Abstract
Accidental radiation exposure is a threat to human health that necessitates effective clinical diagnosis. Suitable biomarkers are urgently needed for early assessment of exposure dose. Existing technologies being used to assess the extent of radiation have notable limitations. As a radiation biomarker, miRNA has the advantages of simple detection and high throughput. In this study, we screened for miRNAs with dose and time dependent responses in peripheral blood leukocytes via miRNA sequencing in establishing the animal model of acute radiation injury. Four radiation-sensitive and stably expressed miRNAs were selected out in the 24 h group of leukocyte miRNAs: mmu-miR-130b-5p, mmu-miR-148b-5p, mmu-miR-184-3p, mmu-miR-26a-2-3p, and five were screened in the 48 h group of leukocyte miRNAs: mmu-miR-130b-5p, mmu-miR-423-5p, mmu-miR-676-3p, mmu-miR-150-5p, mmu-miR-342-3p.The correlation curves between their expression and irradiation dose were plotted. Then, the results were validated by RT-qPCR in mouse peripheral blood. As a result, mmu-miR-150-5p and mmu-miR-342-3p showed the highest correlation at 48h after irradiation, and mmu-miR-130b-5p showed good correlation at both 24 h and 48 h after irradiation. In a conclusion, the miRNAs that are sensitive to ionizing radiation with dose dependent effects were selected out, which have the potential of forming a rapid assessment scheme for acute radiation injury.
Introduction
With the widespread use of nuclear and radiation technology, there are potential radiation damage hazards associated with its use for the benefit of mankind. Acute radiation injury will occur when people are exposed to large doses (>2.0 Gy) of ionizing radiation during a short period of time. The severity of acute radiation injury is significantly and positively correlated with the dose received, which also influences the choice of treatment, making it important to rapidly reconstruct the dose received by the injured person after exposure.
Radiobiometrics is the science of measuring doses and assessing hazards using the biological changes caused by ionizing radiation in the body. One type of biological change with a good dose-effect relationship is called biodosimetry. 1 Radiation dosimetry measurements allow for the early metrological assessment and the prediction of the long-term health effects of the exposed persons after a nuclear accident. Once a nuclear accident occurs, a large number of nuclear radiation injuries will happen, which makes it necessary for radiation biodosimetry to perform fast and simple detection with high throughput. The current radiation biodosimeters are mostly cytogenetic techniques, 2 such as chromosome aberration analysis, micronucleus analysis, and DNA damage analysis. 3 These methods can theoretically cover a dose range of .2–10 Gy, which can basically estimate the dose of most accident casualties. However, the problem of low detection throughput is unsolved, making it difficult to cope with a large number of nuclear and radiation casualties. For example, γH2AX foci have a clear correlation in the dose range of .25–2.0 Gy, but its detection is complex and the number of foci disappears rapidly with the completion of DNA damage repair, which also makes the detection window short. 4 Thus, there is a critical need to develop radiation-specific indicators that are capable of immediately evaluating deterministic effect and stochastic effect of radiation exposure.
