Abstract
Trigger finger release (TFR) is a common hand surgery, historically performed using a tourniquet. Recently, wide-awake local anesthesia no tourniquet (WALANT) has gained popularity due to ostensible advantages such as improved patient pain, satisfaction, lower rate of complications, and decreased cost. This systematic review compares outcomes of WALANT for TFR with local anesthesia with a tourniquet (LAWT). MEDLINE, Embase, CINAHL, Web of Science, Cochrane Central Register of Controlled Trials, and ClinicalTrials.gov were searched. All English-language peer-reviewed randomized and observational studies assessing TFR in adults were included. Quality of evidence was assessed with the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) system. Eleven studies (4 randomized controlled trials, 7 observational) including 1233 patients were identified. In the WALANT group, pain on injection was statistically nonsignificantly lower (mean difference [MD]: −1.69 points, 95% confidence interval [CI] = −4.14 to 0.76, P = .18) and postoperative pain was statistically lower in 2 studies. Patient and physician satisfaction were higher and analgesic use was lower in WALANT. There were no significant differences between groups for functional outcomes or rates of adverse events. Preoperative time was longer (MD: 26.43 minutes, 95% CI = 15.36 to 37.51, P < .01), operative time similar (MD: −0.59 minutes, 95% CI = −2.37 to 1.20, P = .52), postoperative time shorter (MD: −27.72 minutes, 95% CI = −36.95 to −18.48, P < .01), and cost lower (MD: −52.2%, 95% CI = −79.9% to −24.5%) in WALANT versus LAWT. The GRADE certainty of evidence of these results ranges from very low to low. This systematic review does not confirm superiority of WALANT over LAWT for TFR due to moderate to high risk of bias of included studies; further robust trials must be conducted.
Introduction
Trigger finger (TF) is a widespread and potentially debilitating condition characterized by clicking and locking of an affected finger during tendon excursion caused by a size discrepancy between the flexor tendon sheath and annular pulley. 1 The lifetime prevalence of TF is 2.6% in the general population and is even higher in patients with risk factors such as diabetes mellitus.2,3 Currently, TF is one of the most common indications for referral to a hand surgery clinic and a major cause of hand pain and disability.4,5
Operative treatment is routinely performed and is regarded as the gold standard for definitive TF resolution.6,7 Historically, trigger finger release (TFR) involved administration of local anesthesia for analgesic effects as well as a tourniquet to achieve hemostasis. Although effective at producing hemostatic effects, tourniquets have the unfortunate consequence of causing patient pain, thus potentially necessitating sedation or general anesthesia due to the resulting discomfort.8 -10 Besides pain, tourniquet use has additionally been associated with compression neuropathies, muscle tissue damage, and vascular injuries.11,12
Alternatives to achieving hemostasis without tourniquet use have been proposed, including the wide-awake local anesthesia no tourniquet (WALANT) technique popularized by Lalonde in 2005. 13 This approach forgoes the use of a tourniquet by injecting local anesthetic with epinephrine and has been shown to be a safe and effective option for maintaining a clear surgical field. 14 The WALANT technique is gaining popularity in upper extremity surgeries due to the purported lack of tourniquet-associated complications and subsequent anesthetic or sedative interventions, high patient satisfaction, increased patient safety, decreased patient pain, decreased costs, and location flexibility.14,15
The WALANT approach has become common in Canada, while tourniquet use remains high among surgeons in the United States.16,17 Some surgeons have opted to use a hybrid approach including both a local anesthetic with epinephrine and a tourniquet in hand surgeries. 18 Although many studies assessing outcomes of TFR under WALANT have been published in the literature, to the best of our knowledge, there are no systematic reviews available on this topic.
This systematic review attempts to answer the following clinical question: In adult (>18 years) patients with a diagnosis of TF requiring a TFR in the operative or minor procedure setting, is WALANT superior to local anesthesia with a tourniquet (LAWT) for patient pain, patient satisfaction, physician satisfaction, analgesic use, functional outcomes, adverse events, procedure time, and costs?
Methods
This systematic review and meta-analysis were conducted and reported according to the methods outlined in the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guideline. 19 A protocol was registered in the PROSPERO database (CRD42022363080).
