Abstract

Introduction
There have been many studies on the biological effects of high-dose radiation around the world, but the studies on the biological effects of low-dose radiation are still relatively few. Because the biological effects of low-dose radiation are not exactly the same as those of high-dose radiation, the research methods for the biological effects of low-dose radiation should also be different. Given that the occupational exposure of radiation workers in daily life is predominantly characterized by low-dose radiation, it is imperative to investigate the biological effects of low-dose radiation. With the advancement of science and technology, radioactive sources have found wide applications in many fields such as industry, agriculture, medicine and scientific research. 1 Consequently, the number of radiation workers in China has been steadily increasing over the years. According to the China CDC, by the end of 2022, there were 75,000 medical institutions utilizing radiation technology, 15,000 enterprises operating radioactive sources, and nearly one million radiation workers, significantly higher than in 2018. 2 The occupational health of radiation workers has been widely studied. 3 In previous studies, damage among radiation workers has been evaluated using indicators such as peripheral blood cell count or lymphocyte micronuclei and chromosome aberrations. However, these indicators were found to be insensitive to ultra-low doses of radiation exposure. Research indicates that low-dose radiation can influence cell cycle regulation and cyclin expression; and there have been few studies using cell cycle proteins as indicators to investigate the effects of low-dose neutron-γ radiation on cells.4,5 This study aims to investigate the expression levels of cell cycle-related proteins p21 and CDK2 alongside DNA damage-related protein γH2AX in response to low-dose neutron-gamma irradiation.4,6 This Study were not limited to exploring the dose-effect relationship between low dose neutron-gamma radiation and known cycle-related proteins. RNA sequencing analysis of lymphocytes from occupationally exposed workers by our research team indicates that RPS18 is a key differentially expressed gene. Based on this observation, we investigated the correlation between intracellular RPS18 protein levels and exposure dose, thereby improving our understanding of how low-dose radiation modulates the cell cycle. By doing so it may help to elucidate the mechanisms in which cell cycle proteins are involved in low dose neutron-gamma radiation effects, and provide new insights for estimating doses by biological way for large numbers of radiation workers.
Material and Methods
Material


Exposure Plan
The Doses Received by Each Group of Samples in Two Irradiation Experiments (μGy).

The Detection of Cell Cycle, Cycle-Associated Proteins and RNA
The detection of p21, CDK2, p53, ATM, γH2AX and RPS18
Three healthy volunteers, two females and one male, with an average age of 19 years and no history of alcohol or tobacco consumption, were selected. None of the three volunteers had been exposed to medical radiation, antibiotics or toxic chemicals in the past year. The collected blood samples underwent a preliminary cell viability assessment prior to the subsequent experiments. Volunteer recruitment and cell detection were completed in July 2023. Cell testing was done at the Quality Control laboratory, Sinopec Dongying Logging Company.
The volunteers provided written informed consent before the experiment. 36 mL of venous blood was collected from each volunteer, and the blood samples were divided into seven groups and subjected to different doses of irradiation. The irradiation of the different dose groups started at different times and ended at the same time, and the following detection was performed one hour after the end of irradiation. Following irradiation, lymphocytes were isolated by centrifugation, 1 mL of 1×Pharm lysel was added to each sample group for erythrocyte lysis, and clean cells were obtained through subsequent washing steps. The cells were divided into two aliquots, with one aliquot used for cell cycle analysis and qPCR experiments.
The other aliquot of cells for CDK2, p21, γH2AX, p53, ATM and RPS18 detection was fixed with paraformaldehyde, treated with 1 mL of 1×Perm/Wash Buffer perforating solution for 20 minutes, and washed with PBS. The cells used for CDK2, p21, γH2AX detection were stained with CDK2 monoclonal antibody, p21 monoclonal antibody, and γH2AX monoclonal antibody for 30 minutes at 4°C in the dark. After washing with PBS, the cell suspension was detected by flow cytometry. 7 The cells for p53,ATM and RPS18 detection are subjected to primary antibody incubation and washing, followed by secondary antibody incubation and washing prior to flow cytometry analysis.
