Abstract
Research aimed to develop and evaluate biodegradable, pH-responsive chemically cross-linked Pluronic F127 co-poly- (acrylic acid) nanogels for dermal delivery of Terbinafine HCL (TBH) to increase its permeability and as a new approach to treat skin fungal infections. TBH-loaded nanogels were successfully synthesized from acrylic acid (AA) and Pluronic F127 by free-radical copolymerization technique using N,N′-methylene bisacrylamide (MBA) as crosslinker and ammonium persulphate (APS) as initiator. Prepared nanogels exhibited 93.51% drug entrapment efficiency (DEE), 45 nm particle size, pH-dependent swelling and release behavior. Nanogels were characterized using different physicochemical techniques. The ex-vivo skin retention studies through rat skin showed about 42.34% drug retention from nanogels while 1% Lamisil cream (marketed product) showed about 26.56% drug retention. Moreover, skin irritation studies showed that nanogels were not irritating. Nanogels showed improved in-vitro antifungal activity against Candida albicans compared to commercial product. In-vivo studies on rats infected with Candida albicans confirmed superiority of nanogels over 1% Lamisil for eradication of fungal infection. This confirms that TBH loaded in Pluronic F127 co-poly-(acrylic acid) nanogels provided greater targetibility and cure rates of poorly soluble TBH in animal model and hence nanogels could be a potential carrier for effective topical delivery of TBH for skin fungal infection treatment.
Introduction
Keratin rich structures such as nails, skin, and hairs are infected by dermatophytes causing a superficial infection known as dermatophytosis. Due to increasing trends of human fungal infections, these diseases currently have become a worldwide public health issue. 1 Topical treatment of skin fungal infection is preferred over systemic therapy as medication is directly delivered to the infection place resulting in more patient compliance with reduced side effects. 2 TBH is an orally and topically active allylamine derivative that specifically inhibits fungal squalene epoxidase to treat various types of fungal skin infections. 3 Systemic treatment with TBH causes severe side effects such as nausea, vomiting, diarrhea, stomach pain, and hepatotoxicity. Conventional creams and gels used to treat skin fungal infections can avoid side effects, but poor solubility of TBH (both in water and lipids) makes it ineffective to cross the stratum corneum layer of skin. Therefore, it requires a long-term treatment regimen which can result in drug resistance and skin keratinization. 4 Therefore, a nanoparticulate targeted drug delivery system is needed to develop to enhance the stratum corneum penetration of TBH as a new approach to treat fungal infections. It has been reported that polymeric nanosystems like nanogels efficiently deliver cargo at lesion site, thus minimizing systemic toxicity. 5 Nanogels are nanosized three-dimensional aqueous dispersed hydrogel particles, fabricated by physical-chemical crosslinking of the polymer and therefore have the attributes of hydrogel and nanoparticles at the same time. Existence of crosslinks allows nanogels to absorb large amount of water while maintaining structural integrity without undergoing dissolution. The swelling/deswelling property makes nanogels a promising candidate for number of applications. 6 Nanogels are used in dermal and cosmetic products due to their biocompatibility, fine spreading, prolong residence time at site of application and competence of approaching smallest capillary through paracellular or transcellular route with improved intracellular penetration. 7
pH-responsive gels have attracted considerable interest as smart drug delivery systems because of their ability to deliver drug molecules at specific site with improved efficacy. Different monomers and polymers have been employed in nanogel drug delivery systems for site-specific drug release, However, AA, also known as carbomer, because of its biocompatibility and biodegradability is particularly important. It is widely used in fabrication of polymeric gels because it substantially swells in aqueous media, therefore categorized as a superabsorbent polymeric material. Enhanced electrostatic repulsion between ionized carboxylic groups of AA causes swelling of gel. pH and ionic strength of medium are important factors that effects equilibrium swelling of polymeric gel in a medium.8,9 In pluronic family, Pluronic F 127, is the most widely used water-soluble synthetic polymer consisting of poly (ethylene oxide) PEO and poly (propylene) PPO units with 12 600 molecular weight. Sol–gel transformation occurs when it is heated to body temperature at concentrations of more than 20%.10,11 In cosmetic industry, Poloxamer, especially Pluronic F 127 based hydrogels are very promising in wound healing. 12
Keeping in view the above facts, objective of present research work was to develop and optimize Pluronic F127/AA nanogels for topical delivery. Pluronic F127 co-poly-(acrylic acid) gels are biocompatible as both Pluronic F 127 and AA are Food and drug administration (FDA) approved pharmaceutical ingredient and food additive.7,13 The potential of pH-sensitive Pluronic F127 co-poly- (acrylic acid) nanogels for topical delivery of TBH was investigated through in-vitro and in-vivo evaluation including skin retention studies.
