Abstract
Background:
Platelet-rich plasma (PRP) or corticosteroid injections may be used to conservatively treat mild-to-moderate carpal tunnel syndrome (CTS). We evaluated the cost-effectiveness of PRP injections versus corticosteroid injections for the treatment of mild-to-moderate CTS.
Methods:
Markov modeling was used to analyze the base-case 45-year-old patient with mild-to-moderate CTS, unresponsive to conservative treatments, never previously treated with an injection or surgery, treated with a single injection of PRP, or methylprednisolone/triamcinolone 40 mg/mL. Transition probabilities were derived from level-I/II studies, utility values from the Tufts University Cost-Effectiveness Analysis Registry reported using visual analog scale (VAS), Boston Carpal Tunnel Questionnaire Symptom severity (BCTQ-S), and Boston Carpal Tunnel Questionnaire Functional status (BCTQ-F), and costs from Medicare, published studies, and industry. Analyses were performed from healthcare/societal perspectives. Outcomes were incremental cost-effectiveness ratios (ICER) and net monetary benefits (NMB). Willingness-to-pay thresholds were $50 000 and $100 000. Deterministic/probabilistic sensitivity analyses were performed.
Results:
From a healthcare perspective, compared to PRP injections, the ICER for corticosteroid injections measured by VAS: -$13.52/quality-adjusted-life-years (QALY), BCTQ-S: -$11.88/QALY, and BCTQ-F: -$16.04/QALY. PRP versus corticosteroid injections provided a NMB measured by VAS: $428 941.12 versus $375 788.21, BCTQ-S: $417 115.09 versus $356 614.18, and BCTQ-F: $421 706.44 versus $376 908.45. From a societal perspective, compared to PRP injections, the ICER for corticosteroid injections measured by VAS: -$1024.40/QALY, BCTQ-S: -$899.95/QALY, and BCTQ-F: -$1215.51/QALY. PRP versus corticosteroid injections provided a NMB measured by VAS: $428 171.63 versus $373 944.39, BCTQ-S: $416 345.61 versus $354 770.36, and BCTQ-F: $420 936.95 versus $375 064.63.
Conclusions:
PRP injections were more cost-effective than methylprednisolone/triamcinolone injections from healthcare and societal perspectives for mild-to-moderate CTS.
Keywords
Introduction
Carpal tunnel syndrome (CTS) is the most common and widely known compression neuropathy. The median nerve becomes compressed within the carpal tunnel, a passageway comprised of ligamentous tissue and bones at the level of the wrist, resulting in decreased motor and sensory function. 1 The diagnosis can often be made by physical examination findings alone based on 6 of the most common findings associated with CTS. These include numbness in the median nerve distribution, nocturnal numbness, thenar atrophy and weakness, a positive Phalen test, loss of 2-point discrimination, and a positive Tinel sign. If physical examination findings remain unclear, nerve conduction studies may confirm or refute a diagnosis of CTS, assess disease severity, and provide baseline objective measurements for future comparisons. 2 In addition, many hand surgeons may require nerve conduction studies prior to patient consultations and insurance companies may require studies prior to surgical decompression. 2
Following a diagnosis of CTS, treatments often start with conservative measures that may include splinting, analgesic medications, and physical therapy. As symptoms progress, injections may provide alternative treatments prior to surgical interventions, with minimal risks of complications. Injections may include corticosteroids, strongly recommended by the American Academy of Orthopaedic Surgeons to improve patient outcomes or more recently gaining popularity, platelet-rich plasma (PRP). 3 Following unsuccessful injections, surgical decompression often provides symptomatic relief for patients. Surgical decompression of the median nerve may be performed using an endoscopic or open approach. With no notable differences between these approaches at 12 weeks, apart from earlier return to work with the endoscopic approach, either may be appropriate for the treatment of CTS. 4
As the most common compression neuropathy, costs associated with the treatment of CTS have increased from both healthcare and societal payer perspectives. 4 While these costs continue to accumulate over a patient’s lifetime, it is critical to evaluate the cost-effectiveness of newer conservative treatments. For these reasons, the objective was to evaluate the cost-effectiveness of PRP injections versus corticosteroid injections for the treatment of mild-to-moderate CTS. Our hypothesis is corticosteroid injections provide lower costs, while PRP injections provide greater effectiveness.
