Abstract
Background:
The intra- and postoperative effects of dexmedetomidine are not completely consistent and midazolam/fentanyl is most widely used in peripheral surgeries. The objectives of the study were to evaluate the sedative, analgesic, hemodynamic, anti-inflammatory, and antioxidant effects of dexmedetomidine against midazolam in patients undergoing peripheral surgeries with mild traumatic brain injuries.
Methods:
Medical records of patients who underwent peripheral surgeries with mild traumatic brain injury were included in the analysis. Patients received intraoperative midazolam (MDZ cohort, n = 225) or dexmedetomidine (DEX cohort, n = 231). Pre-, intra-, and postoperative characteristics of patients were collected and analyzed.
Results:
After administration of anesthesia, up to 40 minutes, patients of the MDZ group had lower modified observer’s assessment of alertness/sedation score than those of the DEX group (P = .041), but after 40 minutes, patients of the MDZ group had a higher score than those of the DEX group throughout surgeries (P = 0.048). The DEX group has less requirements of postoperative morphine/equivalent doses than the MDZ group (4 ± 1 vs 5 ± 1, P < .0001, q = 18.451).
Conclusions:
Intraoperative DEX offers better sedation, postoperative analgesia, and clinical recovery for peripheral surgeries and suppresses inflammatory response.
Level of Evidence:
III.
Introduction
Hemodynamic stability, weaker stimuli, and intracranial homeostasis are needed to achieve during surgeries. 1 Disordered hemodynamics enables neuroendocrine response or immune response, which leads to the onset of stress hormones and inflammatory response. 2 Inflammatory cascade emerged by neuroendocrine hormones and pro-inflammatory mediators contributes to the pathology of traumatic brain injuries. 3 During the major surgeries, these adverse physiological responses can be observed due to lower perfusion pressure, increased intracranial pressure, surgery-related damage, and/or pain stimuli. 4 Sometimes secondary traumatic brain injuries, for example, cerebral hemorrhage or cerebral edema, may damage brain tissue, which is affected by the recovery from anesthesia and postoperative cognition. 5 Therefore, to alter the balance of inflammation and neuroendocrine responses, to reduce incidences of postoperative complications, and to prevent traumatic brain injuries, it is necessary to consider anesthesia management. 1 Anesthesia and surgery often for causes unrelated to the early phase after mild traumatic brain injuries. 6 A stage in which the brain is vulnerable to the secondary insults could be caused or exacerbated by the procedure. Systemic inflammation and oxidative stress are postulated to have a role in such secondary injuries, and they seem to increase following a surgical procedure. 7
Midazolam (MDZ) is a γ-aminobutyric acid receptor agonist type of benzodiazepine. 8 It is a commonly used anesthetic agent and does not affect neuronal growth. 9 It may protect against necroptosis and neuronal degeneration, which is induced by oxidative and physiological stress. 8 It exhibits dose-dependent respiratory depression. 10 Dexmedetomidine (DEX) has neuroprotective action by inhibiting neuroendocrine and inflammatory responses 1 and has a lack of respiratory depression. 10 However, the sedative, analgesic, and hemodynamic action of DEX is not completely consistent and MDZ with fentanyl is most widely used in peripheral surgeries. Anesthetics need a drug in peripheral surgeries that can be used safely with less severe adverse effects. 11 Therefore, there is a need for retrospective analysis to estimate the sedative, analgesic, hemodynamic, anti-inflammatory, and antioxidant role of DEX and MDZ before nonrandomized, single-blind, controlled trial.
The objectives of the retrospective analysis of prospectively collected data were to evaluate the sedative, analgesic, hemodynamic, anti-inflammatory, and antioxidant effects of DEX against MDZ in patients undergoing peripheral surgeries with mild traumatic brain injuries.
Materials and Methods
Ethics Approval and Consent to Participate
The designed protocol (GPPH/CL/15/19 dated January 12, 2019) of the established study had been approved by the Guizhou Provincial People’s Hospital review board. The study had adhered to the law of China, Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement, and the Declaration of Helsinki (V2008). An informed consent form was signed by all participating patients regarding anesthesia, surgeries, pathology, and publication of the study during hospitalization.
