Abstract
Sulfamethoxazole (SMX) is widely used as an antibiotic in the clinical application with side effects of hypoglycemia. This is because SMX contains the sulfonamide structure, which closes ATP-sensitive potassium (KATP) channels and induces insulin secretion. However, there are no detail reports that measure the effective dose that can close KATP channels and induce insulin secretion. In this study, whole-cell patch clamp recording was utilized to measure the effect of SMX on KATP channel activity on pancreatic β cells. Also, the static incubation assay with mice islets was assessed to measure the insulin secretion capacity of SMX. SMX was shown to inhibit the KATP channel in pancreatic β cell membrane and induce insulin secretion in relatively high concentration. The half maximal inhibitory concentration (IC50) for KATP channel activity of SMX was .46 ± .08 mM. It was also shown that a near IC50 concentration of SMX (.5 mM) was able to nearly fully block the KATP channel when simultaneously applied with low concentration sulfonylurea, tolbutamide (.01 mM). Our present data provide important information for the clinical use of SMX to treat infection in diabetic patients using sulfonylureas.
Introduction
Sulfamethoxazole (SMX), a broad-spectrum antibiotic, is widely prescribed for clinical practice. A combination of SMX and co-trimoxazole (Trimethoprim-sulfamethoxazole, TMP-SMX 1:5 mixture), has been a first-line agent for bacterial infections such as urinary tract infections, prostatitis, and bronchitis.1–3 Moreover, it is also administered for the treatment and prophylaxis of pneumocystis carinii pneumonia in patients with acquired immunodeficiency syndrome (AIDS).4,5
However, serious adverse events related to SMX have been described. In recent years, SMX and its mixed product TMP-SMX have been reported to cause a severe hypoglycaemia in clinical application.6–8 This potentially life-threatening complication eventually results in a decreased level of consciousness and seizure.
9
This is because SMX is a sulfonamide structure containing drug that causes inhibition of KATP channel (Figure 1). Structure of SMX and sulfonylurea drugs glibenclamide and tolbutamide.
The KATP channel plays a major role in the glucose-dependent insulin secretion. In pancreatic β cells, the rise of blood glucose causes an increase in intracellular ATP, subsequently leading to the closure of KATP channel, depolarization of cell membrane, and the opening of voltage-dependent Ca2+ channel, which ultimately stimulates the release of insulin. 10
It is widely believed that Marcel Janbon of University of Montpellier, France, first reported the case on hypoglycemia in sulfonamide treated typhoid patient and thus led to the discovery of antidiabetic agent sulfonylurea. However, sulfonylurea contains S-anylsulfonylurea structure which is different from sulfonamides (Figure 1). The first sulfonylurea drug to treat diabetes was discovered in Germany independently from Janbon’s report, since intensive studies based on sulfonamide glucose lowering effects performed in France from 1940s to 50s were not accessible to Germany due to the war and postwar turbulences.11,12
From pathophysiological point of view, SMX can induce KATP channel closure and induce insulin secretion, independently from glucose dependent insulin secretion, when administered to patients. Therefore, the clinical usage of SMX potentially has risk of developing hypoglycemia. However, cases of hypoglycemia induced by SMX have been reported in patients with renal failure or those who were treated with high dose.13–15 These reports clearly indicate that pathophysiological conditions that increase the circulating concentration of SMX is linked to development of hypoglycemia.
Because sulfonamide drugs such as SMX are no longer used for the treatment of diabetes, its detail aspect of glucose lowering effect is less studied. However, diabetic patients are more prone to infection and therefore there is a higher chance to be treated with the antibiotics such as SMX. Because KATP channel inhibiting sulfonylurea drugs are widely used for the treatment of diabetes, additional usage of SMX in such patient may also increase the risk of developing hypoglycemia. Therefore, detail information such as dose dependent effect of SMX on KATP channel is clinically important.
In this study, by using the whole-cell patch clamp electrophysiology technique and insulin static incubations, we investigated the dose dependent effect of SMX on KATP channel activity and synchronized effect when used together with sulfonylurea, tolbutamide.
