Abstract
Industrial waste is mainly responsible for accumulating Zn (II) in the soil, which needs to be removed to avoid its bioaccumulation and hazardous effects on the environment. In a recent study, the potential of the RY12 strain was evaluated as a biosorbent of Zn (II) ions in an aqueous medium. Different microbiological techniques like biochemical, molecular characterization, and 16S rRNA gene sequencing were used for the identification of RY12. The impact of different parameters such as the initial zinc ion concentration, pH, temperature, and the removal of other metals such as manganese, lead, cobalt, silver, copper, mercury, and chromium was also evaluated on the reduction of Zn (II). Fourier Transform Infrared spectroscopy (FTIR) was also carried out to investigate the role of cellular surfaces in the sorption of Zn+2 ions. Both biochemical and phylogenetic analyses established that strain RY12 Pseudomonas sp. capable of reducing Zn+2 up to 89% at 28°C (pH = 6.5; initial Zn+2 concentration = 200 mg/L). The FTIR analysis revealed that the bacterial cell wall’s amino, carboxyl, and phosphate groups were involved in the reaction with Zn (II). Our findings suggest that Pseudomonas sp. RY12 is a proficient bacterium for removing zinc from industrial waste and could be a valuable bioremediation agent.
Introduction
Heavy metals in soil pollute the environment and pose the most severe health threats to humans. Due to the magnitude of the problem, metal contamination receives significant attention in the remediation sector, and it needs time to develop novel techniques to address it. 1 This pollution is primarily caused by industrial operations such as metal forging, mining smelting, sewage sludge, and fossil fuel burning. Significant levels of Zn are present in these waste products, which accumulate in the soil. As a result, it harms soil physicochemical properties and microbial density, resulting in infertile soils and agricultural productivity. These metals, in general, cannot be broken down into less or more dangerous compounds and remain in the environment. 2
Growing plants in heavy metal-contaminated soils, such as cobalt, copper, manganese, molybdenum, vanadium, strontium, and Zn, house a variety of microbes that can endure high concentrations of these metals and provide multiple benefits to the plant and soil. Microbes in the rhizosphere are involved in various heavy metal interactions with plants in contaminated soil. Plant growth-promoting rhizobacteria (PGPR) receive special attention because they directly improve plant remediation by altering metal bioavailability through pH changes, phytohormone synthesis, and chelator release.3,4 Microorganisms from the rhizosphere are ideal for use as biocontrol agents. The rhizosphere offers the front-line shield for roots against attack by pathogens. Bacterial genera that are believed to have the potential for biocontrol are
Increased Zn levels in soil block various metabolic pathways in plants, resulting in growth retardation and senescence. Zinc toxicity in plants restricts the growth of roots and shoots. Copper (Cu) and manganese (Mn) concentrations in plant shoot also rise as a result of excess zinc. These deficiencies have been attributed to a delay in transferring nutrients from the root to the shoot. The formation of a purplish-red tint in leaves due to phosphorus (P) deficiency is another distinguishing effect of zinc poisoning. 5 In vascular plants, increased zinc concentration and low water levels may cause stomatal closure, decreased CO2 uptake, chlorophyll concentration, and oxidative damage resistance systems activation. 6 A high level of zinc produces reactive oxygen species or dislocates other metals from the active sites of proteins. Zinc poisoning causes chlorosis in young leaves due to Zn-induced Mg or Fe shortage.7,8
Evaporation, chemical precipitation, ion exchange, cementation, electrochemical deposition, membrane process, and activated carbon adsorption are some of the most common methods for metal removal. In contrast, these gadgets are typically only partially solicited due to their high investment, inadequacy, or operating expense. As a result, the biosorption approach has gained popularity. The main advantages of this technique (biosorption) include high efficiency, cheap cost, minimal biological and chemical sludge, biosorbent renewal and metal recovery, and no additional dietary requirements.9-12