Material and Method
Materials
All ingredients used during study were of analytical grade. Pluronic F127 (mol. wt. 12 600 g/mol), AA (mol. wt. 73.06 g/mol) were purchased from Sigma-Aldrich GmbH Merck, Germany. MBA (mol. wt. 154.17 g/mol) and APS (mol. wt. 228.18 g/mol) used as crosslinker and initiator, were purchased from Sigma-Aldrich GmbH, Darmstadt Germany. Sabouraud dextrose agar (SDA) and Hydroxypropylmethylcellulose (HPMC) were supplied by Thermofisher Scientific Waltham, MA, USA, and Sigma-Aldrich GmbH, Darmstadt, Germany, respectively. TBH was kindly gifted by Saffron Pharmaceuticals (Pvt) Ltd, Pakistan.
Methods
Synthesis of nanogels
Feed scheme of Pluronic F127 co-poly-(acrylic acid) nanogels.

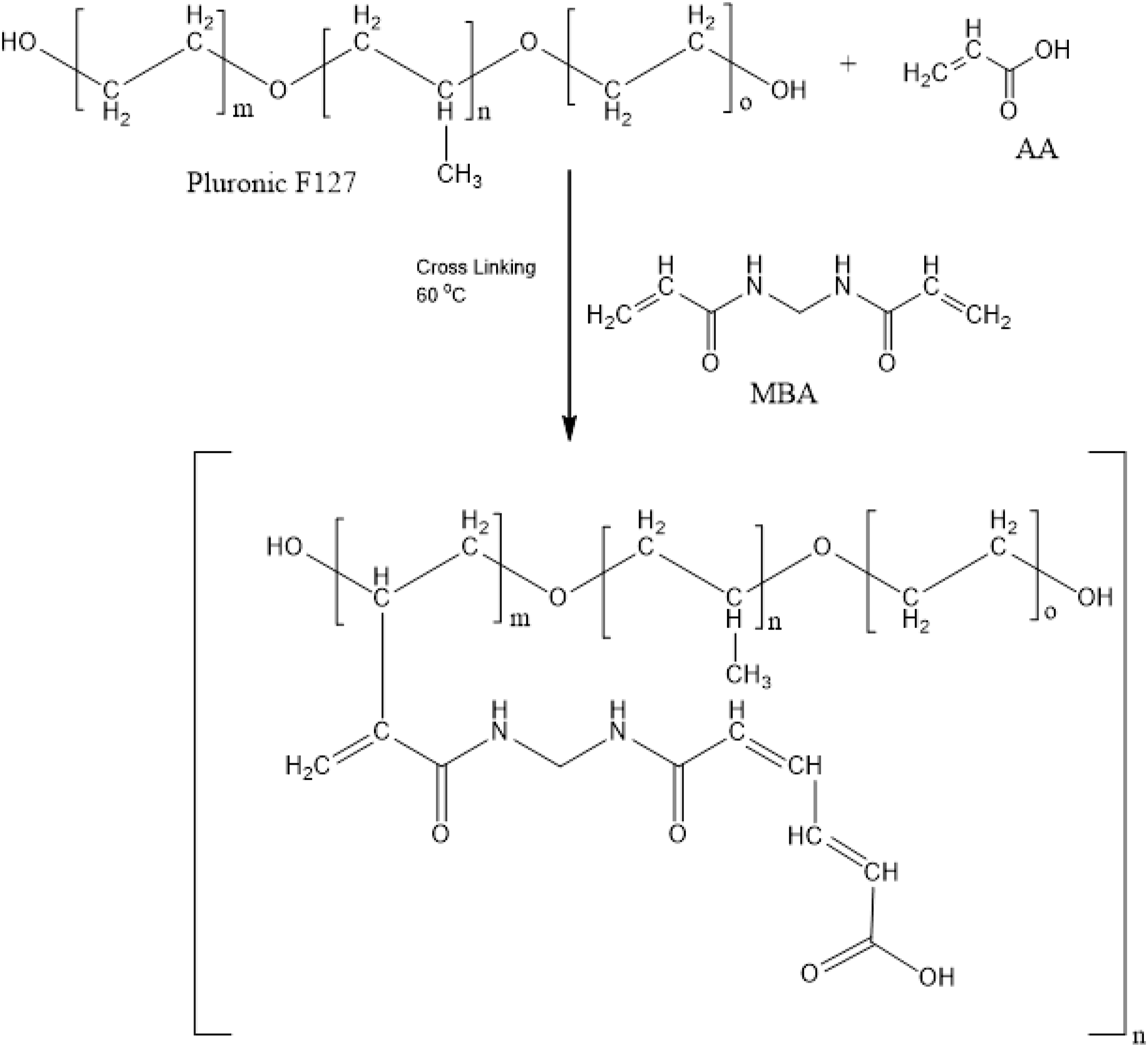
Proposed structure of Pluronic F127 co-poly- (acrylic acid) nanogels.
