Abstract
Background:
In surgery for Dupuytren disease (DD), palmar fascia specimens are routinely submitted for pathological evaluation. The purpose of this study was to determine the rate of discordant diagnosis and the value of, and costs associated with, routine pathological analysis of palmar fascia tissue extracted in surgery for clinically diagnosed DD.
Methods:
All pathology reports for in-house palmar fascia specimens obtained in surgery for clinically diagnosed DD (time period: January 2001 to December 2020) were retrieved from one academic institution. All specimens were classified by a hierarchical free-text string matching algorithm (HFTSMA) and searched for evidence of malignancy. The primary outcome was percentage of concordant, discrepant, and discordant diagnoses. Secondary outcomes included anatomical location and costs. The HFTSMA was used to capture the anatomical location. Costs included professional, laboratory processing, and ancillary fees based on the Ontario Schedule of Benefits.
Results:
The search retrieved 1323 pathology reports, with 1480 palmar fascia specimens, from 1078 individual patients. By diagnosis, 96.1% of specimens (1422/1480) were concordant (fibromatosis), 3.9% (58/1480) were discrepant (scarring/fibrosis, benign fascia/connective tissue, or other benign findings), and 0% (0/1480) were discordant. The most common location was ring finger (n = 381, 48.7%). Ancillary testing was minimal. The cost per palmar fascia specimen was estimated to be CAD $34.57. The institutional costs were approximately CAD $2558.18/year.
Conclusions:
Routine pathological examination of specimens in cases of clinically diagnosed DD does not yield additional clinically important findings and may not warrant their costs.
Introduction
Dupuytren disease (DD) is a benign fibroproliferative disorder of the palmar and digital fascia that can result in deformity and reduced function of the hand due to the development of nodules, cords, and contractures of the digits.1,2 There are many treatment options for the management of DD, including nonoperative and minimally invasive approaches. Surgical approaches, such as partial or total fasciectomy, are recommended when DD has progressed to the point where digital flexion contractures decrease the ability to use the hand effectively, and/or the proximal interphalangeal joint is flexed to 30° or more. 3 A common practice in surgery for DD is to obtain a pathology sample of the cord and send it for evaluation in a pathology lab. 1 The tissue sample is analyzed to rule out neoplastic tissue. 1 Rare cases of epithelioid sarcomas misdiagnosed as DD have been reported 4 ; however, the pathologist will most often confirm benign palmar fibromatosis, in keeping with DD.1,5 Therefore, routine pathologic examination of clinically diagnosed DD may not provide value (health outcomes achieved per dollar spent).
Low value for money spent on health care has been identified as a challenge in North America.6 -9 According to data from the Organisation for Economic Co-operation and Development (OECD), in 2019, the United States spent 16.8% of its gross domestic product (GDP) on health care spending, the highest of all OECD member countries. 10 However, the United States only ranked 28th of all OECD member countries in life expectancy after birth (2016-2020). 11 Canada spent 10.8% of its GDP (ranked 11th of all OECD member counties) on health care spending 10 and ranked 12th on life expectancy at birth. 11 Efforts have been made to reduce health care spending and improve value.6 -9 Efforts include Choosing Wisely, a campaign launched by the American Board of Internal Medicine Foundation with the goal to reduce wasteful or unnecessary medical tests, treatments, and procedures. 12
Previous studies have examined the value and waste of practices in the pathological examination of extracted tissue of gallbladders, 13 knee replacements, 14 tonsils, 15 intervertebral disc, 16 and hernias. 17 When considering the necessity of practices such as tissue sample pathological analysis, it is important to consider opportunity cost. Opportunity cost is defined as the value of resources no longer available for alternative use. 18 In the case of pathological analysis of tissue specimens obtained in surgery for DD, valuable resources consumed include the time of the pathologist, as well as other associated costs. The purpose of this study was to determine the rate of discordant diagnosis and the value of, and costs associated with, routine pathological analysis of palmar fascia tissue extracted in DD surgery.