Cell cycle detection
The lymphocytes used for cell cycle assays were fixed overnight with ice-cold ethanol, followed by thorough washing. Subsequently, each sample was stained with 0.5 mL of PI/RNAse solution for 30 minutes at a temperature of 4°C in the absence of light. Flow cytometry was employed to detect the cell cylce, the PE channel was selected to capture the PI signal. 8
Detection of p21 and CDK2 mRNA expression using qPCR
The total RNA was extracted using ThermoFisher’s RNA magnetic bead isolation kit. The cell samples used for qPCR were lysed with Trizol, followed by the addition of chloroform. After centrifugation, the aqueous phase was mixed with absolute ethanol and transferred to a separation column for further centrifugation.
The mRNA of each sample was normalized to a concentration of 100 ng/μL. Subsequently, a reverse transcription reaction was performed in a 20 μL system containing olig dT, reverse transcriptase, dNTPs, buffer, MgCl2, RNase-free water, and an RNA hydrolase inhibitor. Next, a 10 μL PCR system was prepared in a 96-well plate with four replicate wells for each sample and subjected to 40 cycles of amplification. The CT value for each well was recorded and the relative expression of the target mRNA compared to the internal control was calculated.
Detection the Rate of Chromosomal Aberrations and Micronuclei
Three more volunteers were selected, with similar characteristics consistent with those described in Section 1.3.1. A total of 28 mL of venous blood was collected from each volunteer and divided into seven groups for subsequent gradient dose irradiation.
Chromosome preparation: 0.5 mL of irradiated venous blood was added to 1640 medium containing phytolectin for the detection of chromosome aberrations. Colchicine was added to achieve a final concentration of 0.04 μg/mL, and the cells were incubated at 37°C for 48 hours prior to harvesting. After applying KCl hypotonic treatment, the specimens were fixed with a mixture of methanol and acetic acid (3:1), dropped on slides, dried, and then stained with Giemsa. 9
Micronucleus preparation: 0.5 mL of irradiated venous blood was added to 1640 medium and culture at a temperature of 37°C for 44 hours, then, cyt-B was added to the blood samples with a final concentration of 6 μg/mL, and the incubation continued for an additional duration of 18 hours. After harvesting, the cells were subjected to hypotonic treatment with 7.5 mmol/L KCl solution. Subsequently, they were fixed with a 3:1 mixture of methanol and glacial acetic acid. The cell suspension was prepared under conditions of 70% humidity, followed by the preparation of slides by pipetting at room temperature. The micronuclei were observed as separate entities within the cytoplasm, distinct from the main nucleus shape (round or oval) with smooth edges, and the staining intensity was similar to or slightly lighter than that of the main nucleus, while the size of micronuclei were less than one-third of the main nucleus. 10
Statistical Analysis
Normality of the experimental data, Pearson correlation analysis, independent sample t, One-way ANOVA and curve fitting were performed using SPSS 27.0.
Results
The Expression of Cyclin Exhibited Dose-dependent Variation
