Abstract
Probiotics are live microorganisms present in several foods and nutritional supplements that may prevent or limit the effects of various illnesses and infections and elicit a range of health benefits in physically active individuals. The primary clinical areas of interest with probiotics include metabolism; gastrointestinal, inflammatory, and functional disorders; the respiratory system; and a range of infections and allergies. The gastrointestinal tract is a key element controlling and regulating adaptation to exercise and physical activity. Gut symptomatology such as nausea, bloating, cramping, pain, diarrhea, and bleeding occurs in some highly active individuals such as athletes, particularly in prolonged exhaustive events. A small number of studies examining probiotic supplementation in highly active individuals indicate modest clinical benefits in terms of reduced frequency, severity, and/or duration of respiratory and gastrointestinal illness. The purported mechanisms of action for probiotics include direct interaction with the gut microbiota, interaction with the gut epithelium and mucosal immune system, and via immune signaling to organs and systems including the liver, brain, and respiratory tract. Future research will identify additional biologically and clinically beneficial strains, validate multicomponent formulations, clarify dose–response issues, and inform the development of guidelines for clinicians, health care practitioners, and the general community.
There is growing interest in probiotics in the research community, the food industry, and the general public.1-3 Although the potential applications and benefits of probiotic supplementation have only been explored extensively in the past decade, the discipline of nutritional immunology dates back to the 19th century. 4 The research community is investigating the beneficial effects of probiotics as a treatment of selected acute and chronic illnesses and boosting the immune system in a range of population groups including children, healthy active adults, and the elderly. The food industry is developing a range of food ingredients and products promoting a healthy lifestyle. The consumer is interested in these products for a variety of reasons, including healthy eating and improved health. There is substantial interest is the use of probiotics in healthy physically active individuals who seek guidance and advice on better ways to maintain health and physical performance.5,6
Although the potential applications and benefits of probiotic supplementation have only been explored extensively in the past decade, the discipline of nutritional immunology dates back to the 19th century.
Although most of the clinical and research attention has been directed toward acute and chronic disease, there is increasing interest in the use of probiotics to enhance health and well-being in disease-free physically active individuals. These individuals in a variety of occupational (eg, military, emergency services, construction, and outdoor industries), recreational, and sporting settings typically place a high priority on good health to maintain physical performance. The gut plays a primary role in mediating defense against infection and regulating mucosal homeostasis. 7 Better understanding of the gut, including the immune system and microbiota, is needed to identify and develop more effective methods of diagnosis, treatment, and clinical/lifestyle management. In the context of well-trained athletes, the gastrointestinal (GI) tract is a key element regulating adaptive and recovery processes related to the high levels of physical exertion. 8 Host defense at mucosal surfaces should protect individuals against dietary-related pathogens. However, during prolonged exhaustive and/or intensive exercise immunoregulation may be disturbed, increasing the risk of gut symptomatology such as nausea, bloating, cramping, pain, diarrhea, and bleeding. 9 Nutritional supplementation is one means of maintaining immune competence and limiting the risk of gut problems impairing physical activity and sports performance. In this review, we explore recent advances in our understanding of the relationships between the human gut microbiota, probiotic supplementation, and the human immune response; summarize the outcomes of research examining the clinical and biological effects of probiotic administration in physically active groups; and present some recommendations for clinicians and future directions for researchers.
Human Gut Microbiota
Our understanding of the role of microbiota in gut health and general well-being is increasing rapidly. Biomedical research encompassing clinical medicine, gastroenterology, endocrinology, immunology, and nutrition is furthering our understanding of gut health. The mucosal lining of the GI tract represents the first line of defense against invading pathogens—an important element is the capacity to differentiate between pathogenic and nonpathogenic commensal microbes. This arrangement has led to the public image of the good and bad bacteria. 10 Colonization of the human gut begins at birth and is influenced by a broad range of genetic and environmental factors, although the relative influence of the 2 types is not entirely clear. 11 It is generally considered that diseases such as obesity, metabolic syndrome, and colitis are affected by host genetic and environmental factors.
