Abstract
Nanoparticles are widely used in the pharmaceutical, agriculture, and food processing industries. In this study, we have synthesized green lead nanoparticles (gPbNPs) by using an extract of
Introduction
Nanotechnology has become one of the fastest-growing areas of science and technology and it is helping to considerably improve, and revolutionize, many technology and industry sectors such as medicine, transportation, energy, food safety, and environmental science. Nanomaterials can be used in pollution control and wastewater treatment, making water accessible to all. Not limited to the above fields, nanomaterials and nanotechnology have many applications in every field of science and engineering, leading to greater technological advancements. The products of nanotechnology have a big market, and its value was 39.2 billion dollars in 2016 and it will be 124 billion dollars in 2024. 1 Their innovative physicochemical properties enable their use in textiles, cosmetics, and drugs, thereby increasing the possibility for human and environmental interaction with nanomaterials. 2 The purposeful use of nanomaterials might have happened as a result of ingesting nanomaterials containing drugs. 3 Fortuitous exposure of nanomaterials to the liver occurs directly during the manufacturing and disposal of used nanomaterial products. 4 Workers involved in the development, production, and use of new nanoparticles are already exposed to uncertain levels of toxicity. 5 Silver nanoparticles are frequently used in skin care products. 6 Occupational exposure to NPs could be associated with an increased risk of various cancers. The underlying mechanisms of nanomaterial toxicity are not yet cleared, but there is little evidence that reactive oxygen species (ROS) play important roles in the toxicity and carcinogenetic effects of metals. 7
Thus, nanomaterials produced cytotoxicity in cells due to the accumulation of metal ions. 8 The human breast is an important organ of the human body and has a major role in child feeding. So in this study we used green lead nanoparticles (gPbNPs) to find out their anticancer activity on human breast cancer MDA-MB-231 cell lines. Some reports detected variation in cytotoxicity among normal cells transformed cells and cell lines for NPs. Iron oxide NPs induced reacted toxicity in the lung cancer cell lines instantly and did not in IMR-90 fibroblast line. 8 NP-mediated toxicity involves various mechanisms, in particular the production of excess reactive oxygen species (ROS). As is well known, mitochondrial dysfunction is the major source of ROS overload. 9 Oxidative stress has mainly resulted from the generation of reactive oxygen species (ROS). Excessive ROS causes the imbalance of oxidation and antioxidant system in the body, which may induce lipid peroxidation and change various enzymatic activities. 10 Oxidative stress and apoptotic response are key molecular mechanisms to induce the toxic effects of xenobiotics. 11 To my knowledge, there are no studies that confirmed the adverse effects of gPbNPs on human breast cancer MDA-MB-231 cell lines.
Materials and Methods
Chemical and Reagents
MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide), 2, 7-dichlorofluorescin diacetate (H2-DCFH-DA), neutral red uptake dye, Annexin V-FITC, and propidium iodide were purchased from Sigma-Aldrich (St. Louis, Missouri, United States). Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), and antibiotics were purchased from Gibco, USA. Led acetate was purchased from Merck & Co., Inc. NY USA.
Green Synthesis of Lead Nanoparticle From Ziziphus spina-christi Leaf Extract
After the reaction was ended, the suspension was mixed at 500 rpm for 24 hours at 30°C. Finally, the precipitate was filtered and rinsed with a solution before being washed with distilled water 3 times. The precipitated materials were dried in the oven at 150°C for 4 hours. The Pb nanoparticle was collected and stored at room temperature. The dried powder was ground.
Characterization of Nanomaterials
gPbNPs were characterized using energy-dispersive X-ray spectroscopy (JEM-2100F) (JEOL, Japan), scanning electron microscope (SEM), and transmission electron microscope (TEM) (JEOL Inc., Tokyo, Japan) operated at 110 kV.
Cells and Exposure of gPbNPs
MDA-MB-231 cell line was procured from American Type Culture Collection (ATCC), USA. These cells were sub-cultured in DMEM with 10% FBS and 10,000 U/ml antibiotics in a 5% CO2 incubator at 37°C. The cells at 80% confluence were sub-cultured into 96 well plates, 6 well plates, and 25 cm2 flasks according to designed experiments.
