Abstract
Pulmonary endothelial cell injury is a hallmark of acute lung injury. High-mobility group box 1 (HMGB1) can modulate the inflammatory response via endothelial cell activation and release of inflammatory molecules. Thus, we tested whether induced pluripotent stem cells (iPSCs) can alleviate ischemia/reperfusion (I/R) induced lung injury, and, if so, whether HMGB1 mediates the effect in a male C57BL/6 mouse model. Intravenously injected iPSCs into mice 2 h after I/R showed a significant attenuation of lung injury (assessed by lung mechanics, edema, and histology) 24 h after reperfusion (compared with controls), along with decreases in HMGB1, phosphorylated nuclear factor-κB, inflammatory cytokines [interleukin (IL)1β, IL6 and tumor necrosis factor-α], and the activation of endothelial cells. Furthermore, these effects of iPSCs can be mimicked by blocking HMGB1 with an inhibitor in vivo and in vitro. We conclude that iPSCs can be a potential therapy for I/R-induced lung injury. These cells may exert therapeutic effects through blocking HMGB1 and inflammatory cytokines.
Keywords
Introduction
Lung transplantation is the primary treatment option for end-stage lung disease. Ischemia/reperfusion (I/R)-induced lung injury is a critical syndrome and the cause of high morbidity and mortality rates in the first year after transplantation. 1,2 The mechanisms of ALI may include priming effects of dangerous factors exposure on the pulmonary alveolar epithelium, endothelium or inflammatory cells. 3 The central pathophysiologic concepts in ALI are inappropriate accumulation and activity of leukocytes, uncontrolled activation of coagulation pathways, the increased permeability of alveolar endothelial and epithelial barriers, and dysregulated inflammation. 4,5 Although the clinical treatment strategies are anti-inflammatory agents, antioxidants and mechanical ventilation, their therapeutic efficacy is unsatisfactory. Therefore, a new strategy for preventing and treating I/R lung injury is urgently needed.
Cell-based therapy is currently considered to be a promising therapeutic method for acute lung injury (ALI). 6 Multipotent stem cells (MSCs) have been shown to possess anti-inflammatory properties. There are numerous studies on the role of MSCs in the inflammatory response to lung injury. 7,8 In addition to MSCs, induced pluripotent stem cells (iPSCs) may constitute another option for cell therapy; iPSCs derived from somatic cells, and they are characterized by unlimited proliferation, multi-directional differentiation potential, low immunogenicity and lack of ethical controversy. 9 In 2019, it was demonstrated that iPSCs alleviate paraquat intoxication and lipopolysaccharide-induced lung injury, although the mechanism of action remains unclear. 10 Intravenously administered iPSCs can be homed to lung tissue with paraquat-induced injury and alleviate lung tissue damage, suggesting that iPSCs may be a potentially promising treatment strategy for I/R-induced ALI. However, the critical determinants of the protective effects of iPSCs are poorly understood.
High mobility group box 1 (HMGB1) is a non-histone binding protein and, as the prototypical endogenous danger-associated molecule, it can act as an instrumental effector molecule initiating the innate immune response, by itself or through interacting with cytokines. 11,12 Increased level of HMGB1 is key to the development of hypoxia-induced kidney injury. 13 It was previously reported that HMGB1 is an early mediator in the development of hemorrhagic shock-induced ALI. 14 Intratracheal administration of HMGB1 can lead to ALI, while inhibition of HMGB1 prevents the development of ALI. 15 At present, HMGB1 antagonists, such as glycyrrhizin, have achieved favorable outcomes in sepsis models. 16,17 However, it remains unclear whether the attenuation of lung injury by iPSC transplantation is associated with HMGB1.
The aim of the present study was to investigate the mechanism underlying the protective effect of iPSCs against I/R-induced ALI, and determine whether these effects are mediated by suppressing HMGB1 secretion, in the hope that the findings will provide a reliable experimental basis for further elucidating the anti-inflammation mechanism of action of iPSCs in reducing lung I/R injury.