The linings of the mucosa, including the gut, genitourinary tract, and respiratory tract offer an ecological niche for bacterial communities to thrive. The gut is by far the largest physiological compartment inhabited by bacteria. 12 The GI tract is home to more than 1000 species of bacteria, which can be assigned largely to 2 phylotypes, the bacteriodetes and the firmicutes. 13 The degree to which the composition of the GI microbiota is determined by host genetics and/or environmental influences is an important question to answer in relation to health and disease. Identifying a composition associated with normal gut function and immune homeostasis that promotes good health would open opportunities for the prevention and treatment of disease.
The make-up of the microbiota reaches its final “adult” state during childhood and is considered to be relatively stable throughout the remainder of life. Metagenomic sequencing has revealed that individuals appear to have a core group of bacteria inhabiting the GI tract, now termed the core microbiome.14,15 These findings are consistent with analysis by other methods for investigating the human microbiota. 16 Investigation of familial groups indicates that family members have a more similar composition than individuals not related to each other, suggesting that a shared environment may play a more substantial role in determining bacterial composition. However, studies of lean and obese twins reveals a substantial variation in bacterial diversity. 17 Nutrient load can exert substantial influence on the structure of the bacterial community, 18 indicating that environmental factors may play an important role in determining microbiota composition beyond the core species.
Clinical Issues
The start of the 21st century has seen a shift in mortality from illness and infection to aging and lifestyle diseases. Several chronic diseases are associated with disturbance of the microbiota including obesity, inflammatory bowel disease, and diabetes. 11 The primary question is whether the diseases are caused by disturbances of the microbiota or alternatively that the diseases themselves are responsible for alterations in the microbiota. There is now general consensus that inflammatory bowel disease, Crohn’s disease, and ulcerative colitis are related to an abnormal immune response to microbes in the GI tract. Beyond the GI tract, a range of other illnesses have been linked to immune system dysfunction including Helicobacter pylori infection, atopic dermatitis, and allergic disorders. Two potential mechanisms for the abnormal immune response have been suggested: an increased proinflammatory response to a bacterial component or alternatively a decreased immune regulatory response. 10 In the latter case, an excessive effector immune response might increase the risk of a secondary autoimmune disorder. Probiotics are a potential strategy to correct these aberrant immune disorders.
A substantial proportion of the probiotics literature relates to study of supplementation in children. The evidence for beneficial effects of probiotics in children has been built initially on a large number of randomized controlled trials and some meta-analyses. 19 The increasing numbers of elderly individuals in Western countries provides challenges for clinicians, health and aged care facilities, and government policy makers. Advancing age is associated with an increased prevalence of disease, increased medication usage, and higher health care costs. Immunosenescence is characterized by a decreased ability of the immune system to respond to foreign antigens and decreased ability to maintain tolerance to self-antigens. 20 The modulation of immune effector responses appears to result in an increased susceptibility to common infection and cancer and reduced responses to vaccination. The mechanisms underlying immunosenescence comprise a series of cellular and molecular events involving alteration of several biochemical pathways and different cellular populations. 20 There is substantial interest in whether dietary strategies or interventions involving macronutrients, antioxidants, and/or probiotics 21 might offset the natural rate of immunosenescence. A recent study with Lactobacillus acidophilus NCFM in 51 elderly individuals elicited substantial beneficial changes in the gut microbiota (fecal quantities of phylogenetic bacterial groups) after only 2 weeks of supplementation. 22 Another dose–response study of Lactobacillus plantarum CECT7315/7316 in 50 elderly subjects for 12 weeks found those at the low dose had increased T-helper and B-lymphocytes and antigen-presenting cells, whereas at the high dose there were substantial increases in the percentages of activated T-suppressor and NK cells. 23
The mechanical and metabolic stress of exercise and physical activity is thought to increase the risk of gut problems in some individuals. A number of studies indicate a moderate to high prevalence of GI problems during exercise, especially those individuals competing in prolonged endurance events such as long-distance runners, cyclists, triathletes, and ultra-endurance marathons. 24 The well-known problems of nausea, cramping, bloating, and occasionally diarrhea are thought to relate to redistribution of blood flow from the gut to skeletal muscle and the peripheral circulation for cooling. It is also possible that the mechanical stress of exercise, particularly running, may exacerbate the movement of the gut, increasing the likelihood of gut symptoms. 25 The interaction of exhaustive exercise, adverse environmental conditions (particularly high ambient temperatures and humidity), and interactions between the gut, dietary composition, and fluid intake may also increase risk of gut problems. 26 Probiotic supplementation in combination with other dietary strategies around the intake of macronutrients and micronutrients could be a useful strategy to assist athletes and highly active individuals who have a history of gut problems affecting their performance.