In order to solve the technical bottlenecks including low throughput and cumbersome operation of cytogenetic detection, searching for radiation-sensitive molecular markers with the help of emerging molecular biology technology has become a hotspot in the field of radiation biometrics.5,6 MiRNAs are a class of endogenous small non-coding RNA molecules of approximately 22 nucleotides in length with a single-stranded short hairpin structure. They can silent or enhance the target mRNAs and regulate the expression of target genes in a post-transcriptional manner.7,8 Studies have shown that miRNAs are widely involved in human pathophysiological processes. Specific downregulation or upregulation of some miRNAs leads to cancer progression and their specific expression can be used as tumor markers. 9 For example, miR-143 and miR-145 play an important role as tumor suppressor genes in gastrointestinal tumors, breast cancer, and brain metastasis of lung cancer.10,11
The advantages of miRNA test include simple operation and high throughput. And in the field of radiation damage research, there are also studies suggesting that miRNA can also be used as radiation biomarker to predict the degree of exposure. 12 Wei et al identified that a set of serum miRNAs including miR-183-5p, miR-9-3p, miR-200b-5p, miR-342-3p, and miR-574-5p that are quite sensitive to different radiation types in the early stages after exposure. They are stable in response to environmental changes and common among different species. 13 They also demonstrated that some specific serum miRNAs have the potential of serving as novel noninvasive indicators in special environments like space. 14 Furthermore, there’s also related studies on integration of miRNA and other molecules. Marshleen Yadav et al 15 reported a two-microRNA–based (miR-150-5p and miR-23a-3p) scalable radiation biodosimetry (miR-RAD) assay that allows rapid and accurate estimation of absorbed ionizing radiation dose in pre-clinical studies. Bai et al identified some novel proteins, miRNAs, and tRNA-derived fragments in serum showing great potential as early biomarkers of exposure to energetic heavy ions. And they might be used for dose reconstruction and risk assessment of heavy ion exposure in deep space exploration. 16 Circulating miRNAs reflect the impact of ionizing radiation irrespective of some different species, often in a dose-dependent manner. 17 In brief, these results make miRNAs promising biomarkers of radiation exposure.
Therefore, in this study, we screened for the dose and time dependent miRNAs in peripheral blood leukocytes by establishing the animal model of acute radiation injury in mice under different irradiation dose conditions, and plotted their expression correlation curves with irradiation dose to provide an experimental basis for the screening for potential radiation biomarkers with the help of miRNA sequencing technology.
Materials and Methods
Animals and Reagents
SPF-grade C57BL/6 male mice, 6–8 weeks old, weighing 19–21 g, were purchased from the Experimental Animal Center of the Naval Medical University (Shanghai, China) and were kept in the laboratory animal room under standard conditions (Faculty of Naval Medicine Naval Medical University, Shanghai, China). MiRNA quantitative PCR kits were purchased from TAKARA (Japan).
Mouse Irradiation Model
C57BL/6 mice were irradiated with Co 60 radiation source at a dose rate of 1.0 Gy/min using the Irradiation Centre of the Naval Medical University (Shanghai, China) to establish acute radiation injury model at different doses (0, 2, 4, 6, and 8 Gy).
BMC Transplantation
BMT were established by following the method of Mulligan and colleaques with modifications. 18 Breifly, bone marrow cells from unirradiated mice were used as the donor, and mice receiving different doses of ionizing radiation were used as the recipient. Within 24 h after irradiation, 2*106 nucleated bone marrow cells were transplanted into the irradiated mice, and the survival of the recipient mice was monitored for 30 days.
Analysis of Peripheral Blood Cells
C57BL/6 mice were euthanized with isoflurane, approximately 100 μl of peripheral blood was collected in heparin-anticoagulated EP tubes, fully anticoagulated and then counted in five categories using a small animal haemocytometer, and trends in peripheral blood leucocyte counts were counted.
WBCs Separation and RNA Extraction
The WBCs were separated in a blood separation medium according to the manufacturer’s protocol (Solarbio, Beijing, China). The separated WBCs were washed in 37°C prewarmed phosphate-buffered saline (PBS) 3 times. Then, the miRNAs in WBCs of C57BL/6 mice were extracted by TRIzol method. NanoVue (GE, USA) was used to assess RNA purity. Each RNA sample had an A260:A280 ratio greater than 1.8 and an A260:A230 ratio >2.0.
Small RNA Sequencing and Bioinformatics Analysis
The extracted miRNAs were detected by small RNA sequencing technology (Shanghai Ouyi Biotechnology Co., Ltd.). The changes in miRNA profiles of mice after irradiation were analyzed, and the miRNAs with stable and differential expression were initially screened by statistical analysis.
Those miRNAs that were closely related to radiation dose were analyzed by bioinformatics and the dose-effect curve was fitted.
