Abstract
The aim is to explore the protective effects of Puerarin on radiation-induced vascular endothelial cell damage and its underlying mechanism. The apoptosis and DNA damage of Human umbilical vascular endothelial cells (HUVECs) exposed to radiation alone or in combination with glucose in the exposed group were significantly elevated (P < .05) compared with those in the control group. The Puerarin-treated HUVECs showed significant reduction in the radiation-induced apoptosis and DNA damage (P < .05). Furthermore, X-ray irradiation significantly increased the expression of miR-34a, which was reversed by pre-treatment with Puerarin. Placental Growth Factor (PLGF) was a target gene of miR-34a. The expression of PLGF in the peripheral blood of patients receiving radiotherapy significantly increased with an increase in the cumulative dose of radiation (P < .05), after which it began to decrease at the fourth week (P < .05) and then remained at a low level until the end of radiotherapy. Puerarin exerts a radioprotective effect by decreasing DNA damage and apoptosis through miR-34a-targeted PLGF.
Introduction
Cardiovascular complications induced by chest radiotherapy have been described since as early as the 1960s. 1 In recent years, cardiovascular diseases caused by radiation have received great attention with the development of radiotherapy. Although technological advances have reduced the doses for radiotherapy and number of radiological examinations, small doses of radiation still induce cardiovascular injures. 2 Studies have shown that radiation-induced cardiovascular diseases are mainly caused by ionizing radiation-induced vascular endothelial cell injury. Such dysfunction triggers a series of inflammatory and oxidative stress reactions, causes density and diameter decrease of small vessels and micro-vessels, and leads to insufficient blood supply to tissues and organs, finally resulting in cardiovascular complications. 3 It is critical to develop effective drugs which can reduce radiation-induced damage to vascular endothelium, and can prevent and treat cardiovascular disease after radiation.
Studies have shown that ionizing radiation causes deregulation of microRNAs, which play an important role in tumor resistance to radiotherapy and toxicity to normal tissues.4-6 Increased expression of miR-34a following ionizing radiation exposure has been reported. 7 The miR-34a, a downstream gene of p53, regulates the cell cycle, apoptosis, and differentiation by targeting different target genes. 8 Elevated miR-34a has been reported to aggravate DNA damage and promote cell apoptosis. 9 Our previous study showed that miR-34a is involved in DNA damage repair and apoptosis of vascular endothelial cells.10,11 Among the many target genes of miR-34a, placental growth factor (PLGF) Placental Growth Factor has attracted our interest. PLGF, a member of the vascular endothelial growth factor (VEGF) family, is expressed in the placenta at different stages of pregnancy and is detected in the heart, lungs, thymus, thyroid, and skeletal muscles. Resting vascular endothelial cells secrete a small amount of PLGF. However, upon activation, they secrete a large amount of PLGF. After entering the circulatory system, PLGF may regulate vascular endothelial growth and vascular remodeling via autocrine or paracrine mechanisms. 12 Currently, PLGF is believed to have broad prospects for the treatment of ischemic heart disease. On one hand, PLGF has a structure and functions similar to those of VEGF and enhances the angiogenic effects of VEGF. On the other hand, unlike VEGF, PLGF has no side effects on angiogenesis because it has no osmotic stimulation effect on endothelial cells. 13 However, there are relatively fewer studies on the cardiovascular role of PLGF and its underlying mechanism. We hypothesize that miR-34a stimulates the proliferation of vascular endothelial cells and regulates DNA repair and apoptosis of these cells by targeting PLGF.
Puerarin is an active ingredient that is extracted from the traditional Chinese medicine Ge-gen. It is widely used in clinical practice due to its diverse biological functions. At present, Puerarin is mainly used to treat cardiovascular and cerebrovascular diseases, diabetes, osteonecrosis, Parkinson’s disease, Alzheimer’s disease, endometriosis, and cancer. 14 Current studies have found that Puerarin exerts anti-oxidative stress and plays an anti-apoptotic role in regulating a variety of biologically active molecules and some signaling pathways.15-17 There are few reports on the role of Puerarin played in radiation-induced damage. Previous studies have showed that Puerarin effectively alleviates radiation-induced reduction of peripheral red blood cells and white blood cells, and improves the thymus and spleen indices. Moreover, Puerarin increases the activity of superoxide dismutase (SOD) and decreases the content of malondialdehyde (MDA), a product of membrane lipid peroxidation, thereby exhibiting its protective effect on ionizing radiation-induced damage in rats. 18 However, the molecular mechanism underlying the protective role of Puerarin against radiation remains unclear.
Given that Puerarin has diverse biological functions and miR-34a acts on its target genes to regulate physiological and pathological processes, we hypothesize that there is a link between Puerarin and miR-34a, ie, Puerarin may regulate the expression of miR-34a in endothelial cells exposed to radiation.
