Abstract
This study evaluated the ameliorative and prophylactic effects of 2 different doses of polyphenolic-rich fraction of Garcinia kola (PPRFGk) seeds on the histology and hormones of pituitary–testicular axis of male Wistar rats. Thirty-five male Wistar rats (150-200 g) were divided into 7 groups of 5 rats each. Groups I and II were given distilled water (0.5 mL/day) for 8 days followed by propylene glycol (0.2 mL/d) and 600 mg/kg of PPRFGk, respectively, for 21 days. Group III received sodium arsenate (8 days), left untreated for 21 days. Groups IV and V received sodium arsenate (20 mg/kg) for 8 days followed by PPRFGk (300 and 600 mg/kg, respectively) for 21 days. Groups VI and VII received PPRFGk (300 and 600 mg/kg, respectively) for 21 days followed by sodium arsenate (20 mg/kg) for 8 days. Rats were killed by cervical dislocation 24 hours after the last dose and their blood collected through cardiac puncture. Blood sera were assayed for the levels of follicle-stimulating hormone (FSH), luteinizing hormone (LH), and testosterone using immunoassay techniques. Histology of the pituitary gland and testes was carried out. A significant reduction was observed in the concentration of FSH in groups IV, V, VI, and VII in comparison with groups I and II. The concentrations of both LH and testosterone showed significant decreases in groups IV, V, VI, and VII in comparison with group I. Group III presented with the lowest serum hormonal concentrations. Photomicrographs of the pituitary gland revealed greatly reduced basophils in group III and mildly reduced basophils in groups IV, VI, and VII in comparison with groups I and II. Group V revealed hypercellularized and distorted basophils. Photomicrographs of the testes showed detachment of the seminiferous tubules from the basement membrane and disruption of the interstitial space which was worse in group III, moderate in groups V and VI, and mild in group VII. In conclusion, PPRFGk effected a dose-dependent reversal and prevention of the perturbations caused by arsenate in rats.
Keywords
Introduction
Arsenic acid, a metalloid, is a systemic poison, and chronic ingestion of arsenic has been reported to lead to a wide range of health problems, which are collectively called arsenicosis or chronic arsenic poisoning. 1 Arsenic acid exists either as trivalent arsenite (As3+) or pentavalent arsenate (As5+), is naturally occurring, and is ubiquitously present in the environment. Arsenic compounds are environmental toxins with multiple effects on animal and human populations. 1,2 It is commonly used in agro-allied formulations, such as wood preservatives, herbicides, fungicides, and rodenticides, and may cause air, soil, and water pollution. Humans are exposed to arsenic mainly through oral or inhalation routes. Oral exposure occurs via consumption of contaminated water, food, and drugs, and such exposure can be lifelong. Occupational exposure, on the other hand, occurs mainly through inhalation via nonferrous ore smelting, semiconductor and glass manufacturing, or power generation by the burning of arsenic contaminated coal. 1 The main source of environmental arsenic exposure in most populations is the drinking water in which inorganic forms of arsenic predominate. 3,4 Numerous studies, evidence in both clinical and animal models, have provided experimental evidence that arsenic-induced toxicity is mediated via the generation of reactive oxygen species (ROS). 5
The pituitary–gonadal axis, a subset of hypothalamic–pituitary–gonadal axis, has been reported to be vulnerable to a number of trace metals’ toxicity including arsenate. 6 Inorganic arsenate has been reported to have a suppressive influence on spermatogenesis, androgenesis in the male reproductive system, and ovarian steroidogenesis in the female reproductive system. 7 However, the incipient molecular events leading to these reproductive dysfunctions from exposure to arsenate have not been equivocally elucidated. Garcinia kola Heckel (family, Guttiferae), commonly known as “bitter kola,” is a highly valued ingredient in African traditional medicine. Garcinia kola is regarded as a wonder plant because every part of the plant (bark, leaves, root, and wood) has been found to be of medicinal value. 8 The medicinal importance of bitter kola is based mainly on the phytochemicals of the plant. The seed of G kola has been reported to be rich in flavonoids, a class of polyphenols, 9 which has been reported to be anti-inflammatory, antiproliferative, and antibacterial. 10 Also, several lines of literature have demonstrated that plant-derived polyphenolic compounds do possess antioxidative properties which can mitigate the damaging effect of ROS and by so doing promote good health in humans. 11 Consequently, a number of evidence-based studies have implicated a strong correlation between the ingestion of fruits, vegetables, and medicinal plant rich in polyphenols with antioxidant potential and the reduced risk of oxidative stress-related disorders. 12,13
Medicinal herbs are moving from fringe to mainstream use with a greater number of people seeking remedies and health approaches free from side effects occasionally caused by synthetic chemicals. Therefore, there is a need to explore the effect of polyphenols from a common masticatory, G kola, in West Africa on arsenate-induced damage using rat samples.