Screening of Stable miRNAs for RT-PCR Validation
Primers were designed using the tailing method to detect the expression of the screened miRNAs in mouse by real-time fluorescence quantitative PCR to test the reliability of the dose-effect curve. Since the conventional internal reference U6, β-actin, and GAPDH are not stable enough and often undetectable, miR-16 was chosen as the internal reference.
miRNA Primer Sequences
MIMAT0004583 >mmu-miR-130b-5p ACUCUUUCCCUGUUGCACUACU
MIMAT0017036 >mmu-miR-148b-5p GAAGUUCUGUUAUACACUCAGGCU
MIMAT0004825 >mmu-miR-423-5p UGAGGGGCAGAGAGCGAGACUUU
MIMAT0000213 >mmu-miR-184-3p UGGACGGAGAACUGAUAAGGGU
MIMAT0003782 >mmu-miR-676-3p CCGUCCUGAGGUUGUUGAGCU
MIMAT0017058 >mmu-miR-26a-2-3p CCUGUUCUUGAUUACUUGUUUC
MIMAT0000160 >mmu-miR-150-5p UCUCCCAACCCUUGUACCAGUG
MIMAT0000590 >mmu-miR-342-3p UCUCACACAGAAAUCGCACCCGU
Statistical Analysis
Data were expressed as means ± the standard errors of means. Two-tailed Student’s t-test was used to analyze the differences between two groups. One-way ANOVA was employed to analyze the differences among three groups. The data were analyzed using SPSS ver. 19 software (IBM Corp, Armonk, New York, USA). The graph were made using Graphpad Prism 8.
Results
The Survival Rate of Mice After Exposure to Different Doses of γ-rays
The 30-day survival rate was observed by constructing the mouse model of acute radiation injury under different irradiation dose conditions. It was found that the survival rate gradually decreased with increasing irradiation dose (Figure 1A). In addition, bone marrow cell transplantation was performed on mice under different dose conditions, finding that the salvage ability of a certain number of BMCs decreased significantly with increasing irradiation dose (Figure 1B). It was demonstrated that the degree of acute radiation damage in mice increased significantly with increasing irradiation dose. Therefore, we constructed the mouse model of acute radiation injury at different irradiation doses to screen for potential radiation biomarkers. Changes in survival rate of mice at different doses. (A). The 30-day survival rate was observed by constructing the mouse model of acute radiation injury under different irradiation dose conditions. (B). The 30-day survival rate was observed by constructing bone marrow transplantation models of acute radiation-injured mice under different irradiation dose conditions.
Changes in Peripheral Blood Cell Counts in Mice Exposed to Different Doses