Eligibility Criteria
Studies were included if they: (1) were randomized, quasi-randomized or nonrandomized clinical trials, or observational studies; (2) investigated TFR in adult patients (>18 years) using the WALANT technique compared with surgical release under LAWT control; (3) were published in a peer-reviewed journal with the full text available; (4) were published in English. Studies were excluded if they were reviews, conference abstracts, case series, case reports, letters to the editor, or nonhuman studies. There were no limitations on publication date.
Outcomes
As no core outcome set has been established for effectiveness trials in this patient population, a list of outcomes was generated based on existing systematic reviews and primary observational studies identified through a preliminary search of the literature. Eight outcomes deemed to be critical and/or important by the review authors were included. Outcomes selected include: (1) patient pain; (2) patient satisfaction; (3) physician satisfaction; (4) analgesic use; (5) functional outcomes; (6) adverse events; (7) procedure time; and (8) costs. The primary study authors’ definition of these outcomes was accepted. Any duration of follow-up reported as the study’s primary time horizon was collected; if not specified, data from the final time horizon were used.
Search Strategy
A comprehensive search of the following databases was performed on October 5, 2022: MEDLINE, Embase, CINAHL, Web of Science, and Cochrane Central Register of Controlled Trials (CENTRAL). Clinical trial registries, such as ClinicalTrials.gov, were also searched for unreported or ongoing trials. The search strategy was created in collaboration with a trained medical librarian and can be found in Supplemental Material 1. Reference lists of relevant studies and reviews were screened for additional articles that could meet eligibility criteria.
Study Screening and Data Extraction
Identified articles were reviewed independently and in duplicate by 2 authors (TL and DCTL) for adherence to the eligibility criteria using the Covidence software tool (https://support.covidence.org/help/how-can-i-cite-covidence). 20 Disagreements were resolved by referring to a third blinded author (AT). Data extraction of included studies was done independently by 2 authors (TL and DCTL) into a standardized, piloted, spreadsheet created prior to commencement of the data extraction phase. Extracted data included study characteristics, participant demographics, follow-up period, anesthetic technique, and previously listed outcomes.
Risk of Bias and Quality of Evidence Appraisal
The risk of bias was assessed independently by 2 authors (TL and DCTL). Randomized and nonrandomized studies were evaluated using the Cochrane Risk of Bias 2 (RoB 2) tool 21 and Risk Of Bias In Non-randomized Studies—of Interventions (ROBINS-I) tool, 22 respectively. Quality of evidence was appraised using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) Handbook, 23 and the corresponding summary of findings table was created with GRADEpro GDT software (https://www.gradepro.org/terms/cite). 24
Statistical Analysis
Individual study characteristics and data are presented in tables. Both randomized controlled trials (RCTs) and observational studies are included but were analyzed separately with accompanying narrative summaries in keeping with the Cochrane Handbook for systematic reviews and meta-analyses. 25 All studies reporting on a specified outcome were included in the corresponding synthesis and, where applicable, results are displayed in forest plots. This meta-analysis pooled results using an inverse-variance random effects model and calculated a mean difference with a 95% confidence interval for continuous outcomes. Where appropriate, missing data were calculated in accordance with protocols outlined in the Cochrane Handbook. 25 Study heterogeneity was measured via the χ2 test and I2 statistic. The magnitude of heterogeneity as defined by the I2 value was: 0% to 40% as “might not be important,” 30% to 60% as “moderate” heterogeneity, 50% to 90% as “substantial” heterogeneity, and 75% to 100% representing “considerable” heterogeneity. 26 A random effects model was used for pooled effect estimates. Given that less than 10 studies were included for each outcome, a funnel plot was not used to evaluate the presence of publication bias. 26 The statistical significance threshold was P < .05. Subgroup analysis and publication bias assessment were not performed due to the small number of included articles. Analysis and synthesis were conducted using Review Manager (RevMan) [Computer program]: Version 5.4.1, The Cochrane Collaboration, 2020 (https://documentation.cochrane.org/revman-kb/cite-revman-in-a-reference-list-110242006.html).
Results
Study Selection
The search strategy produced 449 records, of which 160 were automatically removed as duplicates. The remaining 289 studies were screened by title and abstract, and 31 were selected to be analyzed in the detailed full-text review phase. The final 11 articles included in this review were composed of 10 studies9,26 -34 which were identified through the search strategy detailed above and one additional article 35 that was found via citation searching. See Figure 1 for the PRISMA flow chart. Several articles, such as Saleh et al 10 and Gunasagaran et al, 36 assessed TFR under WALANT along with other hand surgeries (eg, carpal tunnel release) but were excluded because they did not report separate data specifically for the TFR group. A table of included studies can be found in Supplemental Material 2.