Materials and Methods
Animal Selection and Care
A total of 35 male Wistar rats (150-200 g) were used for this study. They were obtained from the Animal House of the College of Health Sciences, Obafemi Awolowo University, Ile-Ife, and were allowed to acclimatize over a period of 2 weeks. The rats were housed in plastic cages with food and water provided ad libitum.
Preparation of Polyphenolic-Rich Fractions of G kola
Garcinia kola seeds were obtained commercially from Olubushe, central market in Ile-Ife, and certified at the herbarium, Obafemi Awolowo University, Ile-Ife. A voucher specimen with a voucher no: IFE 17285 was deposited there.
The seeds were air-dried under room temperature in order to prevent the destruction of its important phytochemicals from direct sunlight. A polyphenolic-rich fraction of G kola (PPRFGk) was prepared according to the procedure described by Sakulnarmrat and Konczak, 14 with a slight modification as follows: About 35 g of dehusked G kola seed was ground into a coarse powder, after which 30 g of the powder percolated with stirring in 5 volumes of acidified methanol (80% methanol, 19% H2O, and 1% acetic acid, vol/vol/vol) for 2 hours at 4°C. The mixture was then centrifuged for 20 minutes at 10 000 rev/min at 4°C (Centurion Scientific Ltd, model 8880, Serial No 951184, West Sussex, United Kingdom). The supernatant was collected and the residue extraction was repeated twice. The third extraction was carried out overnight (16 hours). The supernatant from the consecutive extractions was combined and the solvent evaporated under reduced pressure at 40°C using a rotary evaporator (Rota Vapor R-205; Büchi, Zurich, Switzerland). The concentrated crude alcoholic extract was then dissolved in acidified water (99% H2O, 0.1% acetic acid, vol/vol) and loaded into an XAD-16 resin column (450 mm × 25 mm Inner Diameter of the Resin Column [id]) that had been washed with acidified water. The column was then eluted with 80% ethanol. The resulting fraction was dissolved in propylene glycol (0.2 mL/administration) and given to rat according to the designed dosage.
Chemicals
Sodium arsenate salt was purchased from British Drug House, London, United Kingdom and dissolved in distilled water; 0.5 mL of the solution, equivalent to 20 mg/kg, was administered to each rat via oral route.
Experimental Design
Rats were divided into 7 groups of 5 rats each, which were treated as follows: groups I and II were given distilled water (0.5 mL/d) for 8 days followed by propylene glycol (0.2 mL/d) to group I and 600 mg/kg of PPRFGk to group II for 21 days. Group III received sodium arsenate (20 mg/kg) for 8 days and was left untreated for 21 days. Groups IV and V were given sodium arsenate (20 mg/kg) for 8 days followed by 300 and 600 mg/kg of PPRFGk, respectively, for 21 days. Groups VI and VII received PPRFGk (300 and 600 mg/kg, respectively) for 21 days followed by sodium arsenate (20 mg/kg) for 8 days.
Measurement of Food Consumption and Body Weight
During the course of the study, food consumption of the rats was measured daily and their body weights were measured weekly. The amount of food consumed was calculated using the formula: Weight of pellets given to rats per day − Weight of the leftover pellets the next day. The weights were obtained using a sensitive weighing balance (Mettler Toledo, Mg 126, Purchased by Efere M. Obuotor).
