise, and behavioral changes in adults. We will conclude with newer modalities (eg, online interventions) that may further enhance the dissemination and effectiveness of long-term weight control interventions. Although pharmacological interventions and bariatric surgery are effective at decreasing body weight and lowering the risk for type 2 diabetes,17,18 we focus our review on nonpharmacological and nonbariatric lifestyle intervention approaches. Moreover, because obesity and development of type 2 diabetes are significant concerns for children, we refer the reader to several recent, excellent reviews on the topic of pediatric weight control.19-22
Benefits of Weight Loss
Weight loss clearly has beneficial effects on reducing several CVD risk factors, including, but not limited to, lowering blood pressure, triglycerides, and total and LDL cholesterol and increasing HDL cholesterol.6,12,15,23,24 Weight loss has also been associated with a significant reduction in the prevalence of abdominal obesity, 25 sleep apnea, 26 and urinary incontinence. 27 There are also positive effects of weight loss on psychosocial variables, including reductions in depression and anxiety28,29 and improvements in body image. 30
Notably, weight loss also has several established benefits on development and management of type 2 diabetes. Several large randomized controlled trials (see below for more detail) have demonstrated convincingly that lifestyle intervention with a modest amount of weight loss (5%-7%) reduces the incidence of type 2 diabetes.14,31 Moreover, dietary changes, physical activity, and their combination can result in improved glycemic control and CVD risk factors in individuals with preexisting diabetes. 14 Weight loss increases insulin action, improves glucose control, decreases fasting glucose and insulin concentrations, and lowers the dose of diabetes medication.14,16,32-34
Thus, weight loss is highly recommended for overweight (BMI = 25.0-29.9 kg/m2) and obese (BMI ≥ 30 kg/m2) adults at risk for type 2 diabetes or adults with type 2 diabetes. 35 Although larger weight losses produce greater benefits in blood glucose,34,36 small weight losses of 5% to 10% in even very overweight persons will produce significant improvement in diabetes-related health parameters and CVD risk factors14-16 and is achievable and obtainable for most adults through lifestyle interventions. In general, glucose concentrations and other health outcomes improve with reduced energy intake before appreciable weight loss occurs, but long-term weight loss seems to further enhance glucose and diabetes health parameters.14,37
Key Components of Effective Lifestyle Interventions
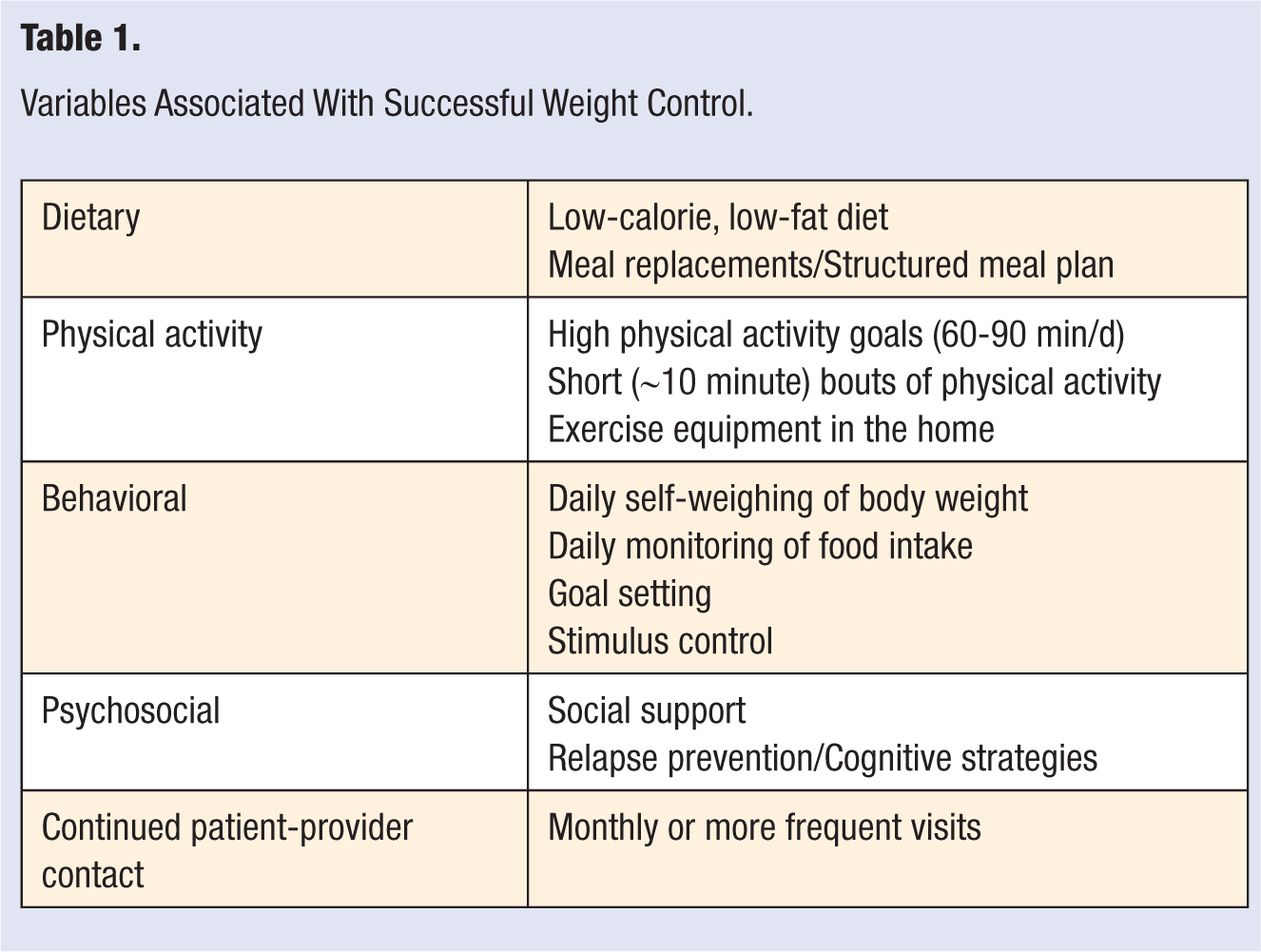
Lifestyle intervention treatment “packages” have been evaluated in several clinical trials and have shown positive long-term results on health outcomes.31,38-41 Most programs achieve average weight loss of 7 to 10 kg (7%-10% of initial body weight) at 6 months—most of which is maintained through 12 months. 42 Longer-term behavior trials, such as the Diabetes Prevention Program (DPP) 31 and the Look AHEAD (Action for Health in Diabetes) trial, 43 have found clinically significant weight losses averaging 4% to 5% (or 4-5 kg) at 3- to 4-year follow-ups. Although it is difficult to tease apart the precise components that contribute to success, research has identified some of the more salient features of lifestyle intervention programs, including dietary, physical activity, behavioral, and psychosocial strategies (Table 1).