Inclusion Criteria
Patients available at trauma care center due to sports injuries (eg, football, basketball, table tennis, and badminton), assaults, and falls (slipping, falling from a staircase, and falling from the ceiling) and underwent anesthesia and the surgical procedure (surgeon and emergency department physicians’ decision) with mild traumatic brain injury (Glasgow Coma Scale 13-15 and the abnormal computed tomography scan like epidural hematoma, contusion, parenchymal hematoma, subarachnoid hemorrhage, subdural hematoma, and/or a skull fracture) were included in the study. Patients who had an American Society of Anesthesiology (ASA) status II or III were only included in the study.
Exclusion Criteria
Patients who faced motor vehicle accidents (automobile, boating, or motorcycle due to major brain injuries), a penetrating head injury (Glasgow Coma Scale less than 13), and duplicate anesthetic records were excluded from the study. Patients younger than 18 years of age and those who had ASA status I (no brain injuries), IV (a constant threat to life), or V (not expected to live 24 hours) were excluded from the study.
Data Collection
For each enrolled patient, the demographic characteristics, preoperative and postoperative pain conditions, type of surgeries, anesthesia used, intraoperative conditions, postoperative parameters, adverse effects, and clinical outcomes during the follow-up period were collected from medical records of institutes after written permission from the competing authorities.
Cohorts
Patients who received 49.9 µG/kg MDZ (Hospira, Inc, Lake Forest, Illinois) and 1 µG/kg fentanyl (Siegfried Hameln, Hameln, GmbH, Germany) in 25 mL of normal saline (Baxter Healthcare Corporation, Deerfield, Illinois) infused over 12 minutes, followed by a continuous infusion of 50 µG/kg/h MDZ in normal saline till the end of the surgery, were included in the MDZ cohort. Patients who received 0.999 µG/kg DEX (Precedex; Hospira, Inc) and 1 µG/kg fentanyl in 25 mL of normal saline infused over 12 minutes and then a continuous infusion of 1.0 µG/kg DEX in normal saline till the end of the surgery were included in the DEX cohort. A total of 100 mL intraoperative paracetamol (10 mg/mL; Accord Healthcare Ltd, North Harrow, United Kingdom) was injected to all patients. The decision of interventions (MDZ or DEX) in the predesign of the study was made based on the age of patients, necessities of sedation and analgesia, availabilities of medications, and the other demographical and clinical condition(s) of patients by anesthesiologists (a minimum of 3 years’ experience) of institute in consultation with surgeons (a minimum of 3 years’ experience) of institute who performed surgeries.
Anesthesia Method
Intravenous access was opened, internal jugular vein and radial artery puncture were performed under local lidocaine (Xylocaine Spray; AstraZeneca UK Limited, Macclesfield, Cheshire, United Kingdom), and a catheter (Roche, Basel, Switzerland) was inserted and central venous pressure and arterial venous pressure were monitored; 2 mg/kg propofol (Diprivan; AstraZeneca, Cambridge, United Kingdom), 0.1 mg/kg vecuronium (Flexivec; Bharat Serums and Vaccines Limited, Thane, India), and 4 mg/kg ketamine (Hospira, Pfizer Inc, Richmond, Virginia) were injected. 7 Anesthesia procedure was performed by anesthesiologists (minimum 3 years’ experience) of the institute. Bispectral index values were targeted 45 to 70 during operation. For recovery of anesthesia, the infusion of normal saline with DEX or MDZ was titrated until the end of surgery. 12
Sedative Effects
The sedation was measured using a modified observer’s assessment of alertness/sedation score. The scores are as follows: 5: response readily to name spoken; 4: lethargic response; 3: response after name called loudly; 2: response after mild to moderate shaking; 1: response to trapezius squeeze. 13 Observer’s assessment of alertness/sedation score was evaluated by anesthesiologists (a minimum of 3 years’ experience) of the institute in consultation with surgeons (a minimum of 3 years’ experience) of the institute who performed surgeries. Bispectral index values and recovery criteria were recorded by anesthesiologists (a minimum of 3 years’ experience) of the institute at various stages of operation.