Materials and Methods
This is a longitudinal descriptive retrospective chart review study conducted in compliance with the REporting of studies Conducted using Observational Routinely collected Data checklist 19 (Supplemental Table S1). Research ethics board approval (from the Hamilton integrated Research Ethics Board [HiREB]) for this project (HiREB Project ID: 13746-C) was obtained.
The project builds on in-house experience in the analysis of free-text pathology reports.20,21 All pathology reports were retrieved from the Laboratory Information System from St. Joseph’s Healthcare Hamilton (Hamilton, Ontario), a McMaster University–affiliated hospital, for the time horizon of the study: January 1, 2001, to December 31, 2020. Custom computer code (written in the programming language Python 22 ) stripped all patient identifiers and replaced the medical record number with an anonymous unique identifier. Subsequently, the report sections were reconstructed to allow complex searches by section.
The primary outcome of this study was the percentage of concordant, discrepant, and discordant diagnoses. A concordant diagnosis was defined as the pathology diagnosis that confirms the clinical diagnosis of DD (fibromatosis).23,24 Discrepant diagnosis was defined as the pathology diagnosis that differs from preoperative diagnosis of DD, but no change in management is required.23,24 A discordant diagnosis was defined as the pathology diagnosis that differs from preoperative diagnosis of DD, and change in management is required.23,24 Secondary outcomes of this study included: (1) anatomical location of tissue sample; and (2) costs (Ontario Schedule of Benefits 25 and the Ontario Schedule of Benefits for Laboratory Services 26 ).
The required sample size was determined a priori through calculations 27 conducted by a biostatistician (E.D.) and was calculated to be approximately 500 (with a 99% chance of observing at least 1 discordant diagnosis), with the expected precision of the 95% confidence interval (±0.01) (see Supplemental Table S2). The study cohort was obtained by searching the “source of specimen” for “dupuytren” or (“fascia” and (“palmar” or “plantar”)). The plantar cases were purged after classification. The computer code also searched the diagnosis section of each report to classify cases. This involved comparing the diagnosis text against a dictionary of terms (see Supplemental Table S3) that were arranged in hierarchy (see Supplemental Table S4).
Block count and ancillary testing were retrieved from the “pathology procedures” section of the pathology report. Informal internal pathology consultations (inferred by the presence of another MD’s name in the report) and formal external pathology consultations were also captured; details are provided in a prior publication. 21 In the 2 cases that consults were made to soft tissue pathologists, the entire reports of the soft tissue pathologist consultations were retrieved (using the pathology case identifiers) from the dump of anonymized pathology reports, such that they could be fully understood.
For all reports, the report text was separated and output into a tab-separated text file (csv) with the diagnostic codes, location codes, blocks, ancillary tests, and workload measures. The csv file was imported into LibreOffice Calc (libreoffice.org) for auditing, further analysis, and tabulation to minimize the risk of bias. Data extracted included diagnosis type and tissue sample anatomical location. The anatomical location was obtained by searching the “source of specimen” section of the report using a second dictionary of terms (see Supplemental Table S5). If the program failed to find any locations in the “source of specimen” section, it would then look in the “diagnosis” section of the report using the same dictionary.
Cost estimate calculations were based on methodology in similar studies.15,23 Cases with 1 specimen were used to estimate the average number of blocks per case, as often cases with multiple specimens include nonpalmar fascia specimens (eg, a case may consist of a “palmar fascia” specimen and a “ganglion cyst”). Data from 2011 to 2020 were used to calculate the average number of blocks per case. The earlier data were excluded as: (1) validation was completed on the 2011-2019 data; and (2) block count data were largely incomplete in the earlier data. Cost per block was then calculated based on the professional component of the pathologic assessment for a palmar fibromatosis specimen (code L863) in the Ontario Schedule of Benefits (July 2, 2021). 25 For laboratory processing fees, the cost per tissue block was estimated based on the Ontario Schedule of Benefits for Laboratory Services 26 (July 1, 2020), capped at 8 blocks per day, per patient.