Variables Associated With Successful Weight Control.
Dietary Prescriptions
Patients in behavioral weight loss programs are given calorie and fat gram goals designed to produce a weight loss of about 2 lb per week. The dietary prescriptions used have been derived from a large number of research studies comparing different degrees of caloric restriction and different macronutrient distributions.44,45 Typically, programs use calorie goals of 1200 to 1800 kcal/d, with the higher goal reserved for individuals who weigh more than 200 lb. Very-low-calorie diets (VLCDs) with <800 kcal/d were once popular and shown to produce weight losses of approximately 20 lb in 12 weeks. 46 However, weight regain often occurred when foods were reintroduced into the diet, and in the long term, patients treated with programs that combined behavior modification with VLCDs had weight loss comparable with those achieved in behavioral programs with balanced 1000- to 1800-kcal/d diets throughout.47,48 Thus, the field today has moved away from the VLCD regimens and now prescribes balanced deficit diets of 1200 to 1800 kcal/d.
The fat goal is typically 30% of calories from fat and is usually prescribed to patients in grams per day to facilitate self-monitoring. A focus on reducing dietary fat consumption is partly because of the evidence that overweight individuals49,50 and individuals with type 2 diabetes 51 have higher consumption of fat. Moreover, fat is more energy dense compared with other macronutrients (ie, fat has 9 kcal/g, and carbohydrate and protein have only 4 kcal/g). Several studies44,45 have found better weight losses when patients were given both calorie and fat gram goals than either goal alone. Thus, most behavioral treatment programs focus on reducing both calorie and fat intake.
Limiting carbohydrates (rather than fat) may also promote weight loss. Several studies have been conducted comparing low-carbohydrate diets (such as the Atkins diet) with low-fat regimens.52-56 One study included a significant proportion of patients with type 2 diabetes. 56 In general, the long-term results from these studies indicate similar weight losses between the different types of regimens (ie, low fat vs low carbohydrate). For example, results of a 2-year study, in which 307 obese participants who were randomly assigned to a low-carbohydrate or a low-fat diet, indicated weight losses of 11 kg (11%) at 1 year and 7 kg (7%) at 2 years, which did not significantly differ between the 2 dietary regimens. 52 What appears to affect weight loss in these studies is not the macronutrient distribution but rather the level of adherence to the regimen (and ultimately the number of calories that are consumed). Although weight losses achieved with very different macronutrient diets appear to be similar, those participants who are most adherent to any of the diets lose the most weight. 57
Promoting adherence to dietary prescriptions
Rather than simply telling patients to eat 1200 to 1800 cal/d, researchers have shown that incorporating frozen entrées or liquid formulas for some of the meals may help improve adherence to the calorie goals and consequently improve weight loss outcomes. Typical meal replacement programs fix the intake of 1 or 2 meals per day with a calorie-controlled, nutritionally balanced liquid shake and allow self-selected additional meals and snacks. In obese patients, diets with meal replacements have proven to be more effective than conventional diets. Wadden and Frey 58 used a balanced deficit diet that consisted of 4 servings daily of liquid meal replacement (150 kcal each) and a prepackaged dinner entrée (about 300 kcal) with salad. Those using this diet as part of an intensive behavioral program lost 14.5 kg after 17 weeks and maintained the weight loss at 1 year. In another study of overweight patients, those on the meal replacement regimen lost 7.3% after 3 months and 8.4% after 4 years, whereas the patients on the conventional diet had lost 1.4% and 3.2% at the same respective time points. 59
Meal replacements also offer a promising strategy for treating obese patients with type 2 diabetes. In one study of overweight patients with type 2 diabetes, patients in the meal replacement group achieved average weight losses of 5.2% at 6 months and 4.4% at 12 months, respectively, compared with 2.9% and 2.4% weight losses among patients prescribed an individualized dietary plan. 59 The ongoing Look AHEAD trial of 5145 individuals with type 2 diabetes has also incorporated meal replacements into the dietary plan and achieved clinically significant, long-term weight losses through 4 years of follow-up.
More structured regimens appear to facilitate greater weight losses by changing the types of food stored in the home, creating a more regular pattern of meal eating, reducing difficulty with estimating portion size and planning meals, and helping control eating when not hungry.60,61
Physical Activity
Regular physical activity generally results in modest, if any, initial weight loss62-66 but is critical in maintaining weight loss and preventing weight regain.67,68 Randomized studies evaluating diet alone (restricting food intake), exercise alone, and the combination of diet and exercise have consistently shown that combining diet and exercise results in the most weight loss. 63 Exercise (~60 min/d) is also the best predictor of weight loss maintenance.62,68 Independent of weight control, physical activity has several additional important health benefits; it increases cardiorespiratory fitness,69,70 increases insulin action,71,72 improves glucose control,14,16 and is related to a decreased risk for type 2 diabetes.73,74 Furthermore, exercise decreases the risk for CVD 75 and promotes a favorable lipid panel (lower triglycerides, total cholesterol, and LDL cholesterol and higher HDL). 76
Physical activity recommendations differ depending on the goals of an individual patient (eg, to lose weight, to prevent CVD risk factors, or prevent weight regain). In 2008, the Physical Activity Guidelines Advisory Committee Report recommended that adults participate in 30 minutes per day, 5 days per week of moderate-intensity aerobic exercise (eg, brisk walking) to reduce the risk for CVD and other metabolic diseases. 77 This physical activity recommendation is corroborated by other health organizations, including the American College of Sports Medicine (ACSM), the American Diabetes Association (ADA), the American Heart Association, and the World Health Organization.62,78-80 However, this dose of exercise (ie, 30 min/d, 5 d/wk or 150 min/wk) is recommended to reduce the risk for disease and is likely insufficient to lose weight or maintain weight loss. The Institute of Medicine 67 currently recommends 60 min/d every day of the week to maintain a healthy BMI and 60 to 90 min/d to promote weight loss maintenance.
Brisk walking is the most common type of exercise and is typically recommended in behavioral interventions and by public health guidelines.62,67,78-80 Some public health organizations recommend strength training in addition to walking (ADA, ACSM). For example, the ACSM developed the first public health guideline for adults to participate in regular strength training (ie, lifting weights).79,80 Even though adding strength training to an aerobic exercise program may not result in greater weight loss, 81 strength training increases blood flow to active muscle, 82 insulin action, 82 and muscular strength79,80 and is a necessary component of a physical activity program.