Animal Sacrifice and Collection of Blood, Brain, and Reproductive Organs
Twenty-four hours after the completion of treatment, the rats were sacrificed by cervical dislocation and their blood collected through cardiac puncture into separate plain bottles. The blood samples were centrifuged at 4000 rpm for 15 minutes using a cold centrifuge (Centurion Scientific Ltd, model 8880, Serial No 951184). The serum was separated and kept refrigerated until when needed. Rats were dissected and their brain tissues and testes excised. The pituitary gland was separated from the brain and fixed in formal saline for histological procedure. The weights of the testes were measured using Mettler Toledo (Mg 126) and fixed also for routine histological procedure.
Estimation of Serum Follicle-Stimulating Hormone, Luteinizing Hormone, and Testosterone
The serum obtained was assayed for the levels of follicle-stimulating hormone (FSH), luteinizing hormone (LH), and testosterone using enzyme immunoassay (EIA) technique. The EIA test kits were purchased from Teco Diagnostics (Anaheim, California), with catalog numbers FSH-96, LH-96, and TEST-96 for the respective hormones. The manufacturer’s procedures were strictly followed.
Histological Procedure
After the testes and pituitary gland were excised, they were immediately fixed in 10% formal saline and processed for routine histology. Paraffin sections of 5-µm thickness were obtained from the tissues using a Leica rotary microtome (Bright B5143, Huntingdon, England). Sections were stained with hematoxylin–eosin and viewed under the microscope with photomicrographs taken with a Leica DM750 microscope and interfaced with Leica ICC 50 camera.
Statistical Analysis
The results obtained were expressed as mean ± standard error of the mean. The data were analyzed using 1-way analysis of variance followed by Tukey multiple comparison tests using GraphPad 5.03 (GraphPad Software Inc, California). The results were considered significant when P < .05.
Results
Percentage Yield of PPRF of G kola Seeds
The mean yield of polyphenolic-rich fraction obtained from the seeds of G kola after 4 extraction runs was 3.22 ± 0.35 g, which represented a mean percentage yield of 6.43% ± 0.70% (wt/wt) based on the dry weight of the seeds (Table 1).
Percentage Yield of PPRF of Garcinia kola Seeds.
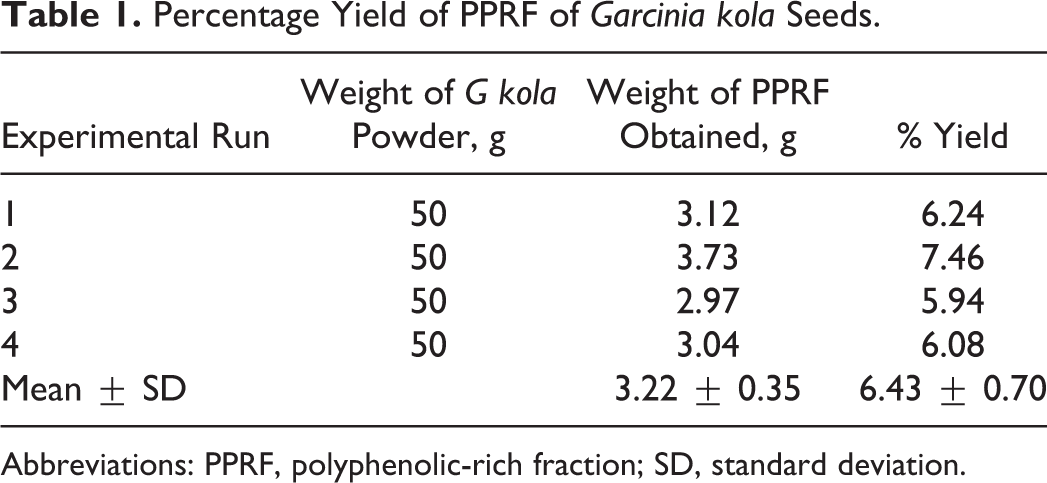
Abbreviations: PPRF, polyphenolic-rich fraction; SD, standard deviation.
Average Daily Food Consumption of Rats
The results indicate that there was a significant (P > .05) increase in feed intake in the group pretreated with 600 mg/kg of PPRFGk (group VII) when compared with the vehicle control group. Likewise, no statistically significant difference was observed in the feed intake of rats in groups II, III, IV, V, and VI in comparison to group I, but the PPRF pretreatment model (groups VI and VII) exhibited a significant increase in feed intake when compared with those in the PPRF posttreatment models (groups IV and V), as shown in Table 2.