Materials and Methods
Animal Care
50 Male C57BL/6 mice were purchased from Shanghai SLAC Laboratory Animal Co. Ltd., and maintained on standard rodent chow and water ad libitum. The animals were used for the experiments at 8-10 weeks of age. The animal experimental protocols were approved by the Animal Care Committee of the Fudan University School of Basic Medical Sciences, in accordance with the Guide for the Care and Use of Laboratory Animals (National Research Council, USA).
Mouse Model of Left Lung I/R-induced Injury
Male C57BL/6 mice were randomly allocated into 4 groups as follows: i) Sham, n = 5; ii) I/R, n = 5; iii) I/R + iPSCs (3 × 106/kg body weight), n = 5; and iv) HMGB1 inhibitor + I/R, n = 5. Left pulmonary I/R injury (LIRI) was induced as previously described. 18 In brief, the mice were anesthetized with 100 mg/kg pentobarbital sodium, then a 20-gauge cannula was inserted into the trachea and the mice were mechanically ventilated. Left thoracotomy was performed and the left pulmonary artery was clamped for 50 min with a 20 g micro-vascular clip (ischemia time). Subsequently, the clip was removed and the blood flow to the left lung was restored. The thoracotomy was closed, and the tracheal cannula was removed. At 2 h after LIRI, iPSCs (3 × 106 cells/kg) were injected via the tail vein. Sham animals underwent the same surgical procedure, but without clamping of the pulmonary artery. The HMGB1 inhibitor -Glycyrrhizin (50 mg/kg, S2302, Selleck Ltd.,) was injected intraperitoneally into the mice once daily for 14 consecutive days prior to lung I/R surgery. At 24 h after reperfusion, left lung tissue and bronchoalveolar lavage fluid (BALF) were harvested.
Measurement of Pulmonary Function
The pulmonary function was measured as previously described. 10 Briefly, the tracheal cannula was connected to a Y-shaped tube and the mouse was placed at the supine position, with its entire body inside the plethysmographic chamber (550 ml volume) to analyze pulmonary function. A rate of 110 breaths/min was provided to the animal through the ventilator. Pulmonary function parameters, including dynamic pulmonary compliance, inspiratory resistance and expiratory resistance, were calculated by the AniRes2005 system (BioLab Tech Co., Ltd.).
Lung Pathological Examination
Tissues from the upper left lung were fixed in 4% paraformaldehyde, followed by embedding in paraffin, serial sectioning at 4 μm, and hematoxylin and eosin staining. Lung injury was scored based on the following criteria 19 : Edema, interstitial inflammatory infiltration, hemorrhage, atelectasis and hyaline membrane formation. The sections were photographed using a DMR Leica microscope (Leica Microsystems, Wetzlar, Germany).
Lung Tissue Edema Analysis
The fresh lower lobe of the left lung was weighed (wet weight), then placed into an oven at 70°C for 72 h and weighed again (dry weight). The lung water content was calculated as (wet weight − dry weight)/dry weight.
Detection of Lung Gas Exchange Membrane Permeability
At 30 min before sacrificing the animals, FITC-BSA (2 mg/kg bw; Biosynthesis Biotechnology Co., Ltd.) was injected via the tail vein. The left lobes were subjected to lavage with 200 µl PBS 5 times. The BALF was centrifuged at 200 × g for 4 °C 15 min. The concentration of FITC-BSA in the BALF was detected via a fluorescent microplate reader (Synergy H1, BioTek Instruments, Inc., Winooski, VT, USA). The total number of nucleated cells in the BALF was also counted.
Mouse iPSCs Culture
C57BL/6 mouse iPSCs were induced from MEFs derived from 13.5-day-old embryos by lentivirus vector carrying Yamanaka factors in our laboratory, as previously described. 10 iPSCs were routinely cultured and expanded under feeder-free conditions with the use of multiple small-molecular inhibitors (CHIR99021 and PD0325901). Briefly, a standard iPSCs medium consists of DMEM supplemented with 0.1 mM non-essential amino acids, 1 mM sodium pyruvate, 0.05 mM β-mercaptoethanol, 50 U/ml penicillin, 50 μg/ml streptomycin, and 0.1 μg/ml leukemia inhibitory factor, 0.5 μg/ml CHIR99021, 0.5 μg/ml PD0325901 and 10% fetal bovine serum. The culture dishes were pretreated with 0.2% gelatin.