According to the flow cytometry results, the average fluorescence intensity of p21, CDK2, ATM, p53 and γH2AX in lymphocytes from three volunteers in each dose group was calculated. The expression levels of cyclin p21, CDK2, ATM, p53 and γH2AX proteins were then plotted against the exposed dose (Figure 1). The expression levels of proteins related to the cell cycle exhibited dose-dependent variations in response to radiation. The expression changes of γH2AX, p21, ATM, p53 and CDK2 are depicted in pictures (A), (B), (C), (D), (E) respectively. The vertical axis in the given images represents the relative expression of cyclin, while the horizontal axis represents the radiation dose. The statistical method was employed to analyze the disparity between each radiation dose group and the “0” dose group. *, P < 0.05; **, P < 0.01, ***, P < 0.001.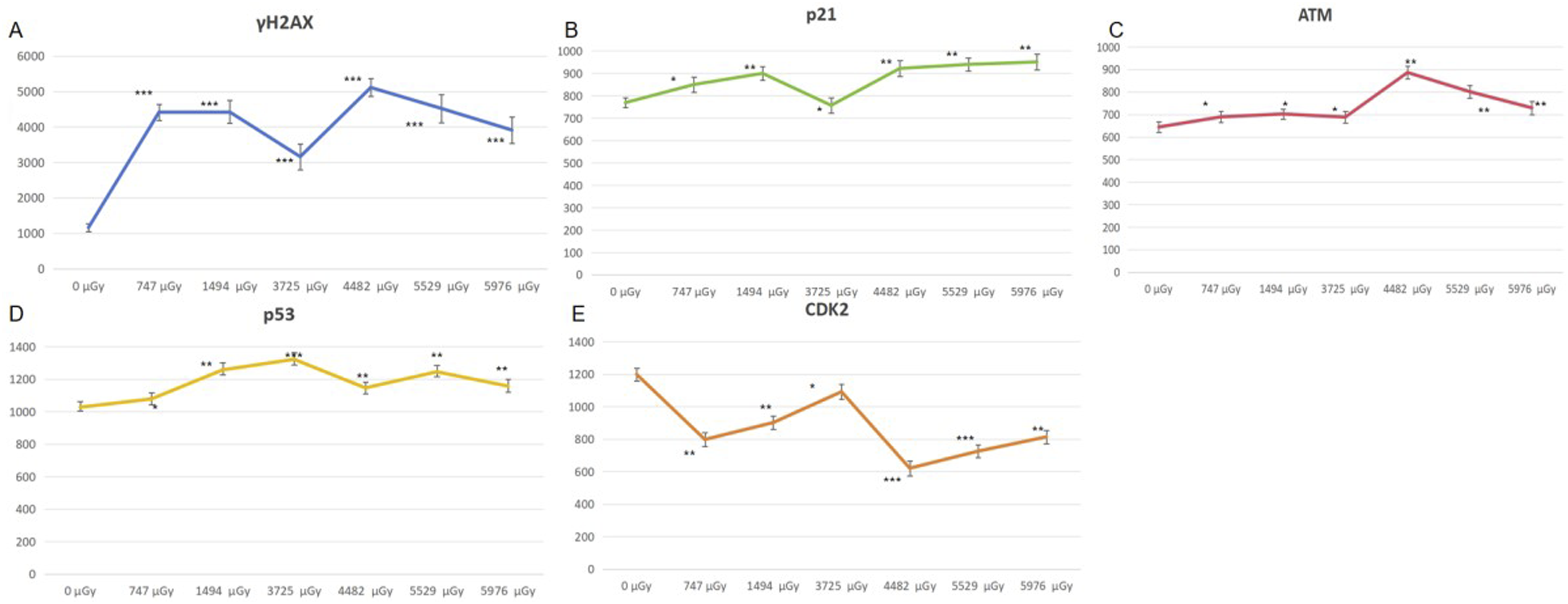
The expression levels of γH2AX, p21, p53, ATM and CDK2 in the irradiated group exhibited significant differences compared to those in the “0” dose group. The results showed that the expression levels of γH2AX, p21, ATM and p53 all showed an upward trend after low dose neutron gamma irradiation. Although the individual values fluctuated, the overall trend was still upward. The change trend of CDK2 expression was contrary to the above four proteins, showing an overall downward trend.
Proportion of Cells in Each Cycle Stage Varied with Dose
The distribution of cells in each cycle phase was analyzed using flow cytometry, and a dose-dependent curve depicting the percentage of cells in G1 and G2 phases was generated (Figure 2). The statistical analysis revealed a significant increase in the proportion of cells in the G1 phase, accompanied by a notable decrease in the proportion of cells in the G2 phase. The proportion of cell cycle alterations varies according to the dosage. The horizontal coordinate showed the irradiated dose, and the vertical coordinate showed proportion of G1 and G2 phases changes with the change of radiation dose. The statistical method was employed to analyze the disparity between each radiation dose group and the “0” dose group, with the distinction being denoted by “*” (n = 3). *, P < 0.05; **, P < 0.01, ***, P < 0.001.
The mRNA Expression of p21 and CDK2 Exhibited Dose-dependent Changes
According to the experimental protocol described in Section 1.3, qPCR experiments were performed to detect mRNA transcripts of p21 and CDK2. The data was graphed as a curve (Figure 3). The mRNA expression of p21 and CDK2 genes exhibited dose-dependent changes. The horizontal coordinate showed the irradiated dose, and the vertical coordinate showed the mRNA expression of p21,CDK2. The statistical method was employed to analyze the disparity between each radiation dose group and the “0” dose group, with the distinction being denoted by “*” (n = 3). *, P < 0.05; **, P < 0.01, ***, P < 0.001.
The significant changes in the expression of the two mRNAs were observed following neutron-gamma irradiation, which demonstrated statistical significance. The mRNA expression levels of p21 and CDK2 in the irradiated group were significantly higher than those in the “0” dose group.
