Abstract
Autophagy has been strongly linked with hormesis, however, it is only relatively recently that the mechanistic basis underlying this association has begun to emerge. Lysosomal autophagy is a group of processes that degrade proteins, protein aggregates, membranes, organelles, segregated regions of cytoplasm, and even parts of the nucleus in eukaryotic cells. These degradative processes are evolutionarily very ancient and provide a survival capability for cells that are stressed or injured. Autophagy and autophagic dysfunction have been linked with many aspects of cell physiology and pathology in disease processes; and there is now intense interest in identifying various therapeutic strategies involving its regulation. The main regulatory pathway for augmented autophagy is the mechanistic target of rapamycin (mTOR) cell signaling, although other pathways can be involved, such as 5′-adenosine monophosphate-activated protein kinase. Mechanistic target of rapamycin is a key player in the many highly interconnected intracellular signaling pathways and is responsible for the control of cell growth among other processes. Inhibition of mTOR (specifically dephosphorylation of mTOR complex 1) triggers augmented autophagy and the search is on the find inhibitors that can induce hormetic responses that may be suitable for treating many diseases, including many cancers, type 2 diabetes, and age-related neurodegenerative conditions.
Keywords
Introduction
Hormesis is a biphasic dose–response to an environmental agent or condition characterized by low dose stimulation with beneficial effect and a high dose inhibitory with harmful effect. 1,2 Hormesis is induced by mild physiological stress caused by a variety of stressors including toxic metals, organic xenobiotics, phytochemicals, drugs, ionizing radiation, nutrient deprivation (ie, fasting and caloric restriction [CR]), hypoxia, and oxidative stress (Figure 1). 3 This type of functional response seems to occur in all eukaryotic organisms, which include yeasts and fungi, protozoans, plants, and metazoan animals. Lysosomal autophagy (self-eating) appears to be strongly associated with the mechanism underlying hormesis 4 -8 ; and Blagosklonny 9 has proposed that the mechanistic target of rapamycin (mTOR) cell signaling system is a crucial feature of hormetic responses, particularly due to its relationship with lysosomal autophagy (Figure 2).
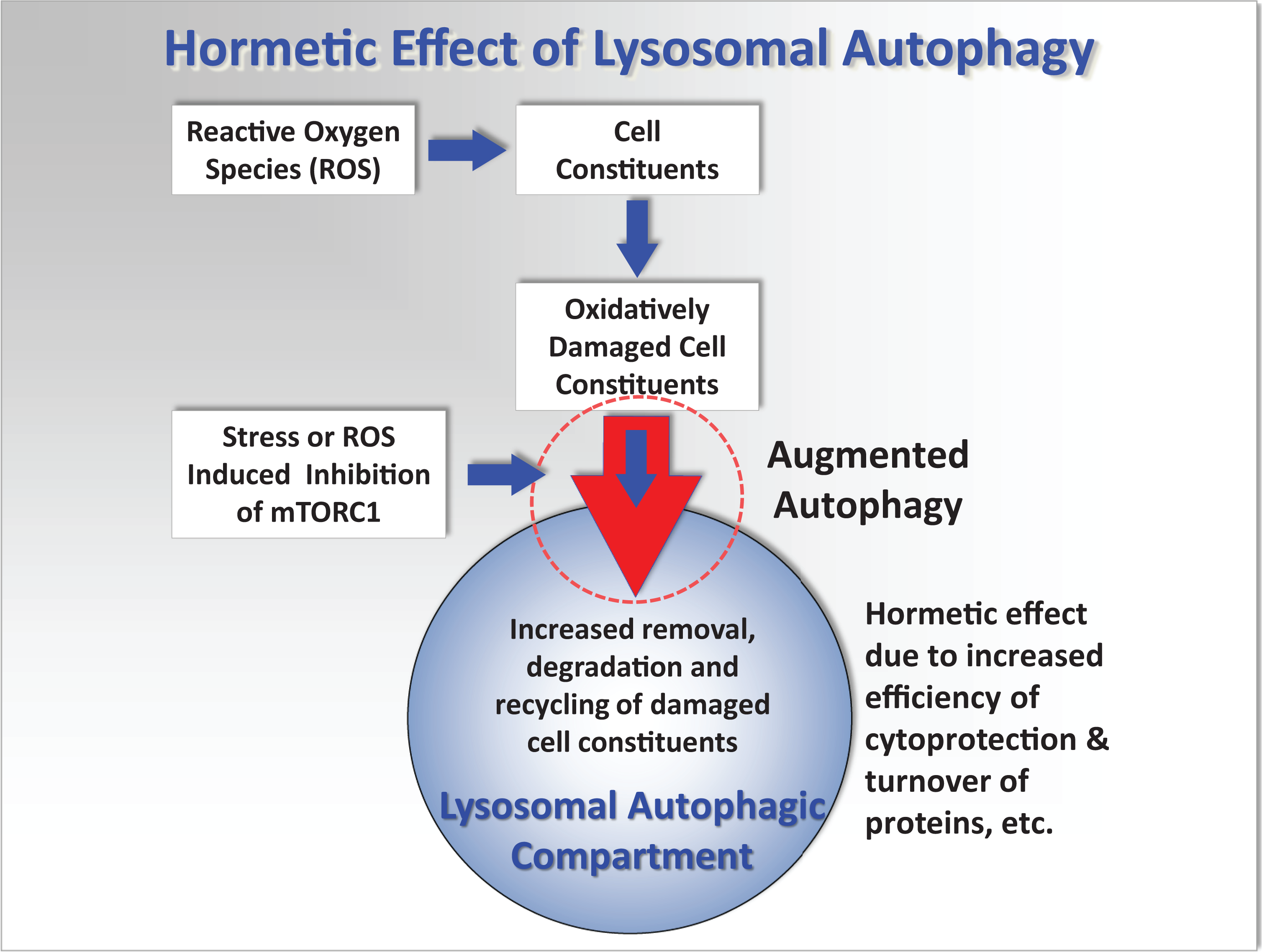
Simplified conceptual mechanistic model showing the interactions of reactive oxygen species (ROS) with cellular components, lysosomal and autophagic processes, that can lead to oxidative stress, cell injury, and pathology. It is hypothesized that repeated stimulation of augmented autophagy (large red arrow circled) by various environmental factors such as fasting and exercise, or low concentrations of toxic chemicals that induce mild stress, will result in a more effective recycling of damaged cellular proteins and organelles; and before major oxidative damage occurs. This will consequently reduce lipofuscin formation that will otherwise accelerate ROS generation and cell injury and thus protect the cell from further oxidative injury. See more detailed diagram in Moore et al. 10

Abbreviated diagrammatic representation of the PI3K, Akt, mTOR cell signaling pathway, and associated pathways, as a major regulator of cell function (see Laplante and Sabatini
11
