Abstract
Background
The relationship between interleukin-8 (IL-8) expression and the prognosis of gastric cancer (GC) patients has been reported, but the results are contradictory.
Aim
To investigate the effect of IL-8 expression on the prognosis of patients with GC.
Method
A comprehensive search strategy was used to search the PubMed, Web of Science and Cochrane Library databases. The total survival time was analysed using the RevMan 5.4 software. Through extensive search and meta-analysis of relevant studies, studies examining the relationship between IL-8 expression and prognosis in patients with GC were conducted to obtain more accurate estimates.
Findings
Eight studies (1843 patients) were included. The combined results of all the studies showed that high expression of IL-8 was a risk factor for poor prognosis in patients with GC (hazard ratio (HR): 2.08; 95% CI: 1.81–2.39). Sensitivity analysis suggested that the pooled HR was stable, and omitting a single study did not change the significance of the pooled HR. Funnel plots revealed no significant publication bias in the meta-analysis.
Conclusion
High IL-8 expression could be a negative prognostic biomarker for patients with GC.
Introduction
Gastric cancer is a common type of cancer worldwide, ranking fifth in incidence and fourth in mortality in 2020, and its incidence is twice as high in men as in women. 1 Currently, surgical resection is the primary mode of treatment for GC.2,3 Adjuvant chemotherapy has been widely used in postoperative patients with locally advanced GC and can improve the 5-year overall survival (OS) rate of these patients from 10% to 15%.4-6 However, many patients with GC miss the opportunity for surgery because of the lack of specific symptoms in the early stages of the disease.
Increasing evidence suggests that chemokines may be associated with cancer and may be a potential biomarker for cancer detection and prediction of prognosis. 7 IL-8 is a member of a family of cytokines known as CXC and is produced by a variety of cells, including neutrophils, macrophages, endothelial cells and cancer cells.8,9 IL-8 effectively modulates the chemotaxis of human neutrophils 10 and attracts neutrophils to inflammatory foci. 11 IL-8 derived from tumours contributes to the chemotactic recruitment of granulocytes. 12 Meanwhile, IL-8 exerts direct pro-tumorigenic effects, including the promotion of angiogenesis, tumour cell dedifferentiation, invasion and/or metastasis. 13 Studies have found that the expression level of IL-8 in GC tissues is significantly higher than that in normal tissues. 14 In addition, elevated levels of IL-8 trigger malignant biological behaviour of cancer cells, leading to poor clinical prognosis. 15 However, the relationship between the expression of IL-8 and the prognosis of GC patients is still unclear, and some research results are contradictory.16,17 Therefore, we conducted this systematic review and meta-analysis of the prognostic significance of IL-8 expression in patients with GC.
Materials and Methods
Data Strategies
This meta-analysis was performed according to the PRISMA 18 guidelines. We selected the relevant studies by searching PubMed, Web of Science and Cochrane databases. We combined MeSH terms and keywords to retrieve the relevant articles. The search terms used were as follows: (Interleukin-8 OR IL-8 OR CXCL8) AND (gastric cancer OR stomach cancer) AND (prognosis OR predict OR prognostic OR survival OR overall survival OR survival rate). No language restrictions were imposed. Two authors independently searched and assessed the eligibility of all the studies. Any differences were resolved through discussion and agreement between the two authors.
Inclusion and Exclusion Criteria
The included studies were required to meet the following criteria: (1) the studies evaluated IL-8 expression in the tissues or serum of GC patients, (2) the studies evaluated the relationships between IL-8 and GC prognosis and (3) the studies provided sufficient data to estimate the hazard ratio (HR) for OS or disease-free survival (DFS). Exclusion criteria included (1) non-original studies, including meta-analyses, reviews, and case reports; and (2) duplicate studies.
Data Extraction and Qualitative Assessment
Two authors independently extracted the data. The extracted data were summarised in a consistent manner to prevent bias. The extracted information included the first author, country of publication, year of publication, method of analysis, sample size, follow-up time, HR, 95% confidence interval (CI) and other data. The quality of the included studies was assessed using the Newcastle–Ottawa Scale (NOS) 19 from 0 (worst) to 9 (best). The Newcastle–Ottawa Scale provides a relatively objective evaluation criterion for case-control and cohort studies, and its effectiveness and credibility have been widely recognised.
