Abstract
Purpose
The present study aimed to compare the dose distributions between conventional three-catheter intracavitary (IC) brachytherapy and combined intracavitary–interstitial (IC/IS) brachytherapy in the management of locally advanced cervical cancer (LACC).
Materials and Methods
A retrospective dosimetric analysis was performed using data from 78 patients with cervical cancer who underwent high-dose-rate (HDR) brachytherapy. To evaluate the dosimetric advantages of IC/IS brachytherapy, patients were stratified into two groups: those treated with IC brachytherapy and those treated with IC/IS brachytherapy. Dosimetric parameters between the two groups were compared.
Results
In the IC/IS brachytherapy group, V150 values for the high-risk clinical target volume (HR-CTV) were significantly higher than those in the IC group (
Conclusions
Compared with conventional IC brachytherapy, combined IC/IS brachytherapy provides superior HR-CTV coverage with higher doses while simultaneously reducing radiation exposure to the bladder and sigmoid colon, provided that the D90 dose constraint for the HR-CTV is met.

Keywords
1. Introduction
In the radiotherapy of locally advanced cervical cancer (LACC), pelvic external beam radiotherapy (EBRT) is typically administered as the initial treatment modality. However, due to the anatomical location of the cervix, the prescribed dose of pelvic EBRT is limited by adjacent organs at risk (OARs), such as the bladder and colorectum. Notably, pelvic organs—particularly the colorectum—exhibit poor tolerance to ionizing radiation, necessitating the delivery of a supplemental boost via brachytherapy. As a critical component of definitive radiotherapy for cervical cancer, brachytherapy plays an indispensable role; consequently, optimizing local tumor control while minimizing radiation-induced toxicity to surrounding normal tissues has become a central objective in the advancement of radiotherapy technologies.
Brachytherapy has evolved from two-dimensional (2D), point-based dosimetry to three-dimensional (3D), volume-based planning. In particular, image-guided brachytherapy (IGBT) provides a reliable foundation for accurately determining both the therapeutic dose delivered to the tumor and the constrained doses to OARs. 1 The proper placement of brachytherapy applicators is essential for achieving tumor control while maintaining acceptable rates of complications. In patients with large tumor volumes, irregular tumor morphology, or parametrial involvement, conventional intracavitary (IC) brachytherapy often results in incomplete coverage of the target volume and suboptimal dose distribution, thereby compromising therapeutic efficacy. Although interstitial (IS) brachytherapy enables conformal implantation tailored to the shape of the target volume, it may fail to deliver an adequate therapeutic dose to the central region of cervical tumors when used in isolation. 2 Combined intracavitary–interstitial (IC/IS) brachytherapy offers a complementary approach: it ensures sufficient dose delivery to the central tumor region while achieving comprehensive coverage of the high-risk clinical target volume (HR-CTV). The IC/IS implantation technique involves initial placement of a uterine tandem, followed by the insertion of multiple interstitial needles into the tumor bed to optimize dose conformity and target coverage. Importantly, IC/IS brachytherapy effectively overcomes the limitations of standalone IC brachytherapy and has emerged as a key technique in modern image-guided brachytherapy for cervical cancer.
Although IC/IS brachytherapy has been increasingly adopted in the management of LACC, relatively few studies have comprehensively characterized its dosimetric properties. In the present study, we compared the dosimetric advantages of IC/IS brachytherapy with those of conventional IC brachytherapy, focusing on two primary endpoints: (1) improvement in target volume dose distribution, and (2) reduction in radiation doses to OARs—including the bladder, rectum, and sigmoid colon.
2. Methods and Materials
2.1. Patient Cohort
A total of 78 patients with cervical cancer were retrospectively enrolled from our institution. Specifically, data from patients diagnosed with cervical cancer who underwent high-dose-rate (HDR) brachytherapy between June 2019 and May 2021 were analyzed. Eligible patients were staged according to the International Federation of Gynecology and Obstetrics (FIGO) 2018 staging system, with disease stages ranging from IIB to IVA. All patients were scheduled to receive EBRT to a total dose of 45–50.4 Gy. Patients receiving concurrent chemotherapy were treated with cisplatin-based regimens. For those who discontinued chemotherapy due to treatment-related toxicity, the reasons for discontinuation and the number of administered cycles were documented; chemotherapy completion status was included as a covariate in subsequent analyses. Indications for combined IC/IS brachytherapy included eccentric or bulky cervical tumors with inadequate target volume coverage achievable by IC brachytherapy alone, as well as tumors with parametrial or paravaginal involvement. All enrolled patients received intensity-modulated radiation therapy (IMRT) in combination with HDR brachytherapy. Brachytherapy was delivered using a remote afterloading microSelectron HDR Version 3.0 unit (Elekta AB, Stockholm, Sweden) with an iridium-192 (192Ir) source. Written informed consent was obtained from all participants, and all study procedures were conducted in accordance with the Declaration of Helsinki and applicable local regulatory requirements.