Drug loading
Drug was loaded in developed nanogels by swelling diffusion method. First, TBH solution was prepared using water: ethanol mixture. Calculated amount of dried nanogels were added into drug solution, sonicated for 15 min, and then placed on magnetic stirrer at 500 r/min for 72 h. After 72 h, drug-loaded nanogels were subjected to lyophilization. 16 TBH-loaded nanogels were then physically and chemically characterized.
Preparation of HPMC gel
Bioadhesive 1% HPMC gel was prepared to produce semisolid system in order to spread drug-loaded nanogel particles onto the skin. HPMC was selected as a gelling agent because of its ability to produce gel with larger pore size with minimal diffusional restriction for the TBH.17,18 Accurately weighed amount of HPMC was add into water and stirred for 60 min at 1000 r/min. Gel pH was adjusted to 5.5 with tri-ethanol-amine (TEA).
Characterization
Particle Size Analysis
Particle size of prepared particles suspended in .22 μm filtered water was checked by using Malvern Zeta Sizer ZEN, UK, utilizing DLS technique. 19
Fourier Transform Infrared Spectroscopy
Fourier transform infrared spectroscopy (FTIR) spectra of pure TBH, Pluronic F127, unloaded and drug loaded nanogels for functional group determination and to confirm any interaction among the formulation ingredients were recorded with FTIR spectrophotometer (NICOLET 380) using attenuated total reflectance (ATR) technology over the scanning range of 4000–500 cm-1. 20
Thermogravimetric Analysis
Thermogravimetric analysis (TGA) of pure ingredients and nanogels were conducted using Thermogravimetric analyzer TA Q600 series, USA to determine thermal stability. Specific amount of sample (3-5 mg) was loaded on platinum pan and analyzed under nitrogen flow rate of 10 mL/min with heating rate of 10 ºC/min in the range of 0°C to 800°C to find out percent decrease in weight with increase of temperature. 21
Powder X-ray Diffraction
Powder X-ray diffraction (PXRD) analysis of pure ingredients and optimized formulations were carried out to determine the crystalline and amorphous structure using X-ray Diffractometer JDX3522, Japan. XRD patterns were measured in scan range of 0–50 with diffraction angle 2 (θ) using monochromatic wavelength of 1.540 Å and Cu Kα radiation source. 22
Scanning Electron Microscopy
Scanning electron microscopy (SEM) of unloaded polymeric nanogels was carried out to evaluate the surface morphology and apparent shape. Samples were staged on clear metal stub with double-adhesive tape, coated with gold, and visualized under SEM. 23
In vitro Swelling Studies
Swelling studies of all nanogels were performed by placing known amount of lyophilized nanogel particles in dialysis membrane (molecular weight cut-off 14 000) which was then dipped into a phosphate buffer solution of pH 1.2 and 7.4, allowed to swell, removed from swelling media at predetermined time, blotted with Whatmann’s filter paper, and weighed again. This procedure was repeated until constant weight of nanogels was achieved.
24
Swelling index was determined by using equation (1). 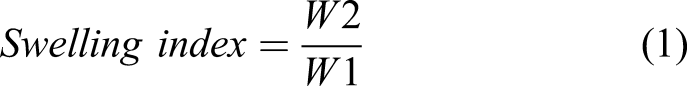
Drug Entrapment Efficiency
Absorption and extraction method was used to determine (DEE) of prepared nanogels at room temperature.
25
Weighed quantity of drug loaded nanogels were added into water and ethanol (1:1) mixture, stirred at 100 r/min for 24 h to obtain drug release. Mixture was then centrifuged at 6000 r/min, supernatant layer was removed and resulting mixture was filtered through .45 μm membrane filter. Filtrate was finally analyzed using double beam UV spectrophotometer at λmax 283 nm. DEE was calculated using following equation
In vitro Drug Release Studies
Drug release study for developed nanogels and commercially available product was performed using open-ended cylindrical glass tube type Diffusion Cell system in phosphate buffers of pH 5 and 7.4. Specified amount of samples were uniformly spread on previously soaked cellophane membrane surface and tied on 1 end of tube. Complete assembly was placed in beaker in such a manner sample containing part of tube slightly submerged into diffusion medium. Diffusion medium temperature was kept 32 ± .5°C and agitated using magnet at 50 r/min. Samples were drawn at selected time intervals and replaced with an equal volume of fresh medium. The release of drug (TBH) was quantified using UV spectrophotometer at specific wavelength of λmax at 283 nm. 26
Ex-Vivo Skin Penetration and Retention Studies
In vitro drug penetration studies were conducted using Franz diffusion cell having diameter of 2 cm and an effective permeation area of 3.14 cm2. Albino rats weighing between 150 and 180 g were sacrificed by giving an overdose of chloroform. Hair from dorsal side of rats was removed using hair removal (veet) cream; shaven part of skin was excised utilizing scissors and forceps, and finally, subcutaneous fat was surgically removed. Skin was washed with 7.4 pH phosphate buffers solution, cut into appropriate sizes, and then kept in a freezer at −20°C until used within 14 days. Skin was fixed between donor and receptor compartment of Franz diffusion cell with the stratum corneum facing towards donor compartment and skin dermal side positioned towards receptor compartment. Receptor compartment was filled with 9 mL phosphate buffer of pH 7.4 set at 32 ± .5°C with constant stirring using magnetic stirrer. Prepared nanogel formulation and commercial product (Lamisil cream) were applied on excised skin. After various intervals of time, samples were taken from receptor compartment via sampling port and each time immediately replaced with buffer. Samples were analyzed using UV spectrophotometer at 283 nm.
