Abstract
Background:
This study aimed to assess the responsiveness of the Trapeziometacarpal Arthrosis Symptoms and Disability (TASD) questionnaire after corticosteroid injections and operative management for trapeziometacarpal osteoarthritis (TMC-OA).
Methods:
This longitudinal cohort study included patients with TMC-OA who underwent treatment (corticosteroid injection or surgery). Measurements occurred at baseline and follow-up time points: 6 weeks, 3 months, 6 months, and 1 year. Patient measures were collected electronically using a data repository platform. The TASD is a validated psychometric patient-administered scale. We examined responsiveness by correlating the TASD scores with another known effective indicator of change (shortened Disabilities of the Arm, Shoulder, and Hand [QuickDASH]). Repeated score measurements over time were analyzed using analysis of variance, and correlations between questionnaires were reported with repeated measures correlation and Pearson correlation coefficients.
Results:
The nonsurgical cohort undergoing corticosteroid injections included 31 (66%) women and 16 (34%) men, and the surgical cohort included 29 (71%) women and 12 (29%) men. Both the QuickDASH and the TASD captured a statistically significant improvement in symptom burden postoperatively (P < .0001 for both questionnaires), whereas no statistical significance was noted after corticosteroid injections (P = .45 and P = .34, respectively). There was a strong correlation between QuickDASH and TASD questionnaires (r = 0.87, P < .0001).
Conclusions:
The TASD is a promising TMC-OA–specific questionnaire to capture responsiveness following treatment, particularly following surgery. The responsiveness of TASD was demonstrated in the TMC-OA cohort. Further research is needed to define a minimal clinically important difference.
Introduction
Trapeziometacarpal (TMC) joint arthrosis is a debilitating condition that imposes significant functional limitations for vocational and avocational activities. 1 Clinical presentation usually involves pain at the base of the thumb, instability, bony deformity, and limitations in function. 2 According to population studies, radiographic TMC joint arthrosis is present in 4% to 33% of patients, with women being more commonly afflicted.3 -5 Management of TMC joint arthrosis is individualized to each patient based on symptomatic burden, disease characteristics, expectations, and preferences. Nonoperative treatment options include temporizing measures such as splinting, intra-articular corticosteroid injections or hyaluronate, activity modifications, and nonsteroidal anti-inflammatory drugs (NSAIDs).6,7 When nonoperative management is unsuccessful, surgery is recommended.
Trapeziometacarpal osteoarthritis (TMC-OA) has significant heterogeneity in reporting outcome measures. 8 A recent systematic review identified 25 outcome measures in assessing TMC-OA. Although patient-reported outcomes were the most common outcome measure, only 10 outcome measures were validated. 8 Thus, assessing the validation and responsiveness of patient-reported outcomes is critical due to their widespread use.
The QuickDASH (shortened Disabilities of the Arm, Shoulder, and Hand) is a patient-reported questionnaire made up of 11 items on a 5-point Likert scale, with higher score reflecting disability. 9 It was originally developed to assess disability regarding physical function and symptoms for upper extremity musculoskeletal disorders 10 and has shown good validity in patients with rheumatoid arthritis. 11 The DASH and QuickDASH questionnaires were the most common methods used in assessing TMC-OA. However, they are not specific to TMC pathologies. Therefore, the use of a validated and responsive instrument to TMC-OA will better characterize the clinical outcomes in future trials.
The complexity of experienced symptoms and disability of patients with TMC joint arthrosis indicate the importance of measuring outcomes with patient-reported and disease-specific tools. The appropriateness of an instrument for the target patient population and intervention is essential as it can be a primary outcome measure in future studies including randomized clinical trials. 12 A recent joint-specific tool, the Trapeziometacarpal Arthrosis Symptoms and Disability (TASD) questionnaire, has been developed and validated with the hope to better discriminate between symptoms and disability caused by TMC joint arthrosis versus other upper extremity diseases. 13 This psychometric scale was shown to have high internal consistency and good convergent, discriminative, and known-group construct validity. 13
Responsiveness is commonly regarded as the ability to detect true differences in an instrument. 14 Actual differences would need to be verified using another instrument to confirm that a difference was observed. 14 This study aims to assess the responsiveness of the TASD after nonoperative (corticosteroid injections) and operative management for TMC joint arthrosis. For the purpose of this study, responsiveness was defined as the accuracy with which the TASD was able to detect change that occurred before and after treatment. 15
Materials and Methods
Study Design and Setting
This longitudinal cohort study took place in a high-volume tertiary care Hand Surgery Program between October 2016 and December 2019. Institutional Research Ethics Board approval and written informed consent from all individual participants were obtained. Patients were followed after undergoing treatment (corticosteroid injection or surgery) at the 6-week (for the surgical group only), 3-month, 6-month, and 1-year time points.