There are new lines of research into the potential benefits of using probiotics as an adjuvant to boost the effectiveness of vaccinations. Live-attenuated influenza vaccine protects against influenza by mucosal activation of the immune system. Studies in animals and adults have demonstrated that probiotics improve the immune response to mucosally delivered vaccines. A randomized double-blind placebo-controlled pilot study showed that 28 days of Lactobaccillus GG (ATCC 53101, Culturelle) supplementation improved rates of seroconversion after administration of live-attenuated influenza vaccine in 42 healthy adults. 27 With the H3N2 strain, 84% of the subjects receiving the probiotic versus 55% receiving placebo had a protective titer 28 days after vaccination. Lactobacillus GG appears to have potential as an important adjuvant to improve influenza vaccine immunogenicity—future studies may identify other species with similar effects. More work is also needed to examine vaccine-naïve or sero-negative subjects and target mucosal immune responses in groups known to have poor response to influenza vaccines. 27
Probiotics as a Nutritional Supplement
The World Health Organization (WHO) definition of probiotics is widely used in the scientific and medical literature. Probiotics are defined by the WHO as live microorganisms, that when administered in adequate amounts, confer a health benefit on the host. A recent review of probiotics reported that more than 700 randomized, controlled studies using probiotics have been conducted in human subjects. 28 A number of reviews conclude there is sufficient evidence for using probiotics in the clinical prevention and treatment of GI tract disorders and the metabolic syndrome. Probiotics can be effective in the prevention and management of acute gastroenteritis and rotavirus diarrhea, antibiotic-associated diarrhea, intestinal inflammatory disorders such as Crohn’s disease and pouchitis, and pediatric atopic disorders. 1 Probiotics consist of yeast or bacteria, especially lactic acid bacteria, and are available in capsule form, as a powder, or in selected dairy products such fermented milk or yoghurt. Probiotics exhibit strain-specific differences in their resistance to acid and bile, ability to colonize the GI tract, clinical efficacy, and in the type and magnitude of benefits to the health of the host.
By definition, probiotic microorganisms are alive at the time of administration. The requirement for live microorganisms raises a whole range of quality control issues relating to product packaging, shipping, storage, and viability. In the research laboratory, the viability of specific strains can be determined, although this is not practical in the clinic and household. Probiotics are generally regarded as safe and well tolerated. Some probiotics may be contraindicated in patients who are immunocompromised or have a severe underlying illness, given the small possibility of causing fungemia and bacteremia. Researchers and clinicians need to consider the likelihood (probability) of both clinical benefit and harm of nutritional (and lifestyle) interventions. 29 New analytical approaches have emerged that permit a detailed investigation of the likelihood of adverse effects, which should also be a priority. In many cases probiotic administration in healthy individuals elicits an increase in mild transient gut issues such as flatulence, changes in bowel habits, and stomach rumbles. 30 Rare cases of more severe side effects and mortality 31 serve as a timely reminder of the potency of viable microorganisms.