Materials and Methods
Cell Culture
Human umbilical vascular endothelial cells (HUVECs) were purchased from the Institute of Cell Biology, Chinese Academy of Sciences, and cultured in high glucose DMEM (Wisent Bio, China) supplemented with 10% FBS (Biological Industries, Israel). The cells were cultured at 37°C in plastic dishes in the CO2 incubator. When the confluency reached 80%, the cells were passaged, on average, every 2 days. The cells were washed in PBS and digested using .25% trypsin (Hyclone, USA). When the cells became rounded, they were transferred to other plastic dishes and cultured at 37°C in CO2 incubator.
Radiation
Human umbilical vascular endothelial cells were cultured in plastic dishes at 37°C in CO2 incubator. After the cells were attached to the wall, they were divided into 4 groups: the control group (control), the irradiation group (R), the irradiation plus glucose group (R + G) and the irradiation plus Pueraria group (R + P). The latter 3 groups were exposed to χ-ray with a total dose of 10 Gy (Philips SL18 medical X-ray accelerator, Berkshire). After irradiation, Puerarin glucose solution was immediately added to the medium (the R + P group), and the same volume of glucose solution was added in the R + G group.
Patients
Four patients were enrolled in this study, including 2 males and 2 females, with an average age of sixty three years. Since one patient was lost to follow-up, the actual number of subjects was three. Two of them suffered from esophageal squamous cell carcinoma and received a total radiotherapy dose of 50 Gy and 60 Gy, respectively. One patient had mediastinal poorly differentiated squamous cell carcinoma with a total radiotherapy dose of 66 Gy. Blood was collected before radiotherapy, at week 2, week 4 after radiotherapy, and at the end of radiotherapy. All patients were given written informed consent, and approved by the ethics committee of Soochow University.
Cell Counting Kit-8 Assay
HUVECs were seeded in ninety six-well plates at densities of 3 × 103 cells and 5 × 103 cells per well and cultured in an incubator at 37°C for 12 h. The cells were divided into 7 groups and treated with Puerarin at 0, 10, 20, 50, 100, 200, and 500 μmol/L. This experiment was conducted with 4 replicates. Cells, at densities of 3 × 103cells and 5 × 103 cells per well, were cultured for 24 h and 48 h, respectively. Then, 10 μL CCK8 reagent was added to each well of volume 100 μL culture medium. After 2 h, the absorbance was measured at 450 nm with a microplate reader.
Annexin V-FITC/PI Double Staining Assay
HUVECs, at a density of 2 × 105cells per well, were seeded in 60-mm dishes and cultured in an incubator at 37°C for 12 h. After adherence, cells were divided into 4 groups and treated with PBS, exposed to irradiation of 10 Gy alone or in combination with glucose or Puerarin. After radiation exposure, 20 μmol/L of Puerarin or an equal amount of glucose was added. After 48 h, the cells were collected, digested by EDTA-free trypsin, and re-suspended in Binding Buffer (.1 mL), and then with Annexin V-FITC (5 μL) and PI Staining Solution (5 μL) for 10 min in the dark. After adding Binding Buffer (.4 mL), apoptosis was detected by flow cytometry in the samples within 1 h.
Western Blot
The total protein was extracted and quantified by the BCA method (Thermo, USA). Protein samples were mixed up with 5 × loading buffer and heated in boiling water for 5 min. Protein (30 μg) was separated by SDS-PAGE and transferred to PVDF membrane (Millipore, USA). The membrane was washed thrice, for 15 min each, in TBST buffer and incubated with 5% skim milk for 1 h. The membrane was incubated with primary antibody against Cleaved caspase-3 (Cell signaling, USA) and γ-H2AX (ab26350, Abcam, USA) at 4°C overnight after being washed again 3 times. The membrane was washed thrice again and incubated with secondary antibody (Multiscience) at room temperature for 60 min. Protein signals were detected using ECL kit (Takara, Japan).
Immunofluorescence
HUVECs, at a density of 2 × 104 cells per well, were seeded in a 6-well plate and divided into 4 groups: control, exposed to irradiation of 10 Gy alone or in combination with glucose or Puerarin. After irradiation, 20 μmol/L of Puerarin or an equal amount of glucose was added. After 48 h, the cells were washed 3 times for 5 min each in PBST buffer, permeabilized in .5% Triton X-100 for 20 min, treated in 3% H2O2 for 20 min, and blocked in 5% goat serum for 30 min at room temperature. The cells were incubated with primary antibody against γ-H2AX (1:500) at 4°C overnight after being washed 3 times in PBST buffer. The membrane was incubated with secondary antibody (Multiscience) at room temperature for 60 min in the dark after being washed 3 times in TBST buffer. After being washed 5 times in PBST buffer, the cells were harvested, incubated with DAPI (Beyotime Biotechnology, China), and observed microscopically to evaluate DNA damage.