2.2. Operating Procedure
The radiotherapy procedure followed a standardized protocol. Prior to each brachytherapy session, routine preparatory interventions were performed before applicator implantation, including administration of a cleansing enema, oral intake of contrast agent, placement of an indwelling urinary catheter, and bladder emptying.
Patients in the IC/IS group underwent implantation with a uterine tandem and interstitial needles, whereas those in the IC group received a uterine tandem combined with ovoid intracavitary applicators. The treatment volume and the number of interstitial needles to be implanted were determined based on findings from clinical gynecological examination and complementary imaging studies (e.g., MRI and CT). Interstitial needles were inserted into the cervix, vagina, and parametrial tumor regions to a depth of 1–3 cm, with 4–6 needles typically used and inter-needle spacing maintained at 0.8–1.5 cm. Vaginal packing with gauze was then performed for applicator stabilization, and the depth and orientation of the interstitial needles were adjusted under CT guidance to avoid perforation of the rectum or bladder. Finally, the patient’s bladder was filled with 150 mL of normal saline, followed by acquisition of a CT scan with a slice thickness of 3 mm.
2.3. Contouring
Target volume and OAR delineation were performed in accordance with the consensus guidelines established by the European Society for Radiotherapy and Oncology.3,4 Contoured structures included the HR-CTV, rectum, bladder, and sigmoid colon. Specifically, the bladder was contoured from the superior bladder fornix to its junction with the urethra. The rectum was contoured from 1 cm above the anal verge to the rectosigmoid junction. The sigmoid colon was delineated along its anatomical course, extending from the rectosigmoid junction to the left iliac fossa (or anterior pelvic region, as appropriate).
All patients received an individualized lymph node irradiation strategy based on FIGO staging and imaging findings (PET/CT or MRI). In patients with suspected or confirmed para-aortic lymph node metastases, the radiation field was extended to include the para-aortic region in addition to standard EBRT. For lymph nodes that were pathologically or radiologically confirmed as positive—defined by a short-axis diameter ≥1 cm and a markedly elevated maximum standardized uptake value (SUVmax) on PET/CT—a simultaneous integrated boost (SIB) or sequential boost was delivered after completion of EBRT, to a total boost dose of 60–63 Gy. Notably, the boost field excluded the uterine body and the upper vaginal segment.
2.4. Treatment Planning
The treatment planning objective was that 90% of the HR-CTV receive 100% of the prescribed dose (6 Gy per fraction), i.e., D90 ≥ 6 Gy. Dose constraints for the bladder, rectum, and sigmoid colon were applied in accordance with the recommendations of the EMBRACE II study group. 5 All treatment plans were generated using Oncentra Brachytherapy Treatment Planning Software, version 4.1 (Nucletron, Elekta, The Netherlands). Dosimetric parameters were compared between the IC and IC/IS brachytherapy groups. Specifically, to evaluate differences in radiation exposure to OARs, the mean doses corresponding to the following volumetric dose metrics were calculated for the bladder, rectum, and sigmoid colon: D0.1cc, D1cc, D2cc, D5cc, and D10cc.