The Variation of Chromosome Aberrations in Peripheral Blood Lymphocytes with Dosage
The chromosome aberration rate of peripheral blood lymphocytes was determined using Method 1.5. Upon exposure to radiation, dicentrics and acentric fragments were observed in peripheral blood lymphocytes (Figure 4), as depicted in Table 2 (Table 2). Exhibited Chromosomal Aberrations in Irradiated Cells. The schematic diagram on the left depicts a acentric fragments, while the schematic diagram on the right illustrates dicentric chromosome. The Findings of Chromosome Aberration Detection.
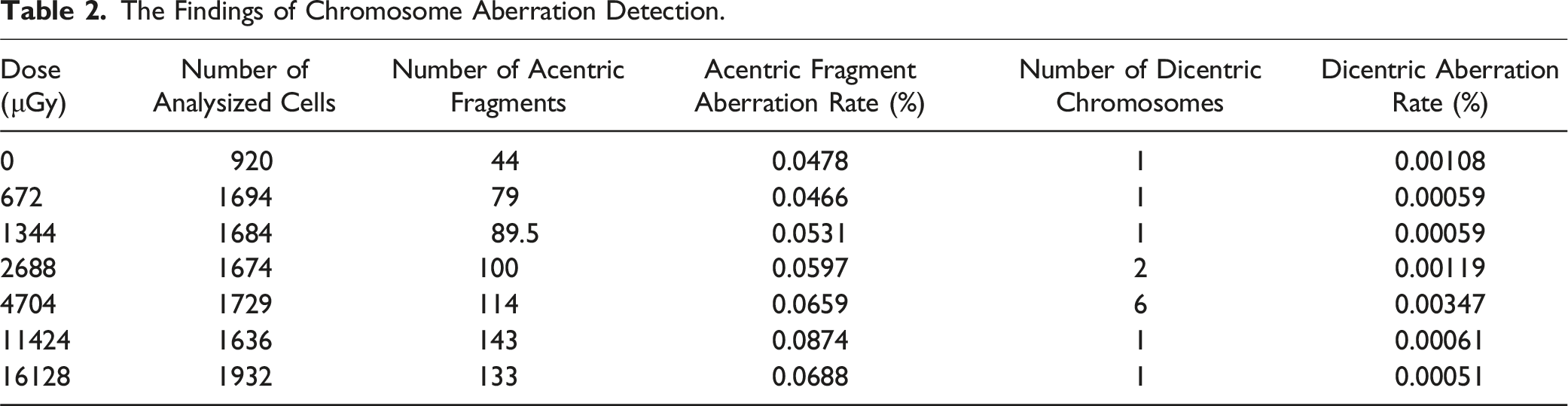
The ANOVA was employed to compare the rate of acentric fragments among all groups (P < 0.001), indicating a statistically significant difference in the change of acentric fragment rates among irradiated cells. Pearson correlation analysis revealed a significant positive correlation between radiation dose and acentric fragment rate (r = 0.785, P < 0.05).
Micronuclei Rate in Peripheral Blood Lymphocytes Exhibited Variation with the Dosage
The presence of one, two, three, and even five micronuclei was observed in binucleated cells (Figure 5). The average rate (‰) of micronuclei in the seven dose groups were listed in Table 3 (Table 3). Exhibited Micronulei in Irradiated Cells. Cells with micronuclei were observed in irradiated blood samples. The normal cells and cells containing 1, 2, 3, or 5 micronuclei are presented in a sequential manner from left to right. The microkernel images correspond directly to the doses indicated below. The Rate of Micronuclei in Each Dosage Group.

The correlation between micronucleus rate and radiation dose was not found to be statistically significant (P > 0.1). The t test analysis of micronucleus rate between every dose group compared to the “0” dose group also showed no significant change after neutron gamma irradiation (P > 0.1).