for a more extensive chart of mTOR-related cell signaling). The mTOR is an atypical serine/threonine kinase that is present in 2 distinct complexes. The first, mTORC1, is highlighted and is composed of mTOR, Raptor, GβL, and DEPTOR and is inhibited by rapamycin. It is a master growth regulator that senses and integrates diverse nutritional and environmental cues, including growth factors, energy levels, cellular stress, and amino acids. It couples these signals to the promotion of cellular growth by phosphorylating substrates that potentiate anabolic processes such as mRNA translation and lipid synthesis or limit catabolic processes such as autophagy. The pathways from mTORC1 leading to activation of autophagy (highlighted) are indicated in red (heavy arrows). Interactions with AMPK (circled) are also shown. Overactivity of mTORC1 is believed to trigger inflammatory processes which can result in pathological injury and processes leading to many cancers and degenerative diseases; and aberrant mTOR signaling is involved in many disease states including cancer, cardiovascular disease, and diabetes. Adapted from Moore
12
and with permission from Cell Signaling Technology, mTOR Signaling Interactive Pathway: https://www.cellsignal.com/contents/science-cst-pathways-pi3k-akt-signaling-resources/mtor-signaling-interactive-pathway/pathways-mtor-signaling. Key to major constituents and symbols: Akt indicates serine/threonine kinase or protein kinase B; AMPK, 5′-adenosine monophosphate-activated protein kinase; mRNA, messenger RNA; mTOR, mechanistic target of rapamycin; mTORC1, mammalian target of rapamycin complex 1; PI3K, phosphatidylinositol-3 kinase; PIP3, phosphatidylinositol 3,4,5 trisphosphate; PTEN, phosphatase and tensin homolog; ROS, reactive oxygen species; activation,
Lysosomes have been linked to hormetic responses for over 40 years. 13 Low level toxic metal stress induced changes in the lysosomal–vacuolar system of a marine coelenterate (Campanularia flexuosa), at concentrations of the metals that were an order of magnitude less than that required for the induction of growth hormesis. This finding was interpreted at the time as a precursor to the stimulation of growth by the metals (copper, cadmium, and mercury) that may have been associated with the augmentation of lysosomal autophagy as a survival strategy. 13 -15 Lysosomes are also notable for their ability to sequester and accumulate many metals, organic xenobiotics (including drugs such as chloroquine), radionuclides, microplastics, and nanomaterials. 16 -23 This sequestration probably has a protective function by compartmentalizing harmful materials from the rest of the cell; although it can result in lysosomal membrane destabilization (ie, permeablization) and release of intralysosomal iron with subsequent generation of reactive oxygen species (ROS) and cell injury. 22,24 Furthermore, it is now apparent that autophagy is triggered by the inhibition of the mechanistic target for rapamycin complex 1 (mTORC1) cell signaling system (Figure 2). 25 Mechanistic target for rapamycin complex 1 is a central focus for regulation of cell function: it acts as a nutrient sensor and is switched off by nutrient deprivation; and it is also inhibited by ROS (Figure 2). 11 Hormesis now appears to be inextricably interconnected with mTORC1 and autophagic turnover of damaged cellular components including proteins and organelles. 9,15,26 -28
Hormesis has been simulated in a cell physiological model using a network modeling method. 29 In this model, health status or homeostasis is represented by connectance: This is essentially the degree of system complexity or connectivity within the interconnected nodes representing the various molecular and cellular processes that constitute a living cell. Mild stress, such as nutritional deprivation, induced an increase in connectance, indicating an increase in health status that is consistent with hormesis. Increasing stress resulted in a decline in connectance, indicative of a loss of complexity and increasing cell injury. 21,29,30 Connectivity associated with autophagy was a significant factor in the increase in connectance induced by mild stress. The outputs from this simulation have since been validated in experimental studies with molluscan hepatopancreas or digestive gland. 10,21,30,31