Statistical Analysis
All the studies were analysed using the RevMan 5.4 software. The effect quantity for evaluating survival condition was the HR and 95% CI. The primary outcomes were OS and progression-free survival. If HR and 95% CI were not reported, we used the method provided by Tierney to calculate the data from the original study, or by consulting the authors. Subsequently, Engauge Digitizer (version 4.1)20,21 software was used to extract the data from the Kaplan–Meier curve according to the scheme provided. Cochran
Results
Study Selection and Characteristics
A total of 202 articles were selected after the initial database search. The titles and abstracts of these articles were reviewed separately by two authors. Finally, 41 out of the 202 study cases were sent for full-text assessment. Eight study cases with sufficient data, or with information that could be calculated indirectly, were evaluated in the current meta-analysis.15,16,22-27 The flow diagram of the literature selection procedure is shown in Figure 1, and the baseline characteristics of the evaluable articles are summarised in Table 1. In the meta-analysis, data from 1843 patients with GC were included, which were extracted from eight selected study cases published between 2001 and 2020. The sample size of these studies varied from 37 to 572 patients. In all the studies, specimens from 6 study cases15,16,23-26 were taken after surgery. Among these, 415,16,23,27 selected study cases were from China, 1
25
case was from India and 322,24,26 cases were from Japan. Five15,22-24,27 studies reported all stages of the disease (I–IV); 316,25,26 studies included stages I to III. Four15,24,25,27 studies assessed IL-8 by immunohistochemistry (IHC), and 222,23 studies investigated IL-8 using enzyme-linked immunosorbent assay (ELISA). The remaining two studies measured IL-8 levels using PCR and biochips, respectively. The follow-up time of the patients ranged from 33 to 150 months. The cutoff value of IL-8 was not available for four studies, and the thresholds for the remaining 4 studies also varied. In addition, 615,16,24-27 studies determined IL-8 expression using GC tissues; 222,23 studies determined IL-8 levels in serum. OS was obtained in 7 studies15,16,23-27, and DFS was obtained in 1 study.