Costs for ancillary tests and consults were also calculated. This included costs for special stains, immunostains, informal consults, and formal consults based on the Ontario Schedule of Benefits for Laboratory Services. 26 Ancillary testing data were only available for the period 2008-2020, as earlier data were not recorded.
Results
The search (January 1, 2001, to December 31, 2020) retrieved 1323 pathology reports with 1480 palmar fascia specimens from 1078 individual patients with clinically diagnosed DD. The pathology diagnostic findings were verified by simple string searches and an audit of 300 specimens; 100% of 300 specimens were correctly classified. The hierarchical free-text string matching algorithm classified all specimens and found no evidence of malignancy. By diagnosis, 96.1% (1422/1480) of specimens were concordant (fibromatosis), and 3.9% (58/1480) were discrepant (1.4% scarring/fibrosis and 2.5% benign fascia/connective tissue or other benign findings). Zero (0%) cases of discordant diagnoses were found. Two cases were sent to a soft tissue pathologist for consultation for second opinion, and both cases were found to be benign. Four additional cases had an informal internal consultation, where another pathologist’s name appeared in the report.
The clinical history contained “Dupuytren” in 881 of 1101 cases. In 165 cases, the history was “not available on requisition” (a placeholder entered when no clinical history is provided); conservatively calculated, a history was missing in 15% of cases (160 of 1101 cases). Most patients (n = 810, 75.14%) had 1 palmar fascia specimen. Most pathology cases consisted of 1 specimen (1190); a breakdown of specimens per pathology case is provided in Table 1.
Numbers of Specimens and Cases.

Note. This table outlines the number of cases that had a total of 1 specimen analyzed, 2 specimens analyzed, 3 specimens analyzed, or 4 specimens analyzed.
Ancillary testing information was available in reports completed in mid-2007 and later. Six cases had deeper cuts, 15 cases had at least 1 immunohistochemical stain, and 1 case had a special stain.
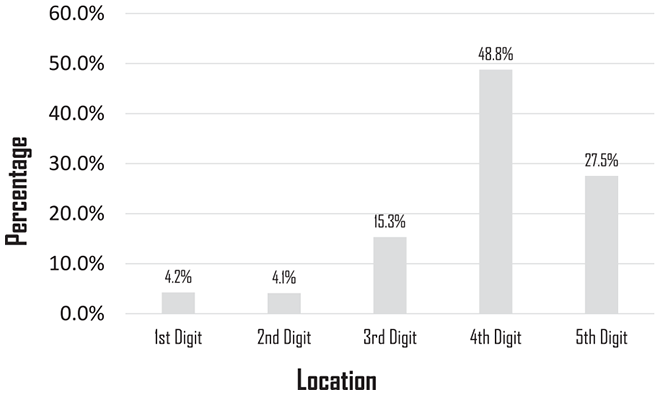
Location information was available for 93% (1385/1480) of specimens. By location, the specimens (where laterality was available) were 51.4% (712/1385 specimens) right hand and 48.6% (673/1385) left hand. The specific finger location available in 53% of specimens (783/1480) are as follows: 48.7% (381) ring finger, 27.8% (218) little finger, 15.2% (119) long finger, 4.2% (33) thumb, and 4.1% (32) index finger (Figure 1).
An average of 1.074 blocks per specimen was determined, based on 706 one-part palmar fibromatosis cases with 758 blocks between 2011 and 2020. The professional component of the pathologic assessment for a palmar fibromatosis specimen (code L863) in the Ontario Schedule of Benefits is CAD $14.30 per block. The laboratory processing cost per block, based on the Ontario Schedule of Benefits for Laboratory Services, is CAD $18.75. 26 Thus, the estimated processing cost per palmar fascia specimen ($18.75 × 1.074 blocks per specimen) was CAD $20.08. The cost of ancillary testing is estimated at CAD $0.19 per specimen block. The total ancillary costs are summarized in Table 2. Thus, the total estimated cost per specimen was CAD $34.57. In the 20-year period, the total cost of DD surgery pathology evaluation was CAD $51 163.60, composed of professional fees, laboratory processing fees, and ancillary testing fees. The yearly average costs were calculated to be CAD $2558.18. The estimated costs per block are summarized in Table 3.