From a clinical perspective there are many questions on the optimum dose, frequency, and duration of administration of probiotics in the wide spectrum of clinical settings. Individual probiotic strains vary in their ability to modulate the immune system. The 2 most extensively studied probiotic species are L acidophilus 32 and lactic acid bacterium or Bifidobacterium lactis. 33 The differential effects of probiotic strains on the immune system means that the efficacy of each strain needs to be evaluated independently in an appropriately conducted experimental trial. Our reading of the literature indicates that a minimum of 2 billion (2.0 × 1012) colony forming units are apparently required to elicit substantial changes in the microbiota to underpin positive clinical benefits. In general, probiotic bacteria are delivered encapsulated to survive gastric transport. A number of studies have investigated the delivery of probiotics with dairy products (yoghurt or milk) or as a powder to be mixed in a drink. Differences in the delivery method could yield variable outcomes. For example, the outcome of interest (eg, GI illness vs the common cold) needs to be considered when deciding on the type, dosage, and route of delivery of a probiotic supplement. Questions also remain on the frequency and duration of supplementation. It is commonly accepted in clinical research that 10 to 14 days of supplementation are needed to elicit substantial changes in the microbiota, and the opposite is true following the cessation of supplementation. It is also unclear whether daily supplementation over an extended period is necessary to sustain the initial changes in bacterial diversity. It is possible that supplementation is more beneficial when used over short durations than longer more continuous periods. Further research is needed to clarify these questions.
The authoritative Cochrane systematic reviews of primary research are highly valued in interpreting the level of evidence for human health care and policy formulation. The Cochrane review for probiotics and upper respiratory tract illness (URTI) concludes that the evidence from 10 controlled trials involving 3451 subjects was weak, but shows a benefit in using probiotics to prevent acute URTI. 34 The mean effect of probiotics was a ~40% reduction in the likelihood of an URTI episode and ~30% reduction in the rate of antibiotic prescription for acute URTIs. Similarly, the Cochrane review for probiotics and acute infectious diarrhea illnesses identified 63 trials involving 8014 people—mainly infants and children. 35 Nearly all studies reported a shortened duration of diarrhea and reduced stool frequency in individuals who received probiotic supplementation compared with a control group. Overall, probiotics reduced the duration of diarrhea by around 25 hours, the risk of diarrhea lasting 4 or more days by 59% after the intervention. However, there was marked variability in the study findings and so these estimates are only approximate. The evidence for benefits in treating acute diarrhea is encouraging but more research is needed to identify clinically useful probiotic species and the cost effectiveness of this treatment.
A recent trend in the food ingredients industry is the focus on multicomponent formulations or synbiotics. The food industry is driving, partly for commercial reasons, the development of combinations of prebiotics and probiotics (and occasionally other ingredients such as lactoferrin and selected immunogobulins) to improve GI tract health and well-being. Some formulations now offer 2 or more different probiotic species presumably to enhance the microbial potency and potential benefits to the gut microbiota and health. Prebiotics are defined as a selectively fermented nondigestible ingredient that beneficially affect the host by selectively stimulating the growth and activity of 1 or more bacteria in the colon having the potential to improve host health. 36 A combination of a probiotic (the active microbial agent) and prebiotic (a fuel source for newly introduced microorganisms) is an attractive proposition. More research is needed to determine the relative merits of single and multicomponent probiotic and synbiotic formulations.