Comet Assay
Cells were collected, washed once with iced PBS, and adjusted to a concentration of 1 × 106 cells/ml. To prepare the first layer of the gel, the slide was heated in water at 65°C and covered by NMA (20 μL, .5% or 7%) at 4°C for 10 min (NMA was weighed and placed in a 1.5-ml EP tube and melted at 95°C for 20 min and then at 65°C). To prepare the second layer of the gel, 10 μL of the cell suspension (about 104 cells) was mixed up in 15 μL of .7% LMA (melted at 37°C for at least 20 min), quickly transferred to the first layer of the gel, and cooled at 4°C for 10 min. After gel formation, the cover slip was removed and 15 μL of .7% LMA, heated at 37°C, was added and then cooled down at 4°C.
For cell lysis, the cover slip was removed, and cells were lysed in iced Lysis Buffer (9 mL of Lysis Buffer in 1 mL of DMSO) at 4°C for 90 min. The slides were washed in PBS and then subjected to electrophoresis at 2.5 V and 300 mA for 20 min. The slide was then neutralized 3 times for 10 min each at 4°C in neutralizing buffer. Finally, the cells were stained with PI solution (20 μL) for 10 min in the dark and observed under a confocal microscope.
Expression of miRNA-34a and Placental Growth Factor in Human Umbilical Vascular Endothelial Cells
Total RNA was extracted from the HUVECs using Trizol reagent (Invitrogen), and total microRNAs were extracted using miRNeasy Mini Kit (QIAGEN). The expression levels of miR-34a-5p and PLGF were determined using quantitative Real Time PCR (qRT-PCR) with specific primers and probes (Roche).
Dual Luciferase Report Assay
Briefly, 293T cells were cultured in DMEM containing 10% FBS (Gibco, USA) in a 5% CO2 incubator at 37°C. Oligo synthesis and vector construction were performed by GenePharma. The sequences for the oligos were as follows- NC: UUCUCCGAACGUGUCACGU, hsa-miR-34a-5p: UGGCAGUGUCUUAGCUGGUUGU. These were then constructed into pmirClo vectors. When the confluency reached 80-90%, cells were washed and suspended in D-Hank’s medium for transfection. Opti-MEM I (150 μL, 50 μL/well), vectors, and oligos (120 pmol) were added to a 1.5-ml EP tube. In another EP tube, Opti-MEM I (150 μL, 50 μL/well) and 6 μL of lipo2000 were added. After 5 min, the contents in each tube were mixed up. After 20 min, the mixtures were added to the 24-well plates (100 μL/well, 3 replicates) and cultured for 6 h. The supernatants were removed, and cells were cultured in a fresh medium. After 24 h, cells were lysed and the supernatants were transferred to an EP tube. The activity of Firefly and Renilla luciferase was determined using a multifunctional microplate reader. The relative luciferase activity was calculated using Renilla activity as an internal control.
Enzyme linked immunosorbent assay