Average Daily Food Consumption of Rats.

Abbreviations: As, arsenate; bw, body weight; PPRF, polyphenolic-rich fraction.
aSignificantly different from group II (P < .05).
bSignificantly different from group IV (P < .05).
cSignificantly different from group III (P < .05).
dSignificantly different from group I (P < .05).
Changes in Body Weight of Rats
It was observed that there was no significant difference in the body weight changes of rats in the control group when compared with the experimental groups. However, a significant increase in body weight changes was observed in group VII when compared to groups III, IV, and V, as shown in Table 3.
Body Weight Gain of Control and Experimental Rats.a

Abbreviations: As, arsenate; bw, body weight; PPRF, polyphenolic-rich fraction.
aMean ± standard error of the mean (SEM).
bMeans significantly different from group VII.
cMeans significantly different from group II.
Changes in Testicular Weight of Control and Experimental Rats
Figure 1 showed that there were no significant (P > .05) differences between groups I and II. From the chart (Figure 1), the bars of the posttreated groups, groups IV and V, and pretreated groups, groups VI and VII, were shorter in comparison to the control rats, but the change was not significantly different (P > .05). Also, there was a significant (P < .05) decrease in testicular weight in group III when compared to groups I, II, VI, and VII.
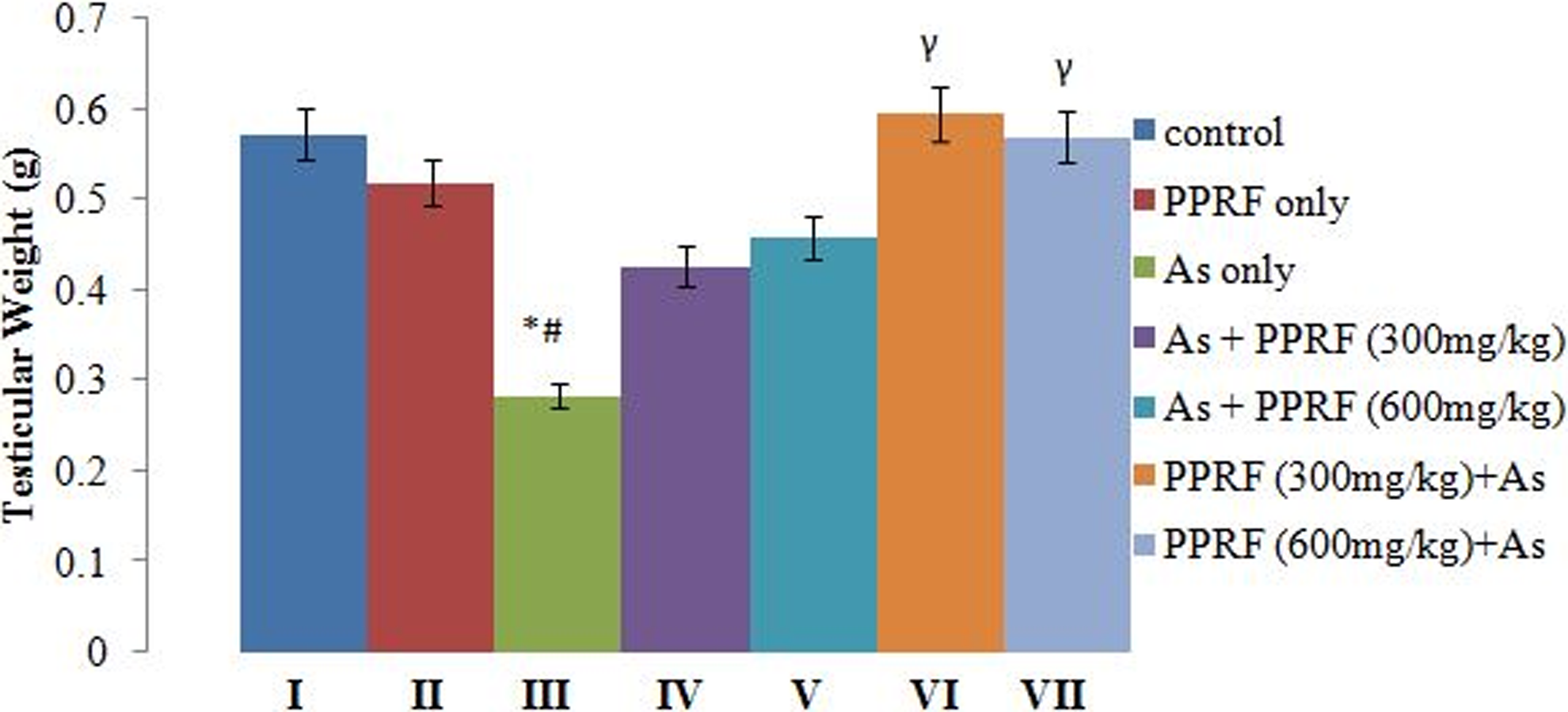
Changes in the testicular weight of control and experimental rats. *Significantly different from group I. #Significantly different from group II. γsignificantly different from group III.
