Abstract
Background:
While concomitant opioid and benzodiazepine use is discouraged due to an increased risk of sedation/overdose, the extent of perioperative opioid utilization in hand surgery patients already using benzodiazepines is unknown.
Methods:
Using an administrative claims database, we identified adults undergoing carpal tunnel, DeQuervain, or trigger finger release, palmar fasciectomies, ganglion/mucoid cyst removals, and hand/wrist soft tissue mass excisions from 2011 to 2021. We identified opioid-naive patients with a benzodiazepine prescription within 90 days before surgery. Outcomes included the percentage of patients prescribed opioids and the amount of opioids prescribed, new persistent opioid utilization, postoperative emergency department (ED) visits, hand therapy utilization, and opioid overdose. We used multivariable regression models to adjust for age, sex, region, insurance, procedure, and comorbidities.
Results:
Patients with benzodiazepine utilization had a higher adjusted odds of perioperative opioid prescriptions, amount of opioids filled, and new, persistent opioid use compared with patients without benzodiazepine use (all P < .001). In the multivariable analysis, patients with benzodiazepine use received +30.8 (95% CI: 28.9-32.8) morphine milligram equivalents and had an odds ratio of 1.48 (95% CI: 1.44-1.52) for developing new, persistent opioid use compared with patients without benzodiazepine use. Patients with preoperative benzodiazepine use also had increased postoperative ED visits and hand therapy utilization.
Conclusions:
Opioid-naive patients undergoing soft tissue hand surgery who are using benzodiazepines were prescribed opioids more frequently/at higher amounts and experienced an increased odds of developing new, persistent opioid use. Surgeons could consider reducing opioid prescriptions in patients already using benzodiazepines in favor of nonopioid pain strategies.
Introduction
Concomitant use of opioids and benzodiazepines is discouraged due to the increased risk of sedation and overdose. For example, clinical practice guidelines developed by the Centers for Disease Control and Prevention caution against co-prescribing opioids and benzodiazepines, encouraging a careful assessment of their risks and benefits. 1 Nevertheless, the incidence of benzodiazepine use in the US population is increasing, with a corresponding increase in benzodiazepine overdose-related mortality. 2 Similarly, the incidence of concurrent opioid and benzodiazepine use is rising, which has been associated with an increased risk of emergency department (ED) visits and inpatient admissions for opioid overdose, death from drug overdose, and all-cause mortality.3 -5 This evidence has led to public health efforts, including by the Centers for Medicare and Medicaid Services and Food and Drug Administration (FDA), aimed at avoiding or reducing the co-prescription of benzodiazepines and opioids (eg, FDA requirement for boxed warnings on the dangers of concomitant opioid and benzodiazepine use).6,7
In light of these data, preoperative benzodiazepine use poses a particular challenge for surgeons given that opioids are a mainstay of treatment regimens for acute postoperative pain. Prior studies have characterized the risks of co-prescribing opioids in surgical patients with preoperative benzodiazepine use. In a large study of opioid-naive patients undergoing 11 different surgical procedures including total joint arthroplasty, preoperative benzodiazepine use was associated with increased postoperative opioid utilization and a greater risk of developing long-term opioid use. 8 Multiple studies in the joint arthroplasty literature have confirmed this finding.9 -12 In comparison, a unique opportunity exists within hand surgery for reducing the co-prescription of benzodiazepines and opioids, as recent evidence has demonstrated the efficacy of nonopioid pain regimens after elective, soft tissue hand surgery.13,14 However, the extent of perioperative opioid prescription in hand surgery patients already using benzodiazepines, as well as the postoperative outcomes associated with preoperative benzodiazepine use, remains unknown.
In this study, we tested the null hypothesis that preoperative benzodiazepine use is not associated with the: (1) percentage of patients prescribed opioids or amount of opioids prescribed perioperatively; (2) incidence of new, persistent postoperative opioid utilization; or (3) incidence of ED visits, hand therapy utilization, or opioid overdose in patients undergoing soft tissue hand surgery.
Methods
Data Source and Cohort Creation
We used the PearlDiver Mariner Patient Records Database, a large national administrative claims database, to identify all patients 18 years old or older who underwent elective, soft tissue hand procedures from 2011 to 2021. These procedures consisted of open and endoscopic carpal tunnel release, DeQuervain release, trigger finger release, palmar fasciectomy for Dupuytren disease, ganglion or mucoid cyst removals, or excision of soft tissue masses, as defined by their respective Current Procedural Terminology (CPT) codes (Supplemental Table S1). Patients were required to be continuously enrolled in the database for at least 1 year before and after the index procedure to allow for accurate comorbidity and outcome capture. If patients underwent more than one of the above procedures, only the first instance was included. Patients were required to have complete demographic data, consisting of age, sex, geographic region, and insurance plan. Only private, Medicare Advantage, and Medicaid managed care plans were included. Patients with a prior history of dependence or abuse of opioids, benzodiazepines, or other substances, as defined using International Classification of Diseases, 9th or 10th Revision (ICD-9/10) diagnostic codes (Supplemental Table S1), were excluded.