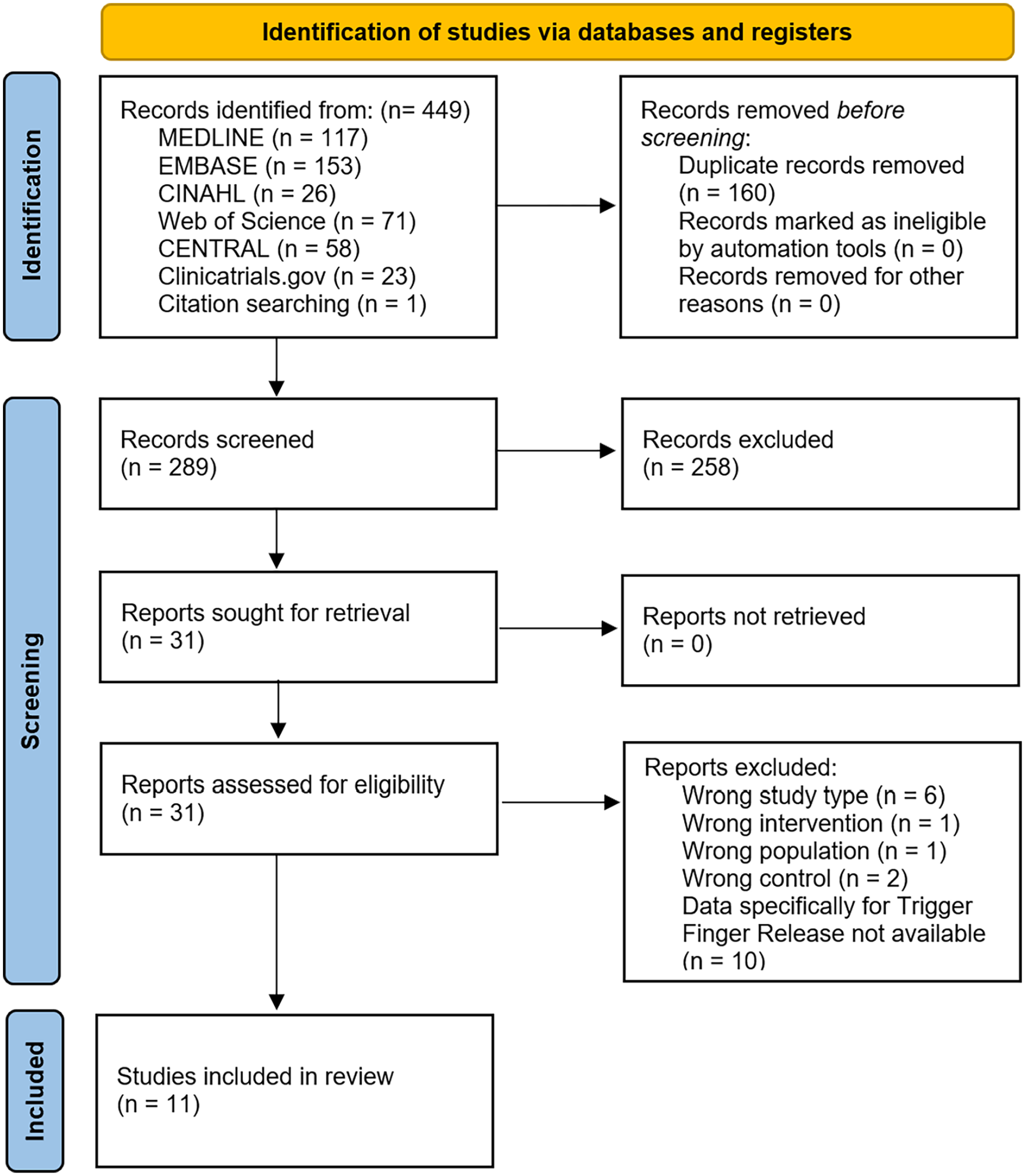
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 2020 flow diagram.