Methods
Preparation of Mouse Islets
Adult (6–8 weeks) male C57Bl/6J mice were used in this study. All mice were housed in a 12 h light/dark cycle with the room temperature maintained at 23–25°C and the relative humidity maintained at 45%–60%. Food and water were provided ad libitum and met all the criteria of the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International. All animal experiments were approved by the Laboratory Animal Care and Use Committee of Fukushima Medical University.
Mice were killed by cervical dislocation, and the pancreas was aseptically dissected. Islets were isolated using liberase digestion according to the procedures reported previously. 16 Isolated islets were incubated in calcium-free Hank’s solution first, and then triturated into single cells in Dulbecco’s Modified Eagle’s Medium (DMEM) containing 10 mM of glucose and 10% fetal bovine serum (FBS). Cells were cultured in an incubator with 5% CO2 at 37°C and used 1–2 days after isolation.
Electrophysiological Analysis
The KATP channel currents were recorded at room temperature (22 °C–25°C) using the whole-cell patch clamp method with an Axopatch 200B amplifier (Axon Instruments Inc., Union City, CA) controlled by Clampex 10.2 software via a Digidata 1320A interface (Molecular Devices). The standard extracellular solution contained 5.6 mmol/L KCl, 138 mmol/L NaCl, 2.6 mmol/L CaCl2, 1.2 mmol/L MgCl2, and 10 mmol/L HEPES. The pH value of extracellular solution was adjusted to 7.4 with NaOH. The pipette solution contained 107 mmol/L KCl, 1 mmol/L CaCl2, 1 mmol/L MgCl2, 10 mmol/L HEPES, and 10 mmol/L EGTA. .3 mmol/L ATP was added to prevent rundown. 17 The pH value of the pipette solution was adjusted to 7.2 with KOH. Glass pipettes were prepared from borosilicate tube glass (Narishige, Tokyo, Japan) and had 2–5 MΩ when filled with the pipette solution. Following gigaohm seal formation, negative pressure was applied to the pipette to rupture the membrane and establish the whole-cell mode. The KATP currents were measured in response to ±10 mV pulses from a holding potential of -60 mV. Data acquisition and storage were conducted with the use of a pClamp 10.2 (Molecular Devices).
Data Analysis
The concentration dependence of SMX on KATP channels was determined by fitting the following formula: I/Ic = 1/(1+[X]/IC50)h. Here, I and I c are the current amplitudes in the presence and absence of SMX, respectively; IC50 is the half maximal inhibitory concentration [X] of SMX; h is the Hill coefficient (slope factor).
Insulin Secretion Experiments
Islets were cultured in DMEM medium overnight before insulin secretion assay. After 1h of starvation in 2 mM glucose, insulin secretion was measured by static incubations (10 islets/well) in 2 mL Krebs-Ringer buffer [KRB solution: 118.5 mmol/L NaCl, 4.74 mmol/L KCl, 2.54 mmol/L CaCl2, 1.19 mmol/L KH2PO4, 25 mmol/L NaHCO3, 1.19 mmol/L MgSO4, and 10 mmol/L HEPES (pH 7.4)] with various concentrations of SMX and .1% bovine serum albumin containing 2 or 20 mmol/L glucose. Samples of the supernatant were measured using a Mouse Insulin ELISA kit (Morinaga, Japan).
Statistical Analysis
All data are expressed as mean ± SEM. Comparison of insulin secretion and KATP channel activity upon application of tolbutamide with/without SMX were analyzed by one-way ANOVA followed by Tukey’s multiple range test. KATP channel inhibition with application of SMX (0.5 mM) alone or SMX together with tolbutamide (.01 mM) was verified using Student’s t-test. Significance was assumed at P < .05.
Results
Effect of SMX on KATP Channel Currents of β Cells
The KATP channel currents of pancreatic β cells isolated from intact mice were recorded using the whole-cell patch clamp technique. The application of sulfonylurea, tolbutamide (.5 mM), a blocker of the KATP channel, inhibited the current to almost complete reduction. Thus, it is assumed that the currents which were detected in this experiment were indeed KATP channel currents. Application of an extracellular solution containing 20 mM glucose had no effect on KATP channel current, indicating that the intracellular complex including the glycolysis system is replaced by the pipette solution. Therefore, factors such as glucose metabolism do not interfere with KATP channel under our experimental condition.