Role of Autophagy in the Economy of the Cell
Autophagy is a cellular “garbage removal” process that is activated in response to various types of metabolic stress, including nutrient deprivation, generation of ROS, growth factor depletion, and hypoxia (Figure 1). 14,15,32 Molecular studies involving targeted mutations resulting in deletions of autophagy-related genes, in organisms as varied as yeast, slime molds, plants, and mice, have shown that autophagy is essential for eukaryotic life. 33
As well as providing an evolutionarily primitive survival mechanism against starvation and other environmental stressors, such as toxic chemicals, lysosomally mediated autophagic processes are crucial for the normal (basal) degradation and turnover of cellular components in autolysosomes. 14,15 Lysosomal autophagy is also implicated in many disease processes, cell injury, cell death, and adaptive responses. 10,14,15,17,18,21,22,31,34 -39 Autophagy comprises at least 3 related cellular processes (ie, macroautophagy, microautophagy, and chaperone-mediated autophagy [CMA]); and it is essential for maintenance of cell homeostasis. 14,15,40 Furthermore, cell morphological evidence has long indicated that lysosomal autophagy is a highly conserved mechanism evolutionarily. 14,33 Damaged cellular constituents and redundant products are removed by lysosomal autophagy, but it is also critically involved, along with other proteolytic systems (eg, proteasomes), in the continuous turnover of intracellular components. 41 Autophagy is upregulated in times of stress or physiological change: by breaking down longer-lived proteins and organelles and recycling the products into protein-synthesis and energy-production pathways, the process allows cells to be temporarily self-sustaining during periods when nutrients are restricted (Figure 1). 14,40
Autophagic lysosomal degradation is the principal route for intracellular protein turnover and clearance. 42 Proteins and lipids targeted for degradation enter the lysosomes via macroautophagy, microautophagy, and CMA. 14,15 Once transported to the autolysosomes, these materials are split into their component molecules. Macroautophagy and microautophagy nonselectively remove entire portions of cytosolic regions and whole organelles, whereas CMA solely targets specific cytosolic proteins. While macroautophagy is induced close to the onset of fasting conditions, evidence suggests that this nonselective degradation can only be maintained for a short period of time in mammalian cells (ca 10 hours 43 ). After this, CMA is the main form of autophagy. This is thought to allow the concurrent degradation of proteins and lipids into the amino acids required for cell survival, while preventing the degradation of molecules critical for basic cell function. Thus, CMA can be designated a stress-induced pathway that is activated not only by nutrient deprivation but also by mild oxidative stress 44 and exposure to toxic compounds. 14,21,31 This was clearly demonstrated by Massey et al 45 who impaired CMA in mouse cells. The survival rates of these cells were unaffected when maintained under optimum conditions and it was hypothesized that the cells were able to upregulate macro- and microautophagic pathways to sustain protein turnover homeostasis. However, these non-CMA autophagic pathways were unable to compensate for the elevated levels of protein damage resulting from exposure to pro-oxidants and ultraviolet as evidenced by an exponential decrease in cell viability, increase in apoptosis, and cell death. 45