Biosorption of heavy metals by instinctual microorganisms from soil and water is a precise approach for producing various biosorbents for heavy metal bioremediation. It was previously unknown whether bacteria, fungi, and algae act as biosorbents to reduce heavy metal toxicity in soil. 13 The contrivance for metal elimination via biomass comprises coordination, complexation, ionic interactions, and chelation between ligands and metal cations. Biosorption is both metabolisms independent like chemical or physiological sorption onto the cell wall or dependent on internal compartmentalization, transport, and extracellular precipitation by metabolites. 14 The potential of bacterial strains to acquire resistance to heavy metals could help with soil fertility and wastewater management, where microbes are involved in biological processes such as organic matter decomposition. 15
The biosorption process is primarily employed to treat wastewater because various metals are present there. The presence of one metal influences the removal of other metal ions. For example, in a solution, biomass like bacteria, yeasts and fungi cannot uptake uranium when cobalt, manganese, copper, mercury, cadmium, and lead are present. On the other hand, Zn2+ and Fe2+ are stimulators for uranium uptake by
In order to well recognition of the complex procedure of biosorption, the studies directing the cell surface structural design, identification of functional groups and acid-base features of the biomass (Biosorbent) are essential for forecasting the metal biosorption behavior and then amending the biosorption property of the metal. Since the metal hazards to microorganisms, plants, and, accordingly to human health, recognize the biosorbing capacity of microorganisms, experiments were performed to illustrate the bacterial strain and estimate the Zn biosorption capability of the strain RY12,
Materials and Methods
Soil Sampling and Culture Conditions
Fifty soil samples (50) were collected from various fields near the industrial area of Faisalabad (Pakistan), irrigated with wastewater from factories. Soil samples (10 g) were suspended in 90 mL of distilled water and serially diluted. Nutrient agar (Oxoid, UK) was prepared, sterilized at 121°C, and at 15-pound pressure for 20 minutes (Samheung, Korea). Nutrient agar was aseptically poured into the sterilized petri plates. Soil samples were inoculated onto the nutrient agar plates and incubated at 30 oC for 5 days. After 5 days, colonies with different morphology were selected. Fifty bacterial isolates were selected and purified by repeat streaks on nutrient agar plates. 17 The 28 isolates were then selected for further study. These bacteria were stored at −80°C in 20% of glycerol.
Determination of Minimum Inhibitory Concentration of Zinc for Isolated Bacteria
The minimum inhibitory concentration (MIC) of the Zn for each isolate was determined by the plate dilution method.18,19 Briefly, the zinc salt ZnSO4.7H2O in varying concentrations ranging from .5 mM to 35 mM (65.38 mg/L = 1 mM) was used to determine MIC. Nutrient agar (Oxoid, UK) with different zinc concentrations was prepared, sterilized at 121°C, 15-pound pressure for 20 minutes (Samheung, Korea). After sterilization, nutrient agar was poured into the autoclaved petri plates and placed on a smooth surface under a laminar airflow cabinet (Thermo Scientific™, UK) to become solid. When the agar became solid, bacterial strains were inoculated with a sterilized inoculating loop. The plates were incubated at 28–30°C for 24 hours in the incubator (Binder, Germany). The zinc MIC against the strain was determined by the lowest concentration of the metal that inhibited microbial growth.
Determination of Bacterial Tolerance to Oher Heavy Metals
The selected bacterial isolate was streaked on Tryptic Soy Agar (TSA) plates with various concentrations of other heavy metals (Mn, Co, Pb, Ag, Cu, Hg, and Cr), ranging from 1 mM to 20 mM, to determine the bacteria’s tolerance for these metals. The plates were incubated at 28–30°C for 5–6 days and observed the growth of bacteria daily. 20
Identification and Biochemical Characterization of Zinc-Resistant Bacteria RY12
Bacterial isolate (RY12) with maximum zinc tolerance was selected from a pool of bacterial isolates. It was identified based on culture characteristics, colony morphology, Gram staining, pigment production, motility, and biochemical tests. Gram staining and spore staining were done, and the results were examined under the microscope. Biochemical tests on motility, catalase, indole production, oxidase, urease, and citrate utilization were performed to identify the strains.21,22
Molecular and Phylogenetic Characters of Zinc-Resistant Bacteria RY12