The hematopoietic system is the most vulnerable tissue to the damaging effects of radiation. Exposure to low or moderate doses (0,2,4,6 and 8 Gy) of radiation leads to a rapid decrease in blood cell counts, including loss of lymphocytes, neutrophils, and thrombocytes, and a severe decrease in hematopoietic progenitors. The decrease in peripheral blood leukocyte count can be used to indirectly reflect the magnitude of the dose to which the body has been exposed. And the change in the absolute value of lymphocytes, in particular, is significantly correlated with the dose to which the body has been exposed, and is mostly used as a key indicator of the severity of acute radiation injury in clinical practice. Therefore, in this study, we constructed the mouse model of acute radiation injury with different doses of irradiation and collected peripheral blood for cell counts at 24 and 48 hours after irradiation. As the irradiation dose increased, the peripheral blood showed a significant decrease in leukocytes, lymphocytes and monocytes, while the number of erythrocytes and platelets did not change significantly. It is worth noting that the decrease in leukocytes, lymphocytes and monocytes did not show a significant dose-effect relationship with the dose of irradiation, and the numbers decreased significantly after doses above 2 Gy (Figure 2A/B). There is no particular pattern of changes in peripheral blood counts, and changes in peripheral blood counts are not only related to radiation. These results showed that peripheral blood count alone is not a good indicator of the exposure dose to which the body has been exposed. Changes in peripheral blood cell counts in mice exposed to different doses. (A). Five classifications of peripheral blood of mice 24 h after different irradiation doses. (B). Five classifications of peripheral blood of mice 48 h after different irradiation doses. WBC(white blood cell),LYM(lymphocyte),MON(monocytes),RBC(red blood cell),PLT (platelet). *, **, and *** represented 
Screening the miRNA Expression Profiles After Exposure
As we all know, serum miRNAs are early indicators of survival after radiation-induced hematopoietic injury. As a radiation biomarker, miRNAs have many advantages. In this study, we collected peripheral blood leukocytes from mice at 24 and 48 h after exposure to different doses of radiation for miRNA sequencing. The results are shown in Figure 3A. The total number of known miRNAs on all samples was 1411 and the total number of newly predicted miRNAs on all samples was 2820, with a total of 8 differential comparison groups ( MiRNA sequencing screening. (A). Differential miRNA genes in mice 24 h and 48 h after different irradiation doses. (B). Boxplots illustrating the distribution of data in mice 24 h and 48 h after different irradiation doses. (C). The differential genetic heat maps between the irradiated groups and the non-irradiated group ( The list of 8 differential comparison groups.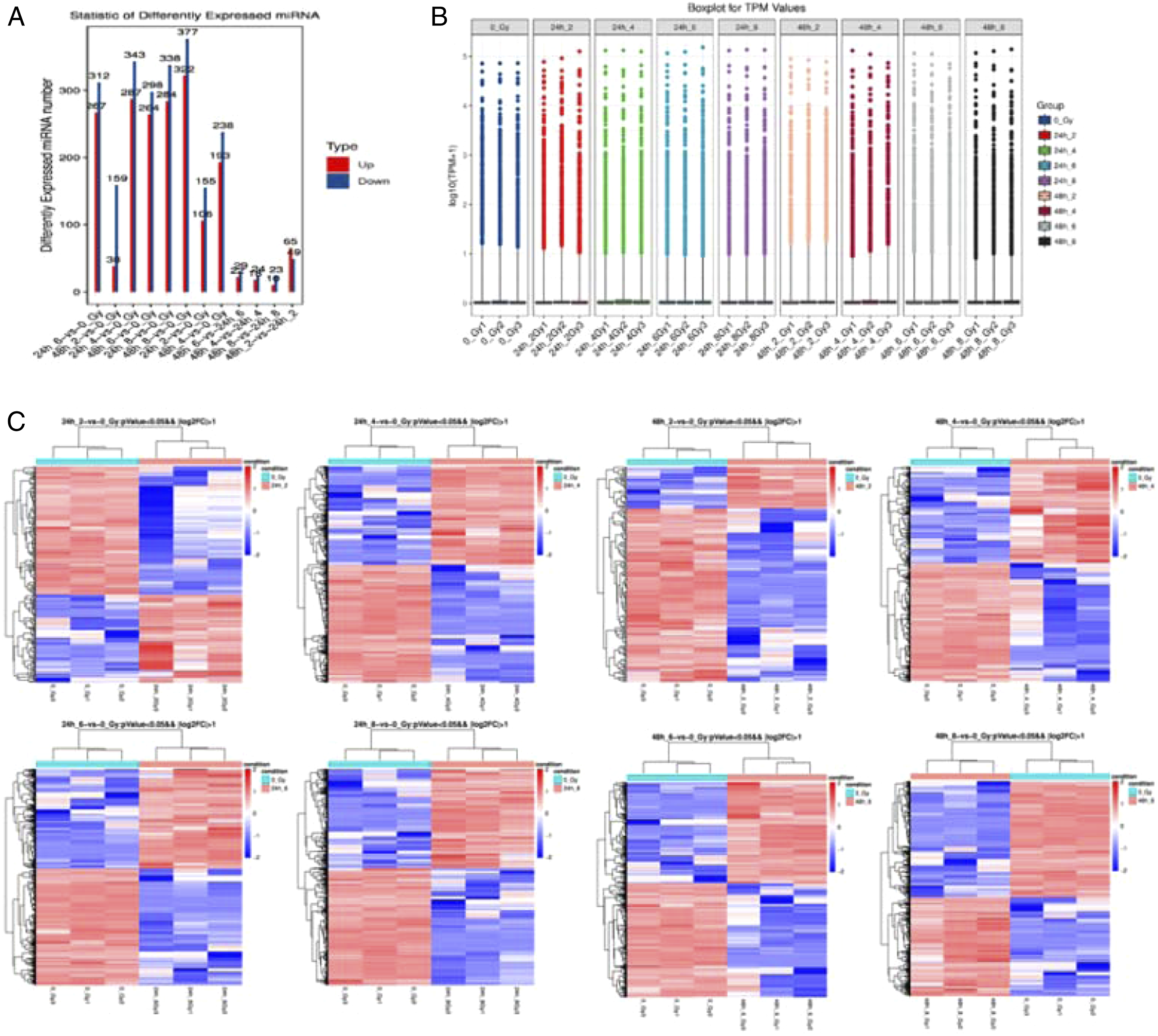

The list of potential radiation dosimeter miRNAs.