To ensure we only captured opioid-naive patients, we further excluded patients who filled opioid prescriptions from 1 year up to, but not including, 31 days before the procedure. Any opioids filled within 31 days (inclusive) before the procedure was assumed to be related to the procedure (eg, prescribed at the preoperative visit to be taken postoperatively), consistent with prior definitions. 15 For the exclusions, our definition included all opioids that are commonly used in the outpatient setting: oxycodone, hydrocodone, morphine, methadone, buprenorphine, tramadol, and hydromorphone, in both generic and brand-name forms (Supplemental Table S1). Patients were then divided into cohorts based on the existence of preoperative benzodiazepine use, which was classified as a filled benzodiazepine prescription within 90 days before the index procedure. The following commonly prescribed benzodiazepines were included: alprazolam, diazepam, lorazepam, temazepam, clonazepam, midazolam, and chlordiazepoxide. Patients without any filled benzodiazepine prescriptions in the 1 year before the index procedure were considered to have no preoperative benzodiazepine use. Patients with filled benzodiazepine prescriptions between 1 year and 90 days before the procedure, but without a filled prescription in the 90 days before surgery, were excluded. A flow diagram of cohort creation is shown in Figure 1.
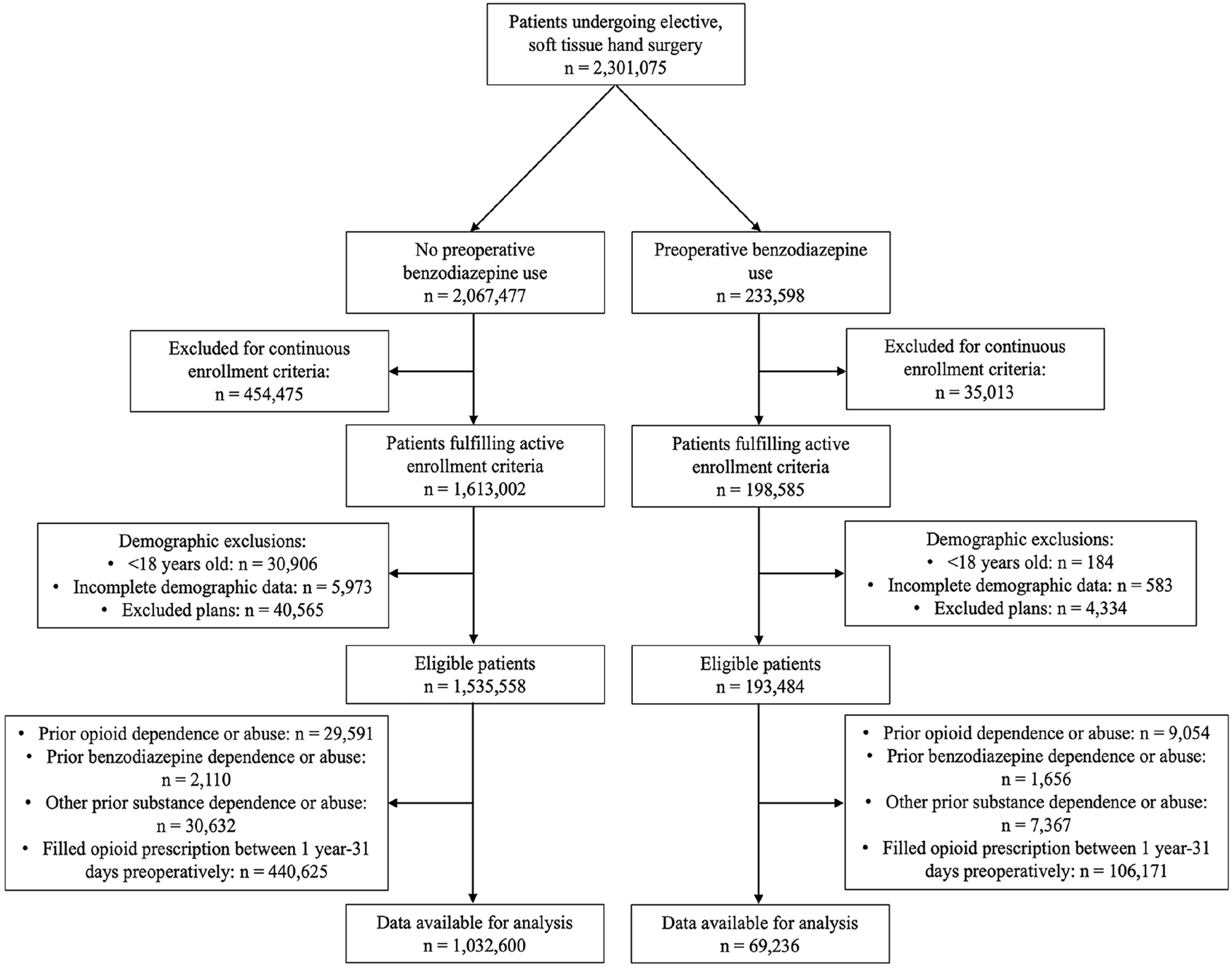
Flow diagram of cohort creation.