Effect of Treatment on FSH Concentration
Result as shown in Figure 2 indicated that no significant (P > .05) difference was observed between groups I and II. However, there was a significant (P < .05) decrease in FSH concentration between animals in group I and groups III, IV, V, VI, and VII. A significant (P < 0.05) decrease was observed in FSH concentration of rats in groups III and V when compared to group II. Group III showed a significant (P < .05) decrease in FSH concentration when compared to pretreatment and posttreatment groups.

Changes in the serum concentrations of follicle-stimulating hormone (FSH) of control and experimental rats. *Significantly different from group I. #Significantly different from group II. γSignificantly different from group III.
Effect of Treatment on Serum LH and Testosterone Concentration
These 2 hormones were affected across groups in the same manner. The changes in serum LH and testosterone concentrations following the different treatment regimens are shown in Figures 3 and 4, respectively. The result showed that there was no significant (P > .05) difference between groups I and II. However, significant (P < .05) increase was observed in the concentrations of these 2 hormones in group II when compared with all the other groups except group IV. Likewise, group III showed a significant decrease in serum concentrations of these hormones when compared to the control and other treated groups, while there was no remarkable difference between the pretreated and posttreated groups.

Changes in the serum concentrations of luteinizing hormone (LH) on control and experimental rats. *Significantly different from group I. #Significantly different from group II. γsignificantly different from group III.