For promoting weight loss and maintenance, many randomized studies have shown that higher doses of exercise result in the best outcomes. Jakicic et al 83 found that women in a behavioral intervention reporting higher amounts of physical activity (ie, >200 min/wk) had greater weight losses at 6, 12, and 18 months compared with women with ≤150 min/wk of exercise (−13.1 kg vs −3.5 kg, respectively). Similarly, Jeffery et al 84 randomly assigned 202 overweight adults to a behavioral intervention focused on a 1000 or 2500 kcal/wk exercise goal. The 2500 kcal/wk exercise group, relative to the 1000 kcal/wk group, reported significantly higher exercise levels and greater weight losses at 12 (−8.5 kg vs 6.1 kg, respectively) and 18 (−6.7 kg vs 4.1 kg, respectively) months. However, after the intervention stopped at 18 months, activity declined in the higher exercise goal group, and there was no difference between groups in exercise and body weight loss at 30 months. 85 Thus, there may be more barriers to exercise with higher doses, and continuing to participate in a behavioral intervention may be necessary to maintain higher amounts of exercise.
Promoting adherence to exercise goals
Higher levels of physical activity are related to better long-term weight loss outcomes, so the question becomes, “How best to encourage overweight individuals to adopt and maintain a higher level of physical activity?” One approach may be to encourage more flexible exercise routines. Jakicic et al 86 conducted a study in which all women received the same diet, the same behavioral program, and the same exercise goals. However, a long-bout group did their exercise in one 40-minute bout, whereas a short-bout group was instructed to exercise in four 10-minute bouts. The short bout exercise prescription led to better exercise adherence; these participants were more likely to exercise on any given day and completed more minutes of exercise over the course of the program. Their weight losses were also somewhat better. Interestingly, the short- and long-bout groups had comparable improvements in cardiovascular fitness. Accumulating multiple daily short bouts of exercise (10 minutes, 3 times/d) may be easier for obese adults to achieve and result in long-term exercise adherence. 83
Another way to improve exercise adherence is to modify the environmental cues for physical activity. Cross-sectional and descriptive studies suggest that exercise levels are higher when exercise equipment is in the home.87,88 Similarly, Jakicic et al 83 showed that overweight women randomly assigned to exercise with home exercise equipment (vs exercise without home exercise equipment) showed higher exercise adherence and experienced greater weight and body fat loss. Taken together, these data suggest that having exercise equipment in the home may be a convenient way for individuals to accumulate multiple bouts of exercise and meet current physical activity recommendations.
Home-based exercise programs also appear to promote better adherence than supervised exercise in a controlled laboratory setting.83,89 Perri et al 89 found that in obese women after 12 months, home-based exercise increased exercise participation and adherence compared with supervised exercise. Also, in obese women, 12-month weight losses were greater with home-based exercise relative to supervised exercise (11.6 vs 7.0 kg, respectively).
Behavioral Strategies
Behavioral strategies typically include a combination of goal setting, self- monitoring with feedback, stimulus control, and relapse prevention/problem solving,39,40 as described below.
Self-monitoring
Self-monitoring of diet is often considered the single most important component of a behavioral weight control program.90,91 Patients are taught to write down everything they eat, to estimate or count calories in those foods by reading food labels or using a resource, and to record a running total of their calorie intake to ensure they stay within their calorie goal. Patients also write down each type of exercise, including minutes of time spent exercising and the number of steps taken each day, as tracked by a pedometer. The patient is then taught to use the diary to evaluate their current behavior, identify problems, and select specific behaviors to target for change. Interventionists review food and activity records and provide specific guidance on choices and supportive feedback to the patient.39,40 A number of clinical trials have found self-monitoring to be significantly correlated with short- and long-term weight loss outcomes.91,92
Participants are also given a weight goal and told to weigh themselves and to track progress toward their goal on a weight graph. Several studies have now shown that frequent self-weighing is a significant and independent predictor of weight loss, less weight regain, and avoidance of initial weight gain in adults.93,94 Daily self-weighing appears to promote the best weight control when combined with behavioral treatment programs.93,95,96 Of note, frequent self- weighing is not associated with an increase in adverse psychological outcomes, including depressive symptoms, eating disorders, or binge eating. 97
Although adherence to self-monitoring tends to wane over time, 98 the advent of new technologies, such as the Internet, PDAs, and smartphones, may make the task of self-monitoring easier for patients. Patients are able to look up the calories in a database and save the data to an online record. Similarly, a variety of instruments are now available that can provide objective measures of physical activity. These instruments may not only make self-monitoring easier but also provide a way for interventionists to monitor patients’ behaviors in real time and provide helpful guidance and support. 99
Goal setting
Typically, in behavioral weight loss programs, participants are given goals for calorie and fat, and physical activity, and asked to monitor and modify these aspects of their behavior. Although the overall behavioral goals are quite general (eg, eat 1500 kcal/d, <30% from fat, and exercise 200 min/wk), more specific daily goals (eg, use jam instead of butter on bread) help break the behavior change into small, achievable steps rather than a general goal, such as to eat less fat.
Stimulus control
Given the role of environmental factors in influencing weight, lifestyle programs typically teach stimulus control techniques to reduce the cues for inappropriate behaviors and strengthen the cues for appropriate eating and activity. Stimulus control techniques are designed to reorganize the environment to support desired behaviors. Patients are taught that it is difficult to consistently make good eating and exercise choices in an environment that does not support these behaviors. Thus, they are instructed to reduce or eliminate cues that encourage overeating/inactivity (eg, remove chips, desserts, or other tempting foods from the house and reduce the number of TVs in the home) and to make it as easy as possible to make good choices (eg, leave exercise equipment in a highly visible area and have low-fat snacks readily available in the refrigerator).
Psychosocial Strategies
Cognitive techniques
Cognitive therapy, when combined with diet and exercise interventions, produces superior weight losses than diet and exercise treatment alone. 38 Thus, the cognitive component is often added to effective treatment packages. Cognitive approaches to weight loss focus on identifying and modifying maladaptive thoughts or self-perceptions associated with overeating and inactivity, prior relapses, or prior treatment failures. Patients are taught to identify and change the maladaptive thoughts and emotions that can contribute to overeating and inactivity, including dichotomous thinking (eg, “If I can’t eat healthy at every meal, I might as well not do it at all”) and rationalizations (eg, “I’ve had a hard day, I deserve a piece of cake”). Patients are taught to replace these negative thoughts with more positive and helpful self-statements.