For this purpose, crude DNA of the selected isolates was extracted following the method. 23 The 16S rRNA gene sequencing and ribotyping were done to characterize identified bacteria by amplifying the gene. Total genomic DNA was extracted by the CTAB method. 24 Polymerase chain reaction (PCR) was used for the amplification of 16S rRNA using 16S rRNA PCR primers, PA (5′-AGAGTTTGATCCTGGCTCAG-3′, and PH (5′-AAGGAGGTGATCCAGCCGCA-3′). 25 The isolate was grown in Luria–Bertani (LB) broth for ribotyping, and total genomic DNA was extracted. PCR products were eluted using a gel extraction kit (Fermentas, Germany) and sent for commercial sequencing (Eurofins MWG Operon LLC, USA). After amplification, 16S rRNA sequences were compared with known sequences in the GenBank database. NCBI blast analysis was used to search for similar sequences, and the phylogenetic tree was constructed using partial 16S rDNA gene sequences through the computer software (MEGA v7.0). 26
Effect of Zinc on the Growth of Strain RY12
The RY12 strain was grown in a nutrient broth medium supplemented with a 1 mM concentration of zinc and incubated at 28°C (constant pH 8.0) for 72 hours. After that, the broth was centrifuged at 10 000 rpm for 10 minutes. Optical density (OD) was measured at different time intervals (6, 12, 18, 24, 30, 36, 42, 48, 54, 60, and 72) hours. 27
Accumulation and Removal of Zinc Metal by Biosorbent (RY12)
A highly zinc-resistant bacterial strain with optimal growth was selected with steady increases in zinc concentrations up to 20 mM or more. Biosorption of zinc was assessed by letting the selected bacterial isolate interact with Zn in MSM broth at pH 7.2. One mL of culture was inoculated in round bottom flasks from overnight grown bacteria (108 CFU/mL) containing 50 mL of sterilized nutrient broth with 500 mgkg−1 of zinc salt (ZnSO4), a soluble form of zinc salt. Flasks were incubated at 30°C for 24 h on shaking. After incubation, the bacterial culture was centrifuged at 10,000 g for 10 min.
28
The zinc content in the supernatant was examined by using atomic absorption spectrophotometer (Perkin Elmer A Analyst 400, USA). The bacterial pellets were stored at 4°C for further study. The percentage of Zn removal by the biosorbents from the culture was calculated by the difference between the initial Zn content in the culture media and sampling. A bacterial strain with high biosorption potential was selected (Figure 1 and 2). A schematic illustration shows how the Growth of bacteria on nutrient agar plate isolated from soil.

The biosorption efficiency of bacterial strain, A %, of the Zn (II) ion was calculated by the equation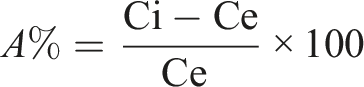
Ci is the initial, and Ce is the equilibrium Zn (II) metal ion concentrations.
Fourier Transforms Infrared Spectroscopy
Fourier transforms infrared spectroscopy was performed to confirm the biosorption of zinc by the bacterial strains. 29
Preparation of Samples
Highly zinc-resistant bacterial strains were grown in nutrient broth (Oxoid, UK) with .1% zinc salt (ZnSO4.7H2O) and without zinc used as controls. Nutrient broth (Oxoid, UK) was prepared and supplemented with zinc, autoclaved at 121°C, 15-pound pressure for 20 minutes (Samheung, Korea). Bacterial colonies were inoculated into the sterilized nutrient broth on the flasks. The flasks were kept at 30°C for 24 hours at 150 rpm, shaking in a rotator shaker (Thermo Scientific™, UK). After 24 hours, flasks were taken out of the shaker and centrifuged test and controlled at 10 000 g (Thermo Scientific™, UK) for 10 minutes. Discarded the supernatant and stored the pellet for FT-IR.
Procedure
The functional groups of the selected strain were analyzed with a Fourier transform infrared spectroscopy (FTIR) spectrophotometer that was a pyroelectric infrared detector (Perkin Elmer Spectrum Two™ equipped with lithium tantalite (LiTa03). 2 mg of the dry mass of the control strains (without zinc) and the test strains (with zinc) were ground with 200 mg dry KBr powder (1:100) in a mortar. The mixture was hard-pressed to prepare translucent sample disks by pressure bench press. The discs were then instantly analyzed within the range of 450-4000 cm−1. Spectrum 10 version 10.3.06 software was used to obtain FTIR spectra.
Statistical Analysis
All statistical analysis was performed using Graph pad Prism version 6.01. All results in this paper were presented as the mean with standard error. The significance of differences was determined by one-way and two-way analysis of variance (ANOVA) with (
Results
Isolation and Determination of Minimum Inhibitory Concentration of Zinc-Tolerant Bacteria
Minimum Inhibitory Concentration of Zinc for Isolated Bacteria.