The Expression of RPS18 Varied with Radiation Dose
The expression of RPS18 was determined following the experimental protocol outlined in Section 1.3, and the acquired data were utilized to construct a dose-dependent plot illustrating the expression of RPS18 (Figure 6). The expression of RPS18 exhibited dose-dependent variation in response to radiation. The horizontal axis represents the radiation dose, while the vertical axis represents the average relative expression of RPS18. The statistical method was employed to analyze the disparity between each radiation dose group and the “0” dose group, with the distinction being denoted by “*” (n = 3). *, P < 0.05; **, P < 0.01, ***, P < 0.001.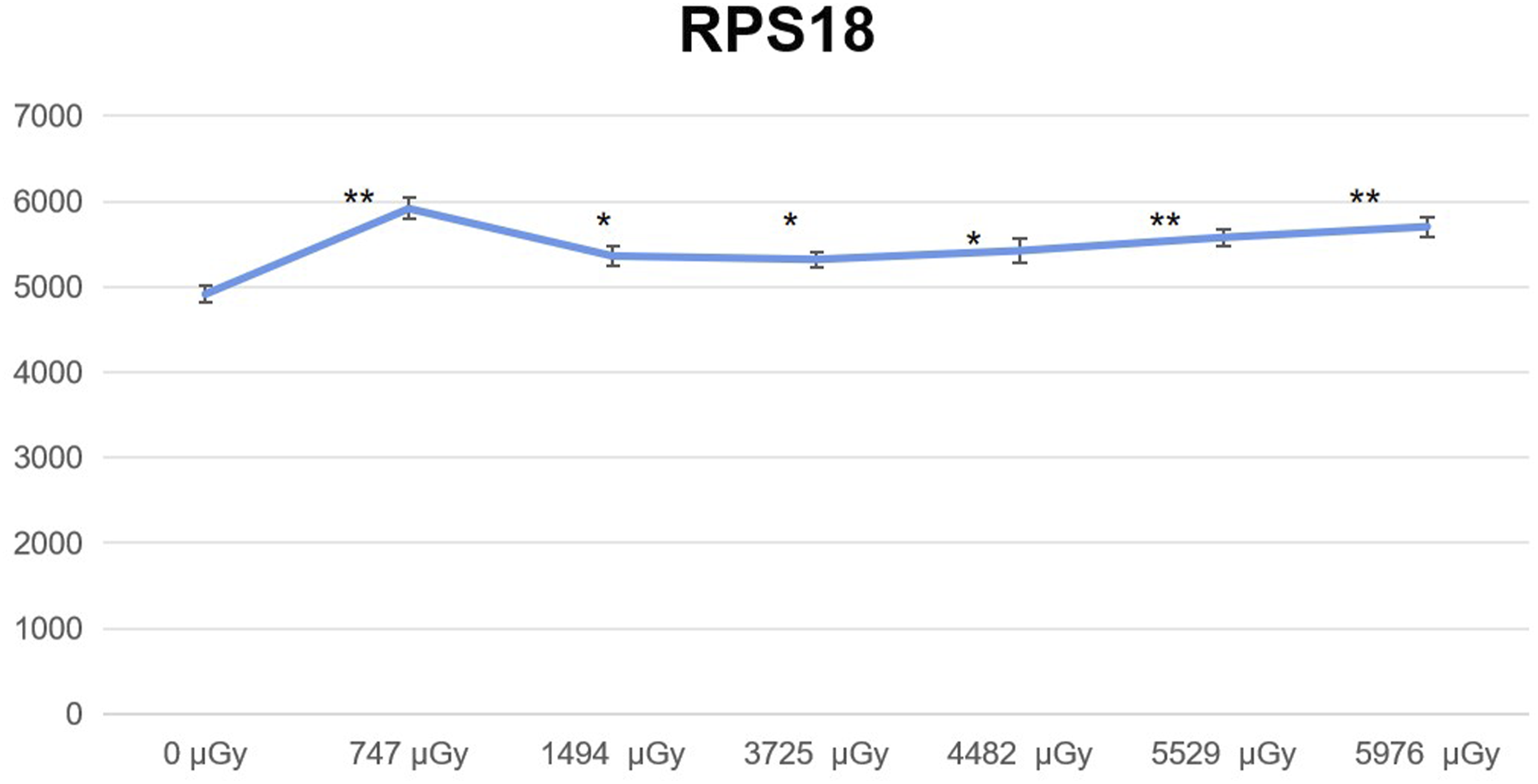
The expression of RPS18 protein exhibited significant alterations following neutron gamma irradiation. The expression of RPS18 in each dose group was significantly elevated compared to the “0” dose group.
Discussion
The effects of low dose neutron radiation have been demonstrated to induce DNA damage and subsequent repair, genomic instability, chromosome aberration, cell cycle arrest, cell apoptosis, and other biological consequences.11,12 Research has shown that low-dose photon radiation is found to have excitatory effects, induce adaptive responses, hyper-sensitivity, and other biological effects. Additionally, long-term low-dose exposure to neutrons can cause biological effects such as damage to the nervous system and changes in peripheral blood cell counts.13-15 The study of this experiment can add to the content of radiation biology. The cellular effects of low dose mixed neutron and gamma irradiation were investigated by studying the changes in cell cycle, cyclin-related proteins, chromosome aberration rate, and micronucleus rate of peripheral blood lymphocytes. Limited by the experimental conditions, three sample sizes were selected in this experiment without sample size/power analysis, which may have a certain impact on the experimental results, but it is still enough to draw the experimental conclusions.