Risk of Bias
Risk of bias assessment was performed for 4 RCTs using the Cochrane RoB 2 tool. None of the 4 studies were registered in trial registries prior to commencement of recruitment and thus have some concerns for bias in the selection of the reported results. Two studies additionally had some concerns for bias in randomization and outcome measurement, whereas 1 study had a high risk of bias due to outcome measurement concerns. Risk of bias assessment for 7 observational studies was done using the Cochrane ROBINS-I tool. All 7 observational studies had a serious risk of bias due to absence of statistical corrections for confounding factors. In addition, 2 of the articles had moderate risk of bias and 1 article had a serious risk of bias for outcome measurement. The complete risk of bias assessment tools can be found in Supplemental Material 3.
Patient Pain
Pain was assessed by 4 studies (2 RCTs, 2 observational). One observational study (Burn et al 27 ) provided only descriptions of reported findings and was not included in statistical analysis. The remaining 3 articles, Mohd Rashid et al, 9 Lee et al, 30 and Rabinowitz et al, 35 measured pain using an 11-point visual analog scale (VAS) ranging from 0 (no pain) to 10 (maximal pain). All 3 studies measured pain on injection of local anesthetic. Combined RCT data suggest a statistically nonsignificant mean decrease in pain scores of −1.69 (95% CI = −4.14 to 0.76, P = .18) in the WALANT group compared with LAWT (see Figure 2). This result is discordant with the findings of the observational study by Rabinowitz et al, 35 which found a 2.68-point increase (95% CI = 2.28 to 3.08) in pain on injection when performing TFR under WALANT. Notably, however, in the observational study, the WALANT group received no sedation during injection, whereas the LAWT group underwent intravenous sedation with propofol, limiting comparability to the RCTs for this outcome.
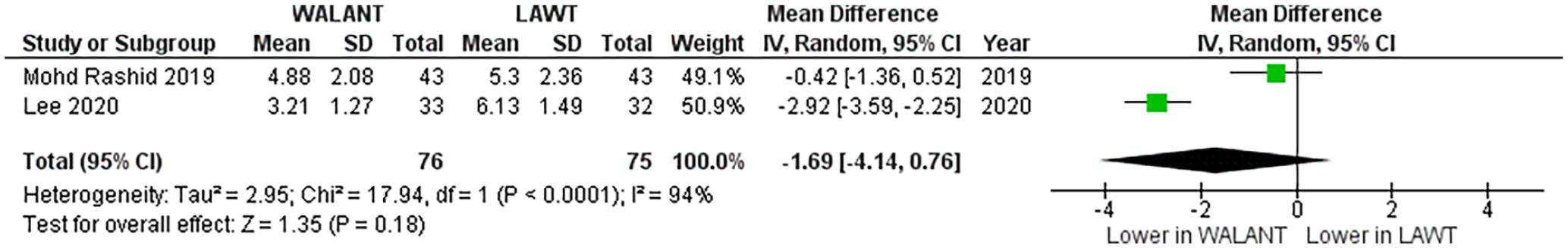
Meta-analysis results for pain (as measured by visual analog scale) during injection, comparing the WALANT approach with LAWT.
Lee et al 30 and Rabinowitz et al 35 additionally reported postoperative pain scores, with both studies finding statistically significant decreases of −2.10 (95% CI = −2.66 to −1.54) and −1.84 (95% CI = −2.89 to −0.79), respectively, in postoperative pain scores of among patients who underwent TFR with the WALANT procedure compared with LAWT.
Patient Satisfaction
Two studies (1 RCT, 1 observational) assessed patient satisfaction. Lee et al 30 administered a 5-point Likert scale (ranging from 1 = very dissatisfied to 5 = very satisfied) during the first postoperative visit 2 days after surgery. Patients in the WALANT and LAWT groups were found to have a satisfaction of 4.18/5 and 3.25/5 (P = .026), respectively. Rabinowitz et al 35 collected survey responses from patients, which found that when asked whether the surgical experience was better, equal, or worse than expected, 86% of the WALANT group (43 patients) said better and 14% said equal, whereas in the LAWT group (33 patients), the responses were 23% better, 71% equal, and 6% worse. When asked how their operative pain was compared with pain during a dental procedure, responses in the WALANT group were: 49% less, 32% equal, 19% never had a dental procedure, whereas in the LAWT group: 29% less, 23% equal, 38% more, 10% never had a dental procedure. When asked which procedure patients would like to undergo for TFR in subsequent cases, 100% of the WALANT group would choose WALANT again, whereas 32% of the LAWT group would choose LAWT and 68% would choose WALANT.