Outcomes
Our primary outcome was the incidence and amount of perioperative opioid utilization. The perioperative period was defined as the period beginning 31 days before surgery to 14 days after surgery, inclusive. Any opioid prescription filled in this period was assumed to be related to the surgery, which is consistent with assumptions used in prior opioid-related studies. 15 To study this outcome, we only included opioids commonly prescribed after surgery: oxycodone, hydrocodone, tramadol, and hydromorphone. The amount of opioid utilization was quantified as the number of morphine milligram equivalents (MMEs) filled. Morphine milligram equivalents were calculated directly by PearlDiver software using standard conversion factors provided by the Centers for Disease Control and Prevention, 1 according to the following equation: strength × units × conversion factor. Secondary outcomes included the incidence of new, persistent opioid utilization in patients who filled perioperative opioid prescriptions, ED visits, hand therapy utilization, opioid overdose, or benzodiazepine overdose, as defined using their respective ICD and CPT codes (Supplemental Table S1). Hand therapy CPT codes were chosen based on prior literature.16,17 The incidences of ED visits, hand therapy utilization, and opioid overdose were assessed in all patients, regardless of whether they had filled perioperative opioid prescriptions. This accounts for opioid overdoses that occurred regardless of the source of these medications. Benzodiazepine overdose was assessed only in patients with preoperative benzodiazepine use. New, persistent opioid use was defined as continued opioid prescriptions in the 91 to 180 days (inclusive) following surgery in patients who had a perioperative opioid prescription, consistent with prior definitions. 15 The cumulative incidence of ED visits and hand therapy utilization was measured in 15-day intervals up to 60 days postoperatively. Given this time constraint, we assumed that these ED visits and hand therapy visits were related to the index hand procedure. All-cause ED visits were analyzed to capture all visits that could be directly or indirectly related to the procedure. The cumulative incidence of opioid overdose was measured in 3-month intervals up to 12 months postoperatively.
Statistical Analysis
Categorical variables were compared using χ2 tests. The Elixhauser comorbidity index was compared using the Mann-Whitney test. The Elixhauser comorbidity index was calculated directly by PearlDiver software by assigning 1 point to each comorbidity present, from a list of validated diagnostic codes. 18 Patients were grouped by whether they had preoperative benzodiazepine use. We created multivariable logistic regression models to evaluate the association between preoperative benzodiazepine use and the categorical outcomes while adjusting for the effects of age, sex, US region, insurance plan, Elixhauser comorbidity index, and the specific hand procedure the patient underwent. A multivariable linear regression model was used to evaluate the association between preoperative benzodiazepine use and the amount of MMEs prescribed, adjusting for the same variables as above. Due to the large sample size afforded by the database, statistical significance was defined a priori as P less than .001.