Materials and Methods
Markov Modeling Analytic Overview
Markov modeling was used to evaluate the cost-effectiveness of PRP injections versus corticosteroid injections for the treatment of mild-to-moderate CTS. Markov models are decision trees that simulate clinical pathways as transitions between discrete health states. 5 These transitions follow transition probabilities. Health states have designated quality-of-life (QOL) values (utilities) and costs. A QOL value was defined as an individual’s quantitation of disease burden on life and rated on a scale of 0 (death) to 1 (perfect health). As the model progresses through cycles, QOL values accumulate in each state, which aggregate into quality-adjusted life-years (QALYs). Costs included direct payments from healthcare perspectives and indirect payments (lost productivity from time off work, unpaid caregiving) added to direct payments from societal perspectives. 6
Model outcomes were incremental cost-effectiveness ratios (ICER), calculated by dividing cost differences by QALY differences for treatments [ICER = ΔCost/ΔQALY], and net monetary benefits (NMB), evaluated against predetermined willingness-to-pay (WTP) thresholds [NMB = WTPxQALY–Cost]. The WTP threshold was defined as the maximum cost society was willing to pay for an additional QALY, set at $50 000 and $100 000 United States Dollars (USD)/QALY. Thresholds were determined using the historical value of $50 000, and incorporated a range value $100 000, recommended by the Second Panel on Cost-Effectiveness in Health and Medicine. 7 If the ICER for the treatment falls below the WTP threshold, then it is cost-effective. The treatment with the greatest NMB value is the most cost-effective. If one treatment costs less and produces more QALYs compared to another, it is the dominant treatment.
Consolidated Health Economic Evaluation Reporting Standards reporting guidelines were followed. Model designs and analyses were performed in accordance with recommendations made by the Second Panel on Cost-Effectiveness in Health and Medicine using TreeAge Healthcare software (TreeAge Software, Inc., Williamstown, MA). 8 Clinical data were derived from randomized controlled trials (RCT) and prospective comparative studies identified through Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)-compliant systematic review strategies. Derivations of clinical parameters, transition probabilities, utilities, and costs were provided (Appendix, Supplemental Digital Content).
Model Design
Figure 1 illustrates the model design. Model base-case parameters were derived from 9 aggregated prospective level-I/II studies.9-17 The base-case assumes a 45-year-old patient, with unilateral mild-to-moderate CTS, unresponsive to conservative treatments (splint, medications, physical therapy), presenting to a hand surgeon’s office for evaluation, and treatment following a referral. Following reevaluation of CTS by physical examination (paresthesia, pain, swelling, or clumsiness of the hand aggravated by sleeping, maintained hand, arm posture, or repetitive hand action alleviated by changes in hand posture; sensory impairment in median nerve innervated territory of the hand; motor deficit or atrophy of median nerve innervated muscles; positive Phalen’s and/or Tinel’s sign) 1 and nerve conduction studies, a single injection of PRP or corticosteroid (methylprednisolone 40 mg /mL = triamcinolone 40 mg/mL) with 2% lidocaine was administered under ultrasound guidance. Following the injection, patients entered an optimal state or suboptimal state, characterized by the need for reintervention. Reinterventions were either a reinjection or surgery (endoscopic or open carpal tunnel release). Over subsequent 12-week cycles, we assumed patients in optimal states continued in optimal states, while patients in suboptimal states either continued in a suboptimal state or entered an optimal state. Once in an optimal state, a patient was considered cured for modeling purposes. Patients may die each year depending on life expectancy data derived from age-adjusted annualized mortality rates. All patients were followed starting at age 45 years over a lifetime, until resolution of symptoms, or until death.