The stock solution of gPbNPs was made in double distilled water at the rate of 10 mg NPs/10 mL deionized water and diluted according to the experimental concentration (0–120 µg/ml). Control cells were not treated with NPs and were considered as controls in each experiment.
MTT Test
The mitochondrial activity was determined by the MTT test. 12 The cells were seeded in 96-well plates and exposed to various gPbNPs concentrations for 24 hour. After being exposed to gPbNPs for 24 hour, the suspension was removed from the plate and the cells were washed in PBS. MTT solution (100 µl) was mixed in each well at a final concentration of .5 mg/mL and incubated for an additional 3.5 hour. The formed formazan crystal was dissolved in isopropanol, and the absorbance was measured at 570 nm using a BioTek Epoch plate reader (BioTek Instruments, Winooski, VT, USA) and Gen5 software (version 1.09).
Half-Maximal Inhibitory Concentration (IC50) and Standardized Dose Concentrations for gPbNPs Were Experimented on the MDA-MB-231 Cell Line.

Neutral Red Uptake (NRU) Assay
A neutral red uptake assay was used to evaluate the viability of cells according to Reference 13. The cells were seeded in 96-well plates and exposed to various gPbNPs concentrations for 24 hour. After being exposed to gPbNPs for 24 hour, the suspension was removed from the plate and the cells were washed in PBS. Then, the plate was mixed with neutral red staining (.5 mg in 10 mL complete media), and the plate was incubated for 4 hour. After 4 hour, the cells were fixed using a fixative solution (.5 gm calcium chloride [CaCl2] and 2.5 mL formaldehyde) and the dye was removed using a solution of acetic acid, absolute ethanol, and water. After 15 minutes, the plate was shaken for 15 minutes at room temperature, and the plate was measured at 540 nm using a BioTek Epoch plate reader (BioTek Instruments, Winooski, VT, USA) and Gen5 software (version 1.09).
Intracellular Reactive Oxygen Species (ROS) Generation
Generation of intracellular ROS in the MDA-MB-231 cell line after exposure to gPbNPs (0, 14, 28, and 42 µg/ml) for 24 hour was measured according to the method in Reference 14. Cells (3 × 104) were cultured in a black bottom culture plate (96 well) and incubated at 37°C in a CO2 incubator for 24 hour for attachment. After exposure, the culture plates were washed with chilled PBS and 10 µM DCFH-DA was added per well and the culture plate was incubated at 37°C in a CO2 incubator for 30 minutes. After incubation, the plate was washed and fluorescence intensity was measured at 485 nm excitation and 535 nm emissions using the microplate reader (Synergy-H1; BioTek). Data were represented as a per cent of fluorescence intensity relative to the control wells. The cells (3 × 104 cells/well) were seeded in a 6-well transparent plate and exposed to different concentrations of gPbNPs for 24 hour. After exposure, cells were washed with PBS and 10 µM DCFH-DA was added per well and the culture plate was incubated at 37°C in a CO2 incubator for 30 minutes. The cell images were captured at 485 nm using a fluorescence microscope (Olympus CKX 41; Olympus: Center Valley, Pennsylvania, USA), with 40× magnification.
Measurement of Caspase 3/7 Activity
After exposure to different concentrations of gPbNPs for 24 hour, the cells were washed with PBS scraped with a scraper in lysis solution and split by using a sonicator. After that, the cell samples were used for the detection of caspase 3/7 activity as per the kit’s instructions (Item No. 10009135, Cayman Chemical 1180 East Ellsworth Road Ann Arbor, Michigan 48108 USA).
Expression of Apoptotic Genes via the Quantitative Reverse Transcriptase Polymerase Chain
Ribonucleic Acid (RNA) Isolation
The MDA-MB-231 cell line was cultured in 75 cm2 flasks and incubated for 24 hour at 37°C in a CO2 incubator. The MDA-MB-231 cell line was treated with gPbNPs (14, 28, and 42 μg/mL, respectively) for 24 hour. After exposure to cells, RNA was extracted from the cell line by using 1 mL of guanidinium thiocyanate (TRIzol) reagent and precipitated by sodium acetate. Then, TRIzol/cell lysate was transferred into the Eppendorf tube and vortexed. After 5 minutes of incubation, 200 µl of chloroform was added. Cells were incubated at room temperature for 2 minutes and centrifuged at 4200 rpm for 15 minutes at 4°C. After centrifugation, three layers were observed. Carefully transferred the 400 µl of the upper aqueous phase, which contains the RNA, into a new tube.