Study Population
We included adult patients with a primary clinical diagnosis of TMC joint arthrosis, defined as tenderness and crepitation with axial pressure or circumduction of the joint. Only patients who underwent management for their TMC arthrosis with either: (1) corticosteroid injections; or (2) operative interventions (e.g. trapeziectomy ± ligament reconstruction and tendon interposition or joint fusion) were included. Exclusion criteria consisted of the: (1) lack of written and/or oral English fluency; (2) inability to provide informed consent; (3) history of inflammatory arthritis; (4) history of crystalline arthropathy; (5) history of posttraumatic arthritis; and (6) history of any prior surgical intervention in the treated hand.
Trapeziometacarpal Arthrosis Symptoms and Disability Questionnaire
The TASD is a validated psychometric scale composed of 2 subscales which was developed in 2016. 13 This patient-administered questionnaire consists of 12 questions on a 5-point Likert scale and measures symptoms (7 items) and disabilities (5 items). Scores are obtained by summing all responses, subtracting 1, and multiplying by 25. 13 Scores range between 0 and 100, with higher scores representing more severe levels of symptoms and/or disability. 13
Data Collection
Patient-reported outcome measures were collected electronically using a data repository platform. The time points for the data collection were similar in both groups except for the 6-week visit, where data were collected only from the surgical cohort. Baseline sociodemographic characteristics, such as work status and medical history, were also collected. Physical examination components such as key pinch strength and grip strength were reported. Finally, TASD and QuickDASH scores were calculated from the self-administered questionnaires.
Assessing Responsiveness
Responsiveness assessment of functional scales should aim to determine the ability to discriminate between patients who improve and those who do not. 16 No standardized methods of assessing responsiveness exist. The approach used to assess responsiveness in this study was correlation with other indicators of change. For example, among the methods used by Beaton and colleagues to test the responsiveness of the DASH questionnaire, a correlation analysis between the changes in DASH and other outcome measures was carried out. 14 In this study, we examined responsiveness with score changes following surgical or nonsurgical intervention, that is, corticosteroid injection. Patient score changes were measured, and the TASD was compared with the QuickDASH.
Statistical Analysis
Descriptive statistics were performed. Baseline patient and disease characteristics were compared between patients who experienced perioperative complications and those who did not. Categorical variables were described as proportions (percentages). The 95% confidence intervals (CIs) were constructed for the mean TASD and QuickDASH scores using bootstrapping methods. Normality of continuous variables was assessed with the Shapiro-Wilk test or QQ-plots. Non-normally distributed continuous variables were reported as medians (interquartile range), and normally distributed continuous variables were presented as means (SD).
Repeated score measurements over time were analyzed using a 1-way analysis of variance (ANOVA), and correlations between questionnaires were reported with repeated measures correlation. The analysis for the repeated measures ANOVA was done after the normality assumption was verified. Mauchly’s test was used to check the sphericity assumption. As post hoc analyses, a determination of differences between time points was done using Bonferroni correction. As sensitivity analyses, Pearson correlation was used at each time point to compare the results of QuickDASH and TASD scores. The standardized response mean (SRM) for the TASD was calculated between the baseline visit and the 6-month follow-up visit for each treatment cohort due to the least amount of missing data. All statistical tests were 2-sided. A P value of <.05 was considered significant.
Results
A total of 88 patients constituted both the surgical and the nonsurgical cohorts. The patients’ sociodemographic characteristics can be found in Table 1. Of the 47 patients included in the corticosteroid group, 66.0% were women (n = 31). The mean (SD) age of participants at the time of enrollment in the study was 63 (8.6) years. Similarly, the surgical cohort included 29 (70.7%) women and 12 (29.3%) men of a mean (SD) age of 60 (8) years (Table 1). Table 2 summarizes the medications used for pain management by the patients in both the surgical cohort and the corticosteroid cohort. There were no major differences between the 2 groups in the use of muscle relaxants and over-the-counter medications at the baseline visit. The use of NSAIDs was more frequent in the nonsurgical group (standardized mean difference = 0.35). However, the use of narcotic medications was more common in the surgical group at the baseline visit (standardized mean difference = 0.26).