Physician Satisfaction
One RCT, Mohd Rashid et al, 9 assessed physician satisfaction by collecting physician-reported hemostasis scores in WALANT versus LAWT. The scoring system ranged from 1 (minimal bleeding) to 10 (profuse bleeding). Scores of 1 to 3 signified good visibility, 4 to 6 moderate visibility, and 7 to 10 poor visibility. Physicians reported that 74% (32/43) of WALANT cases had good visibility compared with 44% (19/43) in the LAWT group (P = .013). Multivariate analysis showed a 78% decrease in the odds of scoring a moderate or poor score versus a good score in the WALANT group with all other variable remaining constant (P = .003).
Analgesic Use
Measurement of analgesic use varied considerably across studies and thus meta-analysis could not be completed. Lee et al 30 (RCT) found that analgesia use in the WALANT group was significantly (P < .05) lower than in the LAWT group up to and including postoperative day 2 (POD2): (POD6 hours: WALANT 0.33 analgesic [nonsteroidal anti-inflammatory drug] doses used vs 0.81 LAWT, POD12 hours: WALANT 0.41 vs 0.68 LAWT, POD1: WALANT 0.94 vs 1.54 LAWT, POD2: WALANT 0.64 vs 0.96 LAWT). After POD2 there were no statistically significant differences. Rabinowitz et al 35 (observational study) reported that 35% (15/43) of WALANT patients did not require any postoperative analgesia, compared with 0% (0/33) of LAWT patients.
Functional Outcomes
Rabinowitz et al 35 (observational study) measured functional clinical outcomes with the Modified Disabilities Arm Shoulder and Hand (DASH) score preoperatively and postoperatively in both WALANT and LAWT groups (values are presented as DASH score ± SD, where higher scores indicate more severe disability). The DASH scores in the WALANT group were 32.4 ± 18.8 preoperatively and 26.4 ± 19.7 postoperatively. The DASH scores in the LAWT group were 49.9 ± 21.7 preoperatively and 36.5 ± 20.9 postoperatively. Preoperative DASH scores were significantly lower in the WALANT group compared with the LAWT group. Statistically significant (P = .001) improvements in DASH scores were seen in both groups.
Adverse Events
Adverse events were reported by 3 RCTs and 4 observational studies. Mohd Rashid et al, 9 Codding et al, 26 Lee et al, 30 Lee et al, 31 and Rabinowitz et al 35 reported no adverse events, while 2 observational studies (Maliha et al 32 and Rellan et al 33 ) described occurrence of adverse outcomes. Maliha et al 32 reported 1 case of persistent postoperative paresthesia in both the WALANT (1/39) and LAWT (1/37) groups, 2 postoperative infections in WALANT (2/39) versus 0 in LAWT (0/37), 2 incomplete releases or recurrent TFRs or incomplete releases in the WALANT (2/39) group versus 1 in the LAWT (1/37) group. None of these differences were statistically significant. Rellan et al 33 reported no infections in either the WALANT or LAWT group, 3 wound dehiscence complications in each group, and 2 wound adherence complications in both the WALANT and LAWT groups. Similarly, these were not statistically significant.
GRADE Quality of Evidence
Outcomes included in the GRADE certainty of evidence were patient pain on injection, postoperative pain, patient satisfaction, physician satisfaction, analgesic use, functional outcomes, adverse events, preoperative preparation time, operative time, postoperative recovery time, and costs. Grades of evidence were determined to range from moderate to very low for the above outcomes, with the majority rated as having low certainty in the quality of evidence. The full summary of findings table can be found in Supplemental Material 4.
Additional Outcomes
Assessments of operative time and cost are provided in Supplemental Material 5.
Discussion
The historical aversion to administration of epinephrine in fingers to achieve hemostasis has led to a long-standing preference to perform TFR under local anesthesia with a tourniquet. 14 This belief has carried forward to the present day, and despite the myth of epinephrine-related finger loss being disproven almost 2 decades ago, there is still some hesitation among surgeons to employ WALANT.13,16 This systematic review attempted to answer whether performing TFR under WALANT is superior to LAWT for patient pain, patient satisfaction, physician satisfaction, analgesic use, functional outcomes, adverse events, procedure time, and costs.