Role of Autophagy and Hormesis in Disease and Aging
In addition to low nutrient scenarios where the autophagic processes break down macromolecules to their constituents, which may be used for energy and growth; autophagy plays a much more important role in the overall homeostasis of the cell. 14,15,29,32,33 The principal intracellular roles of autophagy are:
These 3 mechanisms are implicated in many bacterial, viral, heritable, and aging-related diseases, including atherosclerosis and various cancers, as well as neurodegenerative diseases (eg, Alzheimer disease, Huntington chorea, Motor neuron disease, bovine spongiform encephalopathy (BSE), Creutzfeldt-Jakob disease, and Parkinson disease. 39,48 -53 (Cuervo, 2008). There is also a related autophagic process called mitophagy to cull dysfunctional mitochondria, such as occurs in fibromyalgia. 54,55
With some cancers, it is generally accepted that autophagy can suppress tumor initiation and cancer growth; and since autophagy blocks growth and increases breakdown of proteins, this is quite logical. 56,57 (Cuervo, 2004, 2008). Cancer cells often have much lower levels of basal autophagy than normal cells. Additionally, many oncogenes and tumor-suppressor genes are intimately associated with cellular autophagy (Figure 2). 11 An example of these includes the phosphatase and tensin homolog (PTEN) tumor-suppressor gene, which blocks the phosphatidylinositol-3 kinase (PI3K)/ protein kinase B (Akt)/mTORC1 cell signaling pathway, thus activating an autophagic response in cells (Figure 2). 11 However, mutations to PTEN, which are known to be very common in many cancers, can lead to lower levels of autophagy, with the subsequent elevated risk of cancer. However, there is also a downside, and the relationship between autophagy and cancer appears to be a double-edged sword, 58 since as some cancers progress, autophagy may actually facilitate tumor survival, just as autophagy appears to help the survival of all cells subjected to a stressful environment. 14 Some cancers, which may grow so quickly as to outstrip their own blood supply, may thus be aided by increased autophagy, as this would supply much needed energy and deal with stress. 48 Consequently, autophagy can have both prometastatic and antimetastatic roles and interacts with the other programmed cell death pathways (ie, apoptosis and necroptosis) in tumor progression. 59
The other area of intense research interest in hormesis and inducible autophagic processes is in the investigation of age-related neurodegenerative diseases including Alzheimer disease, Parkinson disease, and Huntington chorea. 32,39,51,60 -63 While all of these pathological conditions manifest differently, they all share one pathological similarity. All of these neurodegenerative diseases are characterized by excessive buildup of proteins and protein aggregates inside neurons leading to cell dysfunction and ultimately disease. 51 Consequently, the failure of protein degradation pathways by autophagy may play a very important role in the etiology of these diseases. However, the exact role of autophagy in these diseases is still uncertain. Further, growing research also implicates mitochondrial dysfunction and associated oxidative stress as a key pathway in the development of neurodegenerative diseases, as mentioned above for fibromyalgia. 54,55 Furthermore, Matai et al 64 have shown that endoplasmic reticulum hormesis improves proteostasis and viability in a mammalian cellular model of neurodegenerative disease.