Correlation Analysis of the Screened miRNAs
Further, we performed correlation analysis on the screened miRNAs. In the 24 h group of leukocytes, four radiation-sensitive and stable miRNAs were screened according to the screening criteria: mmu-miR-130b-5p, mmu-miR-148b-5p, mmu-miR-184-3p, mmu-miR-26a-2-3p. After that, a linear and semi-logarithmic fit were performed on the individual miRNAs for the dose effect relationship (Y: Dose X: Relative expression). The results are shown in Figure 4A. In the 48 h group of leukocytes, five radiation-sensitive and stably expressed miRNAs were screened according to the screening criteria: mmu-miR-130b-5p, mmu-miR-423-5p, mmu-miR-676-3p, mmu-miR-150-5p, mmu-miR-342-3p, and the dose-effect relationships of single miRNAs were fitted linearly as well as semi-logarithmically. mmu-miR-130b-5p, mmu-miR-423-5p, and mmu-miR-676-3p were selected for primary linear fit while mmu-miR-150-5p and mmu-miR-342-3p were selected for semi-logarithmic fit by comparing the fit (Y: Dose X: Relative expression) (Figure 4B). The results showed that mmu-miR-130b-5p, mmu-miR-184-3p at 24 h after irradiation and mmu-miR-130b-5p, mmu-miR-423-5p, mmu-miR-676-3p at 48 h after irradiation were all positively linear correlated with dose. At the same time, mmu-miR-148b-5p, mmu-miR-26a-2-3p and dose were linearly negatively correlated at 24h after irradiation. Meanwhile, the mmu-miR-150-5p, mmu-miR-342-3p and dose were negatively correlated with the logarithm at 48 h after