In vivo iPSCs Quantification
In our laboratory, the Tet-On gene was carried into iPSCs by lentiviruses used for iPSC induction, 10 which may be used as a marker for iPSCs identification and quantification. The Tet-On-specific PCR primers were as follows: Sense, 5′-AGCACAACTACGCCGCACCC-3′ and antisense, 5′-ATGCACCAGAGTTTCGAAGC-3′. PCR was conducted using genomic DNA isolated from lung tissues as a template. The PCR product of 18 S rDNA was used as an internal control.
Mouse Lung Vascular Endothelial Cell Isolation and Culture
Mouse lung vascular endothelial cells (MLVECs) were isolated using a previously described method. 14 Briefly, the mice were anesthetized with 100 mg/kg pentobarbital sodium, and the lungs were perfused with PBS to remove blood. Lung tissues (∼1 mm3) were cultured in a 60-mm culture dish at 37°C, 5% CO2 for 60 h. Subsequently, the tissues were removed and cultured for 3-4 days. The cells were purified using biotin-conjugated rat anti-mouse CD31(ab7388, Abcam, USA) monoclonal antibody and BD IMag streptavidin particles plus-DM, and the immunomagnetic separation system (BD Pharmingen; BD Biosciences) following the manufacturer’s instructions. MLVECs passage between 3 and 5 times were used in the experiments.
MLVEC Model of Nutritional I/R Injury
As the murine model merely interrupts lung arterial flow while maintaining continued alveolar ventilation during the period of ischemia, an in vitro model of I/R injury was used. 18,20 Briefly, MLVECs were cultured in nutrient-depleted PBS for 2 h, then PBS was replaced with DMEM supplemented with 10% calf serum for reperfusion. Nutrient depletion is defined as a cell culture medium without serum, growth factors or glucose. During IRI, reactive oxygen species(ROS) was considered as the main regulator, and directly related to endothelial cell injury. 21 In vitro nutritional I/R of MLVECs promoted the generation of reactive oxygen species. The Figure 1 shown that iPSCs decreased ROS from 0.2836 ± 0.0456 to 0.1069 ± 0.0447 in MLVECs at 12 h after IRI, meanwhile, iPSCs decreased ROS activity to a level (0.0902 ± 0.0283) similar to that of normal MLVECs (0.0810 ± 0.0352) at 24 h.

In vitro I/R-induced ROS production in MLVECs. During IRI, reactive oxygen species (ROS) was considered as the main regulator, and directly related to endothelial cell injury. Briefly, treated-MLVECs were incubated with DHE at 37°C for 30 min, analyzed on a fluorescence plate reader (Synergy H1; BioTek Instruments, Inc., Winooski, VT, USA), and quantified based on an H2O2 standard curve. The data are presented as mean ± standard deviation of the indicated number of experiments, n = 5.
Measurement of Intracellular HMGB1, Nuclear Factor (NF)-κB, and Inflammatory Molecular Expression in MLVECs
MLVECs (1 × 106 per well) were plated on TranswellTM permeable supports with porous filters (6-well plates, 8.0-µm pore size, Corning Inc.) and cultured for 24 h. An in vitro nutritional I/R model was created according to the literature. 18 In brief, MLVECs were washed 3 times with PBS and then cultured in PBS for 2 h. DMEM supplemented with 10% calf serum was added for “reperfusion”. iPSCs (1 × 104 per well) or HMGB1 inhibitor was added to the upper chamber of permeable supports, respectively. The protein and mRNA of MLVECs were collected at 12 and 24 h for reverse transcription-quantitative PCR (RT-qPCR) analysis and western blotting, respectively. All experiments were repeated 5 times independently.
RT-PCR for Inflammatory Factors
Total RNA was extracted from lung tissue using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) according to the manufacturer’s instructions. Briefly, RNA (1 μg) was reverse-transcribed using SuperScript Reverse Transcriptase (Fermentas; Thermo Fisher Scientific, Inc.). qPCR was performed by SYBR Green Real-Time PCR Master Mix (Toyobo Co., Ltd.). The relative expression of inflammatory factors was calculated by the 2-ΔΔCt method. The primer sequences used were as follows: IL-1β: forward 5′-TGCCACCTTTTGACAGTGATG-3′; reverse 5′-AAGGTCCACGGGAAAGACAC-3′; IL-6: forward 5′-GCCTTCTTGGGACTGA- TGCT-3′; reverse 5′-TGCCATTGCACAACTCTTTTCT-3′; TNF-α: forward 5′-CCCTCACACTCACAAACCAC-3′; reverse 5′-ATAGCAAATCGGCTGA- CGGT-3′; 18 s: forward 5′-GAGAAACGGCTACCACATCC-3′; reverse 5′-CACCAGACTTGCCCTCCA-3′.