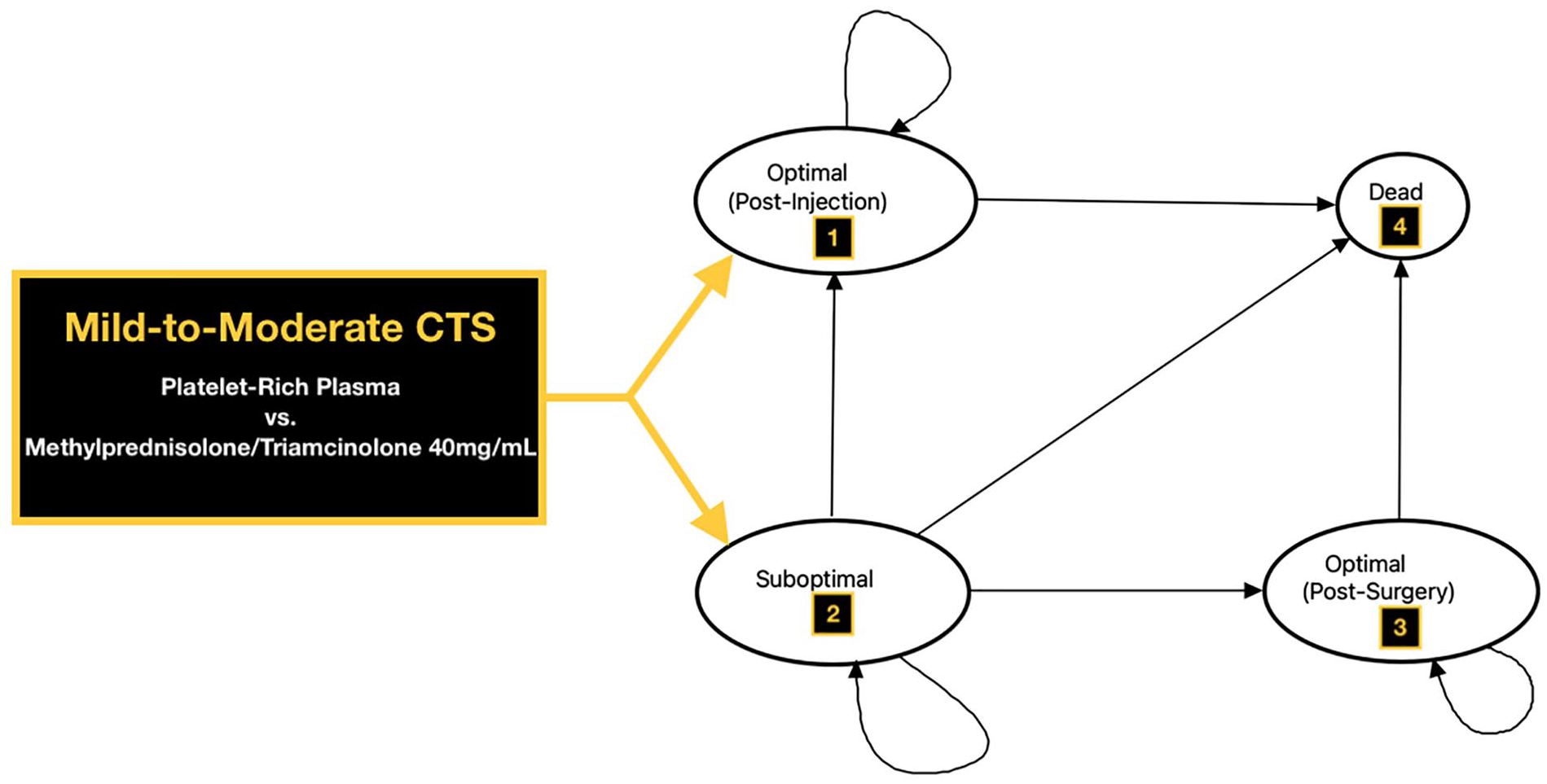
Markov model representation was used to evaluate the cost-effectiveness of platelet-rich plasma (PRP) injections and methylprednisolone/triamcinolone 40 mg/mL injections for mild-to-moderate carpal tunnel syndrome (CTS).
Transition Probabilities
The probability of entering a suboptimal state following the first injection was determined by paresthesia in the distribution of the median nerve at 12-weeks. Following a PRP injection, the probability of entering an optimal state was 89.7% and suboptimal state was 10.3%. 9 Following a corticosteroid injection, the probability of entering an optimal state was 70.3% and 29.7%. 9 We assumed patients with suboptimal states pursued reinterventions over subsequent 12-week cycles for symptomatic relief. Of those receiving reinterventions for persistent symptoms, we assumed 66.7% pursued surgery, while 33.3% pursued reinjections. We assumed probabilities of entering a suboptimal state were consistent over subsequent 12-week cycles (Table 1). Age-adjusted annualized mortality rates were derived from the Centers for Disease Control and Prevention (Appendix, Supplemental Digital Content, Table 1).