Data from 2 RCTs suggest that performing TFR under WALANT compared with LAWT results in a statistically nonsignificant decrease in pain on injection of −1.69 points (95% CI = −4.14 to 0.76, P = .18) on the VAS 0 to 10 scale. Notably, this pooled effect estimate is limited by large heterogeneity and small number of included studies. In addition, 1 RCT and 1 observational study both found statistically significant decreases in postoperative pain scores in the WALANT group. Despite lower pain scores seen in the WALANT group, the certainty of this evidence remains low due to the moderate to high risk of bias in these studies.
Narrative summary in this systematic review identified that both patient and physician satisfaction were higher in the WALANT group than in the LAWT group. These findings are consistent with previous studies describing high patient satisfaction with WALANT procedures due to advantages such as absence of tourniquet-associated complications and avoidance of general anesthesia or sedation as well as their associated risks, side effects, and preoperative preparation.37,38 However, the certainty of evidence in these outcomes is very low due to the moderate to high risk of bias of the studies included, as well as the indirectness of the methods used to evaluate the outcomes. For example, patient satisfaction measures included a patient satisfaction Likert scale and questions such as how the real surgical experience was compared with the anticipated, a comparison of pain during surgery to pain throughout a dental procedure, and whether patients would choose to undergo WALANT or LAWT for future operations. Physician satisfaction was evaluated by 1 RCT via a surrogate hemostasis score and found improved visibility when performing TFR under WALANT compared with LAWT. 9 The article illustrates that scores in the WALANT approach were highly concentrated around good visibility, whereas LAWT had a much more varied distribution. However, the study additionally acknowledges a lack of physician blinding potentially being a source of bias, thus possibly warranting further investigation.
This systematic review also identified reduced analgesic use in the WALANT group compared with the LAWT group while functional outcomes and rates of adverse events were not found to be different between the 2 approaches. Preoperative preparation time was statistically significantly longer by 26.43 minutes (95% CI = 15.36 to 37.51, P < .01; see Figure 3), operative time was similar (mean difference: −0.59 minutes, 95% CI = −2.37 to 1.20, P = .52; see Figure 4), and postoperative recovery time was statistically significantly shorter by 27.72 minutes (95% CI = −36.95 to −18.48, P < .01) in the WALANT group compared with LAWT. The mean operating time among included studies was approximately 12 minutes, which is consistent with the senior author’s clinical experience. Notably, there was significant variability among studies, with mean times ranging from approximately 7 to 24 minutes. Besides differences in measurement methods, it is possible that surgery time is dependent on both surgeon expertise and geographic location. In the senior author’s clinical experience, forearm tourniquet time is typically between 3 and 4 minutes and patients rarely complain of tourniquet pain or discomfort for such a short duration. However, previous studies have mentioned that patients might be embarrassed or uncomfortable to admit to pain to their operative surgeon. 9
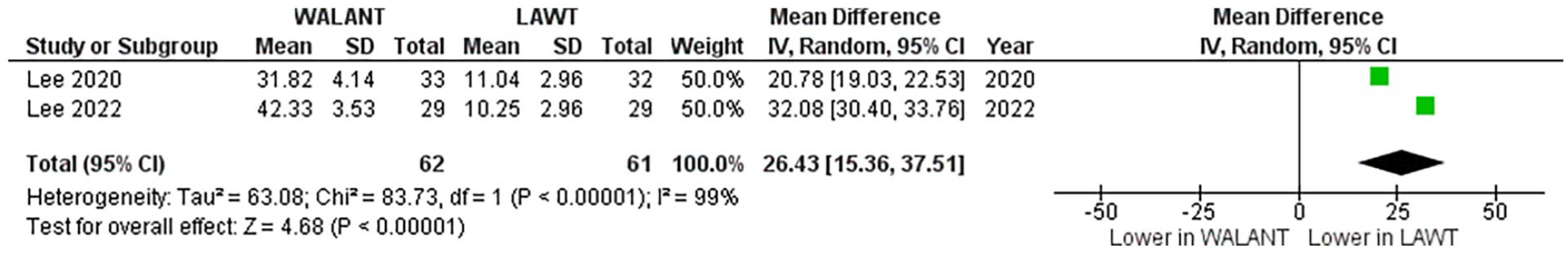
Meta-analysis results for preoperative preparation time, comparing the WALANT approach with LAWT.