Then, the aqueous phase was mixed with 500 µl of isopropanol. The mixture was centrifuged for 20 minutes at 14,000 rpm. After discarding the isopropanol, 1 mL of 75% ethanol in diethyl pyrocarbonate (DEPC) was added, and it was gently mixed. Then, the previous step was carried out twice after centrifugation at 3500 rpm for 5 minutes. After that, the pellet was dissolved in 22 µl of DEPC water after the supernatant had been removed gently. The purity and concentration of the RNA were determined by using a nano-drop spectrophotometer (ND-1000; Thermo Scientific, South San Francisco, CA, USA) and stored at −80°C.
cDNA Synthesis and qRT-PCR
Primer Sequences of GAPDH, Bax, and Bcl-2 Genes.

Statistical Analysis
Data were analyzed by using SPSS 26.0 software (IBM) and expressed as mean ± standard error (SE). Statistical differences between the control group and exposed groups were determined by a one-way ANOVA test with the least-significant difference test. The values of *
Results
Properties of gPbNPs
The nature of gPbNPs as nanoparticles was examined by using EDX, SEM, and TEM. Figure 1A–C showed the EDX spectra, particles shape by SEM, and size of particles by TEM. The shape of gPbNPs was polygonal (Figure 1B). The average size of NPs was 33.8 . 1.95 nm (Figure 1C). After the suspension of gPbNPs in Mille Q water, their size was determined using zeta sizer, and size and zeta potential were 200 ±3.0 nm and ∼12.4 mV, respectively. Characterization of gPbNPs: (A) EDX spectrum, (B) image of gPbNPs by scanning electron microscope, and (C) image of gPbNPs by transmission electron microscope (JEM 1011).
Cell Viability