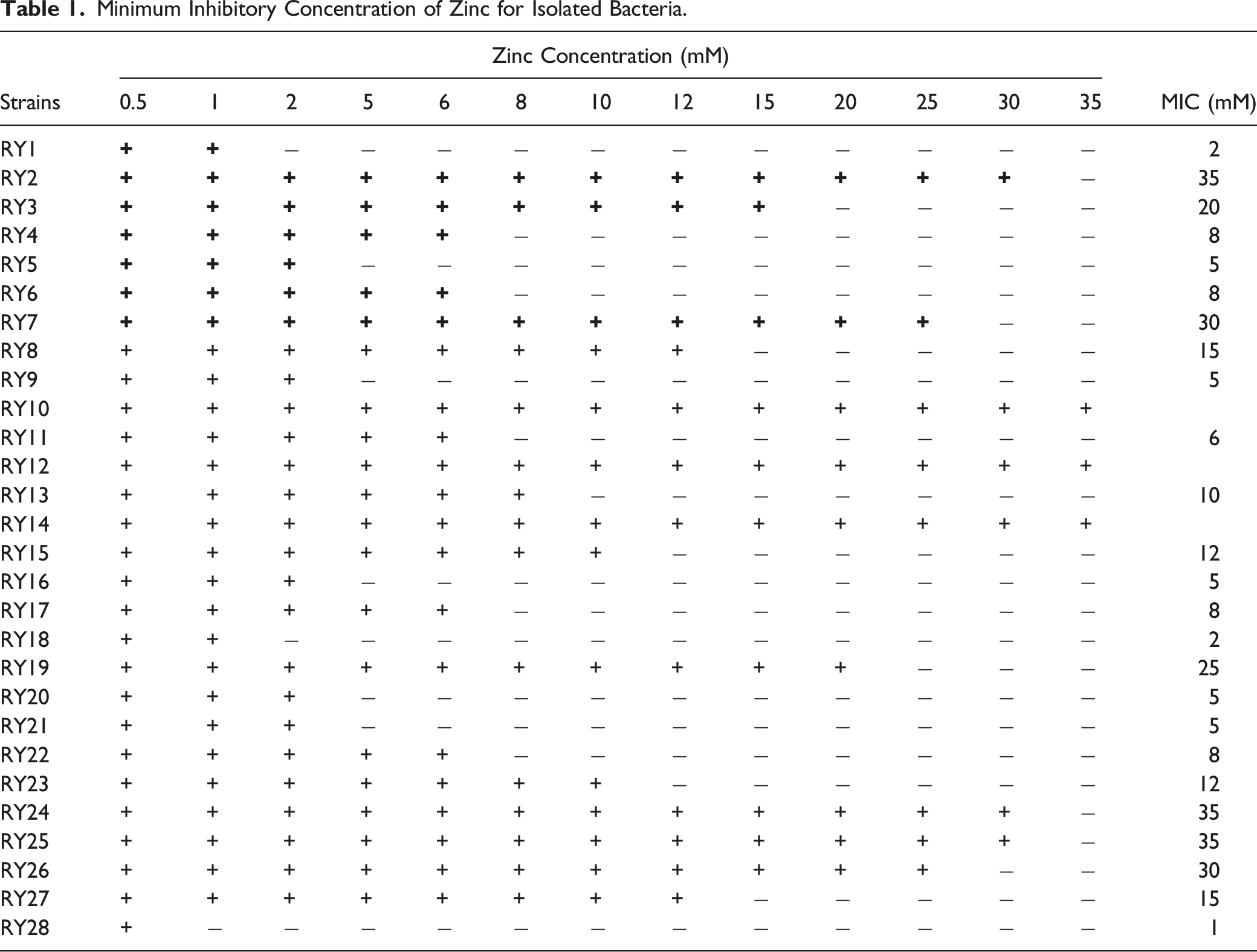
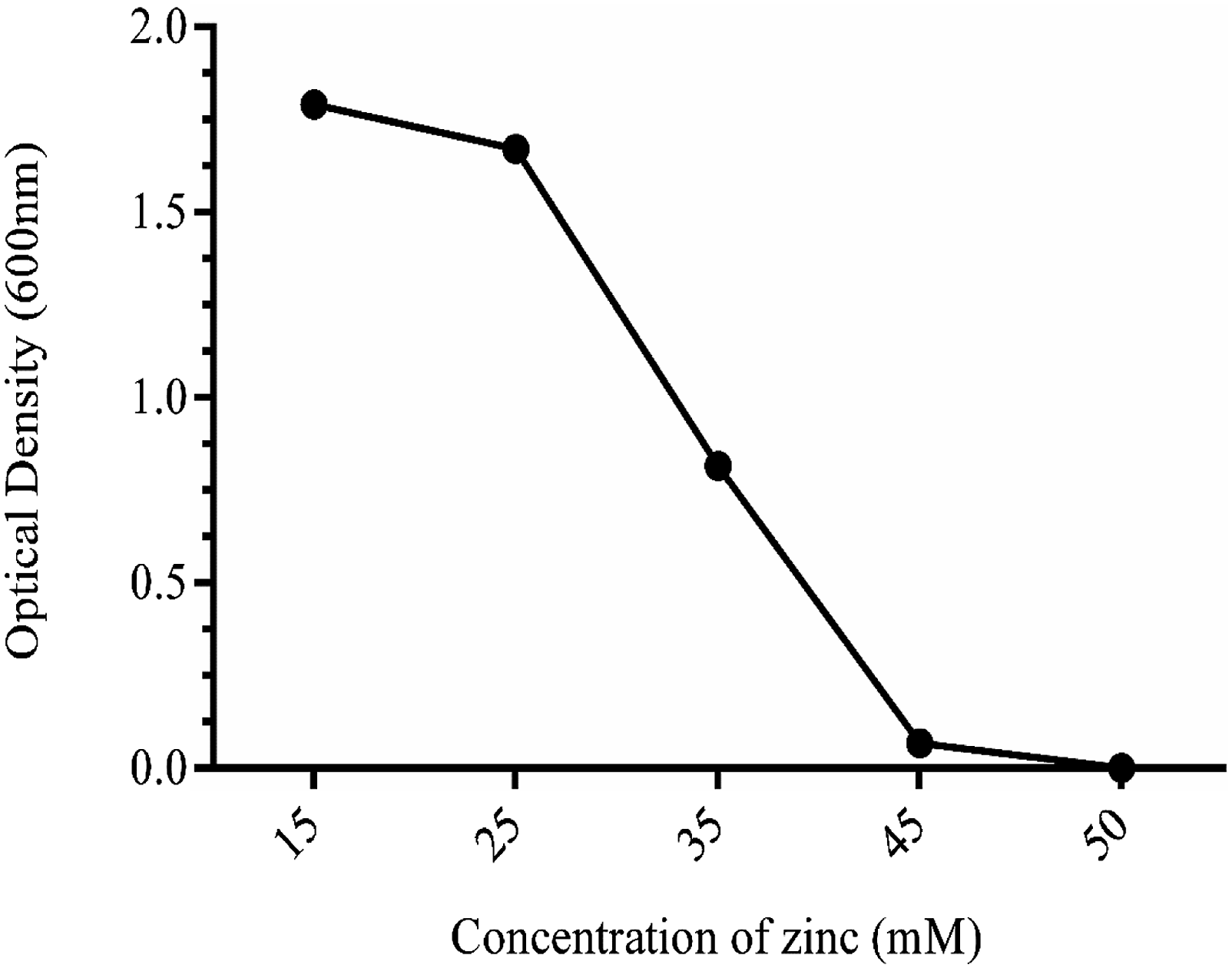
Effect of different concentrations of (Zn2+) on the growth of

Effect of (Zn2+) on the growth of bacterium
Cross Metal Tolerance of Zinc Tolerant Bacterial Isolate RY12.

Identification of Selected Zinc Tolerant Strain RY12
After screening the potential of zinc tolerance, the RY12 strain was selected, having the maximum capability of zinc resistance. RY12 showed light yellow, round, pigmented colonies on nutrient agar plates. Gram staining and spore staining revealed that the strain was Gram-negative and non-spore-forming rods.
Biochemical Characterization of Selected Strain RY12
Morphological and Biochemical Characters of Selected Bacterial Isolate RY12.