Mitophagy is the selective autophagic targeting of defective or dysfunctional mitochondria. If the mitochondria are not working effectively, then the process of mitophagy targets them for lysosomal degradation. 54,55 The key regulators of this process include the tumor suppressor gene PTEN. 11 5′-Adenosine monophosphate-activated protein kinase (AMPK), for example, will also stimulate mitophagy, as well as new mitochondrial growth, essentially replacing old mitochondria with new ones in a subcellular renewal process (Figure 2). 11,65 Consequently, this is one of the reasons that metformin is often proposed as an antiaging compound, not so much for its antidiabetic blood-sugar level effects; but instead, because of its effect on AMPK and augmented autophagy (Figure 2). 11,25,65
As already mentioned, many antiaging interventions may show hormetic features, suggesting that preconditioning might have a preventive medical character (Figure 3 and Table 1). 8,34,66 -72 Hence, mild dietary stress (ie, CR without malnutrition or glucose restriction) achieved through various fasting regimes may exert its beneficial effects on life- and health span, at least in part, through hormetic mechanisms. 10,30,34,73,74 Glucose restriction has been shown to cause an increase in mitochondrial generation of ROS in a nematode model. 74 This mitochondrial ROS may in turn trigger an autophagic response via inhibition of mTORC1 (Figure 2). 75 -78 Furthermore, various repurposed approved drugs have been found to increase life span and health span in rotifers (Table 1). 71 Similarly, physical exercise may counteract aging by virtue of a hormetic dose–response relationship. Consequently, both lack of physical activity and overexercise are harmful, while regular but moderate exercise is beneficial, probably mediated by exercise generated ROS inducing mild oxidative stress leading to preconditioning (Figure 3). 8,66 -68

When exposed to mild stress, cells/organisms respond by a variety of pleiotropic adaptive cellular programs that procure a preconditioned state. 66,67 When an severe stress is applied subsequently, preconditioned but not naive cells/organisms exhibit stress resistance, hormesis, and eventually improved survival. mtROS indicates mitochondrial reactive oxygen species. Adapted from Zimmermann et al. 8
Summary List of Chemical Compounds With Life-Span and Health-Span Extending Properties.a

Abbreviations: Akt, serine/threonine kinase or protein kinase B; AMPK, 5′-adenosine monophosphate-activated protein kinase; AP1, activation protein 1; COX-1, cyclooxygenase 1; DNMT1, DNA methyltransferase 1; EGFR, epidermal growth factor receptor; ERK, extracellular signal-regulated kinase; FAK, focal adhesion kinase; GMP, guanosine monophosphate; GSK3, glycogen synthase kinase-3; LOX, lysyl oxidase; mTOR, mammalian target of rapamycin; NF-κB, nuclear factor κB; NOS, nitric oxide synthase; Nrf2, nuclear factor E2-related factor 2; PGC1-α, peroxisome proliferator-activated receptor-γ coactivator 1-α; PI3, phosphatidylinositol-3 kinase; PLA2, phospholipases A2; PPARγ, peroxisome proliferator-activated receptor γ; PTGS-2, prostaglandin-endoperoxide synthase 2; Scr, homeotic gene sex combs reduced; SIRT1, NAD-dependent deacetylase sirtuin-1; Sirt2, gene coding for SIRT2 - an NAD-dependent deacetylase sirtuin-2; TR, thioredoxin reductase TNF-α, tumor necrosis factor α
Hormesis appears to be executed by a variety of physiological cellular processes, including autophagy that cooperatively interact and converge on enhanced stress resistance and longevity (Figure 3). 8,32,69,81,82 Calabrese et al 68 and Zimmermann et al 8 have postulated that cells and organisms, preconditioned by exposure to mild stress, are protected against more harmful levels of stress and that the relevance of hormesis for both human pathophysiology and specific disease treatment is being increasingly recognized. 83 These authors have argued that this evolutionarily conserved process, hormesis, is directly linked to the capability to cope with pathological conditions, including aging and age-related diseases. They have also identified lysosomally mediated autophagy as a key component of this cytoprotective process. 8
Central Role of mTORC1 and Related Cell Signaling Pathways
The mTORC1 cell signaling system is primarily a nutrient sensor, however, it also interacts with ROS (Figure 2). The PI3K/Akt/mTORC1-related intracellular signaling pathways are essential for eukaryotic cells, since they are the link between nutrient status and growth (Figure 2). Mechanistic target for rapamycin complex 1 is the major cellular nutrient sensor to influence autophagic processes 14 and is inhibited by dephosphorylation. 11 Mechanistic target for rapamycin integrates signals from insulin, nutrients (amino acids or dietary protein), and the fuel gauge of the cell AMPK (all energy including fats) to determine whether the cell should divide and grow or become dormant. 11,65 Excess nutrients may stimulate the mTORC1 system, turning off autophagy and putting the body into a growth mode. This encourages growth of cells, which is generally not beneficial in most adult higher animals. 15 The mTORC1 pathway is also inhibited by ROS and vitamin D (Figure 2). 11,84
Diseases such as obesity, type 2 diabetes, Alzheimer disease, many cancers, atherosclerosis (heart attacks and strokes), polycystic ovarian syndrome, polycystic kidney disease, and fatty liver disease, among others, may be amenable to dietary intervention and mTORC1 inhibitors. 85,86 When the nutrient sensing systems detect low nutrient availability, they signal cells to stop growing and start breaking down unnecessary parts—this is the “self-cleansing” pathway of autophagy. 15,87 Consequently, diseases that involve excessive growth may be countered by reducing positive growth signaling through blocking these nutrient sensors.