Results
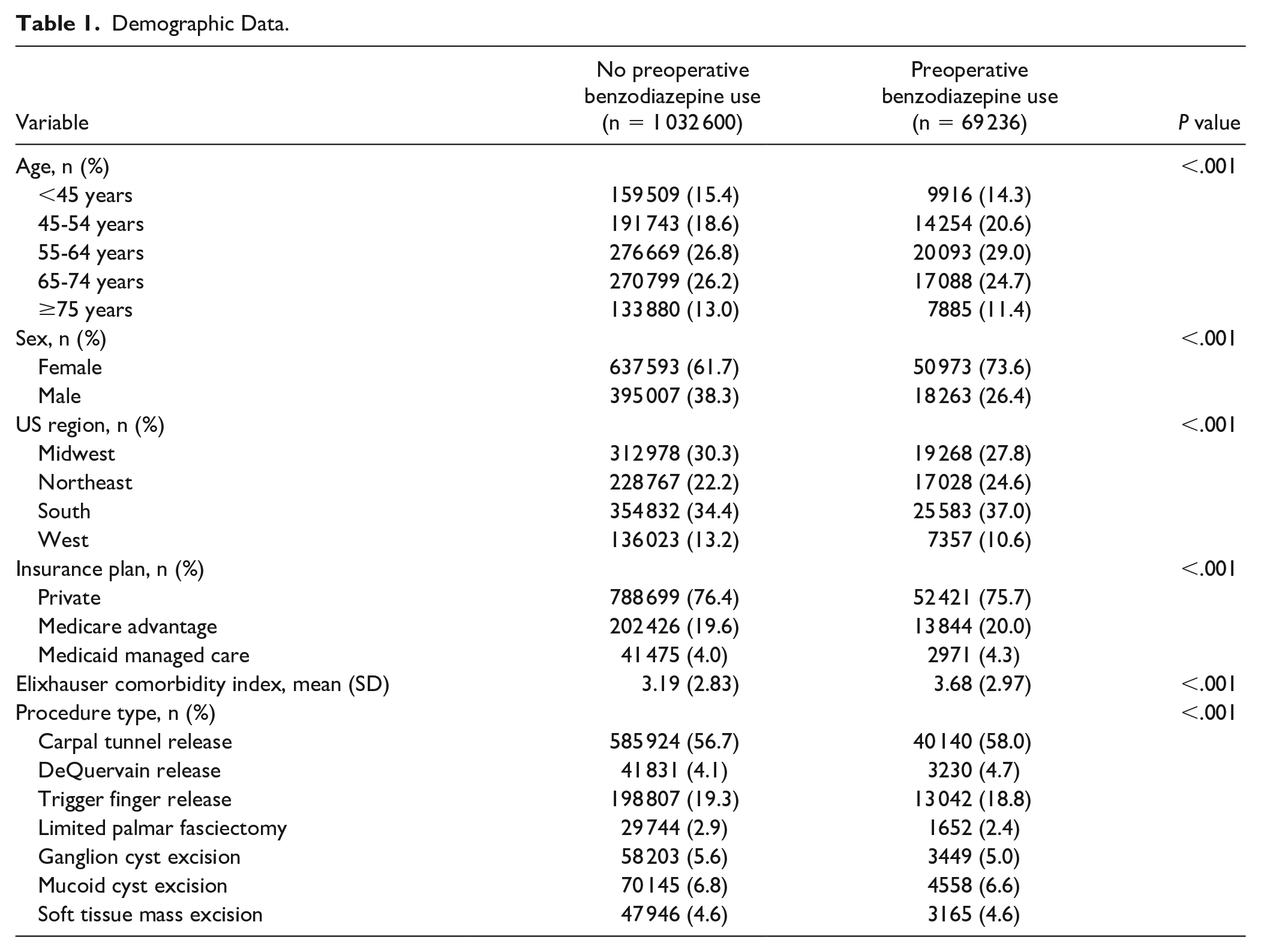
Of 1 101 836 included patients undergoing soft tissue hand surgery, 69 236 (6.3%) had preoperative benzodiazepine use. Patients with preoperative benzodiazepine use differed from those without preoperative benzodiazepine use in the distribution of age, sex, US region, insurance plan, comorbidities, and procedures (Table 1). A higher percentage of patients with preoperative benzodiazepine use were prescribed perioperative opioids compared with patients without preoperative benzodiazepine use (Table 2). After adjusting for the effects of age, sex, region, insurance plan, and Elixhauser comorbidities, patients with preoperative benzodiazepine use experienced an odds ratio (OR) of 2.48 (95% CI: 2.44-2.52; P < .001) for being prescribed perioperative opioids, independently from the specific hand procedure they underwent.
Demographic Data.
Opioid Utilization Outcomes.