irradiation. Correlation analysis of the screened MiRNAs. (A). A linear fit of the dose effect relationship of four radiation-sensitive and stable miRNAs and radiation dose at 24 h after irradiation by screening criteria. (B). A linear and semi-logarithmic fit of the dose effect relationship of five radiation-sensitive and stable miRNAs and radiation dose at 48 h after irradiation by screening criteria. Validation of potential radiation dosimeter miRNAs. (A). Heat map of expression of four miRNAs at 24 h after different irradiation doses. (B). Heat map of expression of five miRNAs at 48 h after different irradiation doses.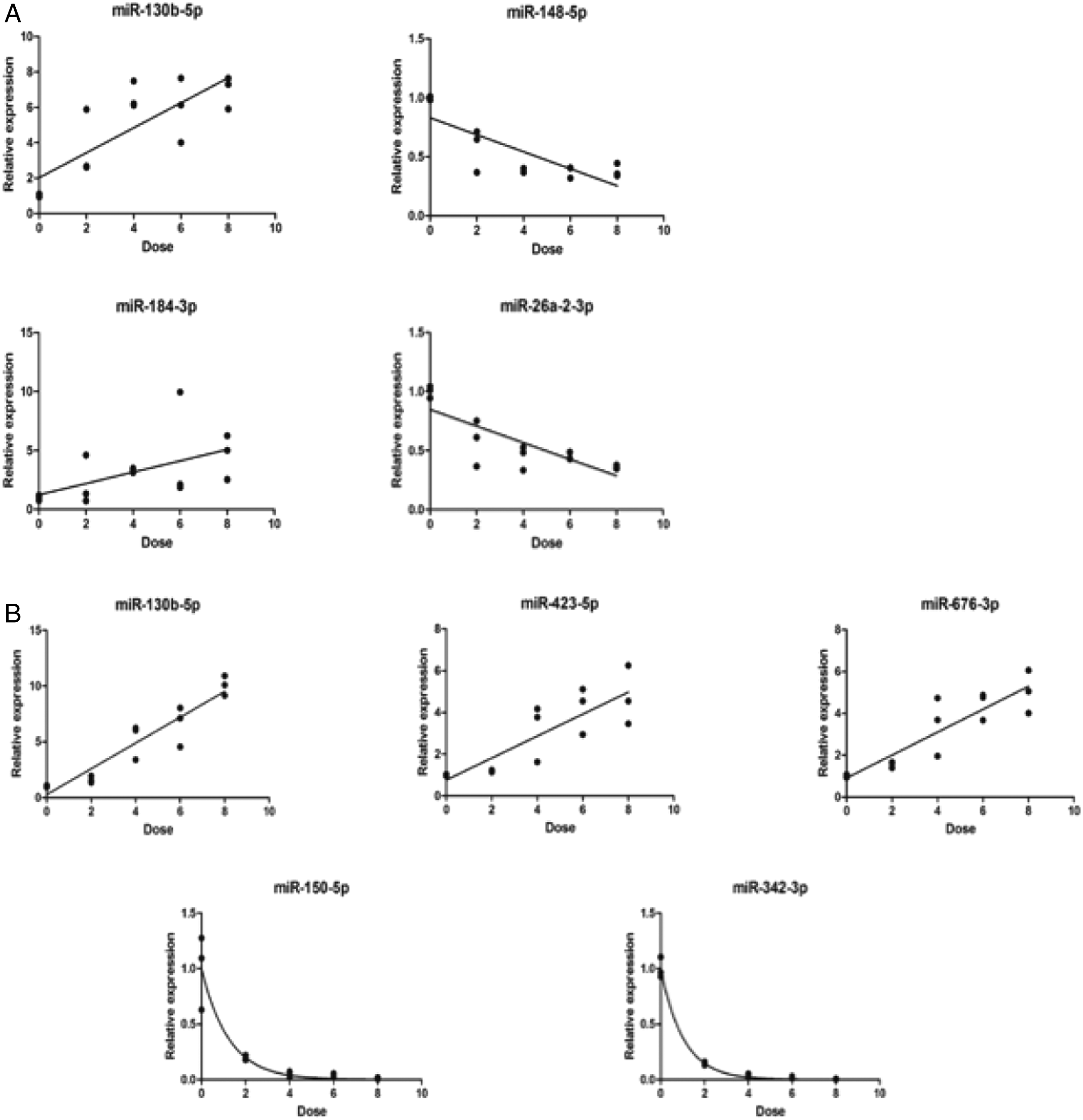

Validation of Potential miRNAs as Radiation Biomarkers
To validate the dose-effect relationship between screened miRNAs and irradiation dose, animal model of acute radiation injury in mice under different doses were constructed again, finding that mmu-miR-150-5p and mmu-miR-342-3p have the highest correlation at 48 h after irradiation, and mmu-miR-130b-5p had a good correlation at both 24 h and 48 h after irradiation (Figure 5). Therefore, mmu-miR-130b-5p was chosen as post-irradiation biological dosimeters 24 h after irradiation. Meanwhile, mmu-miR-150-5p, mmu-miR-342-3p and mmu-miR-130b-5p were chosen as post-irradiation biological dosimeters 48 h after irradiation. The above results show that there is a relationship between the expression of some miRNAs and the irradiation dose, which can be used as radiation biological dosimeters.