Investigations of growth inhibition in humans are problematical because of the multiple interacting cell signaling pathways (Figure 2; Laplante and Sabatini, 2012). 88 However, the clearest evidence usually comes from drugs that target single pathways that can be altered one at a time. The mTORC1 inhibitors (eg, rapamycin, everolimus, various phytochemicals) activate autophagy by blocking mTORC1 (Table 1). 89 Some of these drugs are mainly used for their immune-suppressing effects in organ transplant medicine. However, most immune suppressants increase the risk of cancer, whereas rapamycin does not. In certain cancers, mTORC1 inhibitors have demonstrated anticancer effects (Table 1). 56
Metformin, a drug widely used in type 2 diabetes, also activates autophagy but not directly through the mTORC1 system (Table 1). 72 This drug increases AMPK, a protein molecule that signals the energy status of the cell (Figure 2). 90 High AMPK signals that the cell has insufficient energy and consequently augments basal autophagy. 65 The AMPK senses the ADP/ATP ratio, thus determining the cellular energy status. High AMPK levels directly activate augmented autophagy but also indirectly activate mitochondrial production. 65
Dephosphorylation of mTOR (inhibitory) and the consequent enhancement of autophagic activity are not per se sufficient to explain positive reactions, such as increased growth of the organism, to stressors. However, inhibition of mTOR (specifically mTORC1) does seem to represent a necessary step in order to eliminate the negative effects (ie, molecular and cell injury) of the stressor but the partial dephosphorylation of mTOR must in some way be coupled with a second reaction, perhaps involving AMPK and mitochondrial function, that is able to positively stimulate cell metabolism and physiology. 91 Mitochondrial hormesis (ie, mitohormesis) involving restoration of mitochondrial function and superoxide production via activation of AMPK has now been associated with improvement in markers of renal, cardiovascular, and neuronal dysfunction with diabetes. Consequently, approaches that stimulate AMPK and peroxisome proliferator-activated receptor-γ coactivator-1α (PGC1α) via exercise, CR, and medications result in stimulation of mitochondrial oxidative phosphorylation activity, restore physiological mitochondrial superoxide production, and promote organ healing. 91 The expression of PGC1α is highly inducible by physiological stressors, including exercise, cold, and fasting 92 ; and a central function of PGC1α is its intimate link to mitochondrial biogenesis and the detoxification of ROS. 93
Hormesis and Autophagy in Animal and Human Evolution
Over the geological time course of biological evolution, exposures to low concentrations of foreign or xenobiotic chemicals, both natural and pyrogenic (eg, from forest fires, volcanism, and cooked food), will have undoubtedly acted as selective pressures on the cytoprotective defences of biological systems (see review by Moore). 12 It is probably not unreasonable to propose that hormesis arose as a result of these selective pressures on the cellular interactions of toxic chemicals, their metabolites, and frequently generated reactive oxygen and nitrogen species with cell constituents. Such interactions will have included cell signaling networks that regulate xenobiotic biotransformation (phase I and II, esterases, etc) mitochondrial and lysosomal function, autophagy, and programmed cell death (see review by Moore). 29 Consequently, these interactions have resulted in the integrated toolbox of evolutionarily highly conserved physiological responses that is now recognized as comprising the phenomenon of hormesis.