Patients’ Sociodemographic Characteristics.
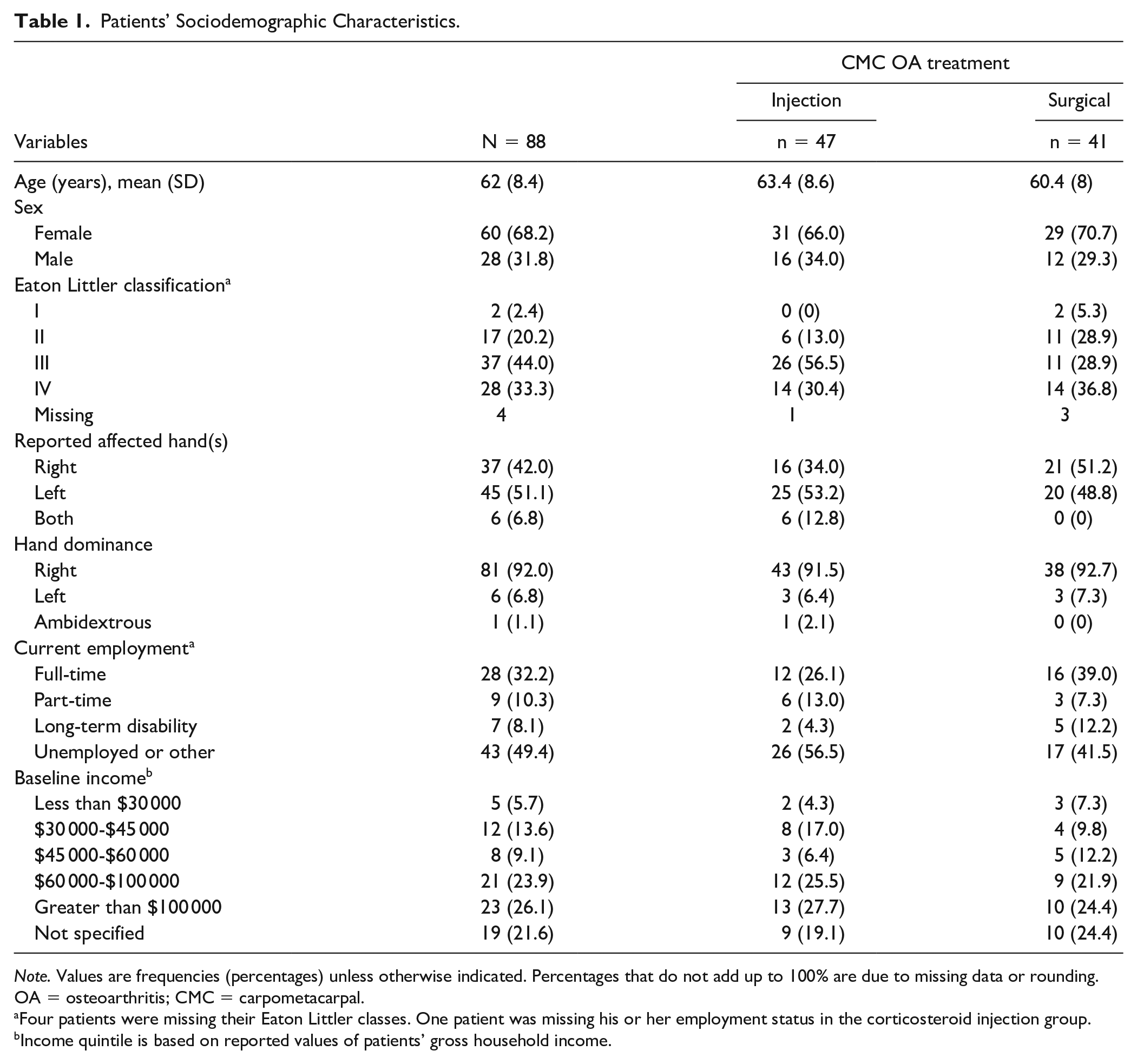
Note. Values are frequencies (percentages) unless otherwise indicated. Percentages that do not add up to 100% are due to missing data or rounding. OA = osteoarthritis; CMC = carpometacarpal.
Four patients were missing their Eaton Littler classes. One patient was missing his or her employment status in the corticosteroid injection group.
Income quintile is based on reported values of patients’ gross household income.
Patients’ Pain Medication Use Characteristics.