Discussion
Biological dose estimation techniques are a key aspect of medical rescue for rapid classification, diagnosis and dose estimation of casualties. Currently, the commonly used methods for exposure dose detection are mainly hematological, cytogenetic, or molecular biological means, and the application of metabolomics, immunomics, and genomics in the field of radiation dosimetry has further enriched the range of radiation biomarkers. However, due to the complex environment of the radiation accident site, the limitations of existing methods and the uncertainty of emerging methods, there are no optimal biomarkers for assessing the exposure dose till now.
Novel radiation biomarkers must be sensitive to radiation, stable, and capable of rapid and repeatable detection in a non-invasive or minimally invasive manner. In contrast, biochemical parameters such as C-reactive protein, amylase and cytokines have disadvantages such as high inter-individual variability and fluctuations with inflammation and infection when used to estimate the irradiated dose. 19 In addition, as the “gold standard” for assessment of absorbed dose is lymphocyte chromosome aberration, such as cracks, deletion, rings et al, which are largely automated in terms of cell harvesting, specimen preparation, and double-stranded chromosome analysis, however, they are still time-consuming, highly technical, and subject to human influence. 20 γ-H2AX, the best biomarker for detecting DNA double-strand breaks, has been gradually being recognized as a molecular marker of radiation damage at home and abroad. But it still cannot avoid the problems of poor dose-effect relationship and narrow detection window. 21 As emerging potential radiation dose estimation biomarkers, gut microorganisms and their metabolites, miRNAs, lncRNAs and other gene transcription markers, radiation metabolites and new measurement techniques provide the feasibility of finding new molecular markers of radiation damage and exploring multi-molecular combinations as rapid radiation biomarkers, but further studies are still needed.
Because of their small size and protection by mechanisms such as Extracellular Vesicles (EVs), miRNAs are not easily degraded by RNA enzymes, which are stable, easy to quantify, and accessible. 22 In addition, the reproducibility of miRNAs in individuals of the same species is good and the source of miRNAs in serum is tissue-specific. Therefore, miRNAs can be used as biomarkers for many physiological responses and pathological stages, including radiation injury. 23
In this study, we first screened the radiation-sensitive and stable miRNAs in the 24 h and 48 h leukocyte groups and obtained the corresponding quantitative efficacy fitting curves after linear and multiparametric fitting of the screened single miRNAs. And then we validated them in mouse and human peripheral blood samples, finding that miR-150/342 have the highest correlation and miR-130 had good correlation in both 24 h and 48 h. It has been confirmed that the miRNAs mentioned above are radiosensitive and have a good dose-effect relationship with radiation dose, and can regulate cell cycle and affect cell proliferation and apoptosis, especially the invasion and metastasis of cancer cells.
Differentially expressed plasma miRNAs have been studied in many models. Existing studies have shown that miR-150-5p is closely related to cell proliferation and apoptosis. It has been showed that miRNA-150-5p promotes hepatic stellate cell proliferation and sensitizes hepatocyte apoptosis during liver fibrosis. 24 In addition, miR-150-5p also plays a role in radiotherapy. Wu found that miR-150 might serve as a potential therapeutic sensitizer through inhibition of the AKT pathway in NK/T cell lymphoma treatment. 25 MiR-130a/b expresses significantly different in different tumors, and both of them play similar roles as proto-oncogenes and oncogenes by regulating key signaling molecules that affect cell proliferation, apoptosis, metastasis and tumor drug resistance in a variety of tumors, and are expected to serve as potential markers for determining tumor metastasis and prognosis.26,27 In addition, miR-130b is associated with cell proliferation and apoptosis.28,29 MiR-342-3p is expressed in a variety of female-related diseases and regulates cell proliferation, invasion and metastasis, suggesting that it plays an important role in the development and progression of those diseases.30,31