As shown in Figure 2, SMX inhibited KATP channel dose dependently. The inhibitory effect of the KATP channel current was maximum at the concentration of 10 mM SMX. The IC50 of SMX for the reduction of KATP channel was .46 ± .08 mM, and the Hill coefficient was 1.8 ± .6. The concentration-response curve for inhibition of KATP channel currents by SMX. The relationship between the SMX concentration and macroscopic KATP channel current is expressed as a fraction of its amplitude in the absence of the drug (I/I
C
). The curve is the best fit of formula (1) to the mean data with IC50 = .46 ± .08 mM, h = 1.8 ± .6.
SMX Stimulates Insulin Secretion
The amount of insulin secretion after SMX application was measured by static incubation of islets. In this experiment, the glucose concentration of 2 and 10 mM were used to represent fasting and postprandial blood glucose level (Figure 3). Under the postprandial condition (10 mM of glucose), SMX significantly stimulated insulin secretion. Even in a low concentration (.1 mM), SMX also increased insulin secretion to reach the peak level (P < .05 vs control). Moreover, the stimulatory effects of SMX were also observed at a glucose concentration of 2 mM (fasting level) but the increase of insulin secretion was observed only in the presence of high concentration SMX (10 mM). (P < .01 vs control). Insulin secretion from islets during static incubation. Insulin secretion was significantly increased at concentration over 10 mM of SMX in 2 mM glucose condition, and .1 mM of SMX in 10 mM glucose condition. *P < .05, **P < .01 vs control.
Effect of Low Dose SMX on KATP Channel Inhibition Together with Tolbutamide
Because SMX was able to induce insulin secretion with low concentration under high glucose condition, it is possible that SMX may induce further inhibition of KATP channel when channel is already partially inhibited. Therefore, we applied IC50 concentration of SMX (.5 mM) together with low concentration of sulfonylurea, tolbutamide (.01 mM). As shown in Figure 4, approximately 50% of KATP channel was inhibited by the low concentration tolbutamide, while addition of SMX (0.5 mM) further inhibited the KATP channel, significantly greater than single application of SMX (P < .05). KATP channel activity with applying both SMX and tolbutamide. KATP channel was recorded by applying in response to ± 10mV pulse from -70mV. Panel A shows the representative recordings of control, tolbutamide (.01 mM), and tolbutamide + SMX (0.5 mM) and B showing the normalized currents (n = 5). *P < .05, **P < .01 vs control.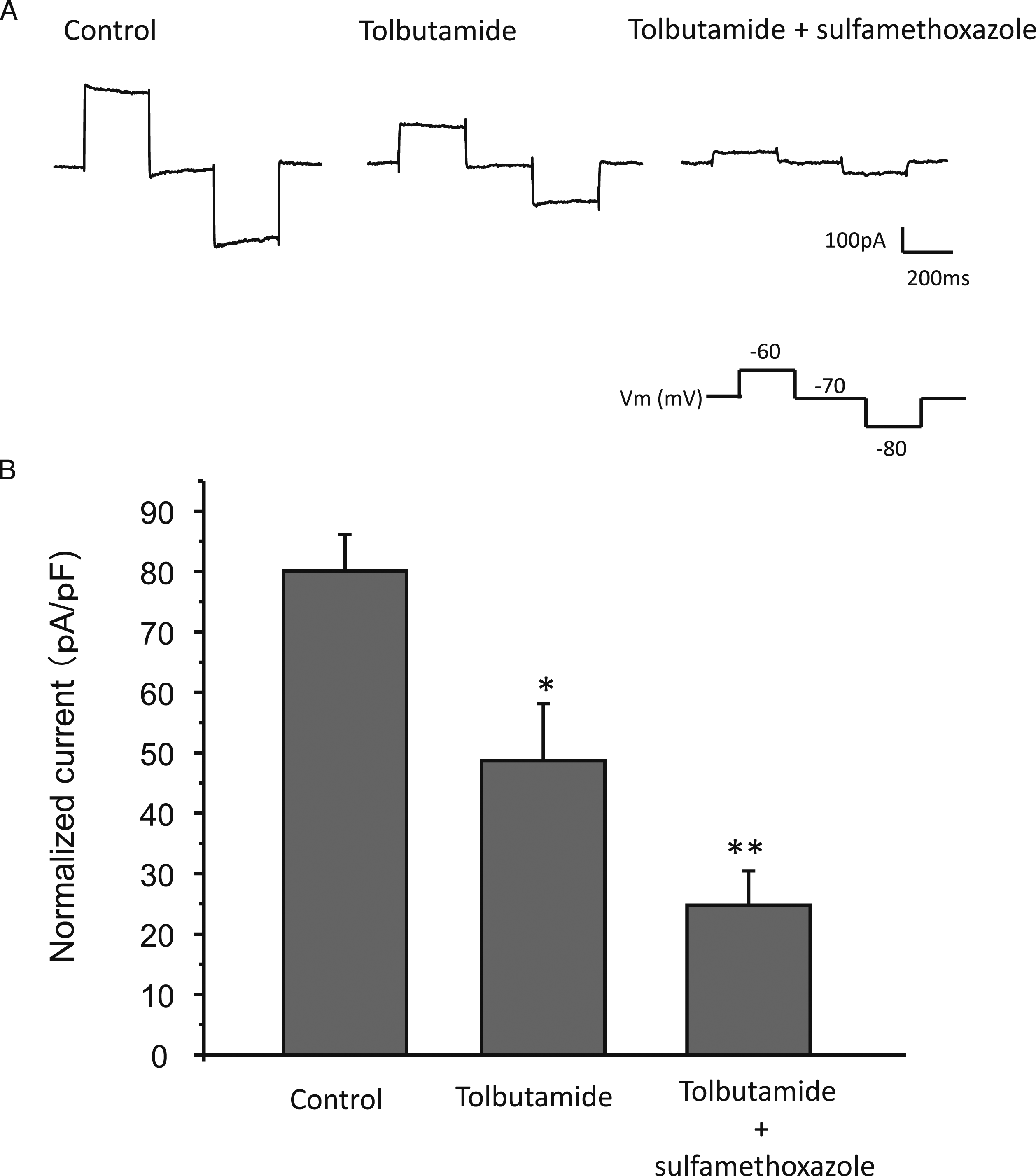
Discussion
Hypoglycaemia can result in a variety of symptoms, ranging from headache, confusion and anxiety, to coma, seizures, and even death. Up to now, several cases reported that the patients who received SMX treatment for bacterial infection have experienced a severe hypoglycaemia. This could be explained by the fact that SMX contains the sulphonamide structure which inhibits KATP channels (Figure 1). KATP channels are ubiquitously expressed on the plasma membrane of cells in multiple organs, including the heart, pancreas, and brain. KATP channels are composed of Kir6.x (Kir 6.1 and Kir 6.2) and sulfonylurea receptor (SUR; SUR1, SUR2A and SUR2B) subunits. 18 SMX could mimic the sulfonylureas action on the KATP channels in pancreatic β cells (Kir6.2/SUR1), depolarizing pancreatic β cells, activating voltage-gated calcium channel, leading to calcium-dependent insulin release.2,10