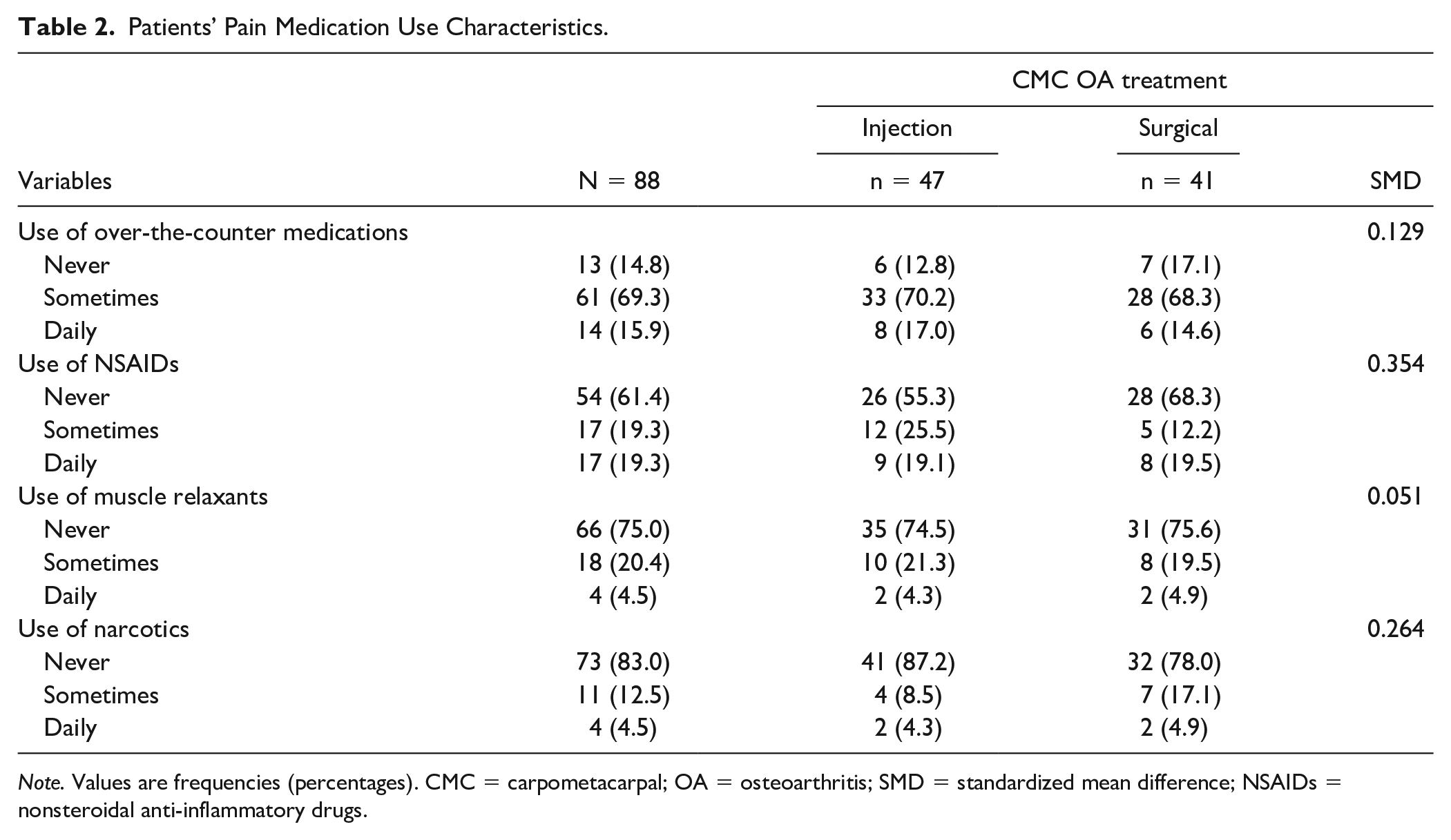
Note. Values are frequencies (percentages). CMC = carpometacarpal; OA = osteoarthritis; SMD = standardized mean difference; NSAIDs = nonsteroidal anti-inflammatory drugs.