Relapse prevention
Based on Marlett and Gordon’s relapse prevention model, 100 patients are taught to identify specific situations that might pose problems for dietary or exercise adherence. Three common types of high-risk situations are situations involving food cues (eg, being in a restaurant or a family meal), situations involving affective cues (eg, anger and depression), and situations involving boredom or transition (eg, watching TV). Patients are taught to use behavioral coping strategies (such as leaving the room) and cognitive coping responses (devaluing the food) to prevent lapses from turning into relapses. Similarly, they are taught to use problem-solving techniques to overcome these and other barriers to their desired goals. 101
Providing participants with training in relapse prevention and with continued therapist contact improves long-term weight maintenance. 102 Relapse prevention training was provided to 1 group during the last 6 weeks of the treatment program; these participants were taught about the process of relapse and practiced skills needed to identify and cope with high-risk situations and to recover from acute lapses. The group that received both relapse-prevention training and continued therapist contact had significantly better weight maintenance than any other group. 102
Social support
Social support has long been studied as a way to improve motivation for long-term weight loss. Overall, findings on the effects of social support suggest a small positive effect. 103 Wing and Jeffery 104 examined the effects of recruiting patients with friends and providing a social support–focused treatment; the best weight losses were found in those individuals who were recruited with friends and provided with the social support intervention. However, other studies have shown more mixed results for social support. A meta-analysis of 21 articles examined the role of family support in weight loss and found inconclusive results; yet spouse involvement appeared to increase effectiveness. 105 Interestingly, Gorin and colleagues 106 showed that there is a ripple effect of participating in a weight loss program; not only do participants in a weight loss program lose weight, but their spouses do too, even though they do not attend meetings. Moreover, the greater the weight loss of the participant, the greater the weight loss in the spouse. Based on findings such as this, a common treatment strategy has been to ask spouses to actually participate in the weight loss program along with the participant and to make the same changes in eating and exercise behavior. Overall, support from social context is often helpful but can, for some patients, interfere with long-term weight management.
Ongoing Patient-Provider Contact
Contact with a practitioner appears to be the most effective approach to helping patients maintain their behavior changes. Several studies have shown the benefits of continuing to attend weight control classes in the long term.48,101,107,108 Continued contact (typically weekly for 6 months and every 2 weeks or monthly thereafter) seems to provide patients with the support and motivation needed to continue to practice weight control behaviors, such as diet monitoring and exercising regularly. The nature of contact (ie, individual vs group meetings) appears to be less important than continuing to see patients, but one randomized controlled trial found that group treatment induced a significantly larger weight loss than individual care, even in patients who indicated a preference for and received individual treatment. 109
Lifestyle Intervention Trials: Long-term Results
As noted earlier, several large, randomized controlled trials have now documented the long-term outcomes of comprehensive lifestyle intervention programs on weight loss, diabetes, and CVD outcomes. The 3 largest randomized controlled trials are the US DPP, 14 the Finnish Diabetes Prevention Study (DPS), and the Look AHEAD trial.16,32,110 All studies included a comprehensive behavioral weight loss program, as described above.
The US DPP 14 is the largest randomized trial to examine whether a comprehensive lifestyle intervention could prevent the progression of prediabetes to diabetes in 3234 overweight/obese individuals with prediabetes with impaired glucose tolerance. Participants were randomly assigned to lifestyle intervention, metformin (most commonly prescribed antidiabetes medication), or placebo (control) groups. The goals for the lifestyle group were to lose ≥7% of body weight by eating a low-calorie (1200-1800 kcal/d), low-fat (<30%) diet and exercising >150 minutes per week. In the lifestyle group, the behavioral intervention included 16 lesson plans incorporating diet, exercise, and behavioral modification to achieve group goals. Study interventionists met with participants weekly during the initial 24 weeks and at least bimonthly for the next 4 years. The metformin group received a dose of 850 mg twice daily plus a standard lifestyle care recommendation. The placebo group received a standard care lifestyle recommendation. Mean body weight loss was greater in the lifestyle group relative to the metformin and placebo groups (−5.6, −2.1, and −0.1 kg, respectively).
The DPP intervention was extremely effective in reducing the risk of diabetes. The crude incidence of diabetes was 11.0, 7.8, and 4.8 cases per 100 person yeas for placebo, metformin, and lifestyle, respectively. Thus, lifestyle intervention reduced the risk of diabetes by 58% compared with placebo and by 39% compared with metformin (Figure 1). Weight loss was the best predictor of a lower incidence of diabetes. 111 Furthermore, with every kilogram of weight lost, there was a 16% reduction in risk for diabetes. 111 Those who met all lifestyle intervention goals had an 89% reduced risk of diabetes. Finally, those who met physical activity goals without meeting the weight loss goal (>7%) decreased the risk of diabetes by 46%.
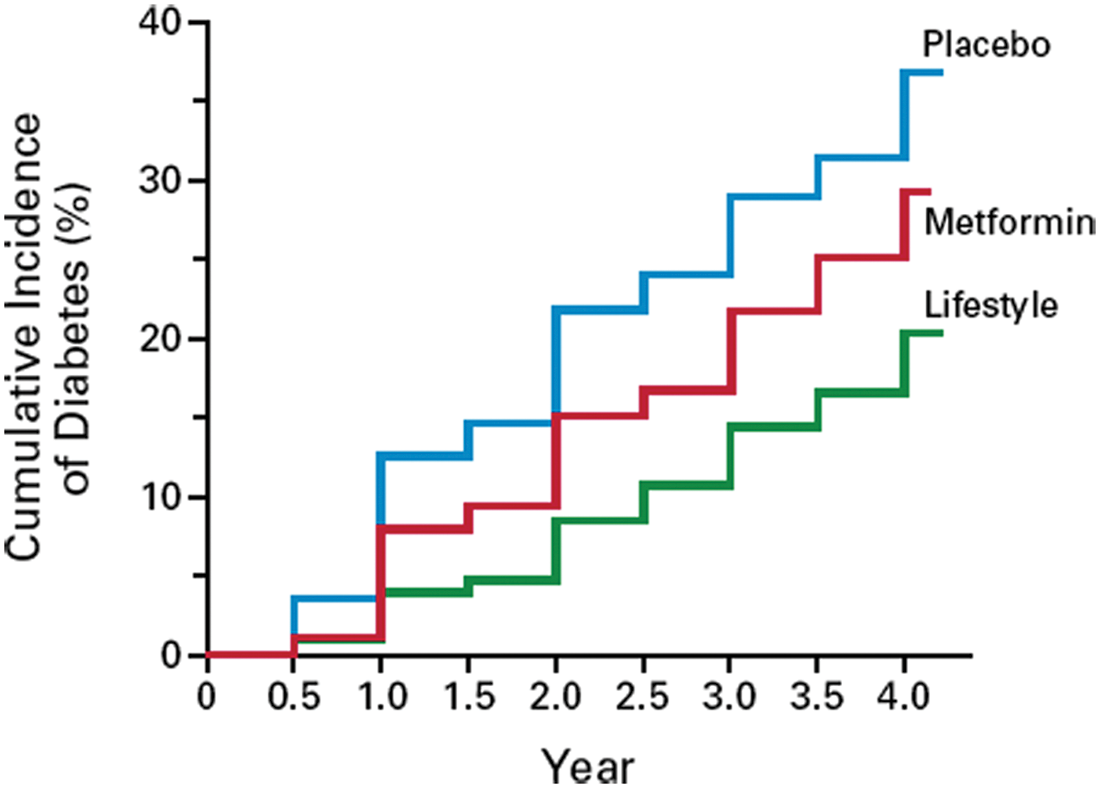
Incidence of diabetes in the placebo, metformin, and lifestyle intervention group after 4 years in the US Diabetes Prevention Program. Lifestyle intervention reduced the incidence of diabetes by 58% and 39% compared with placebo and metformin, respectively (reprinted from Knowler et al 14 ; used with permission).