MTT assay was used to evaluate the cytotoxic activity of gPbNPs by measuring the conversion of tetrazolium dehydrogenase to water-insoluble formazan crystals, and this reduction reaction occurs in the mitochondria of living cells. The MTT results demonstrated that the viability of MDA-MB-231 cells was decreased with gPbNPs interaction and in a dose-dependent manner (Figure 2A). In MDA-MB-231 cells, the cell viability according to MTT assay was observed for gPbNPs at 24 hour as 78, 77, 75, 67, 64, 57, and 43%, as shown in Figure 1A, for concentrations of 2, 4, 8, 16, 32, 64, and 100 μg/mL, respectively, and the cell viability was highly significantly decreased ( Cytotoxicity assessment in the MDA-MB-231 cell line after exposure to different concentrations of gPbNPs for 24 hour: (A) MTI assay and (B) NRU assay. *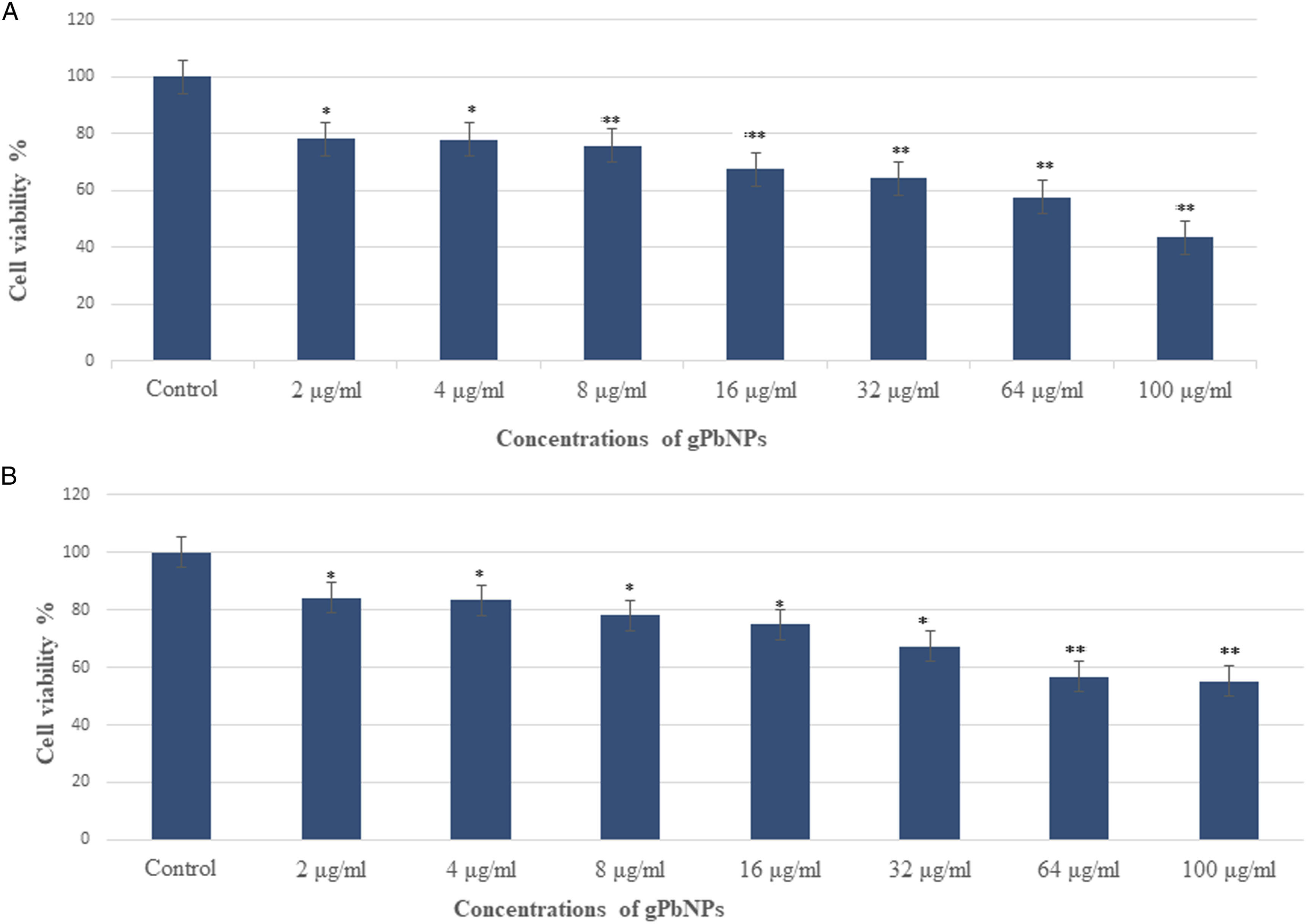
Interpretation of MTT assay results determined the half-maximal inhibitory concentration (IC50)-24 hour of gPbNPs for MDA-MB-231 cells was calculated as the concentration required to inhibit the growth of cancer cells in culture by 50% compared to the untreated cells. The gPbNPs at 55.86 μg/mL decreased the viability of MDA-MB-231 cells to 50%, and this was selected as the IC50-24 hour of gPbNPs for MDA-MB-231 cells (Table 1).
The integrity of lysosomes in MDA-MB-231 cells exposed to gPbNPs was assessed by an NRU assay. This assay provides a quantitative estimation of the number of viable cells to incorporate and bind the supravital dye neutral red in the lysosomes. The cytotoxic effects were also confirmed in control samples and samples treated with gPbNPs via the NRU assay. The NRU assay result is very similar to the MTT assay result for MDA-MB-231 cells and shows a similar trend (Figure 1B). Like the MTT assay, the NRU assay shows that at the initial concentration of gPbNPs, the viability of cancer cells was not significantly affected, but when the concentrations of gPbNPs rise, cell growth reduced. After exposure of gPbNPs to MDA-MB-23 cells, the cell viability by NRU assay at 24 hour was 84, 83, 78, 74, 67, 56, and 55%, respectively, as shown in Figure 1B, for concentrations of 2, 4, 8, 16, 32, 64, and 100 μg/mL.