Western Blotting
Protein was extracted from mouse lung homogenates or cell lysates, then subjected to Western blot. The membranes were incubated with 1:1,000 dilution of purified rabbit anti-HMGB1 polyclonal antibody (ab79823, Abcam, USA), anti-NF-κB monoclonal antibody (8242, Cell signaling technology, USA), anti-phosphorylated NF-κB monoclonal antibody (3033, Cell signaling technology, USA) and anti-β-actin monoclonal antibody (Bioworld Technology, Inc.) overnight at 4°C. The immunoreactive bands were visualized using HRP-conjugated donkey anti-rabbit IgG (1 : 5000, 711-035-152, Jackson Immuno Research Laboratories, Inc.). The blots were quantified using BioRad Quantity One software 4.4.0 (Bio Rad Laboratories, Inc.).
Statistical Analysis
The data are presented as mean ± standard deviation of the indicated number of experiments. Differences among group means were assessed by one-way ANOVA using SPSS 22.0 (IBM Corp.). Differences were considered statistically significant at P < 0.05.
Results
Protective Effect of iPSCs Against LIRI-Induced ALI
At 24 h after left lung I/R, pulmonary compliance decreased by 43.33%, the expiratory resistance increased by 89.46% and inspiratory resistance increased by 1.23-fold compared with the sham group, which significantly improved after iPSC transplantation or HMGB1 inhibitor treatment (Figure 2A-C). We further investigated whether iPSCs affected the pathological morphology of the damaged lung tissue. As shown in Figure 2D, lung exposed to I/R exhibited interstitial thickening, inflammatory cell infiltration and intra-alveolar hemorrhage. To assess the respiratory membrane permeability, FITC-BSA concentration and nucleated cell count in the BALF were analyzed. I/R increased the FITC-BSA concentration and the nucleated cell count by 104.6% and 263.93%, respectively, in the BALF, compared to those in the sham group (Figure 2F and G). Both iPSCs and HMGB1 inhibitor treatment significantly ameliorated these parameters, or even restore them to normal levels. Furthermore, the wet/dry weight ratio was similar to the pulmonary respiratory membrane permeability (Figure 2E). There were no statistically significant differences between the iPSC and the HMGB1 inhibitor treatment groups.

Protective effect of iPSCs and HMGB1 inhibitor against I/R-induced lung injury. (A) iPSC transplantation or HMGB1-inhibitor treatment improved pulmonary function in lung injury induced by I/R, as shown by pulmonary compliance, (B) expiratory resistance and (C) inspiratory resistance. (D) Histological changes involving in hyaline membrane (red arrow) and hemorrhages (black arrow) accompanied by mononuclear cells (green arrow, yellow arrow) infiltration were detected by hematoxylin and eosin staining using a light microscope; (E-G) wet/dry weight ratio, FITC-labeled BSA and nucleated cell count of the BALF were decreased in the iPSC engraftment and HMGB1 inhibitor treatment groups. Data are presented as mean ± standard deviation. I/R, ischemia/reperfusion; iPSCs, induced pluripotent stem cells; HMGB1, high-mobility group box 1; BALF, bronchoalveolar lavage fluid.
iPSCs Attenuate the Expression of HMGB1, Phosphorylated-NF-κB and Inflammatory Factors in LIRI-Induced ALI
The association between iPSCs and HMGB1 was determined. As shown in Figure 3, the injected iPSCs were detected in the damaged lungs after I/R (Figure 3A), with the levels of HMGB1 and phosphorylated-NF-κB decreasing by 45.23% and 40.3%, respectively, compared with those in the I/R group (Figure 3B and C). There were no statistically significant differences between the iPSCs and the HMGB1 inhibitor groups. Furthermore, the effect of HMGB1-NF-κB on downstream inflammatory factor expression was examined. I/R markedly increased the level of IL-1β (265.33%), TNF-α (88.47%) and IL-6 (1388%) in the damaged lung tissue compared with the sham group. Treatment with iPSCs downregulated IL-6, TNF-α and IL-1β levels, which were almost restored to normal. The HMGB1 inhibitor treatment group also produced similar results (Figure 3D-F).