Costs of Ancillary Tests and Consults.
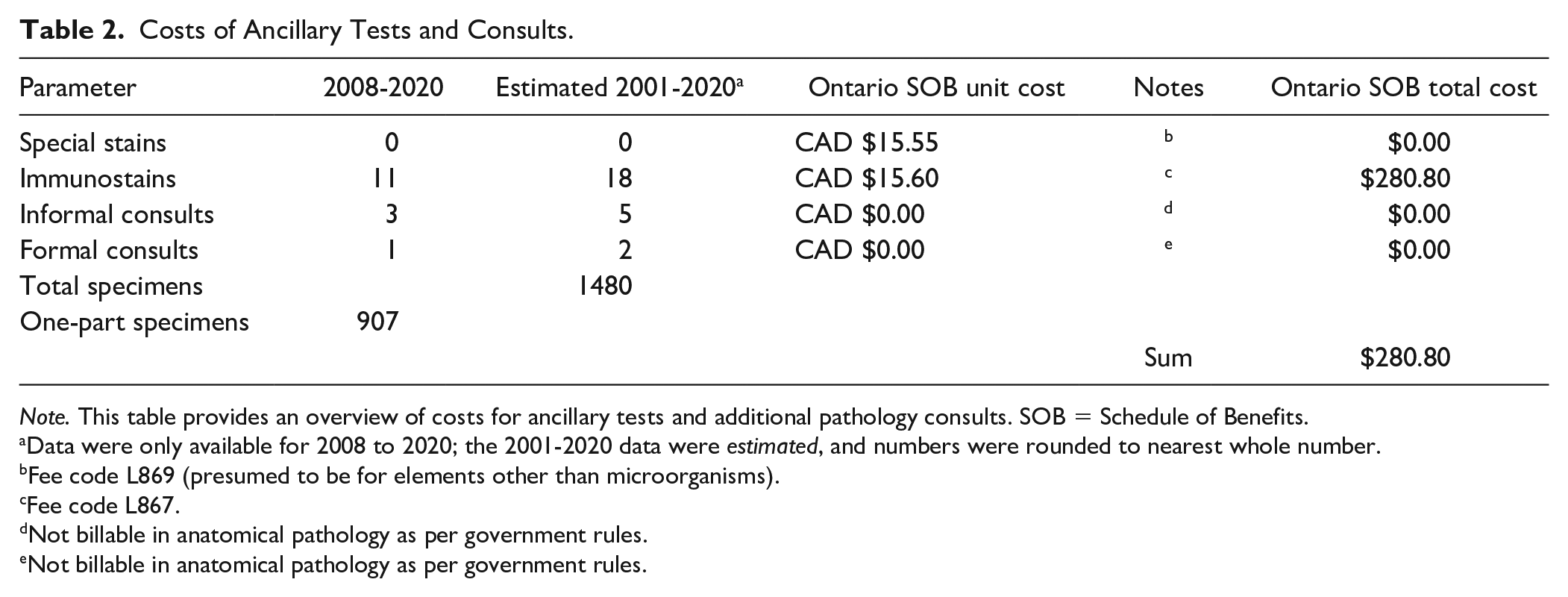
Note. This table provides an overview of costs for ancillary tests and additional pathology consults. SOB = Schedule of Benefits.
Data were only available for 2008 to 2020; the 2001-2020 data were estimated, and numbers were rounded to nearest whole number.
Fee code L869 (presumed to be for elements other than microorganisms).
Fee code L867.
Not billable in anatomical pathology as per government rules.
Not billable in anatomical pathology as per government rules.
Cost Breakdown per Specimen as per the Ontario Fee Schedule.

Note. This table provides an overview of the cost components of the total cost per specimen analyzed in the routine pathological evaluation of tissue extracted in surgery for Dupuytren disease. SOB = Schedule of Benefits.
Not billable in anatomical pathology as per government rule.