22
Flow chart of article selection for systematic and meta-analysis. Characteristics of the Selected Studies. Abbreviations: NOS, Newcastle–Ottawa Scale; PCR, polymerase chain reaction; ELISA, enzyme-linked immunosorbent assay; IHC, immunological histological chemistry; NA, not available; HR, hazard ratio; OS, overall survival; DFS, disease-free survival.

High Expression of IL-8 and OS in GC
Meta-Analysis of Overall and Subgroup Analysis for IL-8 Expression and OS in GC.
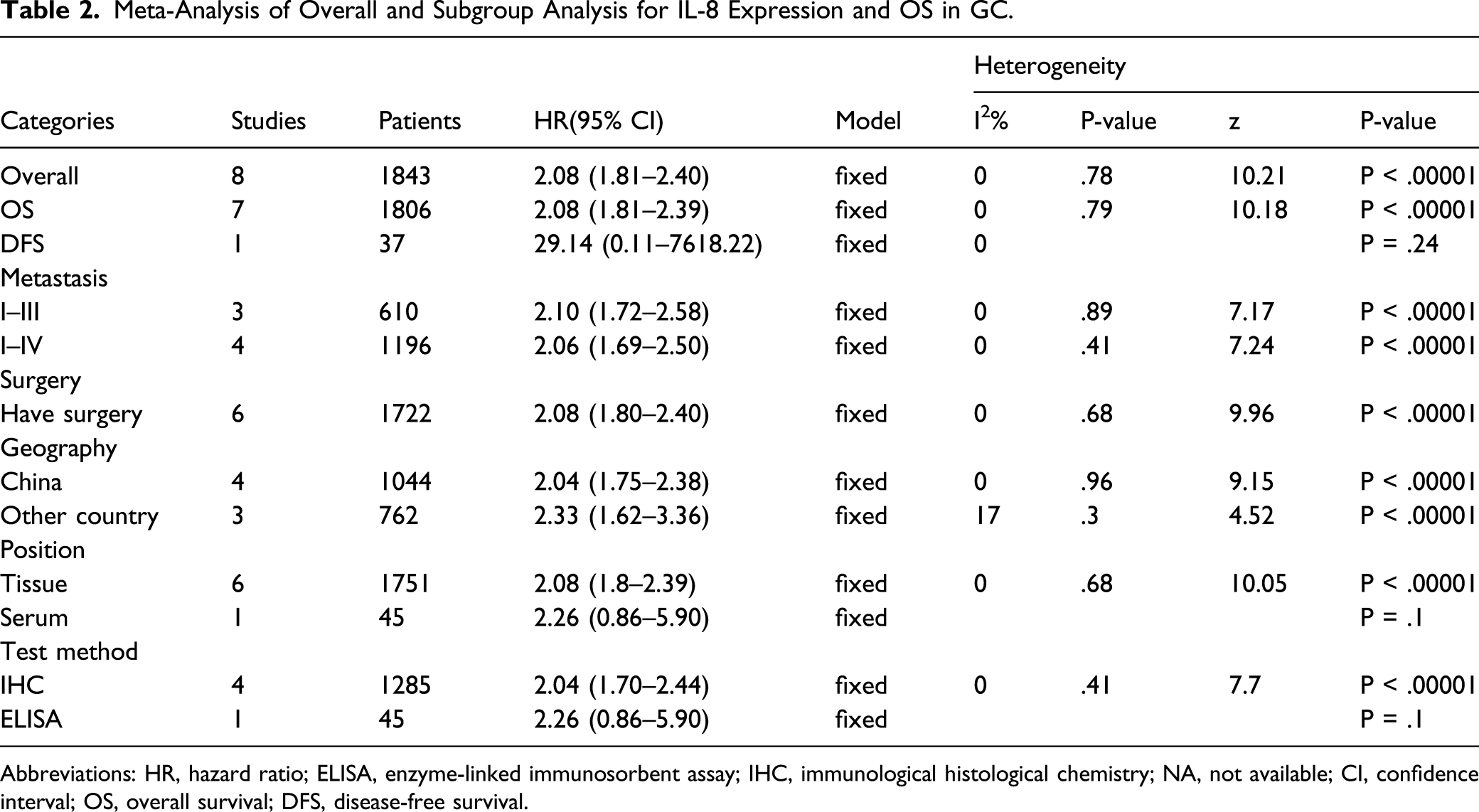
Abbreviations: HR, hazard ratio; ELISA, enzyme-linked immunosorbent assay; IHC, immunological histological chemistry; NA, not available; CI, confidence interval; OS, overall survival; DFS, disease-free survival.

Meta-analysis comparing IL-8 expression and OS in GC patients.

Meta-analysis comparing IL-8 expression and DFS in GC patients.

Funnel plots for the studies.
Subgroup Analysis
Subgroup analysis was conducted according to disease stage, surgery, region, detection specimen and test method. We observed a statistically significant correlation between IL-8 expression and poor prognosis in stage I–III patients (HR: 2.10; 95% CI: 1.72–2.58; Figure 4A) and in stage I–IV patients (HR: 2.06; 95% CI: 1.69–2.50; Figure 4B). Six studies evaluated the relationship between IL-8 expression and surgery. High expression of IL-8 was significantly associated with surgery (HR: 2.08; 95% CI: 1.80–2.40; Meta-analysis between IL-8 high expression and OS of GC patients stratified by tumour stage (A) and (B), region (C) and (D), and surgery (E). Meta-analysis between IL-8 high expression and overall survival (OS) of gastric cancer (GC) patients stratified by detection specimen (A) and (B), and test method (C) and (D).

Sensitivity Analysis
Sensitivity Analysis Results on Overall Survival (OS).

Abbreviations: HR, hazard ratio; CI, confidence interval.