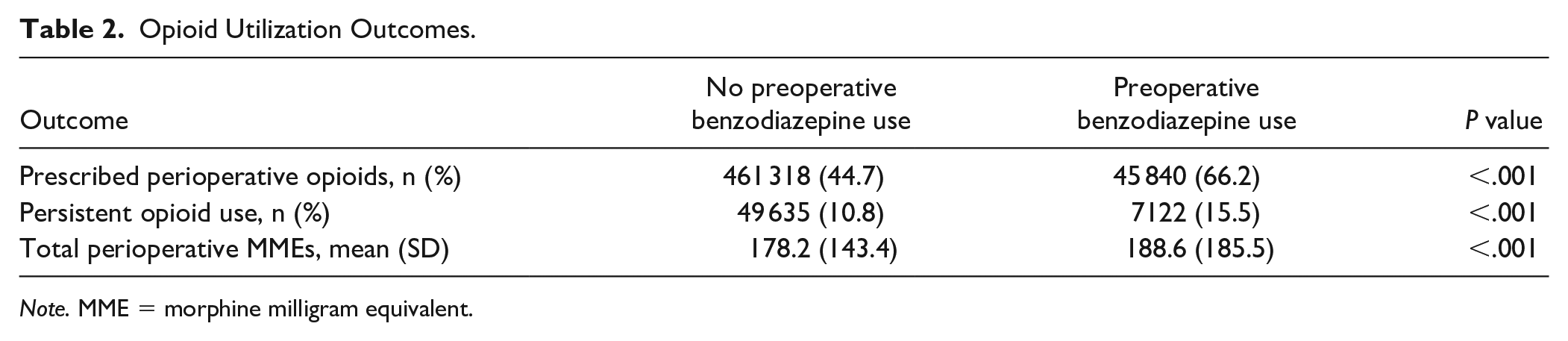
Note. MME = morphine milligram equivalent.
Of the patients who were prescribed opioids, those with preoperative benzodiazepine use were prescribed, on average, +30.8 (95% CI: 28.9-32.8) additional MMEs after controlling for covariates. Patients with preoperative benzodiazepine use had a higher incidence of developing new, persistent opioid use compared with patients without preoperative benzodiazepine use (Table 2). This effect persisted after adjustment for covariates, where preoperative benzodiazepine use was associated with an increased odds of new, persistent opioid use (OR = 1.48 [95% CI: 1.44-1.52]; P < .001).
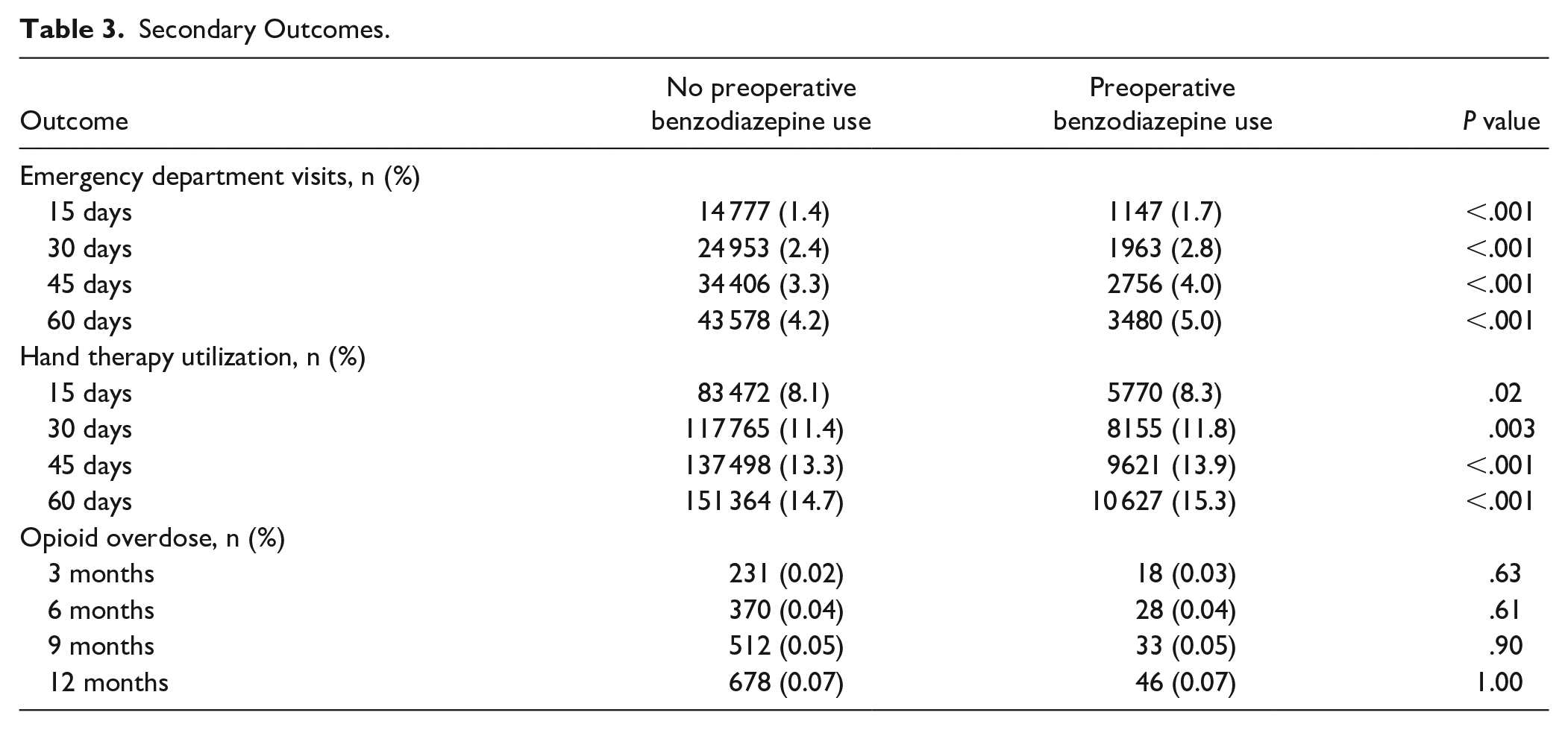