The selected proteins in this experiment, which are associated with DNA damage and the cell cycle, include CDK2 (cyclin-dependent kinase 2), ATM, p53, p21, γH2AX. As a member of the CDK family, CDK2 has been demonstrated to play crucial roles in various cellular processes such as cell cycle progression, apoptosis, and aging. 16 The binding of CDK2 to cyclin E facilitates the G1/S phase transition. 17 In the biochemical reaction of DNA damage, ATM serves as the primary molecular switch that directly detects double-strand breaks in DNA and initiates multiple signaling pathways related to DNA damage. Phosphorylated ATM can activate downstream p53, which, in conjunction with its downstream target p21, establishes a G1 checkpoint.18,19 The p21 protein functions as a CDK inhibitor and serves as a ubiquitous regulator of the cell cycle.20-22 The γH2AX protein is served as a reliable biomarker for DNA double-strand break damage, and its levels are often exhibited a positive correlation with the extent of DNA strand damage.23-25 When cells are exposed to radiation, the expression of γH2AX increases as a result of DNA damage. The cell undergoes cell cycle arrest by regulating cycle-related proteins in order to facilitate repair.
Previous research has demonstrated notable alterations in p21, CDK2, γH2AX, ATM, and p53 subsequent to radiation-induced damage. 26 ATM levels exhibit a significant increase at radiation doses as low as 0.3 Gy. 27 γH2AX exhibits heightened sensitivity to radiation, with a significant increase observed at doses as low as 10 mGy. However, the effects of low-dose neutron radiation are currently less well understood. This experiment further investigated the responses of these cycle-related proteins in low-dose neutron-gamma fields.
Our research team recently performed a RNA sequencing on irradiated lymphocytes and identified the downregulation of mRNAs encoding 69 ribosomal proteins. The analysis of protein-protein interactions shows that RPS18 is at the center of the regulatory network formed by these proteins (Figure 7). Previous research has demonstrated that ribosomal proteins can modulate the cell cycle through various pathways. In this experiment, the expression levels of the RPS18 protein were examined to further investigate the mechanisms by which low-dose neutron-gamma irradiation regulates the cell cycle of lymphocytes. Interaction among proteins corresponding to differentially expressed mRNAs due to low-dose radiation. (A) Protein-Protein Interaction Networks of differentially expressed genes. The colors of the circles represented similar functions or intracellular adjacencies. (B) The 6 most related gene proteins, all of which are ribosomal proteins.
Influence of Low-Dose Radiation on Cell Cycle-Related Proteins and γH2AX
The expression levels of p21, CDK2,p53, ATM and γH2AX in lymphocytes exposed to neutron-gamma irradiation within the range of 0-5976 μGy were found to be significantly different from those in the unexposed control group, based on the experimental data . According to previous studies, a significant increase in γH2AX protein expression can be detected at γ radiation doses as low as 10 mGy. 28 Under the present experimental conditions, even neutron-gamma radiation as low as 0.7 mGy could induce a significant increase in γH2AX expression (Figure 1), further indicating the high sensitivity of γH2AX as an indicator for biological effects caused by radiation exposure. The experimental results demonstrated that each group’s data exhibited fluctuations to some extent, which might be attributed to the weaker biological effects dominated by low-dose radiation. Additionally, individual variations and other factors that might introduce errors could influence the results, but the general trend could still be reproduced.
Our experimental results suggested that neutron-γ irradiation induces a certain degree of DNA double-strand damage, as evidenced by the increased expression of γH2AX. DNA double-strand breaks are known to lead to phosphorylation of ATM, which in turn activates p53. Activated p53 further induces the expression of the target protein p21.29,30 In our experimental results, we observed an increase in ATM, p53, and p21, which may suggest that changes in the cyclin proteins occur through these steps. Conversely, the expression of CDK2 was decreased, suggesting that p21 might inhibit the expression of CDK2. In conclusion, significant changes in the expression of p21 and CDK2 were observed in lymphocytes after low-dose neutron gamma irradiation.