Natural products can have both harmful and beneficial effects; and many of our dietary constituents contain micronutrients, some of which are considered to be nutraceuticals, such as many phenolic, polyphenolic, and isothiocyanate phytochemicals, which are also often biogenic pesticides (see reviews by Moore). 12,94 Many phenolic and polyphenolic phytochemicals and their metabolic derivatives, such as curcumin, resveratrol, dihydroresveratrol, and quercetin, are potent inhibitors of the PI3K/AKT/mTORC1 signaling pathway and inducers of autophagy (Table 1 and Figure 2). 12,80,94 -98
Modification of splicing factor expression by resveralogues (analogues of resveratrol), including dihydroresveratrol, a major gut microbiome metabolite of resveratrol that is readily absorbed, 99 was associated with the rescue of multiple features of senescence, making senescent cells not only look physically younger but start to behave more like young cells and start dividing (Table 1). 79 This hormetic antiaging effect is the first demonstration that moderation of splicing factor levels is associated with reversal of cellular senescence in human fibroblasts and suggests that small molecule modifiers of splicing factor expression could represent promising novel antidegenerative therapies.
Moore 94 hypothesized that inhalation and ingestion (with upper respiratory tract mucus) of certain natural products, such as aerosolized harmful algal toxins (phycotoxins), may have direct effects on the body’s molecular regulatory systems resulting in health benefits (eg, anti-inflammatory, anticancer, antiaging). 100 Recent support for this phytohormetic hypothesis has come from work by Asselman et al, 101 demonstrating that some phycotoxins in natural marine aerosols can inhibit mTOR and induce autophagy in cultured lung cells. Growth inhibition and apoptosis, both linked to mTOR pathway activity, may explain these effects, as yessotoxins were shown to downregulate this pathway. 102 Fungal toxins, such as aflatoxins and ochratoxins, can also induce autophagy and apoptosis and interact via cell signaling pathways; and there appears to be some evidence for hormetic effects. 103
Evidently, hormesis and phytohormesis resulting from exposure to small amounts of toxic pollutants or some naturally occurring biogenic chemicals (ie, from both prokaryotes and eukaryotes) can have a stimulatory effect on the various cellular or cytoprotective processes including autophagy (Table 1). 81,94,104 -107 Currently, this is an area of very active research, particularly in the context of the therapeutic potential for phytochemicals to behave as nutraceuticals and have anti-inflammatory, antidementias, antiaging, and anticancer properties (see reviews by Moore). 12,94
Many toxic materials are natural components of the environment and will have interacted with human biological systems over the time span of human and hominid evolution. Human hormetic cytoprotective systems will probably have benefitted from evolutionary changes in the longer term from exposure to toxic metals, organic xenobiotics, and toxic biogenic products. As mentioned previously, many metals and organic xenobiotics accumulate within lysosomes, where they can cause permeabilization of the lysosomal membrane resulting in the release of intralysosomal iron into the cytoplasm. 17,22,24,26,108 This released lysosomal iron generates ROS that can cause oxidative damage to cellular components 22,24,108 ; however, the ROS will also inhibit the PI3K/AKT/mTORC1 pathway thus inducing augmented autophagy. 21,31 Many of the phytochemicals that are considered to be potentially beneficial at low concentrations are in fact toxic at high concentrations: They are chemical defensive products of the evolution of protective mechanisms in plants to counter pathogens, parasites, and consumption by herbivores. 109 -112 Exposure to these biogenic products, particularly in foodstuffs, will probably have beneficially influenced the evolution of human cellular processes, particularly anti-inflammatory cell signaling mechanisms. 12,25
Exposure to toxins and natural biogenic products, such as phytochemicals, phycotoxins, mycotoxins, and bacterial toxins, has probably had a beneficial role as an evolutionary driver for cellular hormetic cytoprotective systems in humans and many other species (Table 1). 107,112 -117 The phase I and phase II drug metabolizing enzymes that biotransform many of the organic xenobiotics that enter the body, either by ingestion in food and cooked and burnt food (eg, pyrogenic generation of polycyclic aromatic hydrocarbons and arylamines) or by inhalation, have been selected over the course of hominid evolution. 112 -114,117 -119 These selective processes will probably have driven the human capability to detoxify many of the organic chemical pollutants (xenobiotics) encountered during recent industrial history as well as conferring the ability to activate the vast majority of pharmaceuticals used in current therapeutic applications. Antioxidant protection against reactive derivatives of toxic metal and xenobiotic exposure has also been subject to the same type of evolutionary pressures; and this has endowed humans with the remarkable defensive capacity that is a necessary requirement for surviving in a “sea of poisons.” 107,120,121 Perhaps low-level hormesis is actually the real physiological status of most organisms that live in the real environment, including humans and, consequently, are exposed to low levels of a myriad of stressors, rather than under laboratory controlled conditions. 2,94