Studies on Probiotics in Athletes and Physically Active Individuals
A small number of studies have examined the effects of probiotics in athletes or military personnel. A summary of the subjects, probiotic species involved and dosage used, and the clinical and immunological outcomes is shown in Table 1. The most commonly studied species in active individuals are Lactobacillus casei, L fermentum, L acidophilus, and L rhamnosus. The general experimental approach in this type of study is to determine the effects of probiotics on clinical measures (typically the rate or frequency of illness and the severity and duration of symptoms), immune function, and health and well-being. It is important to note that probiotics are unlikely to directly benefit physical performance. The more likely scenario is an indirect benefit with probiotic-induced improvements in respiratory and GI health reducing the risk of symptoms impairing the ability to undertake physical tasks in occupational settings or the ability to train and compete in sporting competitions. 43 Some short-term studies (typically 4 weeks in duration) that have examined the biological effects of probiotic supplementations (changes in gut microflora and immune function) are too short to realistically evaluate longer term clinical implications. Ideally, supplement studies, where possible, should investigate parallel changes in clinical outcomes and the underlying immune function over a period of several weeks to a few months.
Summary of Studies Involving Probiotic Supplementation in Trained Individuals.
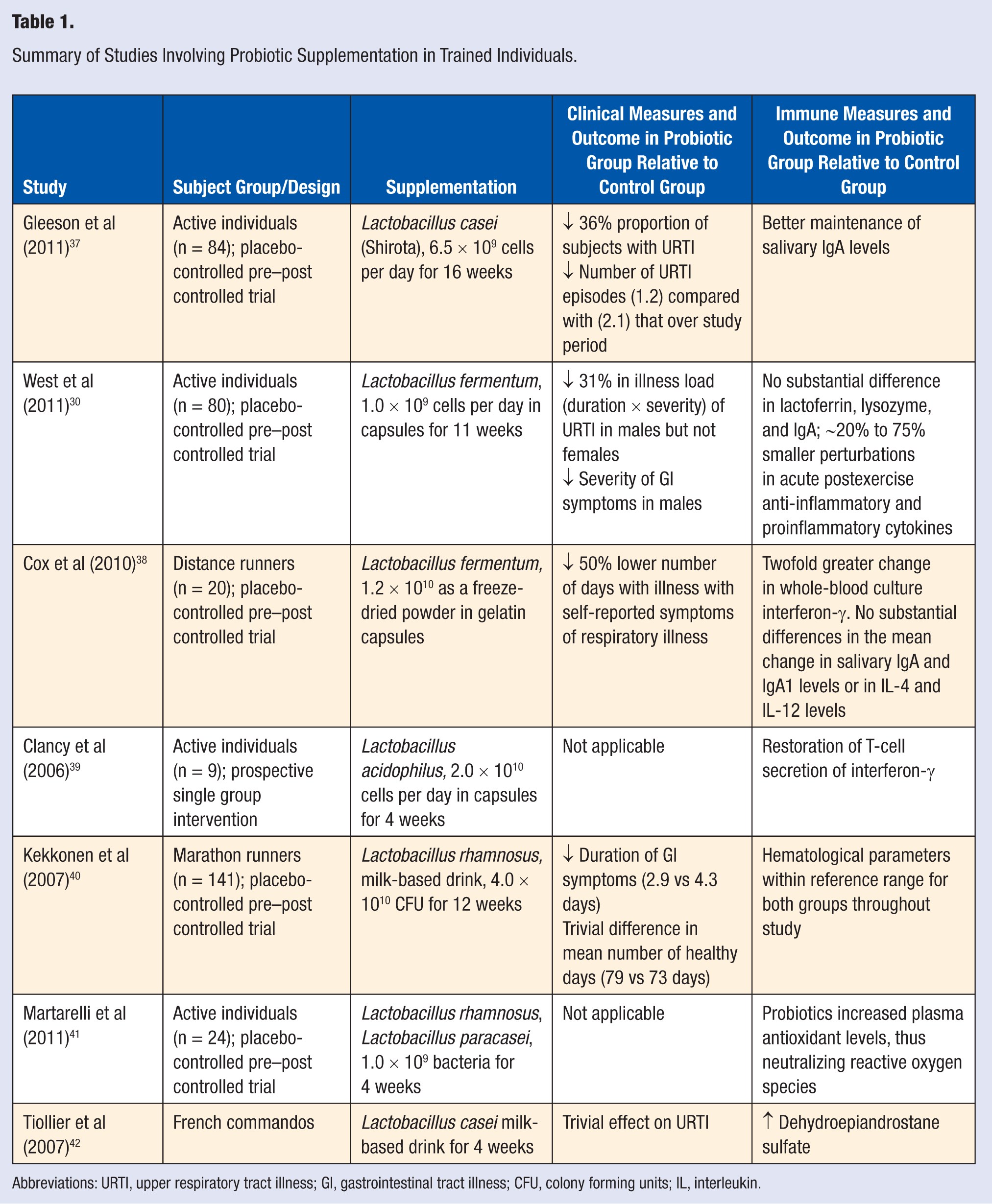
Abbreviations: URTI, upper respiratory tract illness; GI, gastrointestinal tract illness; CFU, colony forming units; IL, interleukin.