The corticosteroid injection cohort had 6 patients (12.8%) who had a repeat injection (4 at the 3-month follow-up visit, 2 at the 6-month follow-up visit) and 1 patient (2.1%) who had 2 repeat injections at both the 3-month and 6-month follow-up visits. No patients in the corticosteroid injection cohort moved to the surgical group during the study period. For the QuickDASH, the data were complete at the baseline visit in the surgical group. However, the data were missing in 6 patients (14.6%), 8 patients (19.5%), 9 patients (22%), and 16 patients (39%) at the 6-week, 3-month, 6-month, and 12-month visits in the surgical group, respectively. For the corticosteroid injection group, the data were missing in 1 patient (2.1%), 13 patients (27.7%), 20 patients (42.6%), and 30 patients (63.8%) at the baseline, 3-month, 6-month, and 12-month visits, respectively. For the TASD scores, the data were complete at the baseline visit for all the study participants. The data were missing in 6 patients (14.6%), 8 patients (19.5%), 9 patients (22%), and 16 patients (39%) at the 6-week, 3-month, 6-month, and 12-month visits in the surgical group, respectively. For the corticosteroid injection group, the data were missing in 12 patients (25.5%), 20 patients (42.6%), and 30 patients (63.8%) at the 3-month, 6-month, and 12-month visits, respectively.
Repeated measures correlation showed an overall strong correlation between QuickDASH and TASD (r = 0.87, 95% CI: 0.83-0.90, P < .0001) (Figure 1). The TASD was strongly correlated with QuickDASH at initial consultation and at 1-year follow-up visit (r = 0.86, 95% CI: 0.79-0.90, P < .0001 and r = 0.84, 95% CI: 0.71-0.91, P < .0001, respectively).
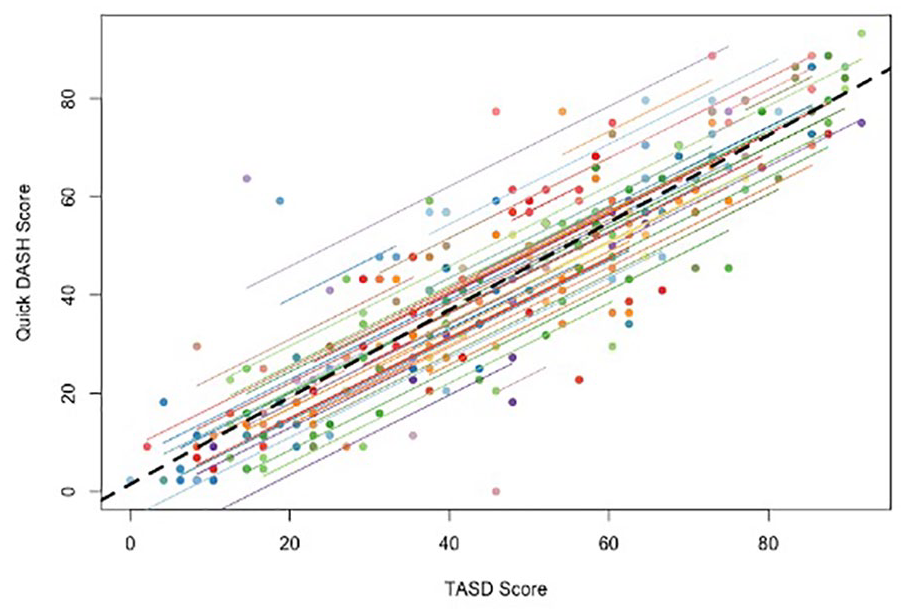
Repeated measures correlation between QuickDASH and TASD scores.
Both the QuickDASH and the TASD captured a statistically significant improvement postoperatively (P < .0001), with an initial increase in scores 6 weeks following surgery and consistent improvement within a year. An improvement in baseline QuickDASH and TASD scores was demonstrated at all time points following corticosteroid injection, yet this was not statistically significant (P = .45 and P = .34, respectively) (Figures 2 and 3). Post hoc analyses showed differences between different time points in the surgical cohort for both QuickDASH and TASD questionnaires (Figures 4 and 5). The differences were significant after applying Bonferroni correction for multiple comparisons between the baseline visit and all subsequent visits except the 6-week visit for the TASD questionnaire (P values are shown in Figure 5). Although the QuickDASH captured a statistically significant difference at 6 weeks compared with the baseline visit, there was no significant difference between the baseline visit and 3-month visit. Other findings in the QuickDASH were relatively similar to those of the TASD questionnaire. The SRM for the TASD at 6 months in the surgical group was 1.31 and in the corticosteroid group was 0.16, indicating higher responsiveness of the TASD following surgery.