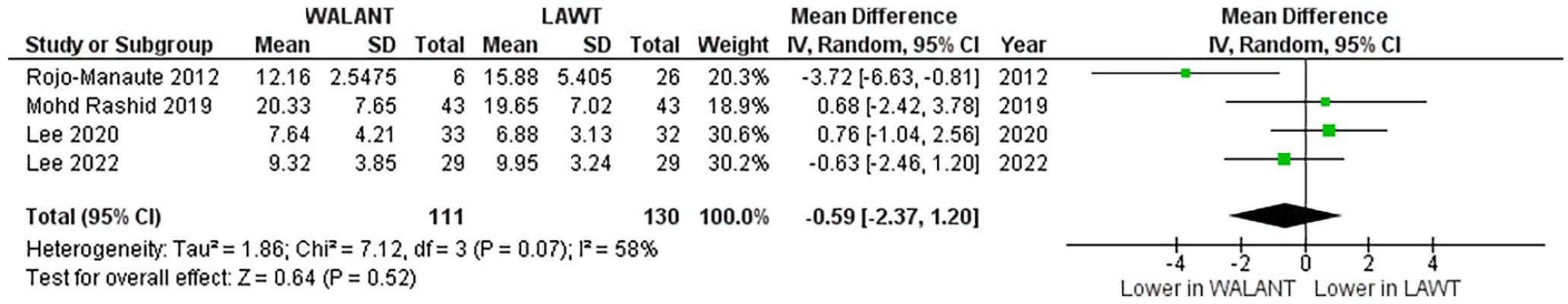
Meta-analysis results for operative time, comparing the WALANT approach with LAWT.
Finally, costs associated with undergoing a TFR under WALANT were found to be lower by 52.2% (95% CI = −79.9% to −24.5%; see Figure 5) compared with undergoing a TFR using the LAWT approach. A limitation of the cost analysis was the large variability and lack of information in the cost reporting of many of the articles included in this analysis. This necessitated performing a cost analysis using only percentage values instead of absolute costs and lacked confidence intervals, standard deviations, weighting, and P values in the provided data. The low degree of confidence in the quality of this evidence limits the external validity of these results to practicing health care systems. In addition, the studies included in this review derive from a variety of different countries including the United States (n = 5), South Korea (n = 2), Argentina (n = 1), Malaysia (n = 1), Spain (n = 1), and Switzerland (n = 1). Proper economic evaluations are needed to find out whether WALANT is a cost-effective procedure compared with LAWT. 39
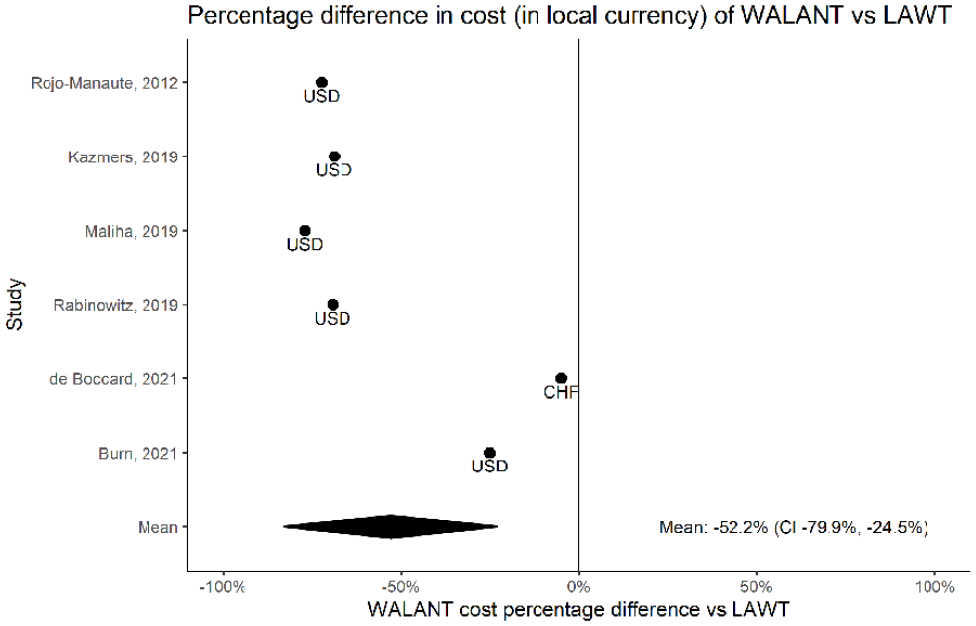
RStudio pooled results cost, comparing the WALANT approach with LAWT. Left of no difference line favors WALANT; right of no difference line favors LAWT.