Publication Bias
Potential publication bias in the meta-analysis was determined using funnel plot (Figure 6). An approximately symmetric funnel plot indicated that the meta-analysis had no significant publication bias.
Discussion
GC originating from the stomach lining is one of the most common malignant tumours in humans. The most effective method for complete rehabilitation of GC is gastrectomy, combined with adjuvant chemotherapy and radiotherapy. However, more than 50% of patients have recurrent peritoneal metastases after treatment. 28 Inflammation, a biological response to tissue damage or infection, is regulated by a class of soluble molecules called cytokines, including interleukins, chemokines and lymphokines. 29 It has been reported that the level of IL-8 expressed by GC cells is directly related to the extent of angiogenesis in the tumour. 30 In addition, the CXCL chemokine family plays an important role in the pathogenesis of GC and can be used as a biomarker for the diagnosis of GC. 31 Moreover, the CXCL8 gene may be highly associated with GC. 32
To the best of our knowledge, this is the first meta-analysis to focus on the association between IL-8 expression and prognosis in patients with GC. The results showed that high IL-8 expression was an independent risk factor affecting the prognosis of GC. Our research included 1843 patients with GC from three countries. We demonstrated that high IL-8 expression might be a marker of aggressive disease associated with poorer OS in GC patients. Indeed, accumulating evidence33,34 indicates that IL-8 is involved in the initiation and progression of GC, and is associated with poor clinical outcomes. In this meta-analysis, we also investigated the relationship between IL-8 expression and GC prognosis in terms of disease stage, geography, surgery, specimen detection and test method. When subgroup analysis was performed by tumour stage, we observed a slightly higher combined HR in stage I–III patients (2.10) than in stage I–IV patients (2.06), suggesting that this adverse prognostic factor is important not only in early GC, but also in late GC. The pooled HR was 2.04 (95% CI: 1.75–2.38) with China, and 2.33 (95% CI: 1.62–3.36) with other countries; both methods were statistically significant. When we performed subgroup analysis based on test specimens, there was no significant difference between serum IL-8 levels (HR: 2.26; 95% CI: 0.86–5.90;
Tian et al 17 performed a bioinformatics analysis which revealed that overexpression of the IL-8 gene was associated with significantly improved prognosis of GC. However, we believe this is not in conflict with our findings, as the expression of IL-8 may be affected by multiple pathways. In the context of tumours, IL-8 plays a role in cancer progression by promoting the recruitment of neutrophils to tumour sites, and the proliferation and migration of tumour cells.14,35 In addition, IL-8 expression may vary greatly depending on the situation. Therefore, the two conclusions are not contradictory.
The present meta-analysis has some limitations. Firstly, the number of studies included was relatively small. This is mainly because the expression of IL-8 is a continuous variable, and some studies did not provide appropriate data. Secondly, since there is no recognised cutoff point for IL-8, we only used the cutoff point given in the original article for definition. Those above the cutoff point were called high expression, and those below the cutoff point were called low expression. The methods and cutoff values used to detect IL-8 expression varied between studies, which can cause heterogeneity. Due to insufficient data, we were unable to conduct a subgroup analysis to explore this effect. To better evaluate the prognostic accuracy of IL-8, further investigation is needed on test methods and cutoff values. Thirdly, precise HR and 95% CI data were not provided in some articles. The calculated HRs and 95% CIs were possibly not as reliable as the given data.
Conclusion
Overall, our meta-analysis showed that high IL-8 expression was significantly associated with adverse clinical outcomes in patients with GC. The subgroup analysis showed that IL-8 in tissues, but not IL-8 in serum, was associated with the prognosis of GC. Our study revealed that IL-8 might be an independent biomarker for GC. To strengthen our conclusion, more standardised prospective studies with high quality are needed to evaluate the prognostic value of IL-8.
Footnotes
Acknowledgments
The authors would like to thank Dr. Chenghua Weng for her help with the revision and guidance in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Discipline Construction Promotion Project of the Second Affiliated Hospital of Soochow University (a team project of application innovation of nuclear technology in medicine) (serial number: XKTJHTD2021001) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