Discussion
North American health care centers have adopted quality management strategies to identify and remove health care waste with the goal to reduce inefficiencies and increase quality.23,24,31 Health care waste is defined as unnecessary processes or services that do not yield value (health outcomes achieved per dollar spent). 32 Unnecessary routine pathological examination of tissue specimens may be a wasteful practice, under the category of low-value care. This study sought to determine the value of routine pathological analysis of palmar fascia tissue extracted during DD surgery. Value was determined by comparing the rate of concordant, discordant, and discrepant diagnoses with the costs associated with pathological analysis. Most specimens (96.1%) were concordant, and no specimens were discordant. Discrepant diagnoses were rare (3.9%) and did not change future management for patients. Of those with discrepant diagnoses, 1.4% were benign fascia/connective tissue and 2.5% were other benign findings. The total approximated cost per specimen in the current study was CAD $34.57 or US $27.97, with a total 20-year estimated cost of more than CAD $51 000.00 or US $41 266.90 (as of the writing of this article).
Many studies have evaluated the necessity of routine pathological examination of tissue specimens.13 -16,24,33,34 A recent study by Liu 34 also evaluated routine pathological examination of specimens for clinically diagnosed DD and found similar results to this study. Using samples from 376 patients, Liu 34 reported that 97.1% had concordant diagnoses, 2.9% had discrepant diagnoses, and 0% had discordant diagnoses (2010 and 2020). Gross cost per specimen was found to be US $512, based on pathology lab fees at 1 institution. 34 In the United States, Current Procedural Terminology (CPT) codes for medical services are used by health care providers for reimbursement by Medicare and other third-party payers. 35 For the pathologic evaluation of tissue specimens obtained in surgery for clinically diagnosed DD, the CPT fees, comprising professional and technical components, is US $42.22. 36
Reducing unnecessary pathological examination of tissue specimens has been an area of attention for many health care associations and organizations.37 -40 In the United States, routine pathological examination of tissue specimens extracted in surgery is recommended, 24 as outlined in the Manual of the Joint Commission on Accreditation of Healthcare Organizations 41 . Currently, in the United States, there are no widely accepted standards 39 ; however, efforts have been made to standardize and provide a framework of defining which types of tissues can be exempt from pathological examination. 38 In Canada, a recent policy developed by the Department of Pathology and Laboratory Medicine and Nova Scotia Health Authority evaluated the requirement of having all removed tissues undergo pathological evaluation. 42 Certain tissues were subsequently deemed exempt from such evaluation; however, exceptions did not include tissue extracted during DD surgery. 42 After implementing the exemption of 34 tissue types in Nova Scotia, it was found that, over 2 years, the total number of analyzed specimens decreased by 10%, resulting in savings of approximately CAD $36 205.28, when considering laboratory staff, pathologist time, and dollar value. 42
The effort to reduce unnecessary routine pathological analysis of tissues is aligned with the goals of the Choosing Wisely campaign, 12 an initiative with the goal to reduce wasteful or unnecessary medical tests, treatments, and procedures. 43 The American Society for Clinical Pathology (ASCP) and the American Society of Plastic Surgeons have both participated in the campaign and recommend reducing unnecessary practices.12,43,44 Although recommendations do not include the elimination of routine pathological analysis of extracted tissue specimens from DD surgery, the ASCP has recommended to “not routinely perform sentinel lymph node biopsy or other diagnostic tests for the evaluation of early, thin melanoma because these tests do not improve survival.” 45 Pathology findings have been used as a quality check in the medical system 46 and can be used to confirm clinical diagnoses and the need for surgical treatment. Although the current study found discrepant diagnoses to be rare, routine pathological examination of tissues from surgically extracted palmar fascia specimens may function as a validation strategy of clinically diagnosed DD and prevent unnecessary surgeries from taking place. 46 The use of pathological analysis, however, could be reduced, and other methods, such as reviewing patient medical history for angle degree deficit in extension of a digit, could be used. In the current study, 0 (0%) discordant diagnoses were found; therefore, the practice of routine pathological analysis for tissue specimens obtained in surgery for DD may not provide value (health outcomes achieved per dollar spent).