Patients with preoperative benzodiazepine use also had an increased incidence of postoperative ED utilization and slightly increased hand therapy utilization compared with patients without preoperative benzodiazepine use at most time points evaluated (Table 3). These small but statistically significant effects persisted in the multivariable analysis (Table 4). Of note, the incidence of opioid overdose did not differ between cohorts. In patients with preoperative benzodiazepine use, receiving a concomitant perioperative opioid prescription was not associated with an increased incidence of benzodiazepine overdose (Supplemental Table S2).
Secondary Outcomes.
Multivariable Analysis.
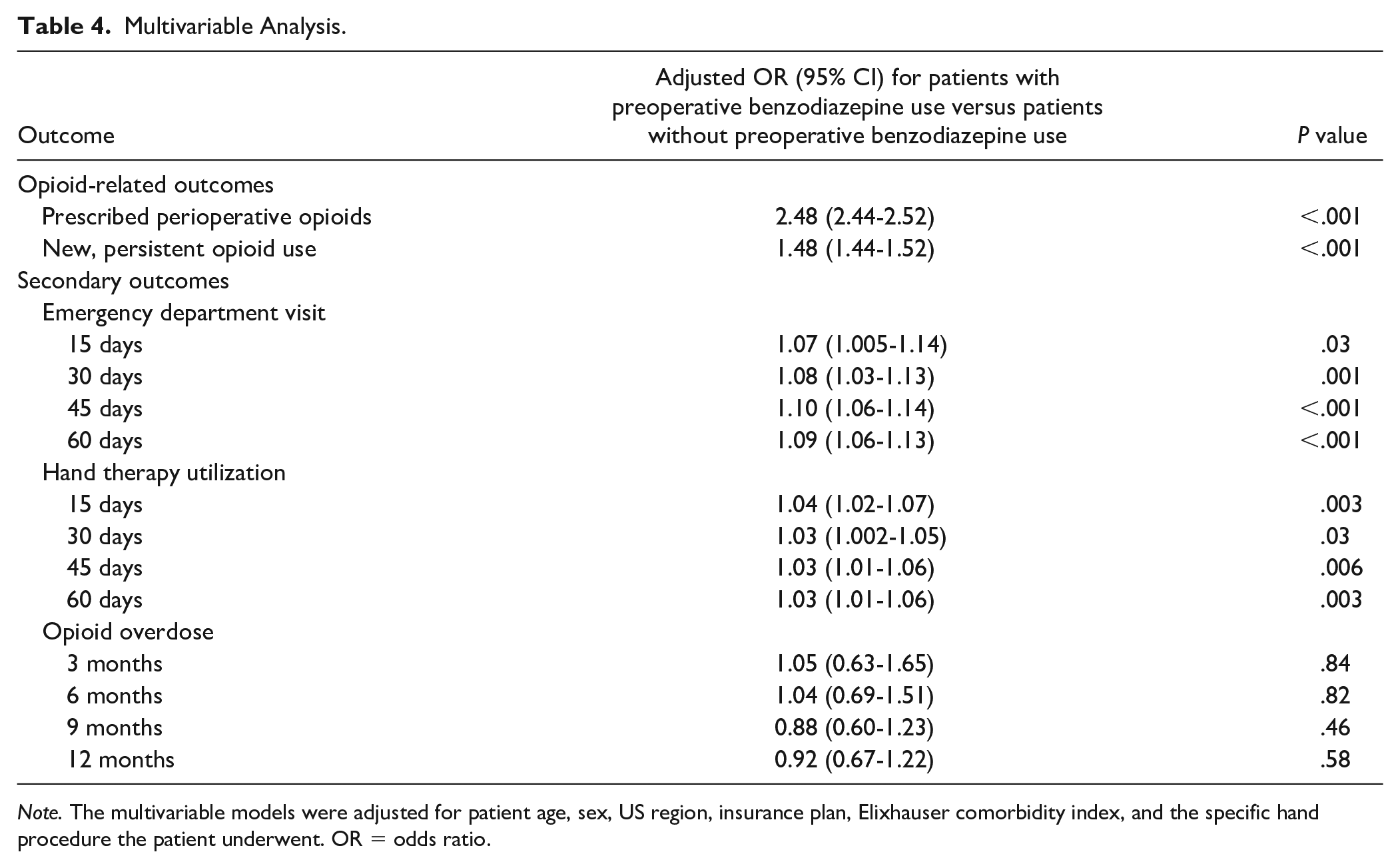
Note. The multivariable models were adjusted for patient age, sex, US region, insurance plan, Elixhauser comorbidity index, and the specific hand procedure the patient underwent. OR = odds ratio.