Model Input Parameters Stratified by Treatment for Carpal Tunnel Syndrome.
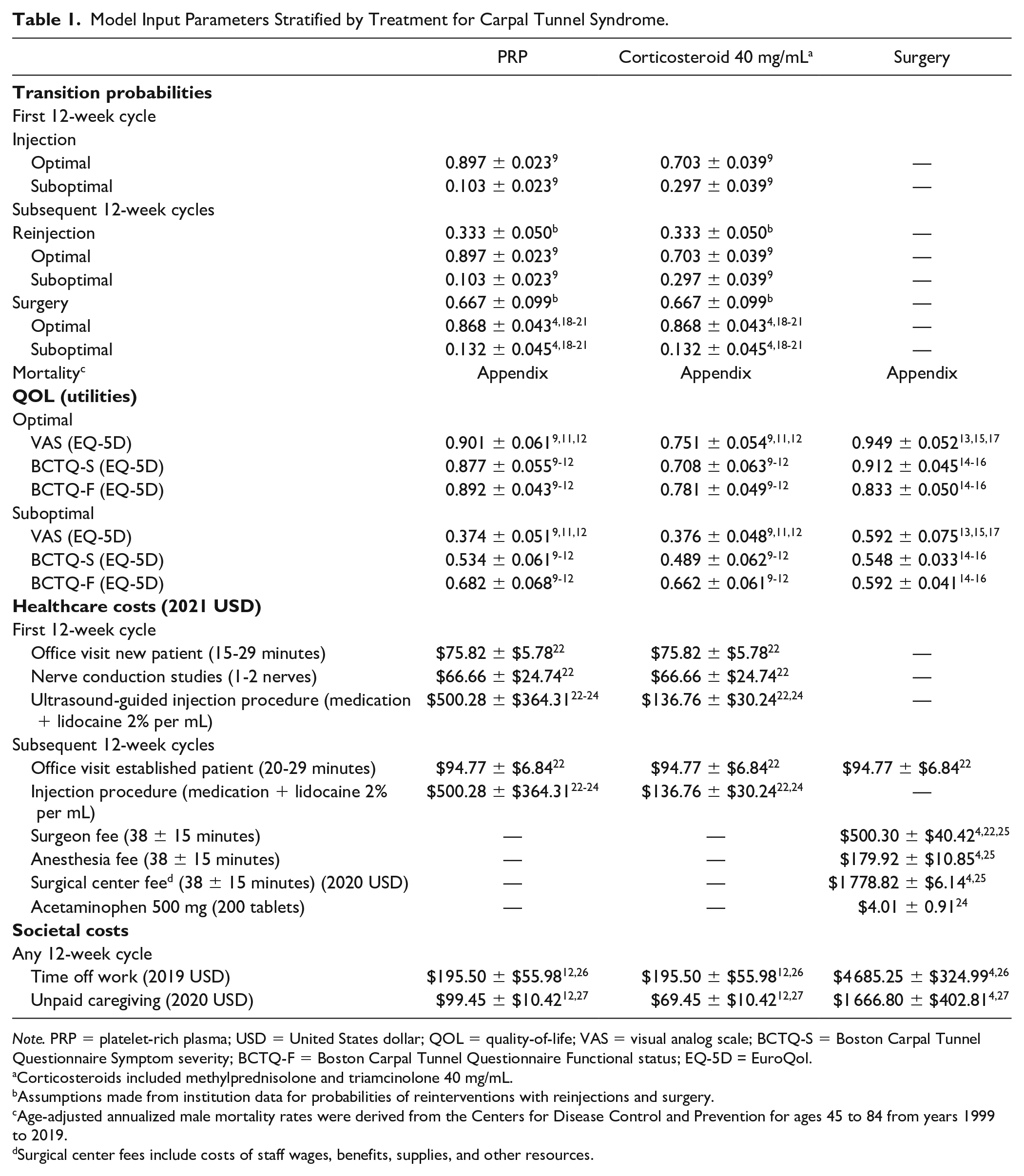
Corticosteroids included methylprednisolone and triamcinolone 40 mg/mL.
Assumptions made from institution data for probabilities of reinterventions with reinjections and surgery.
Age-adjusted annualized male mortality rates were derived from the Centers for Disease Control and Prevention for ages 45 to 84 from years 1999 to 2019.