Especially, as mentioned in the previous section, some differentially expressed plasma miRNAs have been studied as biomarkers for assessment of total-body radiation exposure dosimetry. Evolutionarily conserved miR-150-5p detected in serum and plasma has been shown to exhibit dose response in rodent and nonhuman primate (NHP) models of total body irradiation.15,32 Arnab Chakravarti et al found that miRNA-150 abundant in lymphocytes exhibited dose- and time-dependent reductions in serum. Therefore, Arnab Chakravarti et al suggested it as a sensitive marker for lymphocyte depletion and bone marrow damage. 33 A report by Dinh et al 34 identified miRNAs that showed a dose-dependent expression pattern in the serum (miR-150) of patients with lung cancer undergoing RT. Cha HJ et al found that miR-342 is an ionizing radiation-responsive microRNA in the IM9 human B lymphoblastoid cell line. 35 In addition, Shyam Biswal et al found that high abundance of miR-342 in plasma after ionizing radiation. The high abundance of many circulating miRNAs in the plasma suggests they might have potential biological roles. 36 In addition, Dipanjan Chowdhury et al found that miR-130a-3p increased in serum after irradiation, while miR-150-5p and miR-342-3p decreased, consistent with our results. 12
As non-coding RNA (ncRNA) molecules, miRNAs are involved in various biological activities such as stem cell differentiation, organ development, cell death, cell cycle temporal changes and signal transduction by regulating target mRNA transcription or post-translation and inducing silencing of target gene expression. 37 In recent years, through the combination of high-throughput genomics and bioinformatics with traditional molecular biology techniques and animal models, miRNAs are able to regulate tumor-related gene expression and play a key role in the diagnosis and treatment of cancer and cardiovascular diseases, etc. They can be used as diagnostic markers for cardiovascular diseases, and also helpful for cancer types differentiation and prognosis assessment. Serum miRNAs fall under the emerging “omic” biodosimetry assays and represent a simple technology that may effectively determine whether an individual was exposed to radiation.
Focused on the urgent needs of prevention and treatment of acute radiation disease, this study is dedicated to the formation of a rapid radiation dose assessment protocol, which innovatively targets at screening a group of stable miRNA molecules with obvious expression differences before and after radiation to avoid the instability of single miRNAs. More significantly, we’ve concluded the corresponding quantitative efficacy fitting curves of several potential miRNAs. Meanwhile, the detection process is completed by quantitative PCR with high objectivity, reducing the reliance on manual operation. Furthermore, quantitative PCR can achieve high throughput and rapid detection, which is more suitable for the accident scene rescue.
However, it is still far away from clinical application of circulating miRNAs as radiation biomarkers. Some barriers are still in there. For example, gender has been proved to be important confounding variable in biomarker development that must be incorporated into biomarker discovery and validation. 38 miRNAs are also involved in skin functioning and aging, implying that age may affect the function of miRNAs as radiation biomarkers. 39 What is more, the influences of qualities of radiations, the differences of LET, dose rate, 40 and energy should also be taken into consideration. Thus, it is necessary to know more about the radiosensitive molecules including miRNAs. And the results of this study will be used to make rapid detection kits. We sincerely hope that the systematic study of multi-molecule expression profiling combined with histological techniques and biochemical analysis can be used for early and rapid classification and on-site medical emergency response for different types of nuclear accident casualties.
Footnotes
Acknowledgments
We appreciate the radiation device department of Naval Medical University, which produced gamma-rays. We thank support of research fund BHJ17J009 and GWV-10.1-XK10. We acknowledge the editors and reviewers for their helpful comments on this paper.
Author Contributions
Ying Cheng, Jicong Du, and Cong Liu designed the study. Zhefan Shen, Wei Chen and Lan Fang performed the experiments. Zhenlan Feng and Jianpeng Zhao analyzed the data. Jiaxun Li and Jicong Du wrote the paper. Ying Cheng, Jicong Du, and Cong Liu supported fund assistance. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research fund BHJ17J009, GWV-10.1-XK10 and NSFC81872559. These support the experimental research and data collection of the subject.
Ethics Approval and Consent to Participate
All animal experiments conformed to the National Institute of Health Guide for the Care and Use of Laboratory Animals' (NIH Publication No. 85-23, National Academy Press, Washington, DC, revised 1996), with the approval of the Laboratory Animal Center of the Naval Medical University, Shanghai.
Consent for Publication
Written informed consent for publication was obtained from all participants.
Availability of Data and Material
All data generated or analyzed during this study are included in this published article and its supplementary information files.