In the Finnish DPS16,32 522 overweight men and women with impaired glucose tolerance were randomly assigned to a lifestyle intervention or control group. The behavioral goals of the lifestyle group were to lose ≥5% of body weight, reduce fat intake to <30% of total food intake, and exercise for ≥30 min/d. Those in the lifestyle group were given guidance on how to accomplish these goals and had a visit with a nutritionist 7 times during the first year and 4 times each year after that. Guidance was individually tailored to each participant throughout the intervention. The control group received general information about lifestyle modifications to reduce the risk for type 2 diabetes at the start of the study alone. In the lifestyle group compared with controls, after 1 year, 43% versus 13% of participants reduced body weight by >5%, 47% versus 26% reduced fat intake to <30% of food intake, and 86% versus 71 exercised >4 h/wk. In the lifestyle group, mean body weight loss was 4.7% after 1 year versus 0.9% in the control group. The greater weight loss in the intervention group was accompanied by significantly greater decreases in blood pressure and concentrations of glucose, insulin, and triglycerides relative to controls. Similar to the DPP, the cumulative incidence of diabetes was 58% lower in the lifestyle intervention group compared with the control group after 6 years (Figure 2), and weight loss was the best predictor of remaining free of diabetes. Furthermore, all participants meeting at least 4 of 5 prescribed lifestyle goals remained free of diabetes.
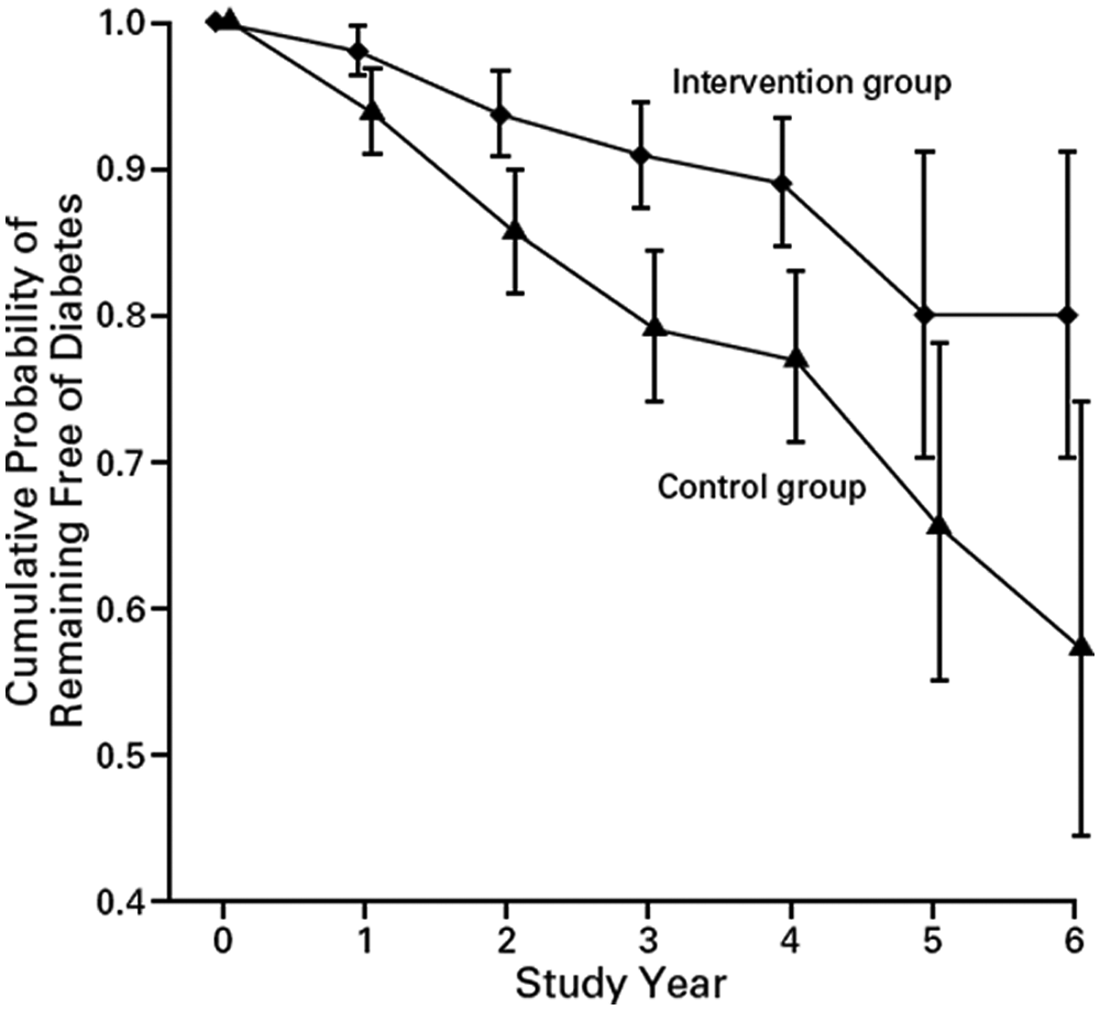
Proportion of overweight or obese participants without diabetes after 6 years in the Finnish Diabetes Prevention Study. The risk for diabetes was reduced by 58% in the lifestyle intervention group compared with the control group (reprinted from Tuomilehto et al 16 ; used with permission).