It is well known that SMX has a capacity to close KATP channel due to the sulphonamide structure. However, no studies have clarified how much concentration can induce hypoglycaemia. In the present study, we have shown that SMX stimulated insulin secretion not only in a high-glucose (10 mM) condition, but also under a low-glucose (2 mM) condition at a concentration of 10 mM. Moreover, the IC50 value for KATP channel inhibition by SMX was .46 ± .08 mM. These findings exhibit a formidable potency of SMX in inhibiting the KATP channel. However, when compared with other KATP channel inhibiting drugs, the IC50 of SMX is high. Typical KATP channel blockers that are used to treat type 2 diabetes, such as gliclazide, tolbutamide, glimepirides, and glinides, show an IC50 of .002 to .004 mM.19–21 Since the serum peak SMX concentration in human subjects after oral administration is .05 to .8 mM, it is unlikely to cause hypoglycemia with a single usage of SMX. 22 However, when used in patients with certain conditions, SMX may become capable of inducing hypoglycemia even with a single administration. One such patient would be pregnant women. According to the previous studies, SMX has been found to have the ability to cross the placental barrier and reach peak fetal levels within 3 hours of administration.23,24 Therefore, SMX could not only affect the blood glucose of pregnant women but also bring hypoglycemia in the fetuses. Also, renal insufficiency such as renal failure or renal transplant is a predisposing risk factor for developing hypoglycemia when using SMX.13,25,26 SMX is excreted from the kidney and its half-life is considered as 8–15 hours and the Food and Drug Administration discourages its use for patients with impaired kidney function.27,28
These clinical reports indicate that patients who may have a background that has higher SMX concentration are prone to experience hypoglycemia. Moreover, SMX inhibits bacterial folate metabolism by competing with para-aminobenzoic acid (PABA) to act on dihydropteroate synthetase and dihydropteroate reductase. The blockage of folate production inhibits the DNA and amino acids synthesis of bacteria. Although SMX is more selective against bacteria than in human, the drug does more or less interfere with human folate metabolism.24,29 Folate plays a critical role in embryonic development and organogenesis, especially in neural tube development. Previous studies have reported some significant associations between first trimester exposure to SMX and congenital anomalies, including neural tube defects, cleft palate, cardiovascular and urinary tract abnormalities, and clubfoot.30,31
Also, as shown in this study, SMX was able to induce an increase in insulin secretion with lower concentration in high-glucose condition, indicating that under the increased intracellular ATP level caused by high glucose and with KATP channel being more closed, SMX may further inhibit the channel and enhance insulin secretion. Consistently, in the existence of low concentration of tolbutamide, SMX was capable of inducing further inhibition of the KATP channel even with a low concentration. There are clinical reports