The serum PLGF for patients was detected using a commercial enzyme linked immunosorbent assay (ELISA) kit for PLGF (Cloud-Clone) according to the manufacture’s protocol.
Statistical Analysis
Data were analyzed using Stata 13.0. Differences between 2 groups were analyzed using Student’s t-test. The expression levels of miR-34a and PLGF were calculated using ΔΔCT method. All tests were two-sided. Values of P < .05 were considered as statistically significant.
Results
Puerarin Attenuated Viability Suppression of HUVEC Induced by X-Ray
HUVECs were treated with Puerarin at 0, 10, 20, 50, 100, 200, and 500 μmol/L, first for 24 h and then for 48 h. As shown in Figure 1, Puerarin treatment for 24 h significantly decreased the viability of HUVEC at its concentration of 200 or 500 μmol/L (P < .05) (Figure 1A). After 48 h of Puerarin treatment, the viability of HUVECs was significantly decreased at its concentration of 100, 200, or 500 μmol/L (Figure 1B). Cell viability of Human umbilical vascular endothelial cells after Puerarin treatment. (A): The activity of HUVECs after treatment with Puerarin at different concentrations for 24 h. (B): The activity of HUVECs after treatment with Puerarin at different concentrations for 48 h. (C): After radiation, the activity of HUVECs treated with different concentrations of Puerarin for 24 h. (D): After radiation, the activity of HUVECs treated with different concentrations of Puerarin for 48 h.* P < .05; ** P < .01.Note: HUVECs=Human umbilical vascular endothelial cells.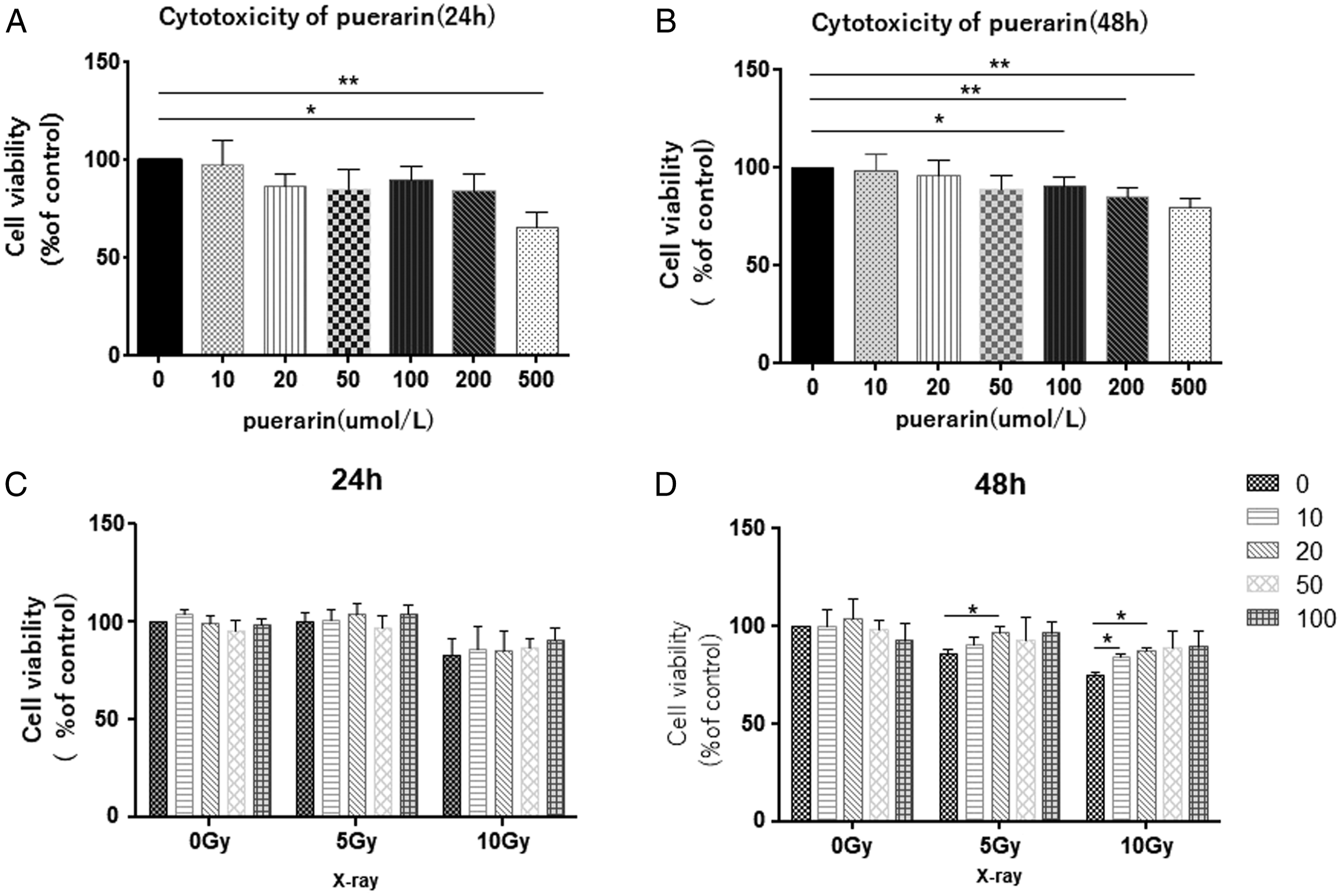
Thus, Puerarin concentrations at 10, 20, 50 and 100 μmol/L were used. HUVECs were exposed to X-ray radiation at 0, 5 and 10 Gy, respectively, and treated with Puerarin at 0, 10, 20, 50 and 100 μmol/L for 24 h or 48 h. As shown in Figure 1, the activity of HUVECs significantly decreased in cells exposed to radiation at 10 Gy for 24 h, but not in cells exposed to radiation at 5 Gy. Treatment with different concentrations of Puerarin had no obvious effects on the viability of cells exposed to radiation at 5 Gy or 10 Gy (Figure 1C). The activity of HUVECs exposed to radiation at 5 Gy or 10 Gy for 48 h significantly decreased. Treatment with Puerarin at 20 μmol/L significantly alleviated the decreased viability of the HUVECs after radiation at 5 Gy (P < .05), while treatment with Puerarin at 10 or 20 μmol/L significantly alleviated the decreased viability of the HUVECs after radiation at 10 Gy (P < .05) (Figure 1D). Therefore, HUVECs were irradiated with 10 Gy X-rays and treated with Puerarin at the dose of 20 μmol/L for 48 h. This was used as the model to study the protective role of Puerarin against X-ray radiation induced damage of endothelial cells.