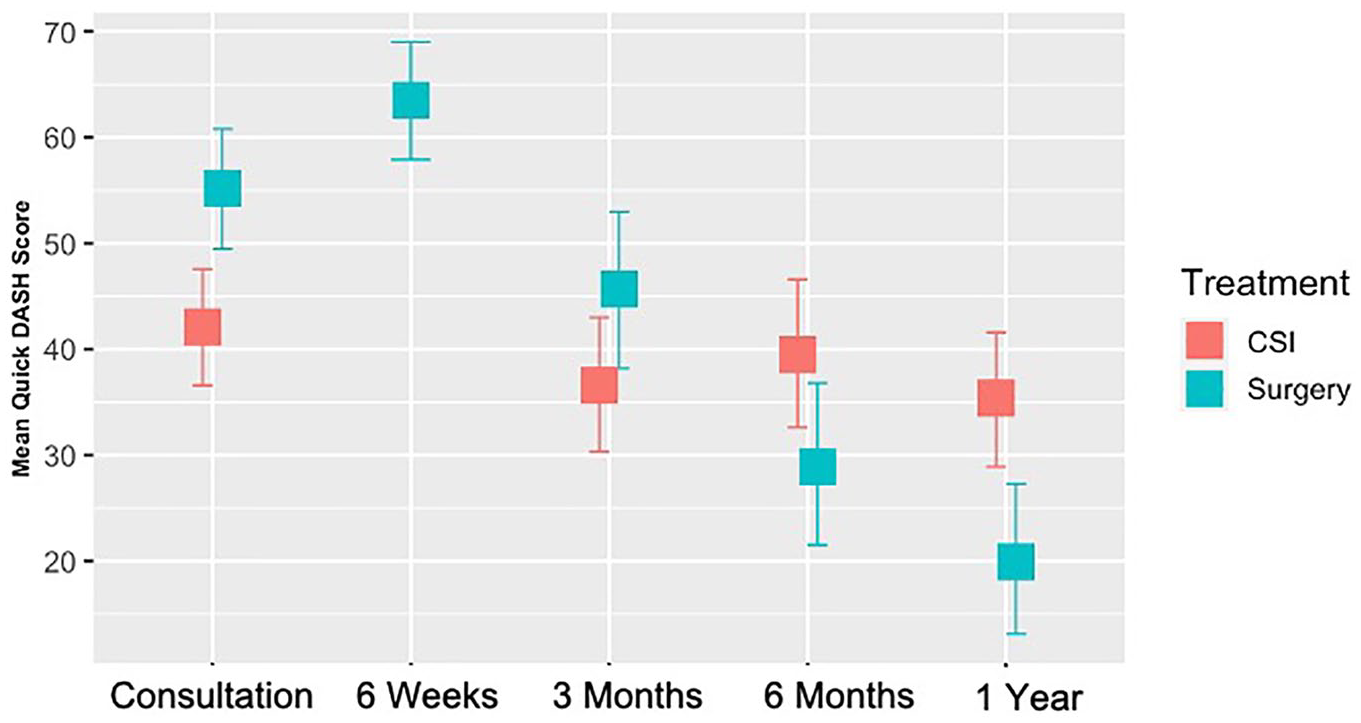
QuickDASH scores in the surgical group and CSI group over time.
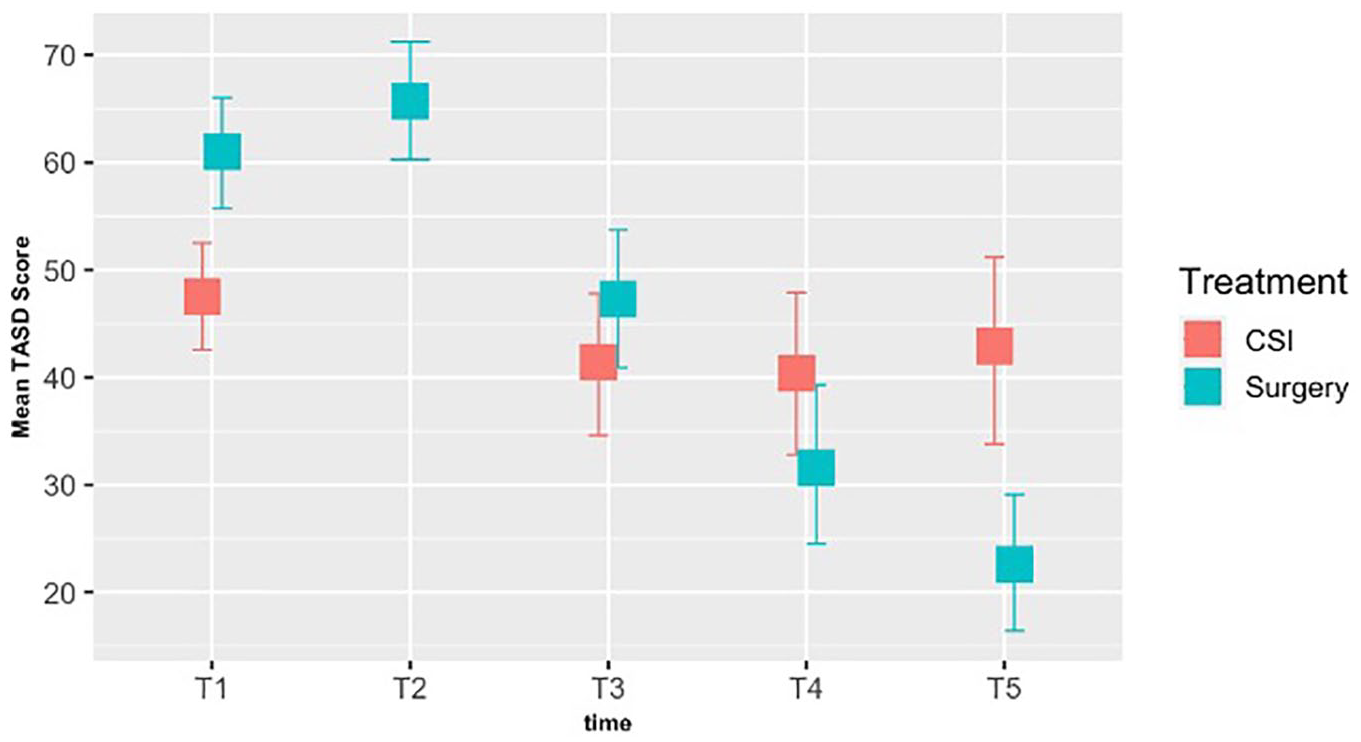
TASD scores in the surgical group and CSI group over time.