Discussion
In this study, we found that patients undergoing soft tissue hand surgery with preoperative benzodiazepine use were prescribed opioids more frequently and at higher amounts than patients without preoperative benzodiazepine use. While the clinical difference in the amount of perioperative opioid use may be small, patients with preoperative benzodiazepine use experienced a nearly 50% increased odds of developing new, persistent opioid use, highlighting the risks of perioperative opioid prescription in this patient population. Given the risks of concomitant opioid and benzodiazepine use, surgeons could consider reducing or eliminating opioid prescriptions in patients who are already using benzodiazepines in favor of nonopioid pain strategies.
Our results are consistent with prior studies on the risks of preoperative benzodiazepine use in orthopedic surgery. In patients undergoing total hip, knee, or shoulder arthroplasty, preoperative benzodiazepine use has been linked with increased postoperative opioid requirements and prolonged opioid utilization.9 -11,19 A study in patients undergoing total knee arthroplasty showed that preoperative benzodiazepine use was associated with a higher risk for revision, subsequent femur fracture fixation, and delirium. 20 In hand surgery, more than 10% of patients undergoing soft tissue or bony hand procedures have pre-existing benzodiazepine use, with greater utilization found in older patients with Medicare insurance. 21 However, only a few small studies have investigated the link between benzodiazepine and opioid utilization in hand surgery. In a study of patients undergoing upper extremity surgery, benzodiazepine use within 6 months before surgery was a strong predictor of prolonged opioid utilization at more than 6 months after surgery. 22 Similarly, in another study of patients undergoing a variety of upper extremity procedures, preoperative benzodiazepine use was associated with more filled postoperative opioid prescriptions. 23 In a larger study of patients undergoing carpal tunnel release, basal thumb arthroplasty, or surgical fixation of distal radius fractures, 16% of those patients had preoperative benzodiazepine use and these patients were more likely to fill more postoperative opioid prescriptions and develop prolonged opioid use. 24 Our data contributes to this literature by showing that in a large, population-based cohort, preoperative benzodiazepine use is associated with increased perioperative opioid use, a higher risk of developing prolonged opioid utilization, and a higher incidence of postoperative ED visits and hand therapy utilization.