Molecular and Phylogenetic Characterization of Selected Strain RY12
Molecular characterization of the selected isolate was carried out using 16S rRNA gene amplification. The bands appeared at 1500 bp. The strain RY12 was identified as Neighbor-joining linear phylogenetic tree of 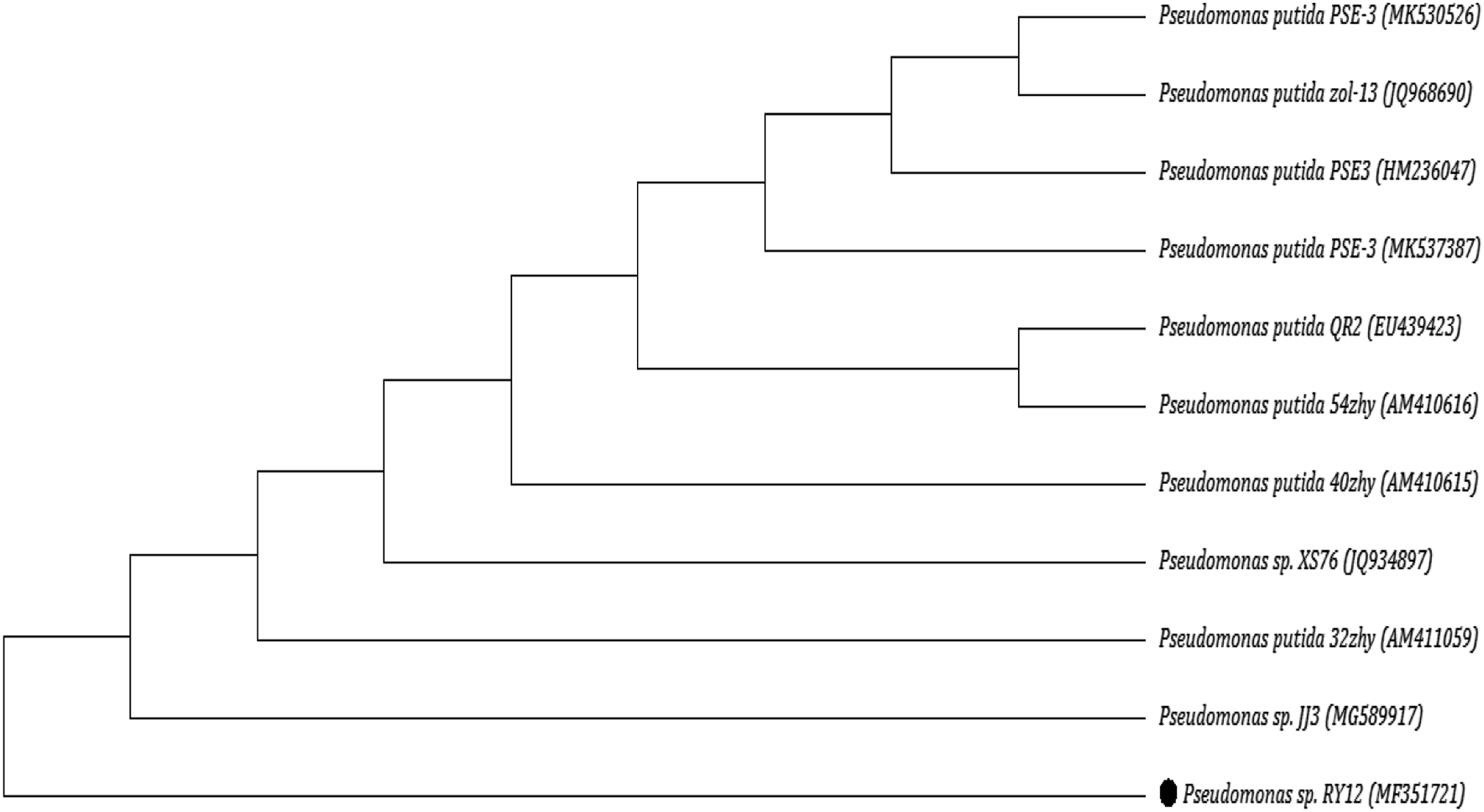
Confirmation of Zn Biosorption by the Biosorbent RY12
After every 0, 2, 5, 10, and 15 days, the samples were centrifuged at 10 000 rpm for 10 minutes. The supernatant was taken to check the decrease in zinc level with the help of atomic absorption spectrophotometer. Statistical analysis by two-way ANOVA revealed that there was a significant (P < .0001) decrease in zinc level with strain RY12 that was 150.9 mg/L, 134.80 mg/L, 99.99 mg/L, 5.13 mg/L & 4.12 mg/L at 0, 2, 5,10, and 15 days of incubation respectively (Figure 6). The change in zinc level was obtained from the difference between the negative control (Broth without strain RY12) and the sample (Broth with strain RY12). With the increase in the incubation period, zinc concentration decreased in broth treated with RY12, while there was no significant change in negative control with the increase in incubation period. Zinc biosorption potential of bacterial strain 
FT-IR Analysis of Strain Pseudomonas sp. RY12
Fourier Transform Infrared Spectroscopy confirmed the biosorption of zinc. The FT-IR is the most useful analytical technique for identifying functional groups. FT-IR analysis was carried out to confirm the difference between the functional groups in relation to biosorption of zinc in bacteria grown in the presence of zinc and control bacteria grown without metal. There was a huge change in the absorption spectra of control and metal-loaded bacterial mass. The infra-red spectroscopic analysis confirmed the active role of various functional groups, including amino, carboxyl, hydroxyl, and phosphate moieties, in metal chelation. The peaks at wavenumber 769 cm−1 recognized as O=C-N, 1033 cm−1 stretching of C-OP bond, 1394 cm−1 symmetric stretching of C=O, 1455 cm−1 corresponds to bending of C-H bond in CH2 and CH3 groups, 1542 cm−1 asymmetric stretching of COO-group, N-H bending and C-N stretching in amide (amide II band), 1627 cm−1 stretching C=O in