The conclusions drawn in this article should be interpreted under the following limitations: This study used a retrospective design with reliance on free-text pathology reports. In 165 cases, the history was “not available on requisition” (a placeholder entered when no history is provided); conservatively calculated, the history was missing in 15% of cases (160 of 1101 cases). This shows the importance of completion of the pathology requisition by surgeons at the completion of the surgery. The report addenda were not searched for diagnostic information. The secondary outcome of cost did not include a full cost analysis. Specific details of costs, such as unit numbers and prices for material costs, were not reported, as those costs are included in laboratory processing fees. 26 In addition, a model projecting and discounting future costs was not performed. A cost analysis should be conducted in the future to determine the value of clinical outcome achieved per dollar spent. In addition, a cost comparison between pathological evaluation conducted in the United States and Canada would be valuable in a future study.
Supplemental Material
sj-docx-1-han-10.1177_15589447221128982 – Supplemental material for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value?
Supplemental material, sj-docx-1-han-10.1177_15589447221128982 for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value? by Kathryn Uhlman, Michael Bonert, Jessica Murphy, Eric Duku, Asghar Naqvi and Achilles Thoma in HAND
Supplemental Material
sj-docx-2-han-10.1177_15589447221128982 – Supplemental material for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value?
Supplemental material, sj-docx-2-han-10.1177_15589447221128982 for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value? by Kathryn Uhlman, Michael Bonert, Jessica Murphy, Eric Duku, Asghar Naqvi and Achilles Thoma in HAND
Supplemental Material
sj-docx-3-han-10.1177_15589447221128982 – Supplemental material for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value?
Supplemental material, sj-docx-3-han-10.1177_15589447221128982 for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value? by Kathryn Uhlman, Michael Bonert, Jessica Murphy, Eric Duku, Asghar Naqvi and Achilles Thoma in HAND
Supplemental Material
sj-docx-4-han-10.1177_15589447221128982 – Supplemental material for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value?
Supplemental material, sj-docx-4-han-10.1177_15589447221128982 for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value? by Kathryn Uhlman, Michael Bonert, Jessica Murphy, Eric Duku, Asghar Naqvi and Achilles Thoma in HAND
Supplemental Material
sj-docx-5-han-10.1177_15589447221128982 – Supplemental material for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value?
Supplemental material, sj-docx-5-han-10.1177_15589447221128982 for Routine Pathological Examination of Clinically Presumed Dupuytren Disease: Does It Add Value? by Kathryn Uhlman, Michael Bonert, Jessica Murphy, Eric Duku, Asghar Naqvi and Achilles Thoma in HAND
Footnotes
Author Contributions
All authors verify that they have made substantial contributions to: (1) participation in the intellectual content; (2) participation in the analysis of data; and (3) participation in the writing and revision of the manuscript. In addition, the authors: (1) certify that they have reviewed the final version of the manuscript; (2) believe it represents valid work; (3) approve it for publication; (4) certify that none of the material in the manuscript has been previously published, is included in another manuscript, or is currently under consideration for publication elsewhere; (5) certify that this article has neither been accepted for publication elsewhere nor have any rights or interests in the manuscript been assigned to any third party; and (6) certify that they have data upon which the manuscript is based and will be able to produce them if the Editor of HAND requests.
Ethical Approval
Research ethics board approval (from the Hamilton integrated Research Ethics Board [HiREB]) for this project (HiREB Project ID: 13746-C) was obtained.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
This was a retrospective review study, and therefore, informed consent was not obtained due to the following reasons:
The research purposes could not be achieved without the information, as it was not possible to extract and verify the integrity of the data without access to pathology reports. It was impracticable to obtain consent due to the number of patient reports that were needed to answer the research question. The information was used in a manner that ensured its confidentiality. All identifiers with the exception of the medical record number were removed from the data set. Individual patients are not identifiable in the results. This work is in the public interest, as it has the potential to lead to reduced health care waste.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