2.5. Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 26.0 (IBM Corp., Armonk, NY, USA). The normality of data distribution was first assessed. Descriptive statistics for OAR dosimetric parameters are presented as mean ± standard deviation (SD). Between-group differences in dosimetric parameters were evaluated using the independent-samples
3. Result
Patient Characteristics
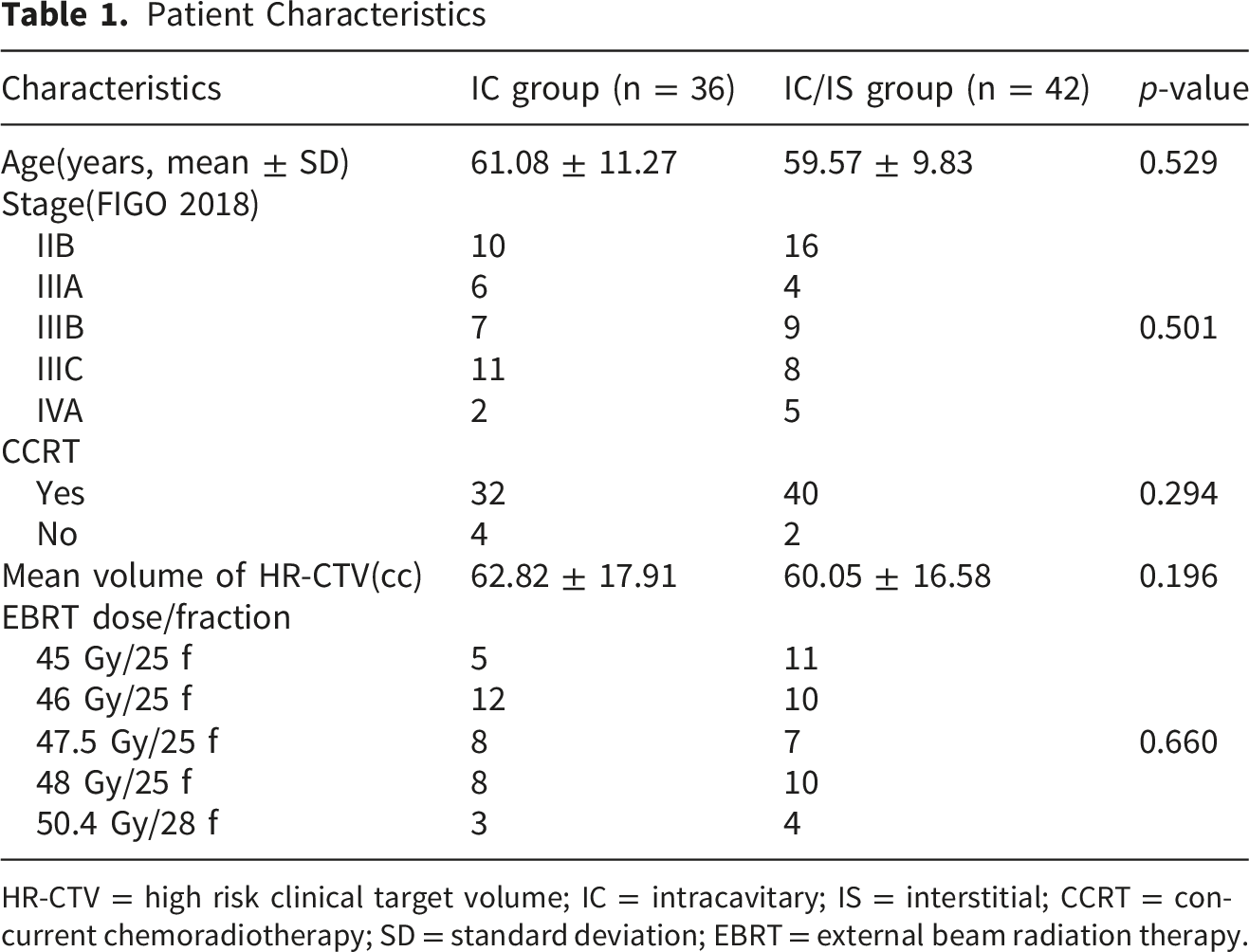
HR-CTV = high risk clinical target volume; IC = intracavitary; IS = interstitial; CCRT = concurrent chemoradiotherapy; SD = standard deviation; EBRT = external beam radiation therapy.
Comparison of HR-CTV Parameters of the IC Group and IC/IS Groups

HR-CTV = high risk clinical target volume; IC = intracavitary; IS = interstitial; SD = standard deviation.