An initial study of probiotic supplementation in athletes centered on a small sample of fatigued athletes presenting at a medical clinic. 39 At baseline the whole-blood culture level of interferon-γ in the fatigued athletes was approximately half that of healthy control athletes. Subjects were supplemented with L acidophilus at a dosage of 2.0 × 1010 cells per day (in capsule form) for 4 weeks. At this point the T-cell secretion of interferon-γ was restored. This study was the first to provide evidence of a T-cell defect in fatigued athletes and of its reversal following probiotic therapy. Neither clinical nor athletic performance measures were recorded in this study. Animal studies examining airway hyperresponsiveness have found that Treg (regulatory) cells in the respiratory tract play an essential role in regulating mucosal immunology. 44 Treg cells suppress inflammation by the expression of interleukin-10 and transforming growth factor-β. Examining whether probiotics induce an active Treg phenotype is necessary to determine whether this regulator of immune activity can reduce the symptoms of respiratory illnesses. Upregulation of discrete surface markers could provide further evidence to support a role for inducible Treg as a key mechanism by which GI microbiota influence immunity in host tissues outside the GI tract.
A double-blind, placebo-controlled crossover trial investigated the use of L fermentum in 20 elite male runners over a 4-month winter training season. 38 Athletes taking the probiotic supplement reported less than half the number of days of respiratory symptoms during the supplementation period (30 days) compared with the placebo group (72 days). Illness severity was also lower for episodes occurring during the supplementation period. However, it appears there may be differences in response to probiotics between males and females. A randomized controlled trial involving a large number (n = 99) of physically active adults reported substantial reductions in respiratory and GI symptoms for males, but not females, with 11 weeks of L fermentum supplementation. 30 The probiotic did not yield a substantial difference in acute postexercise antimicrobial proteins and plasma cytokines at rest but was associated with smaller perturbations in pro- and anti-inflammatory cytokines. There was no apparent explanation for the differential clinical responses between males and females, and this question requires further investigation. Whether the observed differences between the sexes are biological and/or environmental in nature, and can be replicated in other studies, is not clear.
A randomized double-blind intervention study in which 141 runners took either a placebo or L rhamnosus for 3 months leading into a marathon reported no significant difference in either respiratory tract illness or GI symptom episodes in the 2 weeks after the marathon. 40 There was, however, a trend toward shorter duration of GI symptom episodes in the probiotic group. A more recent investigation examined the effectiveness of L rhamnosus in supplementation with a combination of L rhamnosus and L paracasei at a dosage of 2 × 109 cells per day for 4 weeks. The outcomes measures centered on changes in immune function rather than on clinical measures. Probiotic supplementation increased plasma antioxidant levels, thus neutralizing reactive oxygen species.
Two studies to date have examined the effects of L casei supplementation in healthy highly active individuals. Probiotic supplementation with L casei by 47 French commando cadets during a 3-week training course, followed by a 5-day combat course, had little effect on the incidence of respiratory tract illness. 42 Another study of active individuals involved 84 subjects consuming L casei (Shirota) or placebo for 16 weeks. Subjects in the experimental (L casei) group had substantially fewer upper respiratory illnesses compared with the control group. There were 36% fewer subjects reporting illness in the L casei group. Better maintenance of salivary IgA levels was suggested as one possible explanation for the improved clinical picture in the experimental group. The positive clinical outcome is consistent with other studies, but the wide variation in selection of immune measures makes it difficult to identify the biological responses and the specific adaptations to probiotic supplementation.