Oxidative Stress
The ability of gPbNPs to induce oxidative stress was evaluated by measuring reactive oxygen species. The treatment of cells with gPbNPs resulted in the induction of ROS production (Figure 3). As shown in Figure 3A–B, there was an increase in the level of ROS in a concentration-dependent manner. The maximum level of ROS generation was induced by the highest used concentration of 42 μg/mL, while the minimum level of ROS production was closely correlated with the lowest concentration of 14 μg/mL compared to the control group (Figure 3A-B). The results confirmed that ROS-induced gPbNPs were a critical factor for human breast cancer cells (MDA-MB-231) that plays an important role in the induction of apoptosis. The treated MDA-MB-231 cells were examined by using a fluorescence microscope to confirm ROS activity. The images showed that the ROS production was exceeded after treatment with different concentrations (14, 28, and 42 μg/mL of IC50 values-24 hour) of gPbNPs stained with DCFDA fluorescence dye (Figure 3B). The intensity of the fluorescence dye was increased in a dose-dependent manner, as shown in Figure 3B. MDA-MB-231 cells showed a higher density of dichlorofluorescein as a marker of intracellular ROS generation at the highest used concentration of 42 μg/mL, than the control (Figure 3B). (A) Percentage change in intracellular ROS generation in the MDA-MB-231 cell line. (B) Generation of green fluorescence in the MDA-MB-231 cell line after exposure to various concentrations of gPbNPs for 24 hour. *
Apoptosis
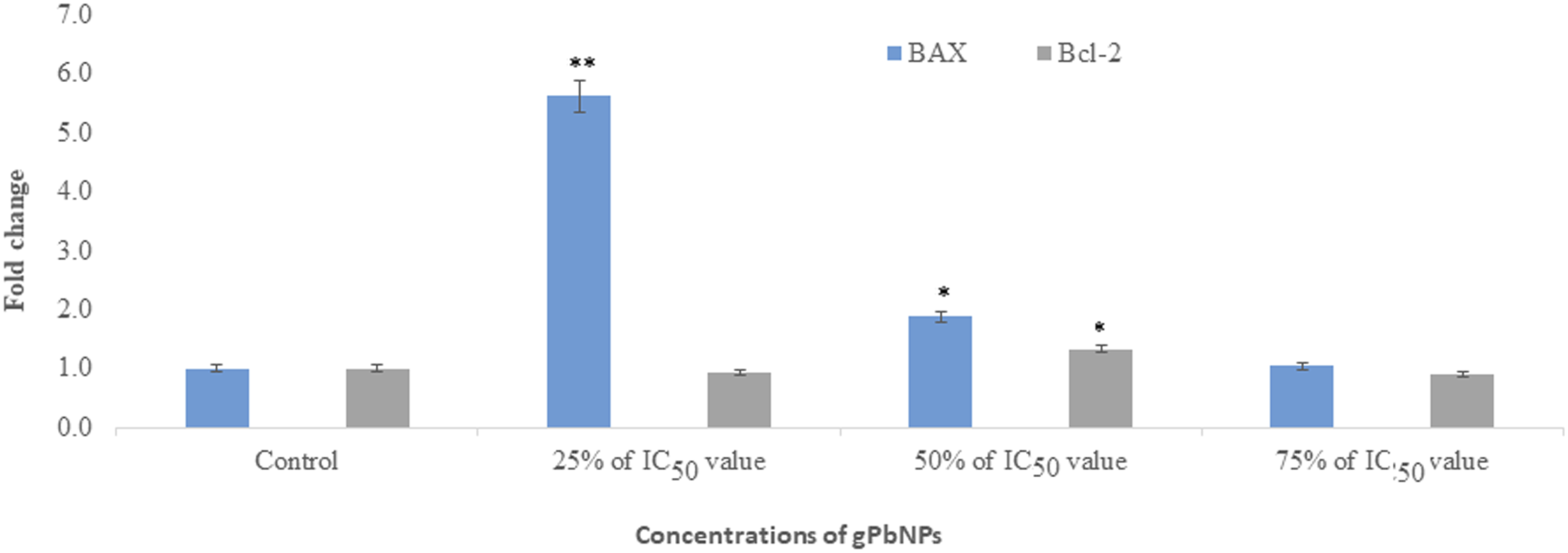
We have determined the activity of caspase 3/7 in MDA-MB-231 cells. Activities of caspase 3/7 were increased according to dose-dependent and its level was increased in MDA-MB-231 cells as the dose of treatment increased (Figure 4). The apoptosis process is regulated by several key genes encoding proteins that trigger cell death. The results of Bax and Bcl-2 gene expression are shown in Figure 5. All data were normalized with GAPDH reference gene expression level. According to the results, a significant increase in Bax ( Caspase 3/7 activity in MDA-MB-231 cell line after exposure to gPbNPs. Each value represents the mean ± SE of three experiments. * RT-PCR assessed and upregulated the expression of Bax and Bcl-2 gene in the MDA-MB-231 cell line after exposure to different concentrations of gPbNPs for 24 hour. *
Discussion
Currently, nanotechnology is a broad field of science that is more beneficial to human life than ever before. In this experiment, we synthesized green lead nanoparticles by using an extract of