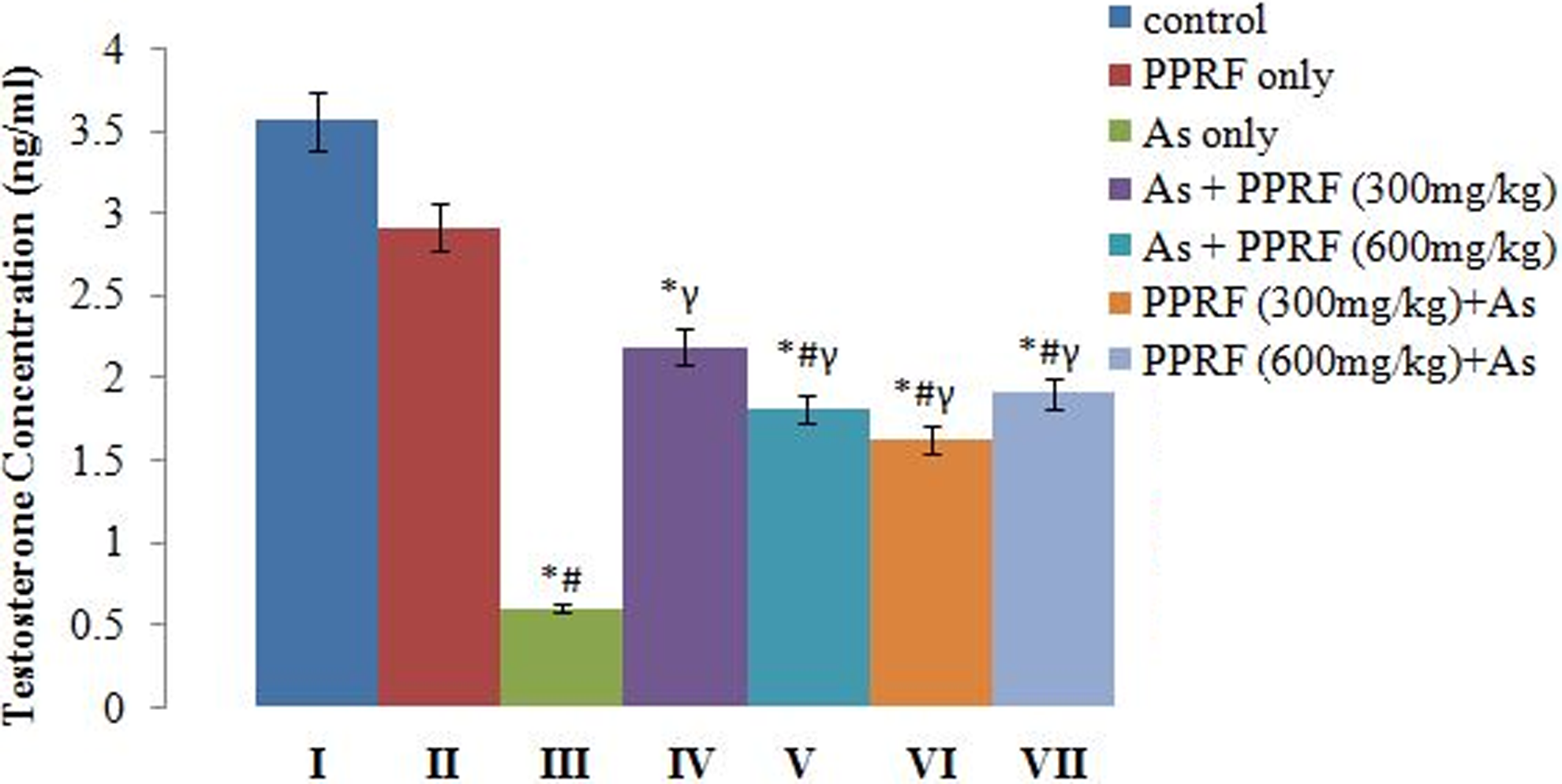
Changes in the serum concentrations of testosterone of control and experimental rats.*Significantly different from group I. #Significantly different from group II. γSignificantly different from group III.
Effects on the Histology of Testes and Pituitary Glands
The effects of the various treatments on the histology of the rats’ testes and pituitary glands are shown in Figures 5 and 6, respectively. The testes revealed normal spermatogenic cell lines across the groups except in group III in which there is disruption of more matured spermatogenic cells. Cellular compositions of the seminiferous tubules appear normal across the groups. Group II showed prominent and healthy spermatogonia. This implies that the extract improved initiation of spermatogenesis. Also, group III showed an enlarged spermatogonia with a whitish deposit that could be as a result of the arsenate intoxication. However, a detachment of the seminiferous tubules from the basement membrane and disruption of the interstitial space were also observed, more pronounced in group III, moderate in groups V and VI, and mild in group VII.

Photomicrograph of testes (magnification ×400) for the different groups. ES indicates enlarged spermatogonia; MSTD, mild seminiferous tubule detachment; NSCL, normal spermatogenic cell line; P & HS, prominent and healthy spermatogonia; STD, seminiferous tubule detachment. The head of the arrow points at the exact spot of interest, that is, where changes were noticed on the photomicrograph, while the text after the arrows give the interpretation of the condition seen as abbreviated.