Taken together, this small series of studies in athletes provide modest evidence that probiotics can provide substantial clinical benefits in highly active individuals. Although some studies suggest favorable clinical outcomes, in part there is little consistency in outcome measures and in the selection of measures of immune function. More studies are needed to resolve these issues. These preliminary studies show promise but substantial work remains to be done in a research setting before definitive clinical guidelines can be formulated.
Mechanisms of Action
Broadly speaking, there are 3 main areas or mechanisms of action that purportedly underpin the effects of probiotic supplementation on discrete aspects of immune function. 45 Modulation of the human immune system by probiotics has received substantial attention in the biomedical literature. 1 A number of human and animal studies have shown that specific strains of probiotics can stimulate and regulate discrete aspects of innate and acquired host defense.
Gut Microbiota
Mucosal surfaces in the microbiota provide a physical barrier to colonization by external pathogens. Furthermore, the bacteria compete for limited resources. The creation of ecological niches to prevent infection is termed competitive exclusion. The stability of the microbiota to displacement by external pathogens is further enhanced by secretion of bacteriocins and other antimicrobial substances. Coupled with humoral secretions of the innate immune/epithelial defense system, such as lactoferrin, lysozyme, and defensins, the microbiota milieu plays an integral role in protecting mucosal linings. 46
Gastrointestinal and Respiratory Epithelial Cell Barrier
Probiotics promote 2 main effects in the mucosa. 47 First, probiotics promote the production and secretion of antibacterial substances such as the mucins from human epithelial cells and defensins from paneth cells in the small intestine. Second, probiotics can enhance the integrity of the epithelial barrier lining the GI tract by strengthening junction proteins, maintaining cellular polarization, and reducing the degree of cell apoptosis. In vitro and animal studies indicate that probiotic bacteria enhance or maintain tight junctions, the space between cells that regulate transcellular solubility, by maintaining ion pump activity and activating MAP kinases. However, in vivo studies in vulnerable populations, in particular the critically ill, have not yet confirmed these benefits, with at least 1 study showing a higher mortality rate following the administration of probiotics. A pilot study examining gut permeability in healthy physically active males over a 21-day period indicated no substantial effect of supplementation (N. P. West, Griffith University, unpublished observations). However, the mean urine lactulose/mannitol ratio in the treatment and control group were within the normal range and it may be that no further enhancement was possible. 45
Systemic and Cellular Immunity
Probiotics are able to influence both innate and acquired immune systems via bacterial–epithelial cell interaction and through the common mucosal immune system. Interaction between bacterial and epithelial cells contributes to the maintenance of mucosal homeostasis by inducing the secretion of anti-inflammatory cytokines that suppress activation of leukocytes in the lamina propria. Beyond direct interaction with the epithelia layer, transcytosis of bacteria in M-cells triggers the activation of dendritic cells and lymphocytes that home to lymphoid tissue and provide protection at distal mucosal surfaces. More recently, there is evidence in animals models that indicate cross-talk between the GI system, the primary site of probiotics effects, and the immune system mediated in part by gut-derived FOXP3+T regulatory cells. 48 Work currently underway will elucidate the role of these suppressor lymphocytes in mucosal homeostasis and protecting host tissues from infection.
Recommendations for Highly Active Individuals
Given the small number of studies that have examined the effects of probiotic supplementation in highly active individuals, it is somewhat premature to formulate definitive clinical guidelines. To overcome the shortage of studies, clinicians and scientists working with highly active individuals need to translate and apply findings of selected studies in closely related fields. Research areas including clinical immunology, nutritional immunology, nutrition, sports medicine, and exercise physiology should yield useful insights. It is difficult to study highly active individuals in their regular occupation and training settings, and interactions between diet, physical activity, and other lifestyle stresses need to be considered. Management of training, lifestyle stresses, and dietary practices is always a challenge in physically active individuals leading busy lives.