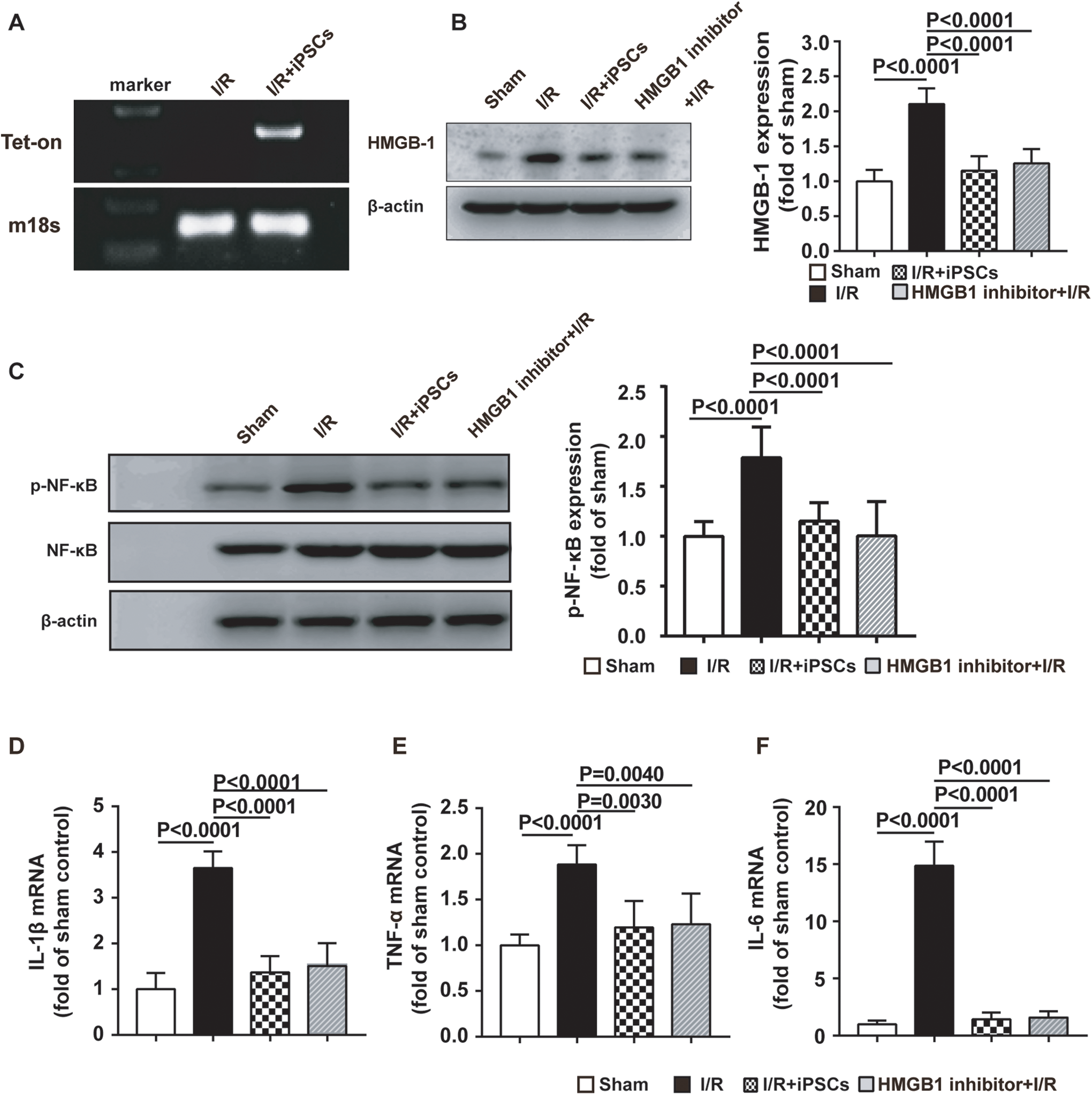
Effect of iPSCs on the HMGB1-NF-κB axis in pulmonary tissue after I/R. (A) Abundance of PCR product from Tet-On genes isolated from lung in the I/R and iPSC engraftment groups after cells injection for 24 h. (B and C) The expression of HMGB1, and phosphorylated-NF-κB was significantly increased in lung injury, and the injection of iPSCs reversed this increase. (D-F) The mRNA expression of inflammatory factors, including IL-1β, TNF-α and IL-6, was examined in the injured lung tissue. Data are presented as mean ± standard deviation. I/R, ischemia/reperfusion; HMGB1, high-mobility group box 1; iPSCs, induced pluripotent stem cells; NF-κB, nuclear factor-κB; IL, interleukin; TNF, tumor necrosis factor.