Co-prescribing of opioids in patients already using benzodiazepines is strongly discouraged, as highlighted by the FDA requirement for a boxed warning on opioid and benzodiazepine medications about the dangers of concomitant use. 7 In our cohort of hand surgery patients, not only was the prevalence of co-prescription high (overall 46%), but also patients with preoperative benzodiazepine use were more likely to be prescribed perioperative opioids than patients without preoperative benzodiazepine use (66% vs 45%). This finding highlights a large potential opportunity for reducing opioid use within hand surgery, especially given the existence of opioid-sparing protocols. A recent randomized controlled trial has shown no difference in postoperative pain between patients undergoing soft tissue hand surgery treated with acetaminophen and ibuprofen versus acetaminophen and hydrocodone. 13 In another study of patients undergoing soft tissue hand surgeries in the procedure room setting, only 5.6% required a prescription pain medication. 14 Therefore, it may be reasonable to avoid prescribing opioids in hand surgery patients with preoperative benzodiazepine use entirely. In these patients, the use of structured pain management education protocols in the perioperative period may further assist in reducing opioid use. For example, 71% of patients in one study who underwent soft tissue hand surgery and received pain management education took no opioids at all. 25
While the full spectrum of benzodiazepine-related adverse effects is broad, those directly attributable to their use, especially in combination with opioids (eg, overdose), could have led to the increased ED visits observed. For example, prior literature has shown an increased risk of ED visits, including for overdose, with combined benzodiazepine/opioid use in the general population.3,26,27 When used in combination with benzodiazepines, opioids can result in a greater than 2-fold higher risk of overdose and account for more than $1100 in additional 30-day postoperative health care costs.3,8 While we did not detect a difference in the incidence of opioid overdose specifically between our cohorts, we were likely underpowered for this outcome given its infrequent occurrence. Furthermore, the increase in ED visits in the benzodiazepine cohort could be attributed to reasons other than overdose, such as altered mental status or syncope. 26 It is thus concerning that concurrent use of these medications is rising along with related mortality, with almost 20% of patients using opioids who also use benzodiazepines.3,8,27 Opioids comprise 67% of the top 10 medications prescribed by hand surgeons, with a 22% increase in the number of opioid prescriptions written by hand surgeons between 2013 and 2019. 28 Therefore, policy efforts are needed to target utilization patterns to reduce opioid-related complications and associated morbidity. Quality measures focused on measuring postoperative opioid use and the utilization of perioperative structured counseling on opioids have been developed for soft tissue hand surgery. 29 These measures could be applied for patients with preoperative benzodiazepine use—a particularly high-risk population—to establish practice patterns and benchmarks, which could then drive further interventions. Across health systems, prescriber-facing alerts could be implemented within electronic medical record systems that are triggered when opioids are prescribed to patients with active benzodiazepine use, allowing surgeons the opportunity to cancel the prescription in favor of alternatives. 30 Similarly, prescribing tools can be used with preset pain management regimens to nudge clinicians toward standardized nonopioid pain medications after soft tissue hand procedures. 31 Such “soft stops” have been shown to be efficacious in altering prescribing practices and reducing opioid use.30,31
Our study should be viewed in light of its limitations. Our findings are subject to the inherent limitations imposed by the use of administrative claims data, including reliance on accurate coding and limited clinical granularity. While we were able to track the amounts of opioids and benzodiazepines filled via prescription drug claims data, we were unable to ascertain the exact amounts of medications actually taken by patients. Some patients may have filled, but not used, their entire opioid or benzodiazepine prescription. Some patients may have paid out of pocket for their medications or used a different insurance for prescription drugs, leading to misclassification of exposure and/or outcome. A minority of patients could have underwent a different procedure within 180 days of their hand surgery and received opioid prescriptions that were unrelated to the hand surgery. While we were able to demonstrate an association between preoperative benzodiazepine use and perioperative opioid utilization, we are unable to prove causation in our retrospective study. Nevertheless, we adjusted for a variety of demographic factors and comorbidities in the multivariable analysis to reduce potential confounding. While patients with preoperative benzodiazepine use were slightly more likely to use hand therapy postoperatively, this effect size was small and likely not clinically important.
In conclusion, preoperative benzodiazepine use is a risk factor for increased perioperative opioid use and the development of new, persistent opioid utilization after soft tissue hand surgery. Given the demonstrated efficacy of nonopioid pain regimens for soft tissue hand surgery, opioids should be used with caution in patients with preoperative benzodiazepine use or avoided altogether if possible. Efforts across health systems are needed to reduce opioid and benzodiazepine co-prescription in hand surgery, which could include prescriber-facing alerts within electronic medical systems and prescribing tools with preset, evidence-based opioid-sparing pain management regimens.
Supplemental Material
sj-docx-1-han-10.1177_15589447241312998 – Supplemental material for Concurrent Perioperative Benzodiazepine and Opioid Utilization in Opioid-Naive Patients Undergoing Soft Tissue Hand Surgery
Supplemental material, sj-docx-1-han-10.1177_15589447241312998 for Concurrent Perioperative Benzodiazepine and Opioid Utilization in Opioid-Naive Patients Undergoing Soft Tissue Hand Surgery by Thompson Zhuang, Ellis M. Berns, Hannah H. Lee and Andrew D. Sobel in HAND
Footnotes
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects as defined by 45 CFR 46.
Statement of Informed Consent
This article does not contain any studies with human or animal subjects as defined by 45 CFR 46. This is an analysis of commercially available, de-identified data, so there was no requirement for informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Department of Veterans Affairs for HHL (CDA-2 1IK2RX003947-01A2; CReATE Motion Pilot grant).
Supplemental material is available in the online version of the article.