Puerarin Reduced HUVEC Apoptosis Induced by X-Ray Radiation
Results of apoptosis are shown in Figure 2. After X-ray radiation at 10 Gy, apoptosis of HUVECs exposed to radiation alone (R), exposed to radiation plus .5% glucose (R + G), and exposed to radiation plus Puerarin at 20 μmol/L (R + P) was significantly increased. However, apoptosis was similar in the HUVECs exposed to radiation alone and in combination with glucose. Treatment with Puerarin significantly alleviated radiation-induced apoptosis in HUVECs (P < .05) (Figure 2A and B). Effect of Puerarin on apoptosis of Human umbilical vascular endothelial cells. (A): Flow cytometry is used to detect the apoptosis of HUVECs. (B): Quantitative analysis of apoptosis of HUVECs. (C): The expression of Caspase-3 in HUVECs detected by western blot. “R”: radiation alone, “G”: glucose treatment, “P”: Puerarin treatment, “R + G”: glucose treatment after radiation, “R + P”: Puerarin treatment after radiation.Note: HUVECs=Human umbilical vascular endothelial cells.
Western blot analysis showed that expression of caspase-3 was significantly increased in HUVECs exposed to radiation alone and in combination with glucose. Moreover, treatment with Puerarin significantly reversed radiation-induced elevation of caspase-3 in HUVECs (Figure 2C).
Effect of Puerarin on X-Ray-Induced DNA Damage in HUVECs
Immunofluorescence staining of γ-H2AX foci showed that irradiation obviously aggravated DNA damage manifested by increased γ-H2AX foci (Figure 3). However, treatment with Puerarin decreased radiation-induced elevation of γ-H2AX foci in HUVECs exposed to radiation alone or in combination with glucose (P < .05) (Figure 3A and B). Consistently, western blot showed results similar to those of the immunofluorescence assay Figure 3C. Comet assay is performed to detect DNA damage in Human umbilical vascular endothelial cells. (A): Immunofluorescence staining of γ-H2AX in Human umbilical vascular endothelial cells.. (B): Quantitative analysis of γ-H2AX foci. (C): Western blot is used to detect the expression of γ-H2AX. (D): Comet assay is used to observe the DNA damage of Human umbilical vascular endothelial cells. in different groups. (E): Quantitative analysis of DNA damage in comet assay. Note: HUVECs=Human umbilical vascular endothelial cells.
Comet assay showed that HUVECs exposed to irradiation alone or in combination with glucose exhibited increased DNA tail length and tail area as compared to the cells in the control group (P < .05). However, treatment with Puerarin decreased the increase in DNA tail length and tail area induced by radiation in HUVECs Figure 3D. Statistical analysis using the CASP software showed that the aforementioned results were statistically significant (P < .05) Figure 3E.
Expression of miR-34a and Placental Growth Factor in HUVECs
X-ray radiation increased the expression of miR-34a in HUVECs (P < .05). The expression level of miR-34a was dramatically increased in HUVECs exposed to radiation alone or in combination with glucose compared with that of the control group (P < .05) and Puerarin treatment significantly reduced it (P < .05) Figure 4A). X-ray radiation increased the mRNA expression of PLGF in HUVECs (P<.05) as compared with that in the control group. Puerarin treatment further increased the mRNA expression of PLGF in HUVECs exposed to radiation alone or in combination with glucose (P < .05) Figure 4B. Western blot showed that the protein expression of PLGF in HUVECs was elevated after radiation treatment, which further increased after Puerarin treatment Figure 4C. miR-34a and Placental Growth Factor expression in Human umbilical vascular endothelial cells.. (A): miR-34a expression in HUVEC in different groups. (B): PLGF mRNA expression in HUVECs in different groups. (C): PLGF protein expression in HUVECs in different groups. Note: HUVECs=Human umbilical vascular endothelial cells.