Postoperative Pain Assessment
Postoperative pain was evaluated using visual analog scale (VAS) 24 hours after surgeries. 0: no pain and 10: the maximum possible pain. 14 Visual analog scale was administered to the patients by nursing staff (a minimum of 3 years’ experience) of the institute who was not involved in surgeries.
Laboratory Parameters
Blood samples were collected from all participants by pathologists (a minimum of 3 years’ experience) of the institute 24 hours before surgery and 24 hours after surgery and serum was isolated from samples. Serum interleukin 6, superoxide dismutase, S100β (S100 calcium-binding protein β), tumor necrosis factor α, neuron-specific enolase enzyme, and malondialdehyde were evaluated using enzyme-linked immunosorbent assay. 7 All assays were performed by pathologists (a minimum of 3 years’ experience) of the institute.
Hemodynamic Parameters
Mean arterial pressure and heart rate were evaluated before anesthesia induction and 5 minutes after tracheal intubation.
Postoperative Pain Management
A total of 100 mL/d postoperative paracetamol (10 mg/mL) for 3 consecutive days was injected to all patients. Postoperative morphine (Duramorph; Baxter Healthcare Corporation, Deerfield, Illinois) or equivalent was administered as and when required (when VAS ≥3; the decision of surgeon(s)). The nursing staff (a minimum of 3 years’ experience) of the institute in consultation with surgeons (who performed surgeries) were involved in postoperative pain management.
Clinical Outcome Measures
During the follow-up period, clinical outcomes of patients were reported at institutes that were used for analysis.
Statistical Analysis
SPSS version 25 (IBM Corporation, Armonk, New York) was used for the purpose of statistical analyses. The Fisher exact test was performed for ordinal data, and Mann-Whitney U test was performed for numerical data 6 between the groups. A 1-way repeated measures analysis of variance was performed for numerical data within a group. Tukey test (considering critical value [q] > 3.314 as significant) was performed for post hoc analysis. All results were considered significant at a 95% confidence level.
Results
Enrollment
From January 1, 2014, to December 12, 2018, medical records of 627 patients with mild brain injuries undergoing anesthesia and peripheral surgeries available at the trauma care center of the Guizhou Provincial People’s Hospital, Guiyang, Guizhou, China, and the referring hospitals were reviewed. Among the available records, 62 patients had faced automobile accidents (major brain injuries), 13 patients had faced boating accidents (major brain injuries), 91 patients had faced motorcycle accidents (major brain injuries), and 5 patients had penetrating head injuries. Therefore, these patients were not included in the analysis because they had major brain injuries. A total of 456 patients were included in the analysis. Patients who received MDZ were included in the MDZ cohort (n = 225) and those who received DEX were included in the DEX cohort (n = 231). The flowchart of the analysis is presented in Figure 1.

Flowchart of the study.
Demographic Characters
There was no significant difference in anthropological, demographical, and clinical conditions of the patients, preoperative pain, heart rate, surgical characteristics, duration of surgery, and volume of propofol used during surgery between the 2 study groups (P > .05 for all) except age. Patients included in the MDZ group were younger than those of the DEX group (P < .0001). A higher percentage of patients with ASA status II (P = .048) and female patients (P = .042) were treated with MDZ (Table 1).
Anthropological, Demographical, and Clinical Conditions of the Patients.a

Abbreviations: DEX, dexmedetomidine; MDZ, midazolam; N/A, not applicable.
a Ordinal data are presented as frequency (percentage) and numerical variables are represented as mean ± standard deviation. The Fisher exact test was performed for ordinal variables and the Mann-Whitney U test was performed for numerical variables for statistical analysis. A P < .05 was considered significant.
b Significant difference.
c 0: no pain and 10: the worst pain.
Sedative Effects
After administration of anesthesia, up to 40 minutes, patients of MDZ had lower modified observer’s assessment of alertness/sedation score than those of the DEX group (P = .041), but after 40 minutes, patients of the MDZ group had higher modified observer’s assessment of alertness score than those of the DEX group throughout surgery (P = .048; Figure 2).

Modified observer’s assessment of alertness/sedation score during operation. 5: Response readily to name spoken; 4: lethargic response; 3: response after name called loudly; 2: response after mild to moderate shaking; 1: response to trapezius squeeze. Observer’s assessment of alertness/sedation score was evaluated by anesthesiologists (a minimum of 3 years’ experience) of the institute in consultation with surgeons (a minimum of 3 years’ experience) of the institute who performed surgeries.