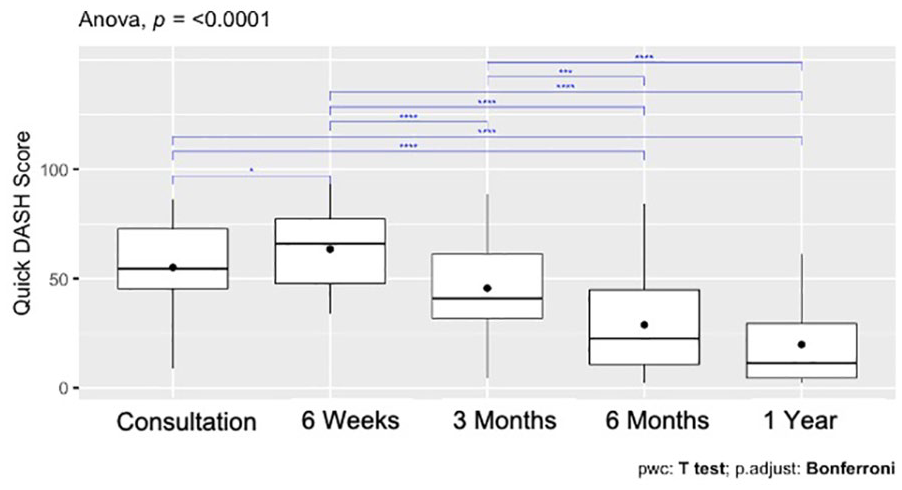
Post hoc analysis of QuickDASH scores in the surgical group.
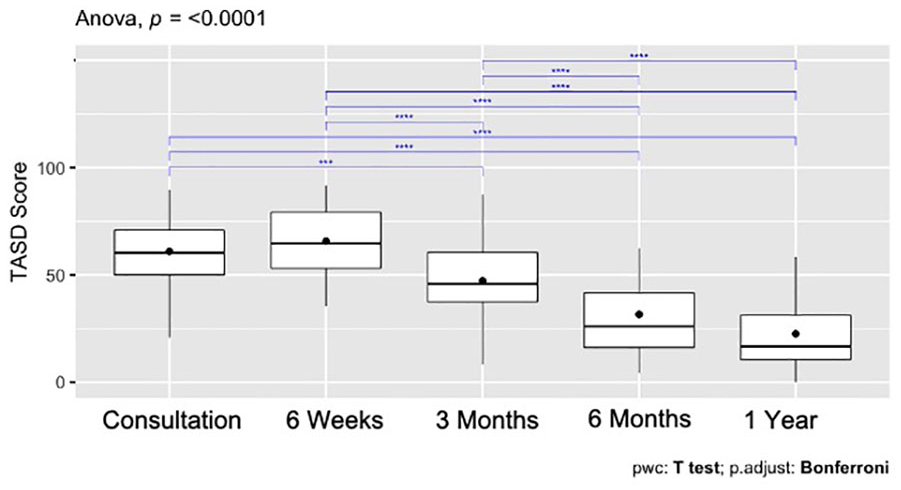
Post hoc analysis of TASD scores in the surgical group.