Surgical center fees include costs of staff wages, benefits, supplies, and other resources.
QOL (Utilities)
Utility values were derived from studies extracted from the Tufts University Cost-Effectiveness Analysis (CEA) Registry. The model applied time-dependent, optimal and suboptimal utility differences following 12-week intervals for PRP injections, methylprednisolone/triamcinolone 40 mg/mL injections and surgery, calculated separately from visual analog scale (VAS), Boston Carpal Tunnel Questionnaire Symptom severity (BCTQ-S), and Boston Carpal Tunnel Questionnaire Functional status (BCTQ-F). Values at 12-weeks were converted from VAS, BCTQ-S, and BCTQ-F to equivalent EQ-5D (EuroQol) utility values using crosswalk strategies for comparisons (Table 1) (Appendix, Supplemental Digital Content, Table 2). 28 EQ-5D values were used in compliance with the National Institute for Health and Care Excellence (NICE) guidelines. 29 Derived VAS, BCTQ-S, and BCTQ-F values for endoscopic and open surgical approaches were averaged into single surgery values at 12-weeks derived from RCTs (Appendix, Supplemental Digital Content, Table 3). Utility values were extrapolated over the course of a patient’s lifetime or until resolution of symptoms.
Costs
Medicare reimbursement schedules were used to estimate unit costs of outpatient office visits, diagnostic studies, medications, anesthesia fees, surgeon fees, surgical center costs, postoperative care, and physical therapy/rehabilitation (Table 1) (Appendix, Supplemental Digital Content, Table 4). Costs of single injections of PRP were estimated at $500.00 from local industry representatives and billing offices (Arthrex ACP® Double Syringe System). 23 Costs of single injections of methylprednisolone 40 mg/mL were estimated at $5.80 and triamcinolone 40 mg/mL were estimated at $18.01 from 2021 Medicare Part B Average Sales Prices. 24 Costs of suboptimal surgeries resulting in additional surgeries were assumed to be equivalent to primary surgeries. Costs of adverse effects from injections were considered negligible, as these resolved without further interventions. Direct costs were expressed in 2021 USD, apart from surgical center costs adjusted to 2020 USD.4,25
From a societal perspective, time-dependent costs associated with lost productivity and unpaid caregivers were extrapolated from cost-effectiveness studies. Productivity costs of missed work were calculated from age-adjusted average 2019 salary incomes for both sexes, multiplied by average days out of work. 26 Unpaid caregiver time-costs were calculated from average 2020 daily salaries of home health aides multiplied by the average days out of work. 27
Discounting
All future costs and utility values were discounted at 3% to account for inflation, opportunity costs, and time preferences. 30
Sensitivity Analysis
One-way and 2-way deterministic sensitivity analyses were performed on all transition probabilities and utilities varying each value from 0 to 1 to determine sensitive parameters. All costs were varied from the base case by published ranges. Costs of PRP injections were varied across ranges from $0 to $2 000. Probabilistic sensitivity analyses were performed using second-order Monte Carlo simulations to simultaneously evaluate parameters across distributions evaluated in 1-way sensitivity analyses. Non-parametric distributions were calculated using means and standard deviations (SD) derived from level-I/II studies.9-17 Beta distributions were used for transition probabilities and utilities, while gamma distributions were used for costs. A total of 10 000 iterations were performed against WTP thresholds $50 000, $100 000, and $200 000. Cost-effectiveness scatterplots were generated to incorporate levels of uncertainty around distributions. Cost-effectiveness acceptability curves were generated to determine the percentage of iterations in which treatments provided the greatest cost-effectiveness.
Results
Base-case
Patient characteristics derived for the healthcare and societal perspective base-cases were provided in Table 2.9-17 No observable differences existed between PRP and methylprednisolone/triamcinolone 40 mg/mL injections for mild-to-moderate CTS. Patients receiving corticosteroid injections required more reinterventions compared to PRP injections. Corticosteroid injections required and average of 1.12 injections and 0.24 surgeries per patient, while PRP injections required an average of 1.04 injections and 0.08 surgeries per patient over a patient’s lifetime. From both healthcare and societal perspectives, PRP injections accumulated more QALYs at lower costs, dominating corticosteroid injections from the outset (negative ICER). PRP injections provided greater NMBs compared to corticosteroid injections for VAS, BCTQ-S, and BCTQ-F.