There were no significant changes for the bispectral index value between both groups at various stages of operation (Table 2). While eye-opening time and time required for the verbal response of patients were lesser for the DEX group than the MDZ group (Table 3).
Comparisons of Bispectral Index Value.a

Abbreviations: DEX, dexmedetomidine; MDZ, midazolam.
a Variables are presented as mean ± standard deviation. The Mann–Whitney U test was performed between groups. A P < .05 was considered significant.
Anesthesia Recovery Criteria.a

Abbreviations: DEX, dexmedetomidine; MDZ, midazolam.
a Variables are presented as mean ± standard deviation. The Mann-Whitney U test was performed between groups. A P < .05 was considered significant.
b Significantly lesser than the MDZ group.
Analgesic Effects
One day after surgeries, MDZ (P < .0001, q = 21.278) and DEX (P < .0001, q = 27.899) were successfully decreased postoperative pain but DEX decreased pain more strongly than MDZ (P = .013, q = 3.552; Figure 3). Also, the DEX group has less requirements of total morphine or equivalent doses administration than the MDZ group (P < .0001, q = 18.451; Figure 4).

Analgesic effects of anesthetics. Variables are presented as mean ± SD. A 1-way repeated measures ANOVA was performed within a group. The Mann-Whitney U test was performed for statistical analysis between groups. Tukey test was used for post hoc analysis. A P < .05 and q > 3.314 were considered significant. *Significantly lower than BL. §Significantly lower than the MDZ group at EL. 0: no pain and 10: the worst pain; BL: 24 hours before surgery, EL: 24 hours after surgery. The visual analog scale was administered to the patients by nursing staff (a minimum of 3 years’ experience) of the institute who was not involved in surgeries. ANOVA indicates analysis of variance; MDZ, midazolam; SD, standard deviation.
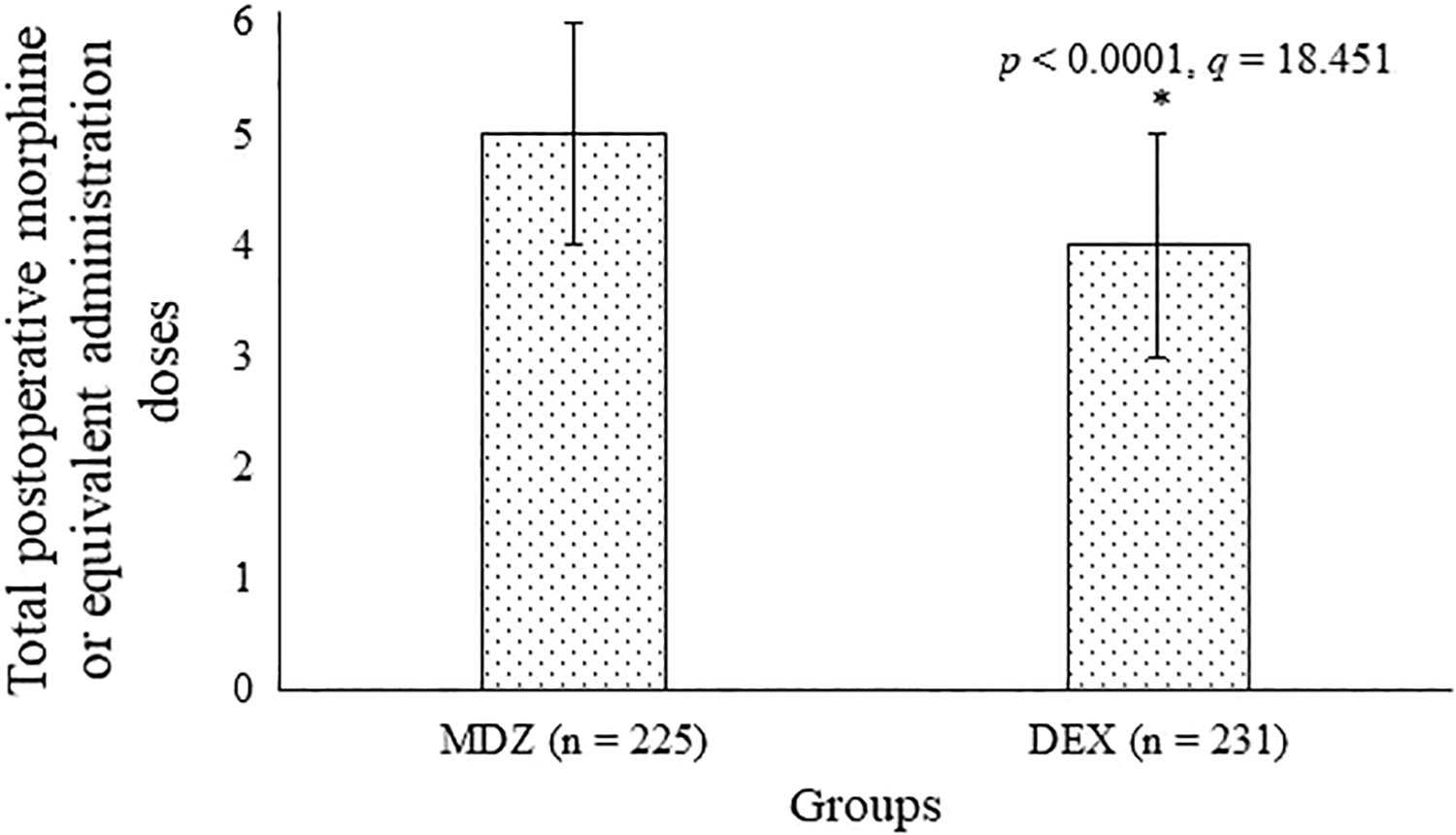