miRNA-34a-Targeted Placental Growth Factor
PicTar software identified that the 3’UTR of PLGF had a 6-bp region that could bind to miR-34. The 3’UTR of PLGF was mutated to construct a luciferase vector that could carry the mutant PLGF (Figure 5A). Dual luciferase reporter assay verifies that PLGF is a target of miR-34a. (A): miR-34a and its binding and mutant sites on PLGF. (B): Luciferase reporter assay reveals the link between miR-34a and PLGF. Note: PLGF=Placental Growth Factor.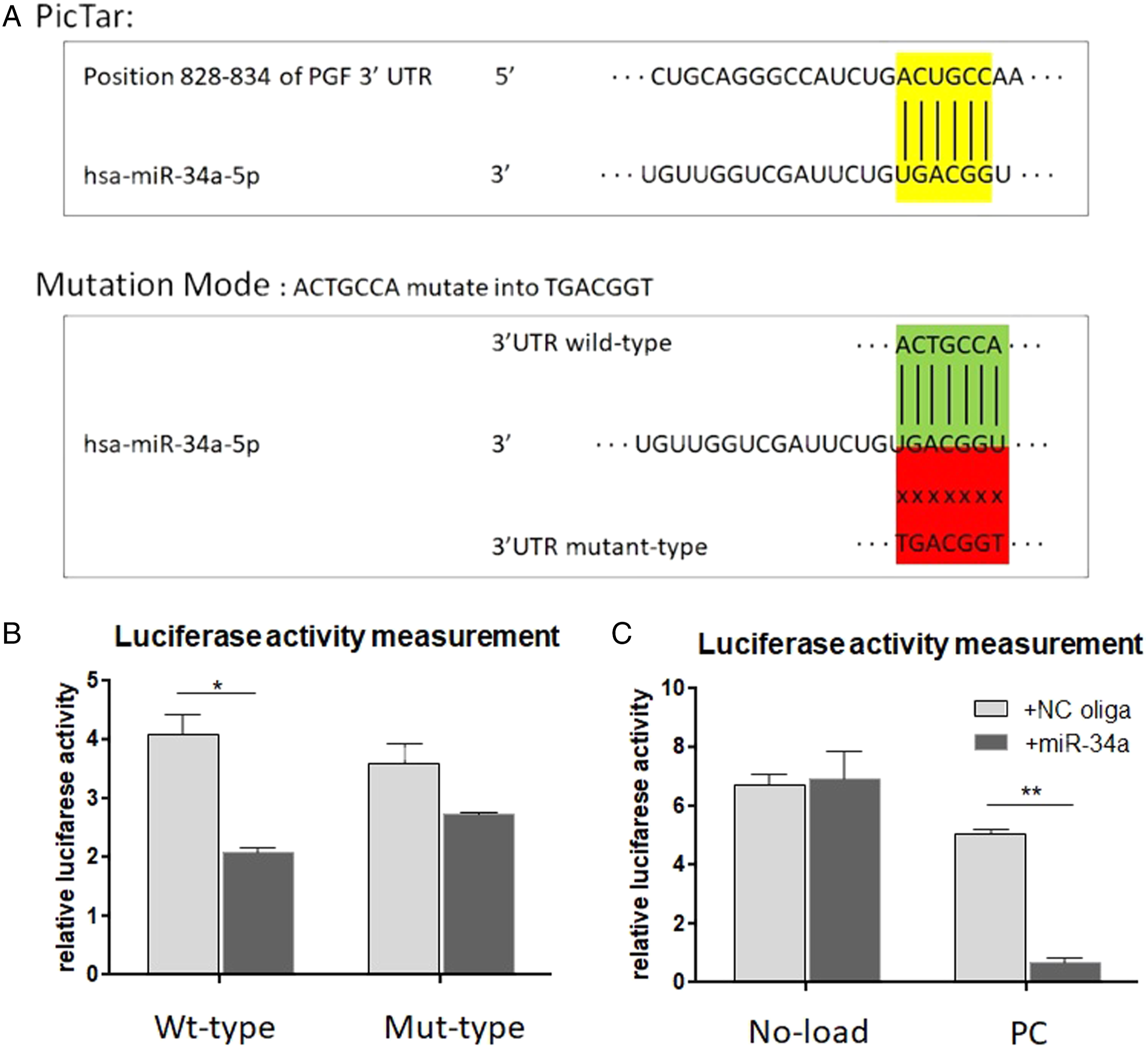
Dual luciferase reporter assay showed that transfection with miR-34a reduced the luciferase activity of the wild-type PLGF-3’UTR reporter plasmid compared with that of the NC oligo (P < .05). However, the luciferase activity of the mutant PLGF-3’UTR reporter plasmid did not significantly change after transfection with NC oligo and miRNA-34a single strand (Figure 5B). Moreover, in the negative control group, the luciferase activity did not significantly change, while in the positive control group, the luciferase activity significantly decreased after transfection of miRNA-34a single strand compared with NC oligo (P < .05) (Figure 5C).