Look AHEAD is a multicenter, ongoing randomized study evaluating whether an intensive lifestyle intervention lowers the risk for cardiovascular morbidity and mortality in 5145 overweight or obese individuals with type 2 diabetes.15,23,33,34,112,113 Overweight or obese adults (BMI > 25 kg/m2) were randomly assigned to an intensive lifestyle intervention group or a standard care (control) group. The 2 main goals of the lifestyle intervention are for participants to lose ≥7% of body weight and increase physical activity to ≥175 min/wk using a comprehensive behavioral intervention. The intervention is similar to the US DPP. Participants in the lifestyle group attended weekly behavioral meetings during the initial 6 months, 3 meetings per month from 6 to 12 months, and ≥1 meeting per month from years 2 to 4. The meetings focused on diet (including use of meal replacements), physical activity, and behavioral approaches to meet the weight loss goals. After 4 years, participants in the lifestyle group, relative to controls, had a greater percentage of weight loss (−6.15% vs −0.88%, respectively) and greater mean change in glycated hemoglobin (HbA1c; −0.36% vs −0.09%, respectively), systolic blood pressure (−5.33% vs −2.97%, respectively), triglycerides (−25.56% vs −19.75%, respectively), HDL cholesterol (3.67% vs 1.97%, respectively), and cardiorespiratory fitness (+12.74 vs 1.96%, respectively). Moreover, in the lifestyle group compared with controls, after 4 years, 35% versus 18% of participants reduced weight by ≥7% weight, and 23% versus 10% of participants reduced weight by ≥10% weight (Figure 3). 114 Furthermore, participants in the lifestyle intervention had lower medication use and lower costs to treat CVD risk factors. 33 These data clearly show that a comprehensive behavioral intervention can promote long-term weight loss and improve CVD risk factors in overweight or obese men and women with type 2 diabetes. The ongoing intervention over the next 7 years will determine whether CVD mortality is lower in the lifestyle group.
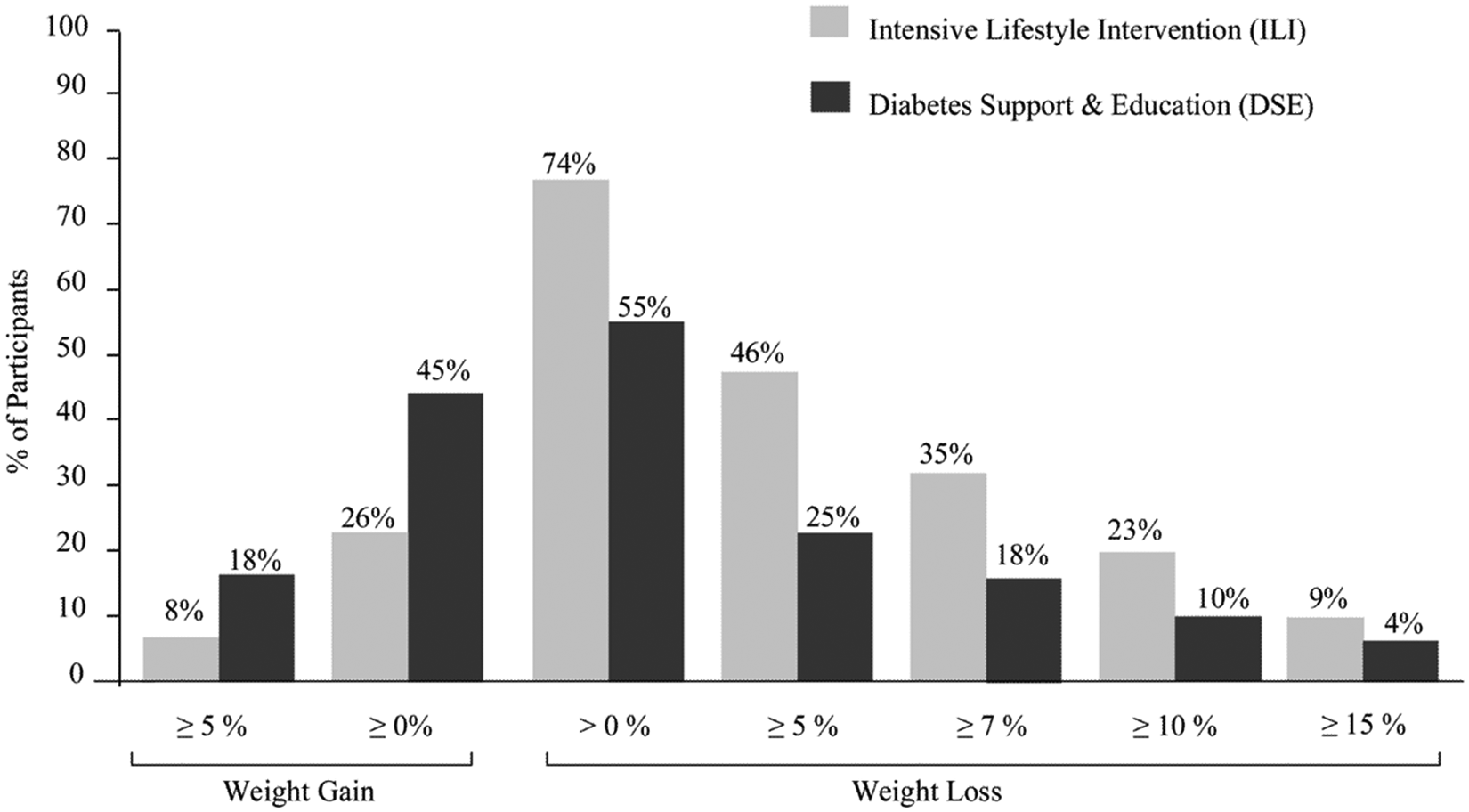
Percentage of participants in diabetes support and education (DSE; controls) and intensive lifestyle intervention (ILI) groups who at 4 years met different categorical weight losses in Look AHEAD. Significantly greater percentage of participants in ILI, relative to controls, met each of the categorical weight losses shown (P < .0001; reprinted from Wadden et al 114 ; used with permission).
Look AHEAD has documented the beneficial effects of weight loss on glycemic control in individuals with type 2 diabetes. However, other randomized studies using less-intensive dietary interventions in individuals with or without diabetes have shown no significant improvements in HbA1c levels with modest weight losses (−0.8 to −4.6 kg).115-119 Brinkworth et al 115 showed that 12 weeks of energy restriction resulted in weight loss of 5.3 kg and lower HbA1c levels. At 64 weeks, the average weight loss was -2.9 kg, yet HbA1c levels had increased to baseline levels. Redmon et al 118 found that a dietary intervention for 2 years resulted in a weight loss of 4.6 kg, but HbA1c levels were not significantly lower than at baseline. These studies suggest that low-intensive lifestyle interventions may have less of an impact on glycemic control compared with more comprehensive behavioral interventions that target both diet and physical activity, as was done in Look AHEAD.