Before exposure of gPbNPs to cells, we characterized the nature and size of nanoparticles using EDX, SEM, TEM, and zeta-sizers. TEM analysis showed that the average size of synthesized NPs was 33.8 S1.95 nm with polygonal and rectangular structures (Figure 1). References 15,16 reported that the size of synthesized nanoparticles depends upon plant extract volume. Nanoparticles are more biocompatible than conservative therapeutic agents, and the substantial study aims to enhance the bioavailability of NPs for therapeutic applications in diseases such as cancer. Reference 17 evaluated the cytotoxicity and apoptotic effects of gPbNPs using MDA-MB-231 cells to determine their toxicity. The MTT assay measured metabolic activity, and the results indicate a decrease in cell viability with increasing concentrations of gPbNPs suggesting that a greater number of gPbNPs could accumulate within cells leading to increased stress and ultimately cell death. These results demonstrated the enhanced efficacy of gPbNPs against breast cancer cells. They inhabited the MDA-MB-231 cells with an IC50 value-24 hour of 55.86 μg/mL, respectively.
Furthermore, the results of the MTT assay were correlated with the results of the NRU assay. The NRU assay measured the integrity of lysosomes because it reflects the ability of viable cells to incorporate the vital dye into these organelles, and the results indicate that the uptake of MDA-MB-231 cells was in a dose-dependent manner, reducing their cell viability at all concentrations. However, the NRU assay showed slightly lower cytotoxicity than the MTT assay. In contrast, these findings were consistent with Reference 18 which reported that the MTT assay indicated high anticancer activity for both Zi-AuNPs and
The majority of cytotoxic agents can induce apoptosis. The excessive production of ROS in mitochondria may contribute to this pathological condition. The ROS production may result in mitochondrial permeability disruption and the release of apoptosis-triggering proteins. Therefore, anti-cancer agents that induce ROS can play a significant role in therapeutic strategy by arbitrating apoptotic pathways. Remarkably, the results showed that gPbNPs increased ROS levels in MDA-MB231 cells in a dose-dependent manner, suggesting that gPbNPs induced apoptosis in MDA-MB231 cells. Reference 19 reported that Cu-ZnNPs induced intracellular ROS generation through mitochondrial pathways in MCF-7 cells, revealing their cytotoxic activity. The pathway involved in apoptotic cell death is crucial in developing potential anticancer agents.
20
The RT-PCR was used to investigate the balance between upregulation of the pro-apoptotic gene (Bax) and downregulation of the anti-apoptotic gene (Bcl-2) in a dose-dependent manner, indicating significant induction of apoptosis in cells treated with gPbNPs in MDA-MB-231 cells. These data may be in part attributed to the increased efficacy of apoptosis in MDA-MB231 cells induced by gPbNPs. As a supportive model, Reference 21 reported that
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Researchers Supporting Project number (RSP2023R27), King Saud University, Riyadh, Saudi Arabia.