Comparison of HR-CTV Parameters of the IC Group and IC/IS Groups
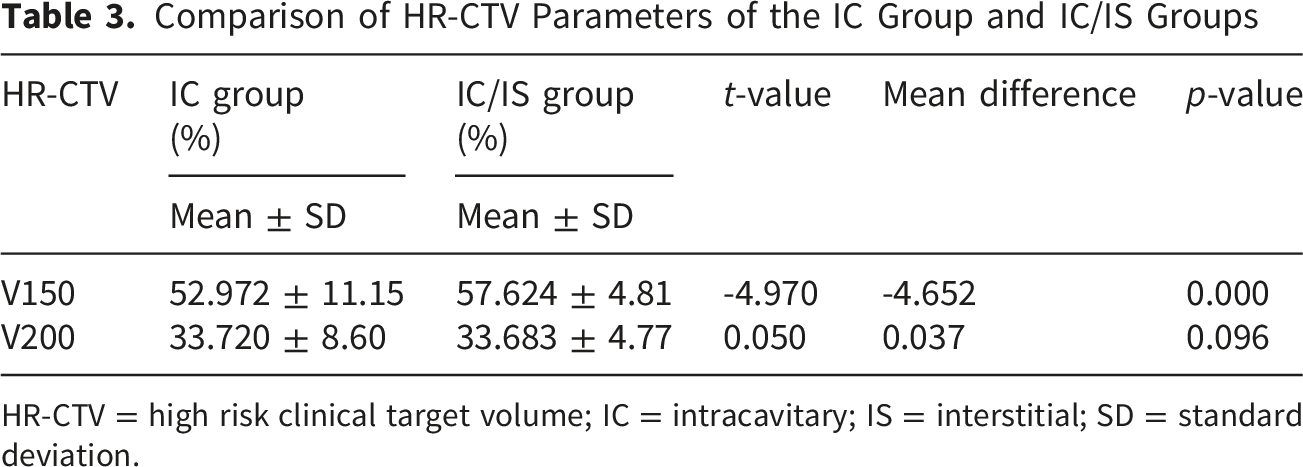
HR-CTV = high risk clinical target volume; IC = intracavitary; IS = interstitial; SD = standard deviation.
With regard to HR-CTV dosimetric parameters, no statistically significant differences were found in D90 or D100 between the IC and IC/IS groups (both Box plot showing the D90 and D100 dose–volume parameters for the HR-CTV in the IC and IC/IS brachytherapy groups. Box plot showing the V150 and V200 of the HR-CTV in the IC and IC/IS brachytherapy groups.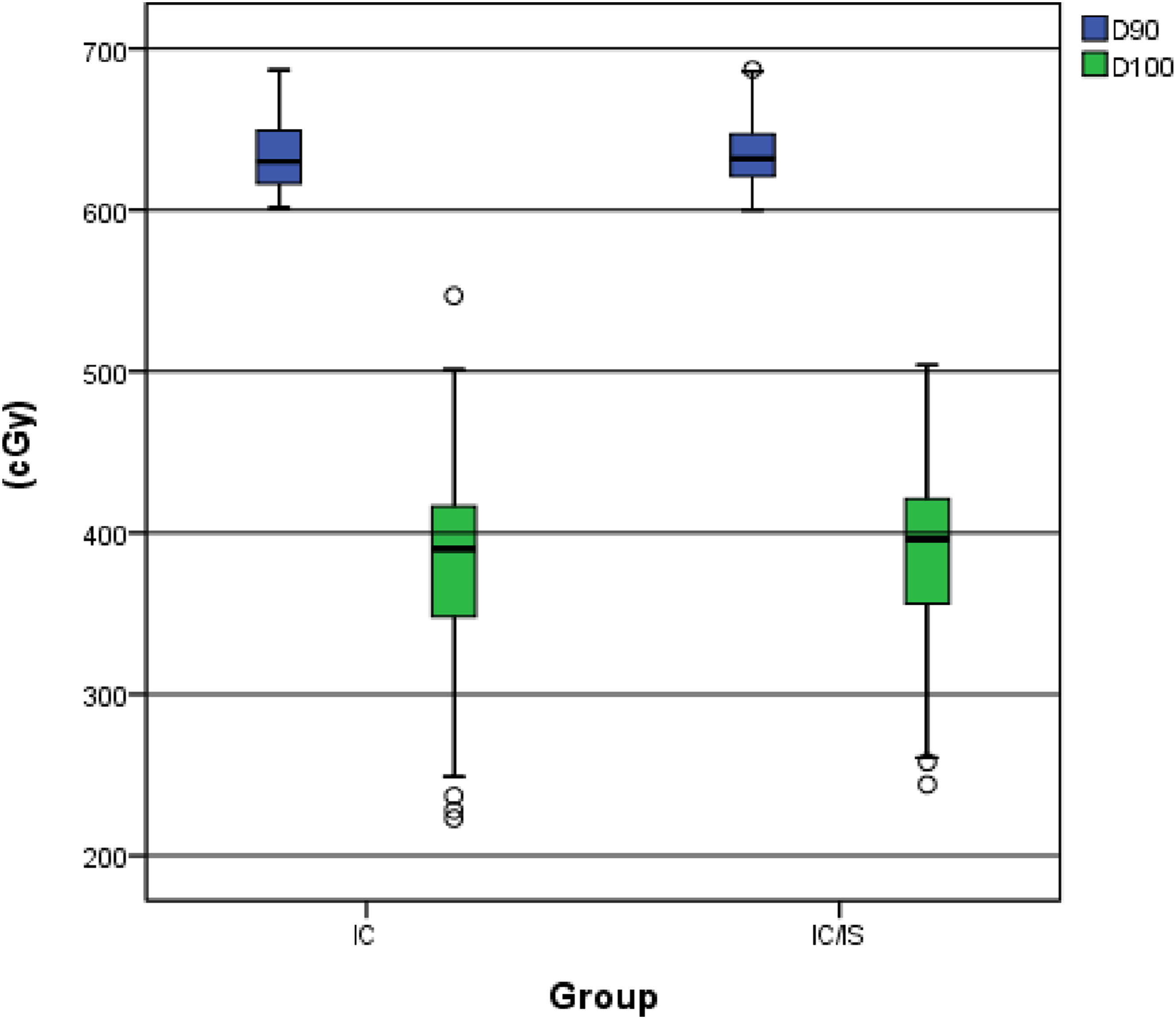

Comparisons of OAR dosimetric parameters yielded the following results:
3.2. Bladder
Dose to 0.1 cc, 1 cc, 2 cc, 5 cc and 10 cc Volume of the Bladder

IC = intracavitary; IS = interstitial; SD = standard deviation.

Box plot showing D0.1cc, D1cc, D2cc, D5cc, and D10cc for the bladder in the IC and IC/IS brachytherapy groups.
3.3. Rectum
Dose to 0.1 cc, 1 cc, 2 cc, 5 cc and 10 cc Volume of the Rectum

IC = intracavitary; IS = interstitial; SD = standard deviation.

Box plot showing D0.1cc, D1cc, D2cc, D5cc, and D10cc for the rectum in the IC and IC/IS brachytherapy groups.
3.4. Sigmoid Colon
Dose to 0.1 cc, 1 cc, 2 cc, 5cc and 10 cc Volume of the Sigmoid

IC = intracavitary; IS = interstitial; SD = standard deviation.
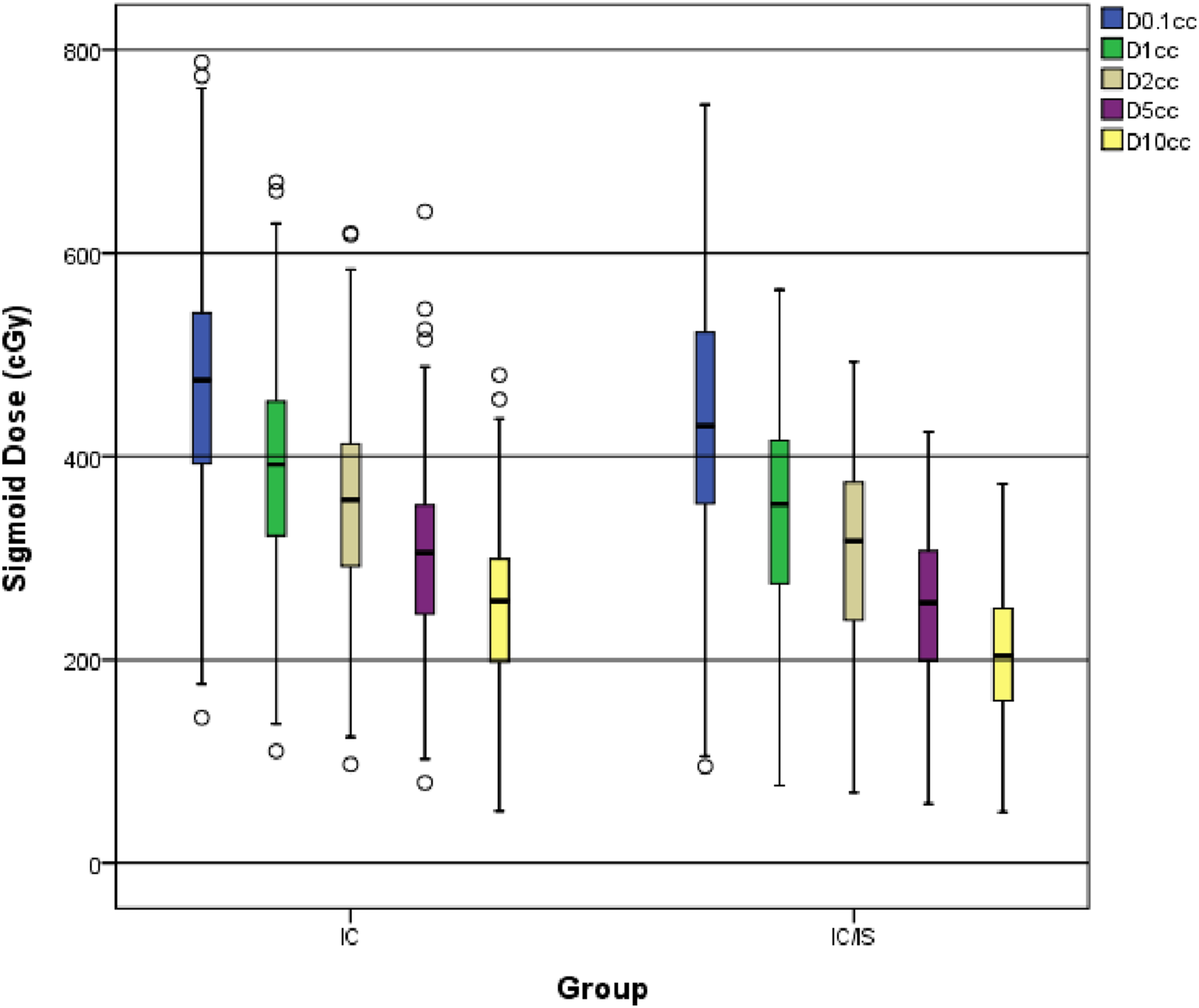
Box plot showing D0.1cc, D1cc, D2cc, D5cc, and D10cc for the sigmoid in the IC and IC/IS brachytherapy groups.