iPSCs Inhibited In Vitro I/R-Induced HMGB1 Release-Mediated Inflammatory Factor Production in MLVECs
Pulmonary MLVECs constitute one of the major cell populations of the lung, and the main source of inflammatory cytokines. 22 HMGB1-mediated activation of NF-κB signaling is a key mechanism responsible for endothelial cell synthesis and secretion of IL-1β, IL-6 and TNF-α, in order to augment inflammation and organ injury. In the present study, MLVECs cultured in nutrient-depleted PBS for 2 h followed by DMEM reperfusion for 12 h induced HMGB1 and phosphorylated-NF-κB increase by 181%, and 87.16%, respectively, compared with the control group, which lasted up to 24 h. However, after co-culturing the iPSCs with in vitro I/R-damaged MLVECs, the levels of HMGB1 and phosphorylated-NF-κB were markedly decreased by 51.32%, and 30.31%, respectively, at 12 h, and by 57.32% and 43.23%, respectively, at 24 h in MLVECs (Figure 4A and B). Moreover, iPSCs almost completely inhibited IL-1β (72.43%), TNF-α (66.99%) and IL-6 (65.51%) expression in MLVEC at 24 h compared with the I/R group (Figure 4C-E).

iPSCs inhibited I/R-induced HMGB1 release-mediated inflammatory factor production in MLVECs. (A and B) MLVEC exhibited increased HMGB1 and p-NF-κB expression following I/R for 12 and 24 h, which was reversed by treatment with iPSCs. (C-E) The expression of inflammatory factors, including IL-1β, TNF-α and IL-6, in MLVEC following I/R for 12 and 24 h in the injury and iPSC treatment groups was detected by quantitative PCR. Data are presented as mean ± standard deviation. I/R, ischemia/reperfusion; iPSCs, induced pluripotent stem cells; HMGB1, high-mobility group box 1; NF-κB, nuclear factor-κB; IL, interleukin; TNF, tumor necrosis factor.
Discussion
I/R-induced lung injury occurring following lung transplantation is characterized by lung inflammation. 23 Intravenously administered syngeneic iPSCs exert therapeutic effects on multiple diseases, and our previous study reported the beneficial effects of iPSCs on paraquat-induced lung injury. 10 However, the mechanisms underlying the protective effects of iPSCs require further investigation. To the best of our knowledge, this study is the first to investigate the mechanism of action of iPSCs in left lung I/R and lung inflammatory response using rodent and cell models.
Left lung I/R in healthy mice was used to mimic lung transplantation-induced lung injury. In this mouse LIRI model, it was observed that I/R severely compromised the respiratory function and increase lung water content, gas exchange membrane permeability and nucleated cell infiltration of the BALF. Importantly, iPSCs transplants acted protectively in mice against I/R-induced lung injury and restored respiratory function and pathological damage. These data suggest that the protective role of iPSCs against I/R-induced ALI may be closely associated with critical mediators of sterile inflammation.