Expression of miR-34a and Placental Growth Factor in Patients After Radiation
The serum miR-34a was elevated after radiation; moreover, miR-34a was further elevated after 4 weeks of radiation than after 2 weeks of radiation (P < .05) (Figure 6A). Consistently, serum mRNA and protein levels of PLGF were significantly increased after 2 weeks of radiation (P < .05). However, the serum mRNA levels of PLGF decreased after 4 weeks of radiation and at the end of radiotherapy compared with after 2 weeks of radiation (Figure 6B). Moreover, ELISA assay showed that serum PLGF contents changed in a similar manner (Figure 6C and D). Expression of miR-34 and Placental Growth Factor in the peripheral blood of patients after radiotherapy. (A): The expression of miR-34a in the peripheral blood of patients at different times after radiotherapy. (B): The mRNA expression of PLGF in the peripheral blood of patients at different times after radiotherapy. (C): Standard curve for measuring PLGF protein. (D): The content of PLGF protein in the peripheral blood of patients at different times after radiotherapy. “Before”: before radiotherapy, “week_2”: after 2 weeks of radiotherapy, “week_4”: after 4 weeks of radiotherapy. Note: PLGF=Placental Growth Factor.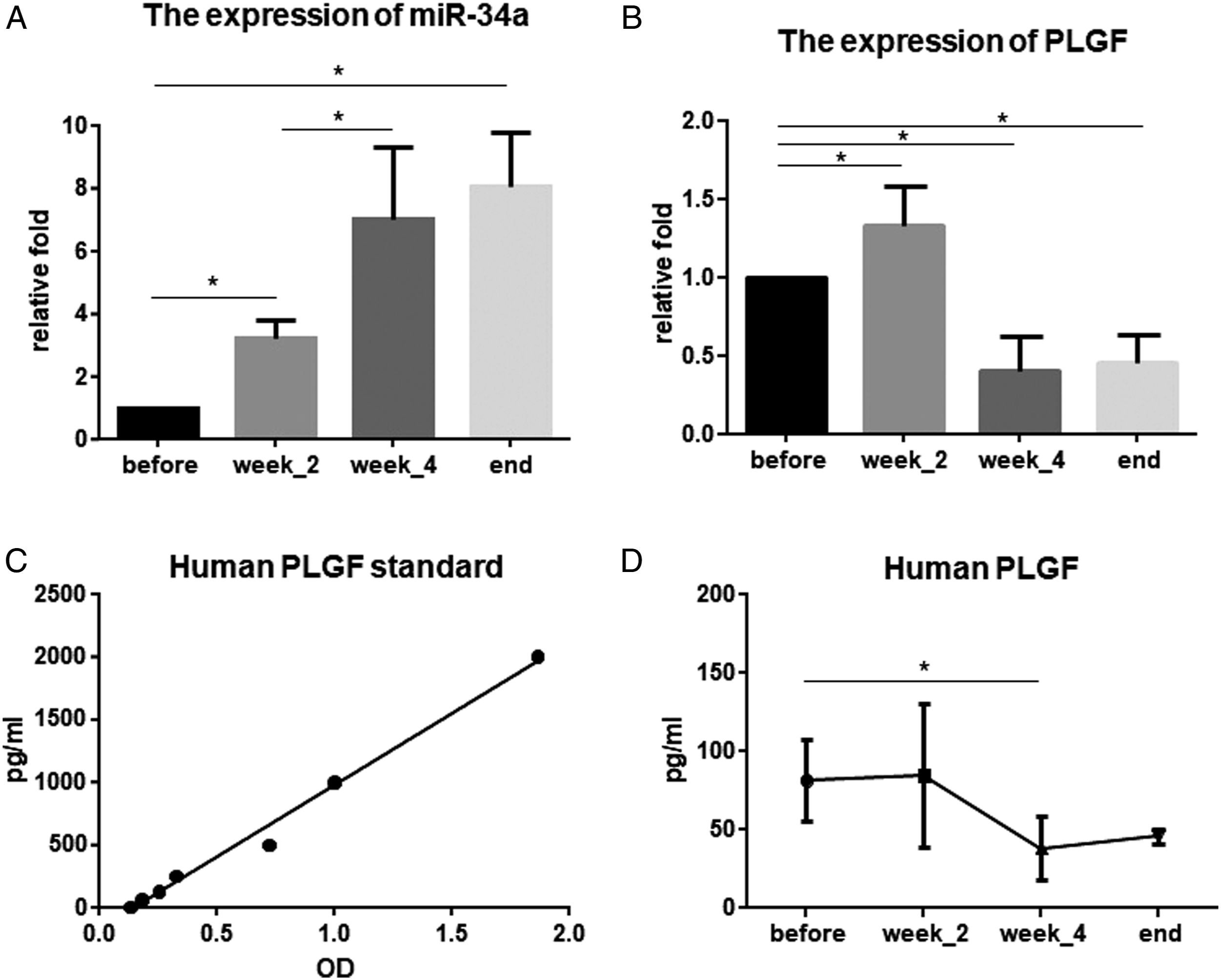
Protective Mechanism of Puerarin Against Radiation in HUVECs
In summary, X-ray radiation increased the expression of miR-34a in HUVECs. Puerarin treatment reduced the expression of miR-34a and increased PLGF, exhibiting its protective role against radiation by regulating DNA damage repair and reducing apoptosis (Figure 7). Protective mechanism of Puerarin against radiation in Human umbilical vascular endothelial cells.