4. Discussion
Brachytherapy plays a pivotal role in the management of cervical cancer across stages I–IV and serves as an essential component of definitive treatment for LACC. While IC brachytherapy yields favorable outcomes in early-stage disease, its efficacy is suboptimal in LACC. This limitation stems from the inherent characteristics of LACC tumors—including large volume, eccentric location, irregular shape, and a high propensity for parametrial invasion—which render standalone IC brachytherapy incapable of achieving optimal target dose coverage and adequate OAR sparing. As early as 1978, transperineal template-based interstitial applicator implantation was first reported, a technique that improved dose distribution uniformity and laid the foundation for modern IS brachytherapy. 6 In 2001, the feasibility of combined IC/IS brachytherapy for LACC was first documented and validated. 7 Subsequent studies have demonstrated that CT-guided 3D brachytherapy delivers higher doses to the central tumor region, with reduced sensitivity to variations in bladder and rectal filling; furthermore, treatment position and respiratory motion do not significantly affect pelvic dose distribution. 8 The present study retrospectively compares dose distributions between CT-guided IC/IS combined brachytherapy and conventional three-catheter IC brachytherapy in patients with cervical cancer. Theoretically, the IC/IS approach enables superior target volume coverage while minimizing radiation exposure to OARs.
The present study identified differences in target volume dose distribution between the two brachytherapy techniques. Consistent with previous findings, 9 combined IC/IS brachytherapy offers dosimetric advantages over standalone IC brachytherapy in the treatment of LACC. Prior studies have reported that 100% of patients treated with IC/IS brachytherapy achieved a HR-CTV D90 of at least 85 Gy, compared with only 50% of those receiving IC brachytherapy alone. Notably, HR-CTV V150 values were significantly higher in the IC/IS group than in the IC group. These results indicate that IC/IS implantation in LACC enables local dose optimization, facilitating both dose escalation within the central tumor region and expansion of the high-dose volume—findings consistent with prior literature.9,10 This effect may be attributed to the additional radiation sources introduced via IS implantation needles. When combined with IC applicators, these needles produce dose superposition near the implantation sites, leading to a marked increase in local V150. In tumors with pronounced lateral extension or parametrial infiltration, IS needles are essential to ensure adequate target coverage; this often necessitates elevation of the peripheral dose, which indirectly increases the overall high-dose volume.