The Effect of Low-Dose Neutron-Gamma Radiation on the Cell Cycle
According to the findings of this experiment, low-dose radiation has been observed to augment the proportion of cells in the G1 phase while concurrently diminishing the proportion of cells in the G2 phase. This phenomenon can be explained by the changes in p21 protein expression. It has been proved that cyclin kinase inhibitor p21 can induce cell cycle arrest at G1 phase and inhibit entry into S phase by deactivating Cdks or inhibiting proliferating cell nuclear antigen (PCNA) activity. Transcription of the p21 gene is activated by both p53-dependent and p53-independent mechanisms, with increased expression occurring when DNA damage activates p53. The results of this experiment suggest that the increased gene transcription and expression of p21 may be accomplished through a p53-dependent mechanism. Elevated p21 can down-regulate a large number of cell cycle-related genes, leading to a corresponding decrease in protein levels, and resulting in cell cycle arrest. 31 The results of this experiment indicated an upregulation in p21 expression and a downregulation in CDK2 expression in the irradiated lymphocytes, suggesting that the downregulation of CDK expression may be associated with cell cycle changes. 32
The Influence of Low-Dose Neutron-Gamma Irradiation on the Transcription of p21 and CDK2 Genes
From our statistical analysis, it is evident that the mRNA transcription levels of p21 and CDK2 were significantly upregulated. Furthermore, the upregulation of p21 mRNA is observed to be consistent with the corresponding alterations in protein levels. 33 Transcription of p21 is often regulated by multiple factors, and DNA damage-induced p21 expression is mainly regulated by the p53 pathway. 34 When ionizing radiation causes DNA damage, p53 expression rapidly increase and become phosphorylated. The activated p53 then regulates p21 expression through transcriptional activation.22,30,35 The mRNA expression level of the CDK2 gene was opposite to the protein expression level, and the reason for this result may be translation inhibition by some pathways. 36
The Influence of Low-Dose Neutron-Gamma Radiation on the Micronuclei Rate in Peripheral Blood Lymphocytes
The micronucleus (MN) is morphologically distinct from the main nucleus and can be easily identified by microscopy. Biologically, micronuclei are chromosomal segments or entire chromosomes that lag during nuclear division.37,38 The micronucleus assay (MN) has been widely used as one of the methods for assessing genotoxicity caused by various chemical and physical factors, including DNA damage induced by ionizing radiation. 39 However, compared to chromosomal aberrations, this assay has limitations such as a high rate of spontaneous micronuclei and poor specificity, which makes it less sensitive in detecting low-dose exposure (detection limit of 0.2-0.3 Gy). 40 No significant difference in the micronuclei rate has been reported between lymphocytes exposed to low-dose radiation and those not exposed to radiation (P > 0.05). 41 Consistent with these findings, it was also observed in our experiment that no significant difference in the micronuclei rate was shown between the exposed group and the “0” dose group, indicating that micronucleus formation is not sensitive to low-dose radiation. Although the micronucleus rate did not rise significantly in this experiment, it still cannot be ruled out that the micronucleus did not respond to the low-dose neutron-gamma field. It is possible that the dose in the experiment was too low to make a significant change in the micronucleus rate. Although a significant increase in the frequency of chromosome fragmentation was observed in this experiment, it is plausible that the micronuclei formed by certain chromosome fragments did not meet the criterion of being 1/16th the size of the main nucleus, Consequently, these smaller micronuclei were not recorded in the experimental results, resulting in a less pronounced change in micronuclei compared to the fragments.
The Influence of Low-Dose Neutron-Gamma Radiation on the Rate of Chromosome Aberrations in Peripheral Blood Lymphocytes
It has been demonstrated that ionizing radiation can induce DNA damage, specifically double-strand breaks (DSBs). Repair of DSBs breaks by nonhomologous end joining is error-prone and can result in the formation of dicentrics or polycentromeres, centromere rings, acentric rings, acentric fragments, microdeletions, reciprocal translocations, inversions, and deletions.42,43 The mechanism underlying chromosomal aberrations can be explained by L.J. Stadler’s break-rejoining hypothesis or S.H.’s interchange hypothesis. For this study, the rate of chromosome acentric fragmentation was selected for analysis due to the difficulty in forming other chromosomal aberrations under low-dose conditions. 44
The rate of chromosome acentric fragmentation in peripheral blood lymphocytes was observed to exhibit significant variation with increasing radiation dose. (P < 0.001). The increased rate of acentric fragment formation suggested an increase of chromosomal damage. 44 The regression analysis revealed a small coefficient of 10−6, indicating that the DNA strand is affected by low-dose neutron-gamma irradiation. However, the influence is not obvious.