amides (amide I band), 2927 cm−1 asymmetric stretching of CH2 group and 3275 cm−1 asymmetric and symmetric stretching of O-H group. Comparison of bacterial envelope after treatment with zinc clearly depicts peak shifting at 765 cm−1/769 cm−1 (O=C-N group), 1033 cm−1/1050 cm−1 (C-OP group), 1372 cm−1/1394 cm−1 (C=O group), 1437 cm−1/1455 cm−1 (C-H group), 1541 cm−1/1542 cm−1 (C=O group), and 2921 cm−1/2927 cm−1 (C-H group). Moreover, alterations in wave numbers 1627 and 3275 represent the importance of the amide group and hydroxyl group in metal chelation (Figure 7). FT-IR spectrum produced by 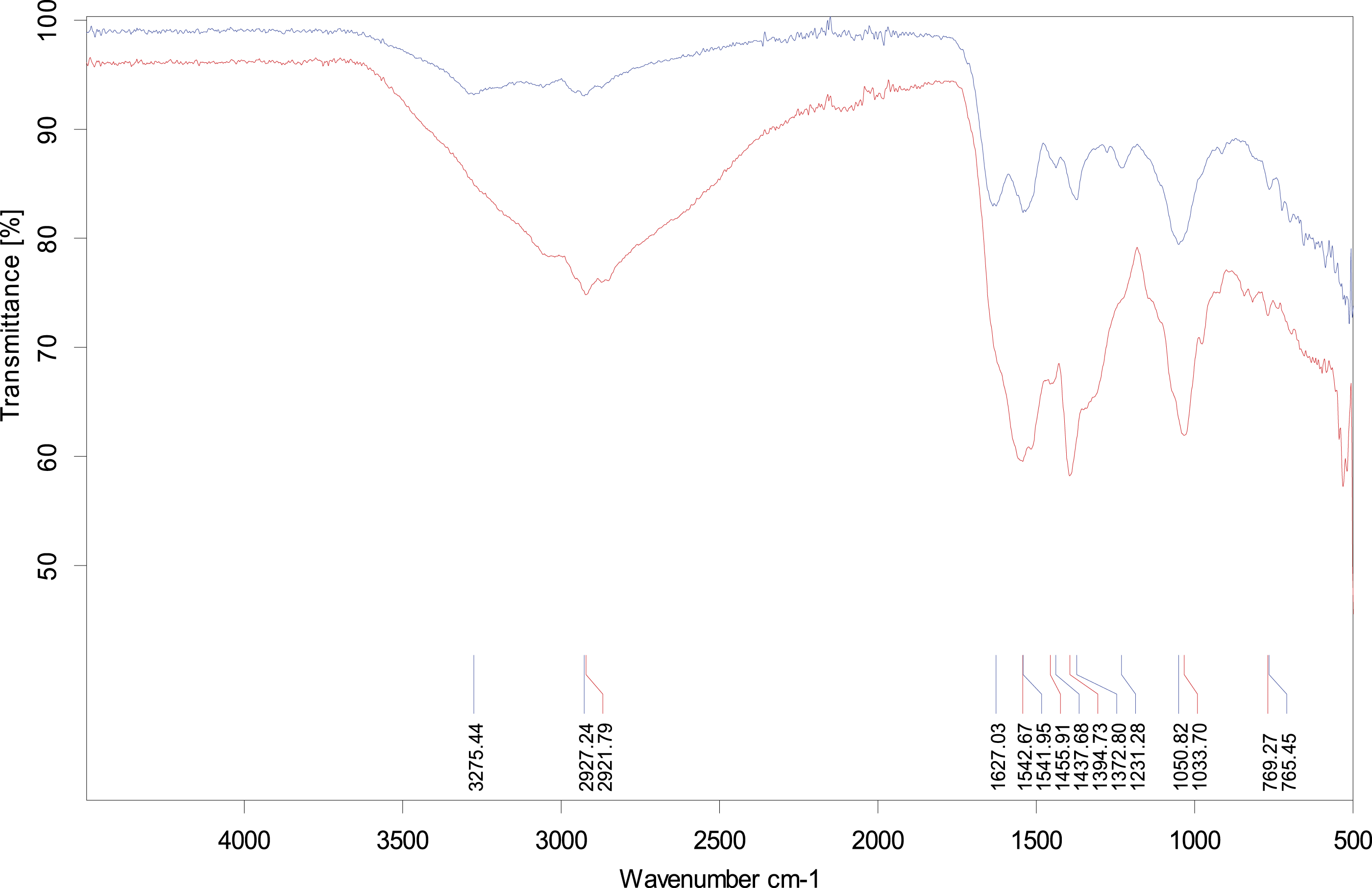
Blue: Untreated (without zinc) cells
Red: Treated (with zinc) cells
Discussion
The proper wastewater treatment not only accomplishes the hassles for agriculture irrigation but also decreases the environmental pollution and protects the public health. Still, industrial wastewater treatment is a well-built engineering science that requires numerous practices and procedures to treat wastewater 2 proficiently.
For that purpose, 50 soil samples were collected from different areas of district Faisalabad, Pakistan, near the industrial areas. Malik et al 30 collected soil samples from various localities, including parks, roadsides, and agricultural and industrial areas of the Sialkot district. In comparison, Mahmood and Malik 31 collected samples from different regions in Lahore, Pakistan. Twenty-eight samples were selected after bacterial growth, and the MIC of these strains was estimated in the present study. Out of 28 bacterial strains, 10 strains having maximum ability to grow under zinc stress at 35 mM were selected.
Islam et al
32
selected
As previously reported in various researches throughout the world, the growth rate of
In some cases, biosorption of these heavy metals by the biosorbents reaches within one hour.
57
Similar results were shown by Tariq et al
38
where