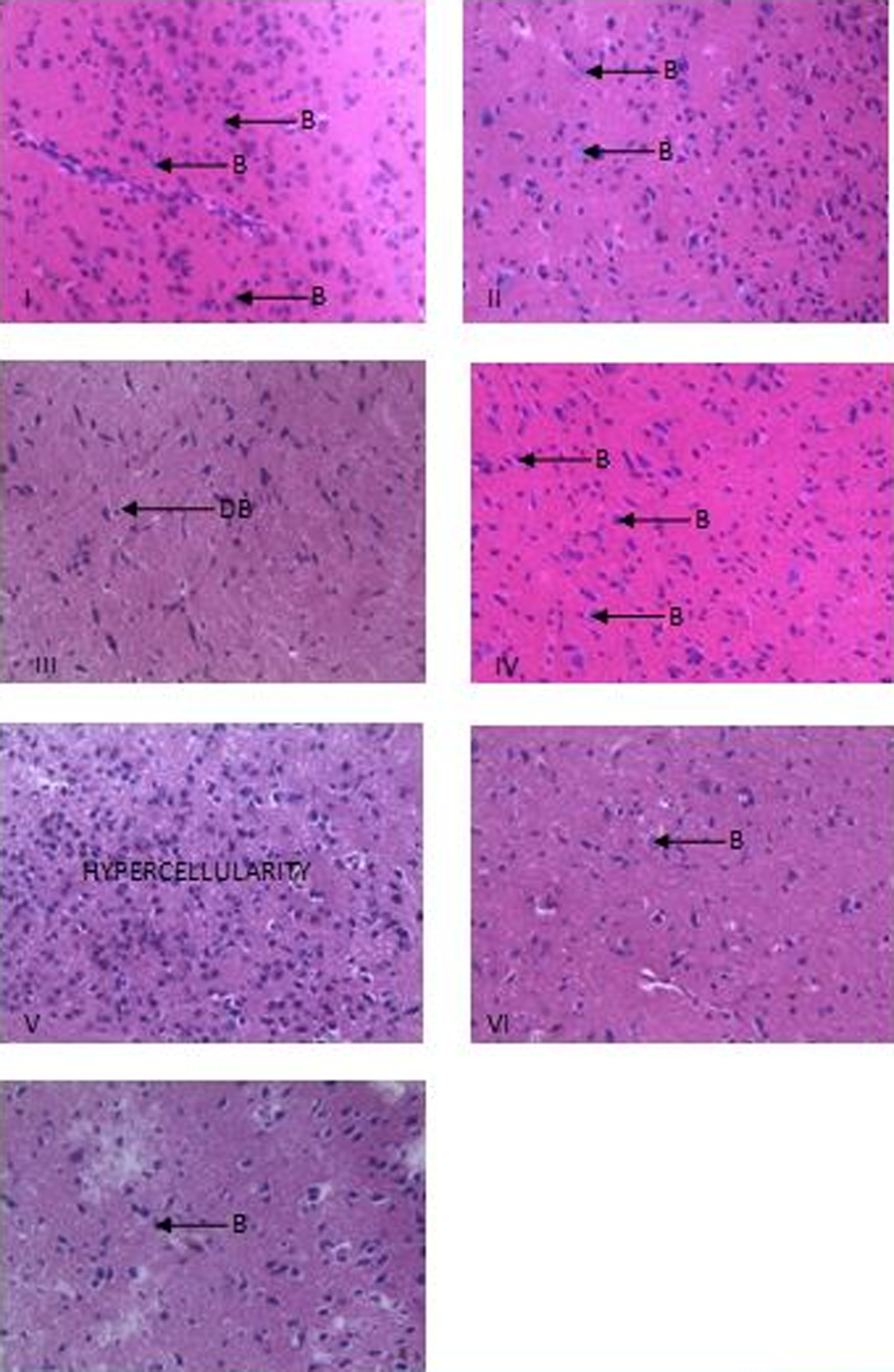
Photomicrograph of pituitary gland of rats (magnification ×400) for the different groups. B indicates basophils; DB, distorted basophils.
The effect of the various treatments on the histology of the rats’ pituitary is shown in plate 2. This plate reveals the presence of basophils (bluish-stained cells) and acidophils. Groups I, II, and IV revealed prominent and healthy basophils when compared with group III which showed very scarce basophils. Hypercellularity of basophils was however noted in the pituitary gland of the rats in group V, while those in groups VI and VII, the pretreatment group, were observed to have reduced basophils when compared to the control group.
Discussion
It has been established that arsenic, an environmental pollutant, impairs the reproductive systems of both male and female rats 6,15,16 and acts as an endocrine disruptor. 16 In this study, it was also observed that arsenate disrupts the serum levels of gonadotropins and subsequently decreases the level of testosterone in the serum of male Wistar rats.