Total postoperative morphine or equivalent administration dose assessment. Variables are presented as mean ± SD. The Mann-Whitney U test following the Tukey test was used for statistical analysis. A P < .05 and q > 3.314 were considered significant. *Fewer requirements than MDZ group. MDZ indicates midazolam; SD, standard deviation.
Hemodynamic Parameters
Changes in mean arterial pressure and heart rate are reported in Table 4. Dexmedetomidine and MDZ both caused hypotension and decreased heart rate.
Hemodynamic Parameters.a

Abbreviations: DEX, dexmedetomidine; MDZ, midazolam.
a BL: Before anesthesia induction, EL: 5 minutes after tracheal intubation. Variables are presented as mean ± standard deviation. A 1-way repeated measures analysis of variance was performed within the group. The Mann-Whitney U test was performed between groups. Tukey test was used for post hoc analysis. A P < .05 and q > 3.314 were considered significant.
Laboratory Parameters
One day after surgeries, MDZ (P < .0001, q = 20.22) and DEX (P = .016, q = 4.081) reduced superoxide dismutase level, but the intensity of MDZ to reduce superoxide dismutase level was higher than DEX (P < .0001, q = 22.87). Midazolam (P = .0003, q = 5.137) increased, but DEX (P = .371) had no effect on serum interleukin 6 level. Midazolam (P < .0001, q = 12.584) and DEX (P = .012, q = 3.641) increased tumor necrosis factor α level, but the intensity of MDZ to increase tumor necrosis factor α level was higher than DEX (P < .0001, q = 6.956). Midazolam (P < .0001, q = 21.169) and DEX (P < .0001, q = 6.314) increased level of malondialdehyde, but the intensity of MDZ to increase the level of malondialdehyde was higher than DEX (P < .000, q = 14.789). Midazolam (P < .0001, q = 80.135) and DEX (P < .0001, q = 42.055) increased level of S100β, but the intensity of MDZ to increase S100β was higher than DEX (P < .0001, q = 35.545). Midazolam (P < .0001, q = 54.591) and DEX (P < .0001, q = 34.046) increased serum level of neuron-specific enolase enzyme, but MDZ increased more strongly serum level of neuron-specific enolase enzyme than DEX (P = .0002, q = 6.024; Table 5).