At completion of diffusion experiment skin surface was wiped with diffusion medium. Wiped diffusion medium combined, filtered and analyzed spectrophotometrically at 283 nm. Drug deposited into skin was also determined after chopping skin into small pieces. All pieces were kept in centrifuge tube containing methanol as drug extraction solvent. Then it was subjected to sonication to extract TBH present in skin to solvent. Finally centrifuged, supernatant collected, filtered and analyzed spectrophotometrically at 283 nm. 27 Experimental procedure for the ex vivo studies was reviewed and approved by the Institutional Pharmacy Animal Ethics Committee, Faculty of Pharmacy, Government College University Faisalabad, Faisalabad, Pakistan.
Antifungal Activity Studies
Cup plate diffusion method was adopted to compare antifungal activity of nanogels and market product (Lamisil cream) against Candida albicans. Clinical isolates of Candida albicans were subcultured on the SDA medium and Petri dishes were incubated at 30°C for 5 days. Initially cultured Candida albicans was identified by examining carefully colonial morphology, then VITEK-2R system (BioM´erieux, Marcy l’Etoile, France) also identified challenged Candida albicans. 28 Few fungal colonies were transferred to sterile saline solution and mixed by using vortex mixture for 1 min. Mixture was allowed to stand for 15 minutes for fungus to settle down. Inoculum was prepared from supernatant and turbidity was adjusted to .5 McFarland standard (106 CFU/mL) employing a hemocytometer. One mL of inoculum was streaked over the surface of solidified SDA medium with the help of sterile cotton swab. Using cork borer, 2 wells were bored into the media plates. Appropriate dilutions pipetted from stock solutions of Lamisil cream and nanogel formulation prepared in dimethyl sulfoxide (DMSO) and distilled water, respectively, were added into wells. The plates were incubated at 30°C ± .1°C for 5 days and antifungal activity expressed as the mean zone of inhibition was measured. Experiment was performed in a sterile area. 29
Skin Irritancy Studies
Skin irritation tendency of optimized formulation was carried out by Draize scoring method. Albino rats with weight (150–180 g) were randomly divided into 3 groups each with 3 animals. Hair on dorsal side of animals was cleared with an electric trimmer 1 day before the study. Group 1 was kept as negative control (no application). Group 2 was treated with standard irritant 1% formalin solution as positive control and Group 3 received optimized formulation. After 24 h rats were evaluated for any signs of erythema and odema and were graded on scale of 0–4.30,31
In vivo Studies
Preparation of immunosuppressed animals
Albino rats weighing 150–180 g were used for skin fungal infection cure rate study. Experiment protocol was reviewed and approved by Institutional Pharmacy Animal Ethics Committee, Faculty of Pharmacy, Government College University Faisalabad, Pakistan. One week before start of experiment, animals were allowed to acclimatize under standard laboratory conditions and provided with healthy diet and water. Immunosuppression of rats was done by administering them intravenous methylprednisolone injection for 3 consecutive days to develop a heavy cutaneous infection.
Preparation of fungal inoculum
Inoculum was prepared from recent Candida albicans cultures. Candida albicans was allowed to grow on SDA medium and incubated at 30°C for period of 5 days. After incubation, 3–5 colonies were moved to test tube containing sterile saline (.9%) and mixed using vortex mixer. To prepare inocula, fungal mixture turbidity was adjusted to concentration of 107 CFU/mL.
Induction of fungal Infection
Hair was removed from animals back by using veet hair removal cream 1 day before getting infected. Skin was disinfected with ethyl alcohol and specific quantity of 107 CFU/mL of Candida albicans intradermally injected to each animal in center of naked skin. After injection, swollen area was rubbed till the disappearance of slight edema formed. After 5 days, fungal infection was confirmed on skin.