Novel industrial chemical and nanomaterial products are being produced constantly, and some of these will undoubtedly present hazards for human and animal health, as the drug metabolizing system and other cytoprotective processes, such as autophagy and hormesis, will be confronted with completely new molecular and supramolecular structures, not previously encountered during the course of human and earlier evolution. 16,122 -124 However, recent research has indicated that metalliferous nanoparticles can be generated naturally in deep groundwater from geological metal ore deposits, 125 so natural exposure to these types of materials has probably been a factor in the evolution of cytoprotection. Risks to health as a result of exposure to nanomaterials, other than combustion particulates, are still largely unknown, although considerable concern has been expressed as to the safety of engineered nanomaterials. 31,39,83,126 Furthermore, there is now some evidence that engineered nanoparticles (eg, C60 fullerene and glass nanofibres) can inhibit mTORC1, induce augmented autophagy, and some may even induce pathological dysfunctional autophagy 21,24,31,108 ; and these materials are finding their way into the environment and may impact on animal and human food networks and possibly on human health. 127
Finally, breakdown of various plastic products can produce both nanoplastic and microplastic contamination of the natural environment; and there are increasing environmental health concerns regarding these materials. 128 -130 Microplastics have been shown to cause cell injury to the lysosomal system in marine animals (mussels) and also can bind potentially harmful persistent organic pollutants that will enter the human food chain with the microplastic. 23
Conclusion
Hormesis is intimately linked with autophagic responses as part of the overall repertoire of lysosomal function. Initiation of augmented autophagy via mTORC1 inhibition or AMPK provides the cell with essential nutrients in stressful scenarios; and these recycled nutrients facilitate survival and growth by supplying energy and building blocks for cellular repair during mild stress. Autophagic removal of unwanted damaged or misfolded proteins, harmful protein aggregates, damaged membranes, and even damaged parts of the genome will protect the cell and/or organism from further damage and aid recovery from environmental insults. 14,15,33,40,131
Autophagy and its regulation by mTOR cell signaling is evolutionarily very ancient and the genes involved are highly conserved from yeasts to humans. 33 This indicates that the interconnectivity between autophagy and hormesis probably emerged very early in the evolution of the eukaryotic lineages.
Finally, agents that can induce autophagy and hormetic type responses, such as fasting, exercise, aspirin, metformin, and phytochemicals as well as mimetics of metformin and rapamycin (rapalogs and CR-mimetics), 65,90,132,133 have considerable potential for significant impact in antiaging, cancer and neurodegenerative disease therapeutics.
Footnotes
Acknowledgments
This article is dedicated to the late Dr Tony Stebbing who introduced me to the concept of hormesis in 1973 and with whom I was privileged to work, during his seminal investigations of hormesis in coelenterates, induced by low concentrations of toxic metals. Credit is also due to my late colleague and friend, Dr Andreas Bubel, who first introduced me to the pathophysiology of lysosomes induced by environmental pollutants 50 years ago: without his keen insight, I would probably not have pursued this area of research for much of my research career. I also want to thank my colleagues of many years, Professor Aldo Viarengo (University of Genoa, Genoa, and Mario Negri Institute for Pharmacological Research—IRCCS, Milan, Italy) and Professor Dr Angela Köhler (Alfred Wegener Institute—AWI, Helmholtz Centre for Polar and Marine Research, Bremerhaven, Germany) for critically reviewing this manuscript. This work was supported by Plymouth Marine Laboratory, the University of Exeter Medical School—European Centre for Environment and Human Health, and the University of Plymouth—School of Biological & Marine Sciences.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received support in kind but no financial support for the research, authorship, and/or publication of this article.