Discussion
Cardiovascular diseases induced by chest radiotherapy have been described since as early as the 1960s. 1 In particular, the diagnosis and treatment via transthoracic radiation directly leads to radiation exposure of the heart and other tissues in the chest, which may cause clinical complications. Transthoracic radiotherapy focuses on diseases such as Hodgkin’s lymphoma and breast cancer. Due to the limitation of technology, ionizing radiation inevitably causes damage to the heart and other tissues in the chest and induces cardiovascular complications. A large-scale epidemiological survey in the United States found that the risk of cardiovascular death in patients with left breast cancer has increased by 1.16-fold than in patients with right breast cancer; such a risk increases with the duration of radiotherapy. 19 A follow-up study of patients with early-stage breast cancer also confirmed such findings. In this study, the risk of cardiovascular death in patients with left breast cancer increased by 1.27 times than in patients with right breast cancer. 20 Other studies also showed that radiation therapy for left breast cancer increases the cardiovascular mortality.21,22 Many studies confirmed that patients with Hodgkin’s lymphoma treated with mediastinal radiation also caught heart disease. 23 Compared with other treatment methods, chest radiotherapy, received by patients with Hodgkin’s lymphoma, increases the risk of cardiovascular disease by 2.2 to 3.1 times, and the risk can even be as high as 7.2 times, as shown in a study. 24
Cardiovascular complications caused by radiation mainly include coronary artery disease, valvular disease, cardiomyopathy, pericardial disease, and vascular disease.25,26 Among them, the most common complication is coronary artery disease, which causes damage to large and small coronary arteries. Radiation mainly results in stenosis of the large blood vessels. Histopathology reveals that fibrosis in the whole layer of the arteries, the loss of middle smooth muscle cells, and the accumulation of lipids are the main causes of stenosis. Damage to the small arteries is caused by myocardial fibrosis, occurring due to the direct impact of radiation on the function of myocardial capillary endothelial cells. In addition to the coronary arteries, all exposed blood vessels are affected by ionizing radiation. Radiation therapy of the head, neck, and mediastinum can lead to vascular disease in areas above the aortic arch, increasing the risk of carotid stenosis and cerebrovascular disease.27-30 Radiotherapy-induced injuries to the subclavian, innominate and axillary arteries have been reported. 31 Thus, radiation-induced vascular injury is considered a key factor in sclerosis, fibrosis and thrombosis of the radiation exposed areas, and persistent vascular endothelial cell dysfunction plays a critical role in the pathogenesis of these changes. Ionizing radiation damages the endothelial function and promotes thrombosis and inflammatory response, eventually leading to tissue damage, fibrosis and cardiovascular complications. In our study, we found that ionizing radiation induced DNA damage and apoptosis in vascular endothelial cells. Therefore, it is critical to reduce radiation-induced damage to vascular endothelial cells to prevent cardiovascular complications after radiotherapy.
In order to find a protective agent for vascular endothelial cell injury, many ingredients with protective roles toward vascular endothelial cells have been isolated from Chinese herbal medicine, such as Notoginsenoside R1 (NGR1), Ginsenoside Rb1 and salvianolic acid B.32-34 Puerarin is the major bioactive ingredient isolated from the root of Ge-gen (Chinese name) in traditional Chinese medicine. It has been widely used in the treatment of cardiovascular and cerebrovascular diseases, diabetes and diabetic complications. Liu B. finds that Puerarin plays a role in inhibiting myocyte loss during Heart failure (HF), partly through ferroptosis mitigation, suggesting a new mechanism of Puerarin as a potential therapy for HF. 35 LPS obviously increased the monolayer permeability, raised the levels of TNF-α and IL-1β in cell supernatant, and reduced the VE-cadherin expression in HUVECs. However, all of the above-mentioned LPS-induced changes were improved by Puerarin pre-treatment. 36 Puerarin facilitated osteogenesis of steroid-induced necrosis of rabbit femoral head and osteogenesis of steroid-induced osteocytes via miR-34a upregulation. 37 In our study, Puerarin exhibited protective effects by reducing DNA damage and apoptosis of HUVECs caused by radiation. On the molecular level, Puerarin reduced the expression of miRNA-34a in HUVECs and up-regulated its target PLGF. In our previous study, we found that miRNA-34a had an important role in radiation-induced DNA damage in HUVECs and might serve as a target against radiation.10,11 The target of miRNA-34a, PLGF is a member of the VEGF family, and is involved in vascular endothelial growth and vascular remodeling. 38 Currently, the mechanism of action of PLGF is unclear, but research shows that PLGF could regulate the inflammatory response, stimulate the proliferation and differentiation of pulmonary vascular smooth muscle cells and endothelial cells, and promote angiogenesis. 39 Moreover, PLGF promotes the proliferation of vascular endothelial cells, especially microvascular endothelial cells, and induces the migration and activation of vascular endothelial cells. 40 However, the effect of ionizing radiation on PLGF expression has not been reported. Our findings show that Puerarin can decrease miR-34a and in turn increase the expression of PLGF, thereby reducing X-ray-induced damage of endothelial cells.
In summary, the present study identifies Puerarin as a novel agent with radioprotective effects on vascular endothelial cells, further clarifies its molecular mechanism, and provides important theoretical significance and application values.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the funding from the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