Experimental design
Four animals groups were formed and each group had 6 rats. Group 1 received no treatment and functioned as negative control (healthy rats). Group 2, after getting fungal infection received no treatment and was kept as positive control. Group 3 animals were treated with the Lamisil cream 1%, and Group 4 animals received TBH loaded nanogel formulation, topically for 10 days. All animals in Group 3 and Group 4 were treated once daily. The therapeutic efficacy of the Lamisil cream and nanogel formulation was accessed on daily basis by macroscopic examination of fungal infection.2,32
Results and Discussion
Particle Size Analysis
Pluronic F127-co-poly- (acrylic acid) nanogels were examined for particle size and zeta potential using Laser Light Scattering Particle Size Analyzer (Malvern Zeta Sizer Nano ZS, Malvern, UK). Zeta sizer curve is shown in Figure 2. Average particle size as depicted by peak was recorded 45.13 nm, which is considered suitable for skin application.
33
Poly dispersity index (PDI) value was found to be .137 and narrow PDI value indicates less aptitude to form clusters. Particle size measurement of nanogels.
Fourier Transform Infrared Spectroscopy
FTIR spectrum of pure Pluronic F127, AA, drug free nanogels, drug and drug loaded nanogel were recorded are shown in Figure 3(a)-(e), respectively. IR spectra of pure Pluronic F 127 are shown in Figure 3(a). Spectrum showed a principle absorption peaks at 2890 cm−1 (C–H stretching), 1340 cm−1 (C–H bending), 1470 cm−1(C–C stretching), and 1100 cm−1 (C–O stretching), respectively.
34
FTIR spectrum of AA Figure 3(b) exhibited stretching vibrations of methylene group at 2973 cm−1. Peak at 1706 cm−1 indicated presence of carboxylic groups. Peaks at 1635 cm−1 correspond to C=O group while peak at peak at 1296 cm−1 presented C–C group. Characteristics peak encountered at 1173 cm−1 giving evidence of carboxylic acid.
35
Figure 3(c) shows that above mentioned peaks were shifted in FTIR spectrum of placebo nanogels due to electrostatic interaction among functional groups of Pluronic F 127 and AA. Peak due to C–H stretching vibrations of Pluronic F 127 at 2890 cm−1 and peak due to stretching vibration of (–CH2) of AA at 2973 cm−1 was overlapped at 2880 cm−1 in placebo nanogel. Peak due to C–C stretching of aromatic ring of Pluronic F 127 at 1470 cm−1 was also shifted to 1460 cm−1 in unloaded nanogels and C–O stretching of ester linkage at 1100 cm−1 was transferred to 1110 cm−1. Similarly, 1650 cm−1 and 1160 cm−1 peaks of nanogel was due to slight shifting of C=O (1635 cm−1) and carboxylic acid (1173 cm−1) stretching vibrations of AA respectively. As shown in Figure 3(d), TBH showed characteristic peaks observed at 1408 cm−1 and 1384 cm−1 for C–N and CH3 groups, respectively. Peak at 2983 cm−1 was assigned to stretching of aliphatic C–H group while peak displayed at 3043 cm−1 indicated aromatic C–H groups.
36
Infrared spectrum of TBH loaded nanogels Figure 3(e) exhibited peaks of TBH at 1390 cm−1 and 1392 cm−1 for C–N and CH3, respectively, and C–H stretching at 2970 cm−1 that is nearly unchanged. This indicated that there was no chemical interaction between TBH and polymer. FTIR of (a) Pluronic F127 (b) AA (c) unloaded nanogels (d) TBH (e) drug loaded nanogels.
Thermogravimetric Analysis
TGA was conducted to investigate the thermal stability of drug, Pluronic F127, unloaded and drug-loaded nanogels within a temperature range from 0 to 800°C. TGA thermogram of Pluronic F127 showed four-stage weight loss started at around 93°C and counted for 19% weight loss till it reached 239°C. Second stage started at 239°C and last up to 441°C, counted for 44% weight loss. During third and fourth stages 19% and 16% weight was lost at temperatures 445°C and 514°C, respectively. Many researchers found that Pluronic F 127 degradation occur between 300°C to 400°C.
37
TBH showed 11% weight loss until it reached 195°C and major degradation of TBH occurred between 200°C and 400°C and near 654°C there is 88% weight loss. TGA profile of drug -free PF127 co-poly- (acrylic acid) nanogel showed an initial weight loss of 20% in temperature range of 117°C to 274°C, indicating more stability over increased temperature range of formed cross-linked network than individual components. Next stage started at around 274°C to 344°C with weight loss of 15%. Near 58% thermal degradation of gel occurred between 344°C to 601°C. A further loss started at 601°C and continued till total degradation of drug free nanogel. TBH loaded nanogel showed 12% weight loss until 137°C. Next stage started at 137°C and counted for 63% weight loss till it reached 317°C. Further degradation happened between 317°C and 549°C, shown in Figure 4 TGA Thermographs of (a) Pluronic F127, (b) TBH (c) unloaded nanogels (d) drug loaded nanogels.