Similarly, the present study demonstrated that dosimetric parameters for the bladder and sigmoid colon—including D0.1cc, D1cc, D2cc, D5cc, and D10cc—were significantly more favorable in the IC/IS brachytherapy group than in the IC group. These findings confirm that IC/IS brachytherapy enables more conformal dose optimization, allowing precise target irradiation while reducing radiation exposure to OARs, thereby demonstrating superiority over conventional IC brachytherapy in this regard. Notably, however, IC/IS brachytherapy did not significantly reduce rectal doses. Potential explanations for this observation include the following: first, the trajectory of IS implantation needles is anatomically constrained by structures such as the pelvic sidewall and ischial spine, limiting the ability to optimally spare the rectum; second, in patients with pelvic stenosis, the restricted operative space increases the technical difficulty of IS needle placement and may compromise dosimetric outcomes. Importantly, IC/IS brachytherapy facilitates dose escalation to the target volume, enabling attainment of clinically required dose levels without increasing radiation exposure to OARs, owing to the precise positioning of the applicator system.
Despite the widely recognized dosimetric advantages of combined IC/IS brachytherapy over conventional IC brachytherapy, its adoption in routine clinical practice remains limited. The primary barriers include its relatively invasive nature, a higher risk of procedural complications, and a paucity of randomized controlled trials (RCTs) evaluating optimal implantation techniques and dose fractionation regimens. 11 Currently, brachytherapy dose fractionation for cervical cancer remains heterogeneous, with no universally standardized protocol; the field is still largely in an exploratory phase. Radiation oncologists typically formulate individualized treatment plans based on tumor characteristics such as location, size, and morphology. Imaging modalities—particularly MRI and computed CT—enable accurate delineation of tumor boundaries and adjacent normal tissues, providing a critical foundation for precise applicator and needle placement. The number of interstitial needles required is determined by the location and volume of residual disease, while insertion depth is primarily guided by the craniocaudal extent (height) of the HR-CTV. Both transperineal template-guided and free-hand implantation approaches are clinically viable for delivering IC/IS brachytherapy. Moreover, 3D printing technology has been increasingly integrated into interstitial brachytherapy for cervical cancer, facilitating the fabrication of patient-specific needle templates that enhance procedural accuracy and reproducibility. 12 Notably, interstitial needle placement is technically demanding. In the absence of real-time image guidance, blind manual insertion or inadequate operator experience may result in tissue injury, vascular puncture, or significant bleeding. At present, most radiation oncology departments employ CT- or MRI-guided 3D brachytherapy for cervical cancer. CT imaging offers the advantage of rapid acquisition, making it well suited for intraoperative assessment and adjustment of uterine tandem position. Furthermore, CT-based planning enables robust optimization of dose distribution according to the prescribed treatment plan. 13 MRI possesses inherent superiority over CT in soft-tissue contrast, allowing superior delineation of pelvic anatomy and tumor extent. However, its broader clinical implementation is hindered by longer scan times and logistical challenges related to compatibility with brachytherapy applicators and workflow. Nevertheless, MRI is frequently used during the preplanning phase to assess the feasibility of brachytherapy and identify potential procedural risks. Importantly, comparative studies have shown that dose distributions and rates of late OAR toxicities are comparable between MRI-based and CT-based brachytherapy approaches. 14
The Groupe Européen de Curiethérapie–European Society for Radiotherapy and Oncology (GEC-ESTRO) defines V150 and V200 as the percentages of the HR-CTV receiving 150% and 200% of the prescribed dose, respectively. For OARs, D0.1cc, D1cc, D2cc, D5cc, and D10cc denote the minimum doses delivered to the most highly irradiated 0.1, 1, 2, 5, and 10 cm3 of tissue volume, respectively. 3 According to recommendations from the American Brachytherapy Society (ABS), when EBRT is combined with brachytherapy, the D90—defined as the dose covering 90% of the HR-CTV—should exceed 80–90 Gy, expressed as the equivalent dose in 2 Gy fractions (EQD2) using an α/β ratio of 10 Gy. 15 Notably, D2cc of the bladder—representing the minimum dose received by the most irradiated 2 cm3 of bladder tissue—is considered the dosimetric surrogate for the region of the bladder wall exposed to the highest radiation dose. This parameter is strongly associated with the incidence and severity of urinary morbidity.16,17 Recent evidence suggests that ureteral stenosis is linked to bladder EQD2 values exceeding 77 Gy. 18 For the rectum, the maximum point dose correlates closely with the dose to the most irradiated 2 cm3 (D2cc). 19 A prior study demonstrated that both the frequency and severity of rectal toxicity are significantly reduced when rectal D2cc is maintained at or below 65 Gy. 20