The reduction in the level of gonadotropins (FSH and LH) in the rats in the untreated group (group III) when compared with those in the other groups occurred probably as a result of derailment elicited in the pituitary–gonadal axis by arsenate. On the other hand, the possibility of low serum level of gonadotrophins in the rats in group III in this experiment may be due to activation of the stress signal pathway by arsenate, the hypophysial–adrenocortical axis, and increase in the secretion of adrenocorticotropic hormone (ACTH) from pituitary as observed by Bernstam and Nriagu. 17 Also, a low serum level of gonadotrophin hormones could also occur as a result of decreased sensitivity of gonadotropic cells in the anterior pituitary gland to gonadotropin-releasing hormone, and this has been reported by Kamel and Kubajak 18 to be caused by elevated levels of corticosterone in the blood. It was also observed by Bombino and Hsueh 19 that high levels of ACTH and corticosterone directly suppress testosterone production and secretion by decreasing the testicular LH receptors that invariably cause reduction in the level of circulating gonadotrophins.
It was observed that the serum concentrations of LH and testosterone were affected in the same pattern in all the experimental groups. This is believed to occur as a result of the direct action of LH on the interstitial cells of Leydig of the testes, which in turn causes secretion of testosterone. Although FSH also affects the production of testosterone by stimulating the development of Sertoli cells which help in nursing developing sperm cells to begin the process of spermatogenesis in the testes, the LH controls the production and secretion of testosterone directly from the testis. 20
Arsenate was observed to induce damage of pituitary and testicular cells in rats which might occur as a result of the ability of arsenate ions to generate ROS 21,22 capable of soft tissue breakdown according to the findings of this study. However, the presence of more basophils in the rats in the posttreatment (groups IV and V) groups when compared to the arsenate group (group III) can simply be traced to the ability of the PPRF of G kola seeds to ameliorate the degeneration caused by sodium arsenate in the system of the rats by causing a regeneration of the lost cells. On the other hand, PPRF of G kola seeds extract was able to prevent cell loss by arsenate as observed in rats in the pretreatment group (groups VI and VII) at both the higher dose and the lower dose of the extract.
The significant reduction observed in the paired testicular weight (a valuable index of reproductive toxicity in male rats) of rats in group III when compared to the vehicle control and other treated groups except groups IV and V could be as a result of detachment of seminiferous tubule as evident in the photomicrographs of the testes. Also, enlarged spermatogonia observed in the photomicrograph of the sodium arsenate–treated group have been linked to the presence of oxidative stress exerted on the testes by arsenate treatment.
However, the significant decrease in the serum level of testosterone of rats in the untreated group (group III) when compared to those in other groups has been observed to be caused by a low serum level of LH, which is a prime regulator of testosterone activities.
Testosterone is required for the attachment of different generations of germ cells in seminiferous tubules, and therefore, low level of serum testosterone may lead to detachment of germ cells from seminiferous epithelium and may initiate germ cell apoptosis. 23 The decreases in serum testosterone concentrations may be explained by the fact that the sodium arsenate treatment causes a decrease in serum LH concentrations. In male rats, circulating LH is responsible for maintaining normal serum testosterone concentration. 20 Likewise, the testosterone level of rats in the control group was significantly higher than those in the other groups except in group II, in which there was no statistically significant difference. This implies that treatment with PPRF of G kola seeds has no adverse effect on serum concentration of testosterone. The findings in this study are in contrast to the work done by Abu et al 24 who reported that aqueous extract of G kola seed may have antispermatogenic property. On the other hand, Farombi et al 25 reported G kola seed to have successfully boost the antioxidant status and exhibit no adverse effect on the liver, testes, and spermatozoa after a long-term oral exposure in rats. The reason for the variation in this present study and that of Abu et al 24 could probably be related to the mode of extraction and the purification of the crude extract of the G kola seeds to obtain its polyphenolic-rich fraction which was used in this study.
It has been proved from this study that the polyphenols obtained from G kola have no adverse effect on reproductive hormones, but instead it was observed to have successfully reversed the reduction in these hormones caused by arsenate administration and polyphenols were able to protect the disruption of the neuroendocrine cycle normally caused by arsenate.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