Powder X-ray Diffraction
X-ray diffraction study of pure Pluronic F127, drug, unloaded, and drug loaded nanogels was conducted to determine their crystalline/amorphous nature which effects physical and mechanical properties. XRD map of pure Pluronic F127 exhibited 2 sharp peaks at (2θ) 15° and 17°, representing crystalline nature of polymer. XRD diffractogram of TBH presented multiple sharp peaks at (2θ) 8°, 21°, 25°, 26°, 28°, and 32°, thus confirming crystalline nature of drug.
38
XRD analysis of unloaded nanogels formulation indicated highly amorphous nature of the system as no characteristic sharp peaks of Pluronic F 127 were present as shown in Figure 5(c), However in TBH-loaded nanogel, all of the corresponding peaks were present but with much lower intensity giving impression of the conversion of crystalline components into amorphous form and establishing successful loading of drug in developed system as shown in Figure 5(d) https://www.tandfonline.com/doi/full/10.1080/25740881.2021.1934019?casa_token=oELrtEXhPT8AAAAA%3AFk5laEkKuCvCQ3_xBf4wrt-a0UwUrFc7qZJnn1Ka4zSSZzXHGMLFFJpgoBXCBKzv8JpUzZhngpicsfw. It can be concluded that prepared nanogel due to its amorphous nature, effectively camouflaged the crystalline nature of individual components because of crosslinking network without any interaction. XRD spectra of (a) Pluronic F127 (b) TBH (c) unloaded nanogels (d) loaded nanogels.
Scanning Electron Microscopy
Developed nanogels were analyzed for their surface morphology by SEM. SEM micrographs of nanogels are shown in Figure 6. Surface of nanogels appeared porous and spongy. Numerous pores present in the system favor the entrance of solvent upon contact, enhanced entrapment and release of the drug from system. Previously, Kifayat At al. found similar results who prepared PEG-4000-Co-Poly (AMPS) nanogels for solubility enhancement of meloxicam. SEM micrographs of Pluronic F127 co-poly-(acrylic acid) nanogels at different magnifications.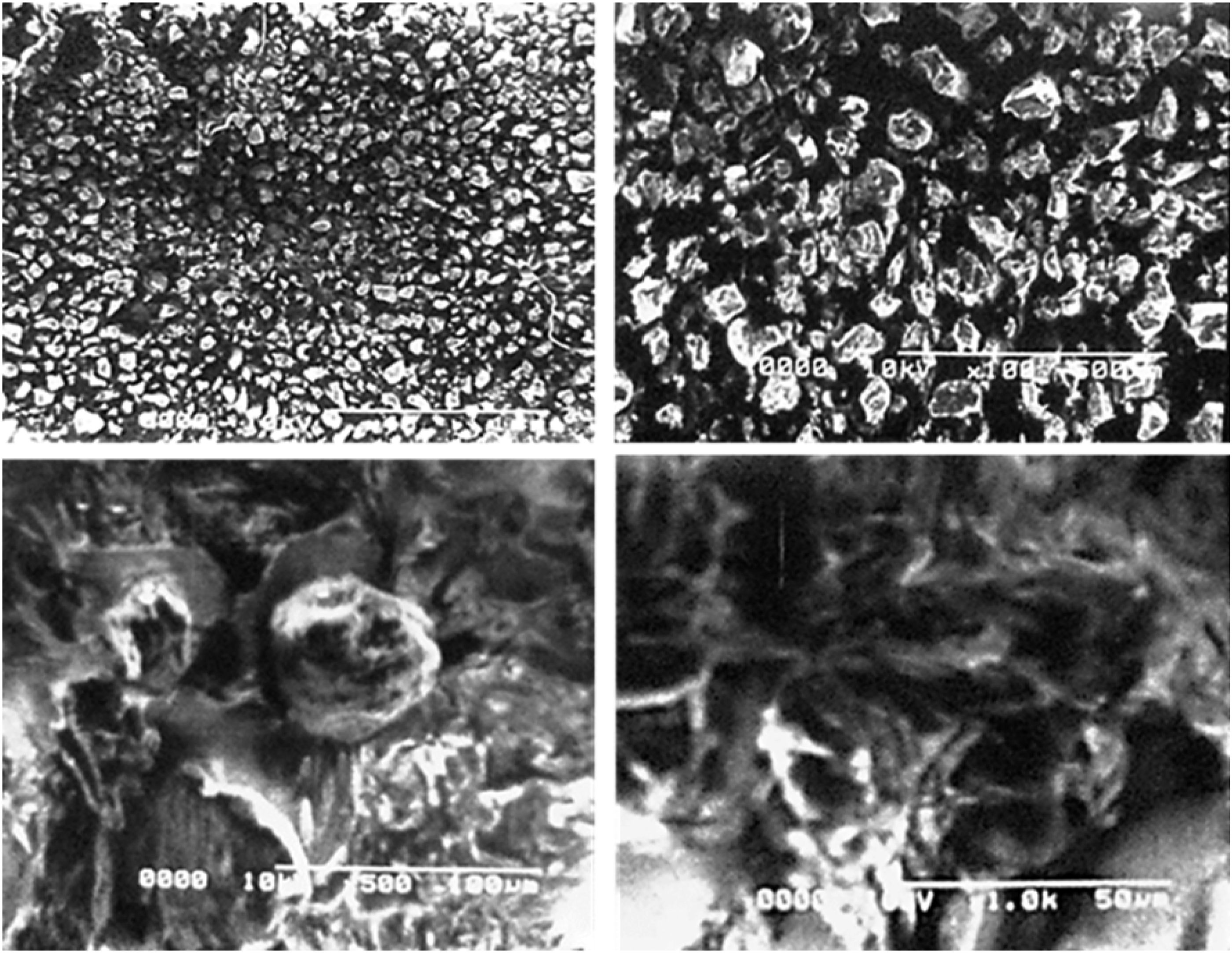
In vitro Swelling Studies
Figure 7 Swelling behavior is a critical attribute of nanogels as it imparts its effect on drug release rate and can be modified by changing amount of nanogels ingredients used. Dynamic swelling index of prepared nanogels was evaluated at pH 1.2 and 7.4. pH has substantial effect on swelling kinetics of nanogels. Interaction among ether functional group of Pluronic F 127 and carboxylic functional groups of AA was accountable for swelling of developed nanogels. Swelling index was increased with an increase in pH of buffer solution from 1.2 to 7.4 due to ionization of COOH groups of AA. When pH of buffer solution was increased than that of pKa of COOH, availability of ionized carboxylic groups increased, resulting in electrostatic repulsion forces between COO− increased osmatic pressure leading to expansion of network structure. Study results revealed that formulations with higher Pluronic F 127 content showed lower swelling. This is due to formation of 3D polymer network construction with less