Notably, IS brachytherapy is associated with an increased risk of normal tissue injury and visceral perforation. Consequently, the incidence of late adverse events was slightly higher in patients treated with IMRT combined with HDR IS brachytherapy than in those receiving IMRT plus conventional IC brachytherapy. These late toxicities primarily included enterocutaneous fistula, urinary fistula, rectal stenosis, and small bowel obstruction. Although IS brachytherapy addresses the dosimetric limitations of standalone IC brachytherapy, the integration of IC and IS techniques in LACC treatment achieves mutual complementarity, thereby optimizing therapeutic outcomes. It must be emphasized, however, that interstitial implantation serves only as an adjunct to intracavitary brachytherapy—not as a replacement. Not all patients with LACC require interstitial implantation; the decision should be individualized based on specific tumor characteristics, including size, location, extent of infiltration, and anatomical constraints. Importantly, IS implantation demands rigorous quality assurance measures, including verification of radiation source placement accuracy and dose delivery precision. To minimize patient discomfort and ensure the accuracy and reproducibility of needle trajectory, position, and depth, implantation should be performed under image-guided localization whenever feasible, with appropriate anesthesia administered.
The present study is among the limited number of retrospective cohort studies evaluating differences in dose distribution between IC brachytherapy and combined IC/IS brachytherapy in patients with LACC. Nevertheless, several limitations must be acknowledged.First, the sample size was relatively small, and the study was conducted at a single institution. Moreover, subgroup analyses stratified by tumor location, tumor size, vaginal involvement, and parametrial extension were not performed due to insufficient statistical power. Second, the analysis focused exclusively on dosimetric comparisons of OAR exposure between the two techniques and did not include an assessment of subsequent clinical toxicity or adverse event incidence. Notably, investigating the correlation between the dosimetric profiles of these two brachytherapy approaches and the occurrence of treatment-related toxicities represents a critical direction for future research.
5. Conclusion
In summary, the clinical rationale for IS brachytherapy aligns with unmet needs in the management of LACC. With the widespread adoption of image-guided brachytherapy, the development of novel applicators, and advances in 3D printing technology, the future design of patient-specific templates and applicators is anticipated. These innovations are expected to enable truly individualized, precision brachytherapy, ultimately improving long-term survival outcomes and quality of life for patients with LACC. Nevertheless, standardized, large-scale, multicenter RCTs are still warranted to generate high-level evidence supporting the broader clinical implementation of combined IC/IS brachytherapy.
Supplemental Material
Supplemental Material - Dosimetric Comparison of Intracavitary Versus Combined Intracavitary-Interstitial Image-Guided Brachytherapy in Locally Advanced Cervical Cancer
Supplemental Material for Dosimetric Comparison of Intracavitary Versus Combined Intracavitary-Interstitial Image-Guided Brachytherapy in Locally Advanced Cervical Cancer by Xi Yang, Bo Zhao, Guiyong Yu, Zhuokai He, Meilian Liu, Rensheng Wang, Kai Hu in Dose-Response
Footnotes
Acknowledgements
We gratefully acknowledge Dr. Jianfeng Luo and Dr. Zuyue Bi for his statistical support and Dr. Xin Zhang for her valuable guidance on OAR delineation in the contouring atlas.
Author Contributions
Xi Yang: Conceptualization, Writing-original draft, Data curation; Guiyong Yu: Formal analysis; Bo Zhao and Zhuokai He: Methodology; Meilian Liu: Project administration; Rensheng Wang: Supervision; Kai Hu: Writing-review and editing, Funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (No. 82060019); Joint Project on Regional High-Incidence Diseases Research of Guangxi Natural Science Foundation (No. 2024GXNSFDA010042); and Guangxi Natural Science Foundation (No. 2024GXNSFBA010244).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data are available from the corresponding author for reasonable requests.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