that support our present finding. As early as in 1975, Mihic et al 32 reported that a single administration of SMX to a diabetic patient had no effect on blood glucose or insulin concentration, whereas administration of SMX to a diabetic patient under hypoglycemic agent treatment showed an increase in blood insulin concentration. Also recent reports indicate an increased risk of developing hypoglycemia when using SMX together with KATP channel inhibitors such as repaglinide, gliburide, and glipizide.33–35
Similar to this effect, we have also reported in the past that the anti-arrhythmia drug disopyramide, which closes KATP channel only in high concentration, can almost completely block the channel in low concentration when applied together with low concentration sulfonylureas. 36 Thus, disopyramide and SMX have the ability to show an additive effect on KATP channel inhibition together with sulfonylurea drugs. Our previous report also presented the case of disopyramide induced hypoglycemia in a type 2 diabetic patent taking a low dose glimepiride. 36 Therefore, SMX may increase the risk of causing hypoglycemia in a diabetic patient under sulfonylurea treatment.
Because diabetic patients are more prone to infection, and sulfonylureas are widely used as an antidiabetic agent, our present data present important information for assessing the possible risk of hypoglycemia when using SMX in such patients.
However, our present study has limitations. We have performed the experiments in isolated islets and β cells. All the data are obtained from an in vitro condition. Because electrophysiological data were obtained from a whole-cell configuration, intracellular metabolism is excluded. The animal model data and human data are not available in this study.
As for the future perspective, drug metabolism and pharmacodynamics, in vivo animal data such as glucose tolerance tests with glucose level and insulin level measurement are required to evaluate the risk of developing hypoglycemia due to the usage of SMX. Also, human data for pharmacodynamic effects are also required to clarify and evaluate the potential risk of developing hypoglycemia with SMX.
Conclusions
In summary, we demonstrated that SMX cause a KATP channel inhibition and insulin secretion in high concentrations. KATP channel is an important factor that regulates insulin secretion. Its closure can induce insulin secretion independently of blood glucose levels. Although a high dose of SMX was known to induce hypoglycemia, our present study practically showed the dose dependent effect of SMX on closing KATP channels and insulin secretion for the first time. Our present data explains the underlying mechanism of high dose SMX inducing hypoglycemia in patients. Because SMX is metabolized in the liver and excreted by the kidneys, the blood concentration of SMX may become high enough to cause KATP channel inhibition in patients with liver or kidney disfunction, and special caution is required in using SMX in such patients. Also, our data indicate that the use of SMX in diabetic patients under sulfonylurea treatment may also have a risk of developing hypoglycemia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant-Aid for Scientific Research (C) (18K08483 to Y. M, 26461366 to K. S) from the Japan Society for the Promotion of Science (JSPS).