In sum, the Finnish DPS, US DPP, and Look AHEAD provide strong support that long-term comprehensive lifestyle modification can prevent or delay the progression of prediabetes to diabetes and improve type 2 diabetes and CVD risk factors. Taken together, these data suggest that (1) lifestyle modification, with a modest amount of weight loss (5%-7%), reduces the incidence of diabetes by up to 58%; (2) weight loss is the best predictor of reduced risk for diabetes; (3) meeting prescribed lifestyle goals substantially lowers the risk for diabetes; (4) increased physical activity, with modest weight loss, independently lowers the risk for diabetes; and (5) in adults with type 2 diabetes, modest weight loss improves glycemic control, improves CVD risk factors, and decreases medication use.
Long-Term Successful Weight Loss
Even though many overweight or obese individuals are able to achieve weight loss, many regain lost weight within 2 to 5 years after treatment termination.120-122 Whether the high degree of recidivism after weight loss is a result of biological or behavioral factors remains unclear. Some investigators conclude that weight regain is inevitable as a result of strong biological pressures to return to an obese body weight. 123 For example, weight loss generally alters peripheral hormones in a direction expected to stimulate food intake and promote body weight regain.124,125 Cummings et al 124 observed that a 17% weight loss was associated with a 24% increase in ghrelin concentrations, indicating more stimulus to eat. Sumithran et al 125 found that 1 year after 14% weight loss, the hormonal response was altered (higher ghrelin, lower leptin, and insulin) in a direction expected to stimulate food intake and promote weight regain. Taken together, these data suggest that overweight or obese individuals who lose weight have a strong physiological drive to regain lost body weight. On the other hand, other researchers believe that weight regain is a result of the inability to maintain behavior changes in the long term 126 or a result of environmental relapse, in which it is impossible to maintain appropriate diet and physical activity patterns within an environment that promotes energy intake and discourages physical activity. 127 Clearly, there is more work to be done in this area of understanding the relative role of behavioral and metabolic factors in predicting weight gain and weight maintenance.
It is interesting to note that although there is great pessimism surrounding the issue of long-term successful weight loss, this belief does not appear to be well substantiated. In the Look AHEAD lifestyle intervention, for example, 37% lost at least 10% of their body weight at year 1 and 23% continued to maintain a weight loss of at least 10% of their body weight at 4 years (Figure 3). Similarly, several epidemiological studies suggest that approximately 1 out of 3 overweight individuals are successful at maintaining a 5% to 10% weight loss for 1 year or more.128,129 Thus, there may be greater levels of success than is often recognized.
In an effort to learn more about successful weight loss maintenance, Hill and Wing established the National Weight Control Registry (NWCR) in 1994. 130 The registry enrolls individuals who are older than 18 years and who report having lost at least 30 pounds and kept it off for at least 1 year. There are currently 10 000 individuals in the NWCR. Although this is not a representative sample of all successful losers, it provides a rare opportunity to examine weight loss maintenance. The registry members are 77% women, 82% college educated, 95% Caucasian, and 64% married; average age is 46.8 years. Participants report having lost an average of 33 kg, reducing from a BMI of 36.7 kg/m2 to 25.1 kg/m2 and maintaining a weight loss of at least 30 lb for an average of 5.7 years; 13% have maintained the weight loss for at least 10 years. Thus, by any criterion, these individuals are clearly successful.
Nearly all the NWCR members reported using both diet and physical activity to lose weight and keep it off. 130 Reported calorie and fat intake were low (averaging 1380 kcal/d with 24% as fat), and exercise levels were very high (2800 kcal/wk). These data underscore the importance of these 2 behavioral strategies for long-term weight control. The successful weight loss maintainers also reported frequent self-weighing—typically daily 131 —consistency in their diet across weekdays, weekends, and holidays, 132 and low levels of television viewing. 133 They also report a healthy home food and exercise environment with significantly more pieces of exercise equipment and healthy foods in visible locations in the home than an overweight control group. 134 Findings from the NWCR suggest that successful weight loss maintenance is possible through hard work and dietary and exercise vigilance.
Newer Treatment Modalities
Short- and long-term behavioral interventions are clearly successful at reducing body weight. However, these interventions typically require substantial face-to-face visits, are not available to the public, and are difficult to disseminate and costly to implement. Internet-based interventions may potentially lower the costs of traditional face-to-face behavioral interventions. In one of the first randomized, short-term studies, Tate et al 135 assigned 91 overweight adults to an Internet education (control) or Internet behavioral intervention group. The Internet behavioral intervention consisted of 24 weekly lesson plans via e-mail and weekly online submission of food diaries. Weight loss was significantly greater at 3 and 6 months in the Internet intervention relative to controls (4.1 kg vs 1.6 kg, respectively), suggesting that an online behavioral approach can produce clinically significant weight loss.
Subsequent studies have compared online versus traditional face-to-face interventions. In the weight loss maintenance trial, 136 participants who had lost at least 4 kg during the initial 6-month program were randomly assigned to either an interactive technology–based maintenance program, a personal contact intervention (monthly phone contacts and quarterly in-person sessions), or a minimal contact control. The in- person program produced the best long-term results, with no difference between the other 2 conditions. Similarly, face-to-face contact was also most effective in the STOP Regain trial. 137 In this trial, participants were recruited after having lost weight either on their own or through any type of program. They were then offered an 18-month self-regulation program designed to help them maintain their weight losses. The program taught participants to self-weigh daily and to use the information from the scale to determine if adjustments in diet and activity were needed. Those who were randomly assigned to receive the program through monthly face-to-face meetings had better outcomes than those who received the program via the Internet, with the poorest results in those in the control group.
Although greater weight losses are often seen in face-to-face compared with online modalities, Internet-based interventions nonetheless produce clinically significant weight losses (>5%) that are maintained over time.85,99,138-142 Moreover, an appealing aspect of Internet interventions is the cost-effectiveness relative to standard behavioral interventions. Krukowski et al 143 recently found that a standard behavioral intervention with substantial face-to-face contact cost $372 per person, whereas an Internet intervention cost $158 per person. The main difference in cost was participant travel time, and the Internet intervention substantially lowered this burden. Thus, preliminary data suggest that Internet behavioral interventions are successful at lowering and maintaining body weight in a cost-effective manner.
Other emerging approaches to promoting weight loss and maintenance include use of text messaging and smart phone applications. Patrick and colleagues 144 evaluated the efficacy of a weight loss intervention delivered via text messaging on mobile phones and found, after 4 months, that the intervention group lost more weight than the control group (2-kg difference). Other ongoing randomized studies145-147 are evaluating the effects of smart phone applications designed to alter diet and physical activity. Coons et al 145 compared whether a smartphone weight loss system, designed to promote self-regulation and adherence, is better than a standard face-to-face behavioral intervention. Others are currently assessing whether interactive digital games and new movements (exergaming) may increase energy expenditure and promote weight loss. 147 Clearly, this line of research is in its infancy but exciting for its potential to increase dissemination and, potentially, the efficacy of existing interventions.