Discussion
This study is the first to our knowledge to explore the responsiveness of the TASD. The TASD is a thumb-specific patient-reported outcome, 13 and it was found to be able to be responsive following surgical intervention and is highly correlated to another commonly used upper extremity outcome measure. The TASD instrument demonstrated expected differences between the 2 cohorts in the study, with a higher SRM following surgery compared with corticosteroid injection.
Responsiveness has been described and interpreted heterogeneously in the literature. Beaton et al 16 introduced a specific taxonomy to clarify the concept of responsiveness. They present a description of several axes that we followed in our study. Our study analyzed change over time for patients with TMC-OA categorized by treatment method. Our study also examined changes “within-person” over time. Future “between-person” differences could be used to detect an estimation of the minimally clinically important difference in TASD scores. Finally, we determined the change before and after treatment (corticosteroid and surgical intervention). For future investigations, evaluating the measured change that is deemed to have an important improvement specifically for patients would have a great clinical relevance.
Although there remains a lack of standardization in the assessment of responsiveness to change, its consideration is necessary prior to introducing a functional scale to clinical practice. 17 Further evaluation of responsiveness is indeed essential to assess the discriminatory power of a scale. In other words, responsiveness allows researchers and clinicians to gauge the ability of a scale to detect a clinically important change. Due to the lack of universal methodological approach to assess responsiveness, we decided to proceed and use a correlation with a known indicator of change, in this case the QuickDASH. Further statistical analyses may be required in future studies with larger sample sizes and different population groups to analyze the diagnostic utility of the TASD. For example, future studies could determine the sensitivity and specificity of the scale through the use of receiver operating characteristic curves.
Trapeziometacarpal OA can be assessed with different outcome measures for disease severity. 8 Imaging does not constitute a good surrogate to determine the severity of symptoms experienced by patients. Patient-reported outcomes must be appropriate to the disease or patient population of interest. The findings of our study demonstrate that the TASD is responsive to treatment. Specifically, the TASD was responsive to treatment, and it discriminates between 2 treatment strategies. Although we did not see the same change in symptom burden among the nonsurgical group, this finding is not surprising. A single corticosteroid injection may lead to short-term relief of symptoms, and often patients return for clinical assessment when their symptoms return. As such, we would not expect the same consistent improvement in symptoms demonstrated in the surgical group. The different TASD trends between the 2 cohorts further support the demonstrated responsiveness of the TASD as it matches what is seen clinically. Misclassification error may be present when interpreting group-level data to individuals with unique characteristics and should be considered when using the TASD. As the study used routine clinical practice in follow-up visits, we did not assess patients in the corticosteroid injection cohort at 6 weeks. Nevertheless, this difference between the 2 cohorts did not change the interpretation of the results. Although not all the participants in the 2 cohorts completed the study, we were still able to demonstrate the responsiveness of the TASD.
Cross-validation of the psychometric properties of the TASD is needed with larger sample sizes and from different patient populations. Caution is required when interpreting such data. Group data, patient characteristics, and treatment characteristics are all elements that can make clinical translation more complex. Overall, we would recommend the use of the TASD in clinical and research setting for patients with TMC-OA as well as reporting of the collected data while using a comprehensive interpretation of its findings. The wide use of TASD can allow knowledge synthesis of TMC-OA treatment across different studies and future meta-analyses. The TASD is a promising tool for TMC-OA.
Conclusion
The TASD instrument demonstrated responsiveness following treatment, particularly following surgery in the TMC-OA cohort. Identifying the minimal clinically important difference is warranted for the interpretation of the results. The responsiveness of TASD supports its use as a candidate outcome measure for TMC-OA.
Footnotes
Author’s Note
Moaath M. Saggaf is also affiliated to Division of Plastic and Reconstructive Surgery, Department of Surgery, King Abdulaziz University, Jeddah, Saudi Arabia.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Statement of Informed Consent
Informed consent was obtained from all individual participants included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