The key benefits of iPSCs therapy is the ability to preferentially located in damaged tissues. Using the lung injury model induced by intratracheal instillation of LPS 10 or I/R, we found significantly more iPSCs in damaged lungs than in other organs and damaged-free animal. One of the key mechanisms underlying iPSCs homing is facilitated by integrin. iPSCs express integrin α4β1, which binds to VCAM-1 of endothelium. We have demonstrated that integrin beta-1 (integrin β1) could be responsible for the selective trafficking of intravenously iPSCs to the activated vasculature. 24
iPSCs have a powerful therapeutic capacity for lung injury, which owing to their self-renewal and paracrine effect. In 2019, we demonstrated that iPSCs trafficked into paraquat-induced pulmonary injured tissue and only a small portion of the cells differentiated into type 2 pneumocytes. 10 Thus, endo/paracrine may be the major mechanism by which iPSCs ameliorated lung injury in the early phase. This is consistent with our in vitro result that iPSCs inhibited ROS production in MLVECs.
HMGB1 is a mediator of organ injury of sterile inflammatory responses. 25 Injurious stimuli in the lung promote HMGB1 release, which augments the inflammatory response. 26 Intratracheal administration of HMGB1 alone caused organ dysfunction in the form of ALI. 27 The glycyrrhizin is a triterpene glycoside which can bound directly to both HMG boxes of HMGB1, thereby reducing the concentration of HMGB1 and inhibiting its chemoattractant and biological activities. 28 -30 In addition, glycyrrhizin can weaken the pro-inflammatory effect of HMGB1 through different signaling pathways. Tan et al demonstrated that the interaction between cell receptors and HMGB1 may be blocked by glycyrrhizin, leading to the inactivation of downstream MAPK/NF-κB signaling pathways. 28 Therefore, glycyrrhizin was used in the present study as a HMGB1 inhibitor. As shown in Figure 3, the administration of HMGB1 inhibitor ameliorated pulmonary pathological injury, alveolar epithelial capillary permeability, nucleated cell influx into the alveolar lumen, and proinflammatory cytokine levels. iPSCs (3 × 106 cells/kg body) were intravenously administered at 2 h after LIRI, and iPSC engraftment was effective in suppressing LIRI-induced HMGB1 expression, followed by amelioration of ALI and restoration of lung function. Taken together, these findings indicate that iPSCs have the ability to reduce LIRI-related injury through attenuating HMGB1 secretion.
In ALI, HMGB1 as a damage-associated molecular pattern that is recognized by the key pattern recognition receptors on endothelial cells. 31 Activated lung endothelial cells secrete cytokines to induce lung inflammation, which is partly mediated through the HMGB1-NF-κB signaling pathway. 32,33 The binding of HMGB1 results in the activation of the NF-κB, which plays a key role in modulating the expression of pro-inflammatory molecules (IL-1β, IL-6 and TNF-α). 34,35 Our previous and other studies demonstrated that HMGB1 is associated with enhanced production of IL-1β, IL-6 and TNF-α. 36 The cell I/R experiment demonstrated that HMGB1 inhibitor administration markedly reduced the expression of NF-κB and associated inflammatory cytokines. However, studies on rodents and humans demonstrated that the glycyrrhizin is poorly absorbed by the gastrointestinal tract, and can occur an enterohepatic circulation of glycyrrhetic acid side-effects. 37,38 Thus, utilizing glycyrrhizin is mainly to demonstrate the pathological conditions involved in HMGB1. In the present study, iPSC engraftment significantly decreased the levels of HMGB1 and phosphorylated NF-κB in the MLVECs I/R model.
Consistently with the results of the in vivo and in vitro experiments, it was originally reported that iPSCs attenuate pulmonary edema, pathological tissue destruction and the inflammatory response, which is likely mediated by suppressing HMGB1 secretion, thereby alleviating the phosphorylation of NF-κB and the production of IL-1β, IL-6 and TNF-α in endothelial cells.
In conclusion, iPSCs play a role in attenuating HMGB1, thereby decreasing the expression of inflammatory molecules and protecting against I/R-induced pulmonary injury. iPSCs can be a potential therapy for I/R-induced lung injury.
Footnotes
Author Contribution
Yijun Li, Shun Wang, and Jinbo Liu Contributed equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by General Program (81970074 to Meng Xiang) of the National Natural Science Foundation of China. Basic Research Project of Science and Technology of Baoan District, Shenzhen (2017JD109) and Joint Research Fund of Guangdong Medical University (L2016009) to Xiaoming Li.