in number and size of porous channels for buffer due to inter-polymer complex formation between Pluronic F 127 and AA. Higher AA contents in formulation amplified swelling of nanogels due to higher concentration of carboxylic acid groups available in reaction system.39,40 Higher concentration of MBA reduced the swelling property. This is because cross-linker promotes cross-linking density of polymeric matrix with hydrophobic character that inhibits buffer penetration.41,42 (a) Effect of pH 1.2 and 7.4 on dynamic swelling
Drug Entrapment Efficiency
Drug loaded nanogels (F1-F9) with different compositions of constituents as shown in Figure 8 were evaluated to determine encapsulation efficiency. Entrapment efficiency ranged from 58.13 to 93.51%. After swelling of polymeric system at high pH, drug got entrapped. Results revealed that entrapment efficiency was increased with gradual increase in the monomer contents and decreased with increase in polymer contents. Similar results were observed for swelling index phenomena. Thus, when swelling and porosity are enhanced, it resulted in a higher percentage DEE. While MBA being used as cross-linker decreases the percentage of DEE when used in high concentration due to formation of compact and dense network structure with smaller mesh size.39,43 DEE of different nanogel formulations.
In-Vitro Drug Release Studies
Figure 9 (a) and (b) showed comparative In vitro drug release of all developed nanogels and marketed product carried out at pH 5 and 7.4 to simulate the skin and fungal infection conditions. . In all nanogels, % drug release was higher at pH 7.4 which corresponds to COOH group of AA. Due to pH-dependent nature of acrylic acid (pKa = ∼4), at pH 7.4 more COOH groups of AA was ionized than at pH 5, that increased electrostatic repulsion among negatively charged COOH groups and developed osmotic pressure on nanogels that resulted in enhanced porosity of gel matrix and drug release. More the swelling of nanogels, more will be the drug release.44-46 Nanogels formulations containing high amount of AA in their composition showed higher drug release rate due to abundance of negatively charged COO−groups in buffer medium as showed in Figure 5(d). Figure 5(c) showed contrary relation between Pluronic 127 concentration and release of drug from nanogels because of maintenance of strong intermolecular interaction of Pluronic F127 and AA, less space available for drug release and also less amount of buffer penetrated into nanogels.
47
Cross-linker (MBA) influences drug release from nanogels as cross-linking density of polymeric network increases or decreases with change in concentration of cross linker. Drug release from nanogels was decreased with increase in MBA concentration as showed in Figure 5(e). Drug release from marketed product was slower at both pH when compared with nanogels at all-time points. (a) Percent drug release from nanogels and marketed product at pH 5 (a) and pH 7.4 (b) (c) Effect of Pluronic F 127 (d) AA (e) MBA wt% on drug release.