Laboratory Parameters.a

Abbreviations: DEX, dexmedetomidine; MDZ, midazolam; N/A, not applicable.
a BL: 24 hours before surgery, EL: 24 hours after surgery. Data are presented as mean ± standard deviation. A 1-way repeated measures analysis of variance was performed within the group. The Mann-Whitney U test was performed between groups. Tukey test was used for post hoc analysis. A P < .05 and q > 3.314 were considered significant.
b Not increased compared to BL.
Adverse Effects
Dexmedetomidine cause hypotension (P = .033) and bradycardia (P = .019) and MDZ leads to respiratory events (P = .003) as adverse effects during hospitalization (Table 6).
Adverse Events During Hospitalization.a

Abbreviations: DEX, dexmedetomidine; MDZ, midazolam.
a Ordinal data are presented as frequency (percentage) and numerical data are presented as mean ± standard deviation. The Fisher exact test was performed for ordinal data and the Mann-Whitney U test was performed for numerical data. A P < .05 was considered as significant.
b Significant dexmedetomidine-emergent adverse effect.
c Significant midazolam-emergent adverse effect.
Clinical Outcome Measures
Wound healing time was less in the DEX group than the MDZ group (P < .0001) and professional activity recovery was higher in the DEX group than in MDZ group (P < .0001). The other clinical outcomes had no significant difference between both groups during the follow-up period (Table 7).
The Intermediate- and Long-Term Sequela During the Follow-Up of Patients.a

Abbreviations: DEX, dexmedetomidine; MDZ, midazolam.
a Ordinal data are presented as frequency (percentage) and numerical data are presented as mean ± standard deviation. The Fisher exact test was performed for ordinal data and the Mann-Whitney U test was performed for numerical data. A P < .05 was considered as significant.
b Significant difference than the MDZ group.
Discussion
After administration of MDZ, up to 40 minutes, patients had lower modified observer’s assessment of alertness/sedation score than those who received DEX, but after 40 minutes, patients who received MDZ had a higher score than those who received DEX throughout the surgery. Also, anesthesia recovery criteria were better for the DEX group than the MDZ group. The onset of action time for MDZ was 0.5 to 1 minutes, and it takes 5 minutes to reach peak plasma MDZ concentration, but the onset of action time for DEX is 10 to 15 minutes and it takes 25 to 30 minutes to reach peak plasma DEX concentration. 14 The results of the current study were in parallel with the results of prospective randomized study 12 and a randomized trial of patients who planned for total knee arthroplasty. 15 Dexmedetomidine could provide the desired level of sedation than MDZ without significant respiratory distress.
Dexmedetomidine had higher control over postoperative pain than MDZ. The results of the study were in parallel with the results of a randomized clinical trial of patients undergoing dental implant surgeries. 14 Midazolam has itself no analgesic property, but due to γ-aminobutyric acid agonist action, it assists the analgesic effect of fentanyl. 16 While DEX has an analgesic effect because it has selective α-2 agonist action. 14 Dexmedetomidine offers better postoperative analgesia.
The study reported that hemodynamic parameters were not stable in patients who received DEX or MDZ. The results of the study were in parallel with the results of a randomized clinical trial on glioma patients undergoing craniotomy resections, 7 double-blind prospective clinical study in supratentorial craniotomies, 17 prospective randomized studies on elective surgery, 12,15 but were not consistent with the randomized clinical trial of patients undergoing dental implant surgeries. 14 In normal physiological conditions, there is self-regulation in the cerebral blood flow, but surgeries can disturb self-regulation in the cerebral blood flow; as a result, the perfusion of the brain decreases and intracranial pressure increases and therefore damages the brain. 7 Dexmedetomidine is a highly selective α-2 agonist and a selective α-1 agonist. 18 Dexmedetomidine acts on α-1 receptors of peripheral vascular smooth muscle, causes peripheral vasoconstriction, and transiently increases blood pressure when administered very quickly. 7 Dexmedetomidine and MDZ are not stabilized arterial pressure and heart rate.
The study reported that the intensity of DEX to increase tumor necrosis factor-α level was lower than MDZ (P < .0001). Also, DEX had no effect on serum interleukin 6 level. The results of the study were consistent with randomized clinical trials on craniotomy resections and dental surgeries. 7,14 Surgeries can cause local trauma, which can stimulate chemotaxis of monocyte, lymphocytes, and neutrophils that express inflammatory factors and induce the inflammatory cascade by direct or indirect pathways, which can lead to brain injury. 1 The expression levels of inflammatory cytokines induced by surgeries can effectively prevent DEX.