Fourier-Transformed Infrared (FT-IR) spectrum analysis was performed to record the purified EPS and the chemical binding of Zn2+. The uptake of zinc metal is due to the functional groups present in the bacterial cell wall and metal chemistry. The cell walls with primary functional groups like phosphate, carboxyl, and amine are capable of zinc sorption and deprotonated at high pH. 60 The dominance and alteration in the peak intensity ranged from 1370 to 1394 cm−1 denoted the existence of carbon and phosphorous-containing oxygen atoms and directed their interaction with zinc ions. 61 It gives the impression of bacterial cell oxidation in zinc biosorption that resulted in changes in the overall IR spectrum. 62 The functional groups in bacterial strains have previously been stated to interact with metals in cationic form. Metal ions coordinate with peptide bonds with either carboxyl or amino side chains or oxygen and nitrogen. 63 In the current study, a shift in wavenumber from 2927 cm−1 asymmetric stretching of the CH2 group and 3275 cm−1 asymmetric and symmetric stretching of the O-H group represented membrane proteins' contribution in zinc binding. 64 The speedy reduction of divalent zinc might be endorsed to surface binding because of several functional groups on the bacterial cell wall and internal diffusion (bioaccumulation), as previously reported.65,66
The results of this study are in line with the findings of Ngabura67, who assumed that amino groups might have a chief role in zinc binding with the cell membrane, as they were the main components of carbohydrates, protein, and hexosamine existing in cell membranes. Additionally, a similar altered FTIR spectrum was observed for zinc-treated cells, proposing the development of zinc complexes with membranous proteins.
68
These alterations in the FTIR spectrum of
Conclusion
The current study showed that bacteria thrive in contaminated environments which may keep agriculture and environment friendly talent even under heavy metal stress. Heavy metal resistance ability of these bacteria makes them possible to survive in a polluted environment. In present study, Pseudomonas sp. RY12 is used to bioremediate Zn heavy metal from agricultural soil polluted with the heavy metal-contaminated water and this indigenous bacterial stain has biosorbent potential towards Zn heavy metal. Finally, this potent strain can be used to bioremediate the heavy metal-contaminated agricultural soils and to achieve the eco-friendly, low-cost goals of sustainable agriculture.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