Ex-Vivo Skin Penetration and Retention Studies
Purpose of study was to investigate skin penetration and retention of selected nanogel formulation in comparison with the commercial cream Lamisil using Franz diffusion cell. Amount of TBH permeated through skin was 6.57% as compared to 13.45% from Lamisil as shown in Figure 10. After 12 h, 42.34% and 26.56% of drug were left in skin from nanogels and Lamisil, respectively. Increased dermal retention of TBH was associated with increased interaction of hydrating nanogels with the stratum corneum components, modifying its hydration level, creating channels that allowed enhanced skin penetration.
48
Hence, it can be claimed that higher TBH retention in skin was largely due to carrier (nanogels), their highly elastic and deformable particles properties. Little drug permeation through skin in case of nanogels than Lamisil also hinted nanogel skin targeting property, which is needed for effective fungal infection treatment. By developing Polymeric nanogels, drug molecules can be delivered at specific sites with improved efficacy and less side effects. Now, it can be concluded that nanogel drug delivery system may have significant potential for topical treatment of various skin diseases.
49
Ex vivo drug skin penetration/retention comparison (1) drug penetrated in receptor compartment (2) deposited in skin layers (3) drug left over skin.
Antifungal Activity Studies
Antifungal activity of optimized nanogels against Candida albicans was evaluated in comparison to commercial product Lamisil cream 1% using cup plate method. Antifungal potential screening was measured with regard to breadth of zone of inhibition. Figure 11 showed outcomes of antifungal activity; it was observed that breadth of zone of inhibition of optimized nanogels (35 mm) was more than Lamisil cream 1% (25 mm), which may be due to smaller particle size of developed nanogels enhancing its penetration into fungal cells, accumulating squalene and becoming deficient in ergosterol, a key component of fungal cell membranes. Higher intracellular squalene concentration leads to fungal cell membranes lysis and cell death. This study showed that TBH-loaded nanogels could improve the in vitro antifungal effect on Candida albicans. It is concluded that lower TBH doses will be required for Candida albicans infection treatment when drug is loaded in nanogels, which could decrease incidence and severity of adverse effects and better patient compliance that can lead to improved therapeutic outcome.
26
Antifungal activity showing breadth of zone of inhibition for developed formulation (F) and marketed product (M) Lamisil cream 1%.
Skin Irritancy Studies
Skin irritation study results on rats measured after 24 h.

Erythema scale: 0 is none, 1 is slight, 2 is well defined, 3 is moderate, 4 is scar formation
Edema scale: 0 is none, 1 is slight, 2 is well defined, 3 is moderate and 4 is severe
In Vivo Pharmacodynamic Studies
In vivo antifungal activity of optimized formulation in comparison with the Lamisil cream was evaluated against most widely used fungus Candida albicans. All rats before getting infected, exhibited typical normal skin missing any sign of skin infection, for example, redness, swelling, skin rupturing, or color change, as shown in Figure 12. Group 1 which served as negative control did not show any cutaneous candida growth while animals of Group 2, 3, and 4 after development of fungal infection showed yellowish or purple patches, swelling, scaling, and skin splitting. Inflammation, edema in Group 3 rats were healed after treatment with Lamisil cream 1%, but some yellowish scars were still not cured. Oppositely, optimized nanogel treated (Group 4) animals showed normal skin lacking inflammation and yellowish marks, as represented in Figure 12. Skin of rats before fungal infection induction, after Candida albicans fungal infection induction and after infection treatment.
In vivo study results showed that Group 4 rats treated with TBH-loaded nanogel formulation showed better pharmacodynamic activity than Group 3 rats treated with commercial product Lamisil. This might be because of the enhanced TBH deposition into skin layers.2,50
Conclusion
In current study, nanogels were synthesized using different ratio of AA, Pluronic F127, and MBA by free-radical polymerization technique in order to improve physicochemical characteristics of TBH for its topical application against skin fungal diseases. Fabricated nanogels showed high DEE, nano particle size, pH-dependent swelling, and drug release behavior. FTIR and SEM confirmed the successful loading of TBH in amorphous nanogels without any interactions, while TGA scans indicated higher thermal stability. The absence of any sign of skin erythema, odema and inflammation during skin irritancy studies indicated safety and biocompatibility of nanogels. Ex vivo skin retention studies exhibited targeting properties of nanogels as compared to 1% Lamisil cream. TBH loaded nanogels showed better in vitro and in vivo antifungal activity against Candida albicans than commercial product. From this study, we concluded that hydrophobic drugs like TBH can be successfully incorporated and formulated as nanogels to overcome the permeability and efficacy problems.
Footnotes
Acknowledgments
The authors are thankful to the Government College University Faisalabad, Pakistan, for providing the research facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