Weight Loss Counseling in the Clinic Setting
For practitioners treating an obese patient with or without type 2 diabetes, a critical question is, “How do I incorporate these behavioral interventions into my practice?” Practitioners face multiple barriers to effectively delivering behavioral treatment, including limited coverage by insurance, limited time and training in behavioral techniques and obesity treatment, competing needs to handle co-occurring medical issues, and perceived lack of interest on the part of their patients. 148 Although available data do not yet indicate how best to incorporate providers in more intensive approaches to achieve successful weight loss, 149 exciting innovations are under way.150,151 Moreover, practitioner advice can have a powerful impact on a patient initiating weight loss efforts. Galuska and colleagues 152 found that physician advice to lose weight led to a 3-fold increase in patients’ attempts to address their weight. Several government bodies and panels have come up with recommendations on how physicians and other practitioners can best address overweight in clinical practice.153-158 Here, we highlight the following key points:
In discussing weight with patients, consider the terms used to describe excess weight. Wadden and Didie 159 conducted a survey of obese patients and found that patients’ least desirable terms for referring to their weight included Fatness, excess fat, obesity, and large size. The more desirable terms were weight, heaviness, BMI, and excess weight.
Make specific calorie recommendations (eg, 1200-1800 kcal/d) and progressive exercise goals leading to the ultimate goal of 60 to 90 minutes of activity per day. Diet and exercise goals should be achievable, with the initial goal of modest weight loss (7%-10%). Also, patients should be encouraged to start monitoring daily food intake, exercise, and weight.
Practitioners should refer obese patients to other practitioners (eg, dieticians, psychologists, and exercise physiologists) or commercial weight loss programs for additional support and guidance. In a multicenter study, Heshka and colleagues 160 found that participants in Weight Watchers lost 5.3% of their initial weight at 1 year and maintained a loss of 3.2% at 2 years, compared with 1.5% and 0%, respectively, among those who received self-help materials. Commercial programs remain a potential avenue for supplemental help, although additional research is clearly needed in this area. 161
Continue to meet with patients and discuss weight and behavior changes on a regular basis. Practitioners can support patients’ weight loss efforts by reviewing changes in weight and health complications at regularly scheduled office visits and by monitoring patients’ efforts to improve their eating and activity behaviors. 161 Success in weight loss should be reinforced and praised, and setbacks should be met with empathy and with encouragement to maintain weight control efforts.
The National Institutes of Health is currently funding ongoing interventions focused on clinic-based support for patients to lose body weight.150,151 The premise behind this funding is that physicians may have a profound impact on the success of their patients’ weight loss, but contact with the patient is limited and typically involves many other medical concerns or priorities. Christian and colleagues150,151 have developed a brief, computer-based program that provides feedback to patients on improving barriers to diet and physical activity. Preliminary data are encouraging because overweight adults achieved an average 1.6% (−1.6 kg) weight loss over 12 months. Moreover, 26.3% of adults in the intervention sustained weight loss of ≥5%. In the not-so-distant future, computer and other programs similar to this one may be available to providers to help their patients lose weight and reduce the risk for chronic disease.
Conclusions and Future Directions
Comprehensive lifestyle interventions are clearly successful at promoting long-term weight control and lowering the risk for type 2 diabetes and associated complications. Modest weight losses (5%-10%) may have a profound impact on the risk for diabetes in adults. Moving forward, there are several promising areas of investigation. Here, we highlight 3 areas.
Prevention
There are several areas to pursue for the primary prevention of obesity and type 2 diabetes. Although not the focus of this review, prevention of obesity in children has become a major area of interest. 162 Obesity in childhood is associated with obesity in adulthood; the older the child, the stronger the association between childhood and adulthood weight status. Moreover, childhood obesity is associated with the same health consequences as adult obesity, including increased risk of developing type 2 diabetes. There are some promising interventions for prevention of weight gain in children and adolescents 20 but new advances and more effective and disseminable programs are clearly needed. 21
Given the high prevalence of obesity and type 2 diabetes in minority populations, development of effective interventions for at-risk, underserved populations is also critical. More research is needed to examine the impact of culturally adapted weight loss and weight gain prevention approaches, taking into consideration the ways in which minority populations interact with the health care system, school system, and community settings.
Finally, the time surrounding pregnancy is another understudied and potentially powerful “teachable moment” for obesity and diabetes prevention. 163 Gestational diabetes (GDM) is a strong risk factor for the development of type 2 diabetes in women. An estimated 3% to 70% of women with GDM will develop type 2 diabetes within 15 years after the birth of their child.164-167 Women with recurrent GDM and obesity are at even greater risk of developing type 2 diabetes.168,169 Preliminary research from epidemiological and bariatric surgery studies suggests that development of GDM may be prevented by reducing prepregnancy body weight.170-173 However, future research is needed to examine the effects of lifestyle interventions before pregnancy to reduce body weight and prevent GDM. More research has been done during pregnancy, and initial findings suggests that helping women gain the recommended amount during pregnancy through healthy eating and physical activity could make a major contribution to preventing postpartum weight retention, particularly in normal weight women.174-176 However, more randomized controlled trials are needed to identify the effects of lifestyle interventions on maternal/fetal outcomes and the most effective and disemminable interventions during pregnancy, particularly for obese women.
Maintenance of Long-term Weight Control
Weight loss is clearly effective in preventing and treating a variety of health problems, including type 2 diabetes. Moreover, over the past several decades, our ability to help overweight/obese patients lose weight has significantly improved. Nonetheless, over time, there is gradual weight regain, with an average weight loss of 4 to 5 kg at the 3- to 4-year follow-up. Many individuals fail to maintain a ≥5% weight loss. It is critical that we better understand the environmental, physiological, and psychosocial variables that make it difficult to maintain weight loss in the long term and lead to this gradual regain. Efforts to overcome these barriers with new behavioral approaches may lead to better long-term outcomes.
Dissemination
Lifestyle programs can have important health benefits, but they are still not readily available to many overweight or obese individuals. Further research is needed to determine how best to disseminate these programs. As noted earlier, of particular interest is the role of technology in expanding the audience who could participate. Whether these programs should be covered by insurance programs is another important public health issue. Efforts are also needed to document the outcomes that participants experience in commercial weight loss programs, including both face-to-face and Internet approaches, in order to determine what role these programs can play in combating the obesity epidemic.
It is critical that practitioners continue to work with scientists and policy makers using a multidisciplinary approach to further understand how best to prevent and treat obesity and its profound health consequences, including type 2 diabetes.