Probiotics appear to be useful in targeting a range of common GI illnesses. Most studies to date have been directed toward acute illnesses such as infectious diarrhea or chronic diseases such as colitis, inflammatory bowel disease, or Crohn’s disease. In context of otherwise healthy active adults undertaking demanding physical tasks as part of their occupation, recreational, or sporting pursuits, the goal is to reduce the inconvenience of mild to moderate symptoms such as stomach pains or cramps, bloating, nausea, and unexpected changes in stool frequency or composition. A particular issue is the increased risk of GI problems when undertaking international travel, particularly to countries with challenges related to food hygiene and associated risk of gut illness. 49 Prophylactic supplementation with probiotics for individuals and athletes travelling to these regions or areas should be considered as part of an overall illness prevention plan. Probiotic supplementation should commence approximately 14 days prior to arrival, or before a major sporting competition, to allow for adequate colonization of beneficial bacteria in the gut microbiota. Mild transient abdominal bloating, flatulence, and cramping occasionally experienced subjects during the initial stages of probiotic supplementation are most likely adaptive responses to changes in the microbiota. 30 Although probiotics are most well known in relation to their purported effects in moderating common GI illnesses, the purported benefits in the respiratory system 30 could be useful for athletes experiencing recurrent or persistent common cold and flu-like symptoms.
Future Research
A 3-phase research and development model is suggested for developing and promoting probiotic additives and supplements. The initial phase of research typically involves laboratory identification of new strains using phenotypic and genotypic methods, characterization of functional effects in animal and laboratory (in vitro) models, and evaluation of safety issues in animal and human studies. Much of this research is funded by the food ingredients industry where there is considerable commercial interest in isolating and characterizing novel probiotic strains that offer defined benefits to consumers. 33 The second phase involves multiple independent research studies employing the usual requirements of a double-blind, randomized placebo-controlled trial with general or specific populations (eg, immunocompromised individuals, the young or elderly, individuals in highly active occupations, athletes undertaking intensive training programs) with appropriate sample size, methodology, and analysis to determine the real-world effectiveness of a particular probiotic supplement. The predominance of short cross-sectional studies of the acute effects of exercise rather than long-term prospective studies of athletes in training over weeks, months, or years is another issue. When a sufficient number of short-term and long-term clinical trials have been conducted, a meta-analytic approach is needed to summarize the outcomes of these trials.
The final phase addresses issues such as the appropriate packaging and labeling of products, the typical shelf life under standard retail and home conditions, the training and education of clinicians and practitioners, and development of corporate and individual consumer information on specific applications, dosage, and possible contraindications. Research teams should include where possible expertise in areas such as microbiology, sports medicine, and clinical educators. Education needs to be targeted to a range of different groups broadly categorized as professional (eg, research scientists, clinicians, and allied health care practitioners) and public (general public, individuals with various illnesses, children, the elderly, and healthy physically active individuals).
Conclusion
There is emerging evidence that probiotic supplementation can yield beneficial effects in promoting health and treating various illnesses and diseases. Probiotics may assist highly active individuals in occupational, recreational, and athletic settings by reducing the risk of respiratory and GI illness during stressful periods of intensive physical activity. Overtrained athletes or those with compromised immune function may also benefit from probiotic supplementation. The clinical benefits of probiotics appear to be mediated by changes in gut microbiota, upregulation of mucosal barrier integrity in the GI and respiratory tracts, and both innate and acquired immune systems. Although existing studies are showing the promise, more research is required to clarify issues on strains, dose–response, short-term and long-term safety, and underlying mechanisms of action before definitive clinical guidelines can be issued.