Dexmedetomidine had a weaker effect on contrasting malondialdehyde but had a stronger effect on increasing superoxide dismutase due to surgery when compared to MDZ. The results of the study were consistent with randomized clinical trials on craniotomy resections and dental surgeries. 7,14 Malondialdehyde and superoxide dismutase are antioxidants and protect the body from oxidative damage. 7 Midazolam recovers oxidative stress, hypoxia, and brain injuries. 8 Midazolam and DEX may be effective in the reduction of brain injuries.
The intensity of MDZ to reduce neuron-specific enolase enzyme level and S100β was lower than DEX. The results of the study were in parallel with the results of a randomized clinical trial on glioma patients undergoing craniotomy resections. 7 Neuron-specific enolase enzyme level and S100β are biomarkers of brain injuries and express the level of brain injuries. 7 Dexmedetomidine could have stronger neuroprotectant effects than MDZ.
The study reported hypotension and bradycardia due to DEX. The results of the study were in parallel with the results of randomized clinical trials on dental implant surgeries, 14 imaging performance study, 19 critically ill patients who require mechanical ventilation, 20 and patients during spinal anesthesia. 12 A detailed study is required for the use of DEX in hypertensive patients during peripheral surgeries.
There are several limitations to the study, for example, the study is a retrospective cohort study and lack a prospective, randomized trial. The placebo control group is required to ascertain the “naive” increase in biomarkers due to surgery, 7 but the lack of a control group. The other limitations are the single-center study, the lack of other medications to compare such as propofol, the lack of data on brain injury intensity, lack of brain monitoring. Age, sex, and clinical conditions have a significant impact on intraoperative and postoperative conditions of patients, for example, older patients may have more hypotension and bradycardia and perhaps have a different inflammatory response than younger ones, 4 but the study did not evaluate the effects of demographical and clinical characters on outcome measures. The results did not clearly indicate the relationship for the betterment of surgical procedure, which provided only mechanistic clues. An in-depth study is required to elucidate the complete mechanism of DEX.
Conclusions
Midazolam offers rapid intraoperative sedation and suppresses oxidation responses. Dexmedetomidine offers better intraoperative sedation, postoperative analgesia, and better clinical recovery with manageable adverse effects and suppresses oxidative and inflammatory response during major peripheral surgeries. Dexmedetomidine may be an alternative sedative to MDZ in peripheral surgeries. The hypothesis is required to test for other surgeries.
Supplemental Material
Supplemental Material, STROBE_Statement - Intraoperative Dexmedetomidine Versus Midazolam in Patients Undergoing Peripheral Surgery With Mild Traumatic Brain Injuries: A Retrospective Cohort Analysis
Supplemental Material, STROBE_Statement for Intraoperative Dexmedetomidine Versus Midazolam in Patients Undergoing Peripheral Surgery With Mild Traumatic Brain Injuries: A Retrospective Cohort Analysis by Jing Peng, Fujuan He, Chenguang Qin, Yuanyuan Que, Rui Fan and Bin Qin in Dose-Response
Footnotes
Authors’ Note
Jing Peng and Fujuan He contributed to this work equally. All authors read and approved the manuscript for publication. J.P. and F.H. both were equally contributed to the project administration, conceptualization, and literature review of the study and drafted, reviewed, and edited the manuscript for intellectual content. C.Q. contributed to software, formal analysis, validation, and literature review of the study. Y.Q. contributed to resources, data curation, investigation, and literature review of the study. R.F. contributed to software, data curation, validation, and literature review of the study. B.Q. contributed to data curation, software, formal analysis, and literature review of the study. The authors agree to be accountable for all aspects of work, ensuring integrity and accuracy. The data sets used and analyzed during this study are available from the corresponding author on reasonable request.
Acknowledgments
The authors are thankful for the medical and nonmedical staff of Guizhou Provincial People’s Hospital, Guiyang, Guizhou, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Youth fund of guizhou provincial people's hospital (No: GZSYQN[2018]13).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