There are several limitations to this systematic review. First, moderate to high risk of bias was noted in all included studies. Among RCTs, there were concerns for bias in randomization, outcome measurement, and selection of reported results. In addition, all observational studies had a serious risk of bias from potential unaddressed confounding factors, and several had concerns in the outcome measurement domain. Due to the methodological faults of the primary studies, the synthesis in this systematic review consequently also has a moderate to high risk of bias. Second, all pooled results suffered from moderate to considerable heterogeneity that could not be further explored due to a limited number of included studies. Finally, due to a limited number of included studies, the authors were unable to evaluate for the presence of publication bias using a funnel plot.
This systematic review consolidates results from the available evidence on TFR under WALANT and describes some of the limitations of the available literature. Due to the outlined concerns regarding the validity of the evidence presented, a high-quality RCT in which the WALANT approach is compared with the LAWT approach for TFR surgeries is warranted. In addition, such a study should be coupled with an economic evaluation. 40
Conclusions
The existing evidence appears to suggest that TFR under WALANT compared with LAWT shows statistically nonsignificant improvements in pain on injection and statistically significantly decreased postoperative pain. Narrative summary of the evidence found higher patient and physician satisfaction scores, lower analgesic use, and no difference in functional outcomes or rates of adverse events using WALANT. Preoperative time was statistically significantly longer, operative time was similar, postoperative recovery time was statistically significantly shorter, and procedure cost was lower in WALANT compared with LAWT. However, the moderate to high risk of bias of included studies as well as limitations to cost calculations and generalizability of the results ultimately diminishes the level of certainty in this evidence. Overall, the existing randomized and observational study evidence does not establish the superiority of the WALANT approach over LAWT. Thus, the definitive answer can only be ascertained on the execution of further robust studies.
Supplemental Material
sj-docx-1-han-10.1177_15589447231222517 – Supplemental material for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-han-10.1177_15589447231222517 for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis by Tal Levit, Declan C. T. Lavoie, Emily Dunn, Lucas Gallo and Achilles Thoma in HAND
Supplemental Material
sj-docx-2-han-10.1177_15589447231222517 – Supplemental material for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-2-han-10.1177_15589447231222517 for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis by Tal Levit, Declan C. T. Lavoie, Emily Dunn, Lucas Gallo and Achilles Thoma in HAND
Supplemental Material
sj-docx-3-han-10.1177_15589447231222517 – Supplemental material for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-3-han-10.1177_15589447231222517 for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis by Tal Levit, Declan C. T. Lavoie, Emily Dunn, Lucas Gallo and Achilles Thoma in HAND
Supplemental Material
sj-docx-4-han-10.1177_15589447231222517 – Supplemental material for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-4-han-10.1177_15589447231222517 for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis by Tal Levit, Declan C. T. Lavoie, Emily Dunn, Lucas Gallo and Achilles Thoma in HAND
Supplemental Material
sj-docx-6-han-10.1177_15589447231222517 – Supplemental material for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-6-han-10.1177_15589447231222517 for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis by Tal Levit, Declan C. T. Lavoie, Emily Dunn, Lucas Gallo and Achilles Thoma in HAND
Supplemental Material
sj-pdf-5-han-10.1177_15589447231222517 – Supplemental material for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis
Supplemental material, sj-pdf-5-han-10.1177_15589447231222517 for Trigger Finger Release Using Wide-Awake Local Anesthesia No Tourniquet Versus Local Anesthesia With a Tourniquet: A Systematic Review and Meta-analysis by Tal Levit, Declan C. T. Lavoie, Emily Dunn, Lucas Gallo and Achilles Thoma in HAND
Footnotes
Supplemental material is available in the online version of the article.
Ethical Approval
This article did not require review by institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
No informed consent was obtained as this article does not contain any studies with humans.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