Effect of Low Dose Neutron Gamma Irradiation on RPS18 and Cell Cycle
The perception of ribosomal proteins (RPs) as static components of the ribosome has changed significantly in recent years, with recent discoveries shedding light on their regulatory roles in a wide range of biological processes, including cell cycle regulation, DNA repair, cell migration and invasion, and cell differentiation.45,46
In recent years, a large number of studies have found that RPs can regulate cell cycle through the MDM2-p53 axis by binding to MDM2. MDM2 protein can negatively regulate p53 in two ways: the first way is that MDM2 directly binds to p53, thereby masking the transcriptional activation domain of p53; Secondly, MDM2 acts as a ubiquitin ligase of p53, mediating the binding of ubiquitin to p53 and accelerating the degradation of p53.47,48 In our experimental results, we found that the expression of RPS18 was significantly increased when lymphocytes were exposed to low dose neutron gamma irradiation. In our experiment, it was found that the overall change trend of RPS18 was consistent with the expression of p53 and the proportion of cells in G1 phase, which means that RPS18, like other proteins, may alter the cell cycle by modulating the p53 protein.
The Present Protocol Was Compared to the Conventional Protocol
This experiment assessed the protein expression levels of p21, CDK2, and γH2AX, as well as the related RNA expression levels of p21 and CDK2 genes. Additionally, the study investigated the variation in cell cycle distribution as a function of radiation dose, and traditional biological dosimetry measures such as the rate of chromosomal aberrations and micronuclei in peripheral blood lymphocytes in response to low-dose neutron-γ irradiation. It was observed that conventional rate of micronucleus in peripheral blood lymphocytes was not suitable as a biological dosimeter in low dose neutron-gamma field conditions. The rate of acentric fragment aberration in peripheral blood lymphocytes continued to exhibit a favorable response to the radiation dosage. Notably, the expression levels of CDK2 protein, γH2AX protein, and p21 mRNA displayed significant changes by radiation exposure, suggesting their potential as novel biological dosimeters with faster response and increased sensitivity compared to traditional methods. In addition, the less studied ribosomal proteins have also been shown to be involved in cell cycle regulation after radiation damage, although the molecular mechanism requires further experiments.
Compared to traditional chromosomal aberration and micronucleus assays in peripheral blood lymphocytes, the use of monoclonal antibodies to detect cell cycle-related proteins undoubtedly involves higher costs and more complex development techniques. However, with the expanding applications of monoclonal antibodies, this may pave the way for the widespread use of monoclonal antibodies in radiation biodosimetry in the near future.49,50
Conclusion
Even at low doses of neutron-γ radiation, lymphocyte cell cycle arrest and alterations in cycle-related protein expression can be induced. These significant changes may serve as indicators for assessing the extent of cellular damage caused by radiation. Of greater significance, we have identified a previously overlooked protein, RPS18, which may play a role in cell cycle regulation following exposure to low doses of neutron-γ radiation. Further investigations are needed to elucidate the mechanisms by which this protein is involved in cell cycle regulation.
Footnotes
Acknowledgments
The authors are grateful to the volunteers who provided blood samples in this experiment, and to the laboratory managers of Sinopec Dongying Logging Company who provided irradiation conditions. We thank Sun Hao, Liu Changcai, Li Hailiang, and Yang Zifeng for their assistance in this experiment.
Author Contributions
Jinghong Fu was responsible for the experimental operation and the writing of the paper; Weiguo Li was responsible for experimental design and review. Haiqing Wang and Guangfen Du were responsible for completing the experimental procedures. Chenxing Liao, Yuyan Zhang were responsible for reviewing the literature and collating the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: 1. Innovation and Entrepreneurship training Program for college students of Shandong First Medical University (2024104390558) 2. The Medical and Health Science and Technology Development Plan Project of Shandong Province (202202080554) 3. The Development Fund of the Institute of Radiology, Shandong Academy of Medical Sciences (2023-1). 4. Innovation Project of Shandong Academy of Medical Sciences. 5. Collaborative Innovation Project on Radiation-Resistant Materials, Tumor Radiation Effects, and Science Popularization Education (340375).