Characteristics of Patients from Included Studies Stratified by Treatment for Carpal Tunnel Syndrome.
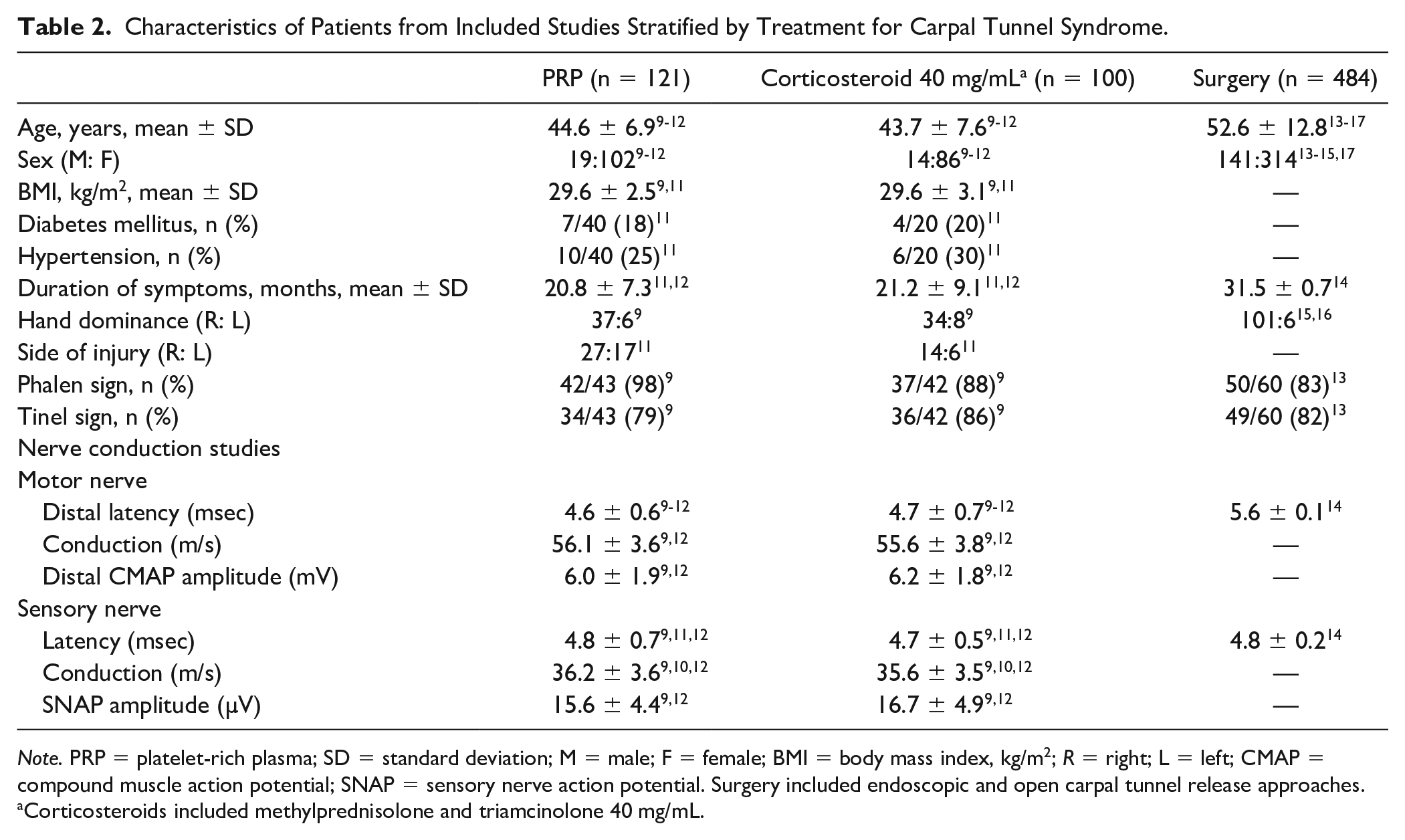
Corticosteroids included methylprednisolone and triamcinolone 40 mg/mL.
Cost-Effectiveness Rankings Over a Patient’s Lifetime at WTP Threshold $50,000 Stratified by PRP and Methylprednisolone/Triamcinolone 40 mg/mL Injections for the Treatment of Mild-to-Moderate Carpal Tunnel Syndrome.
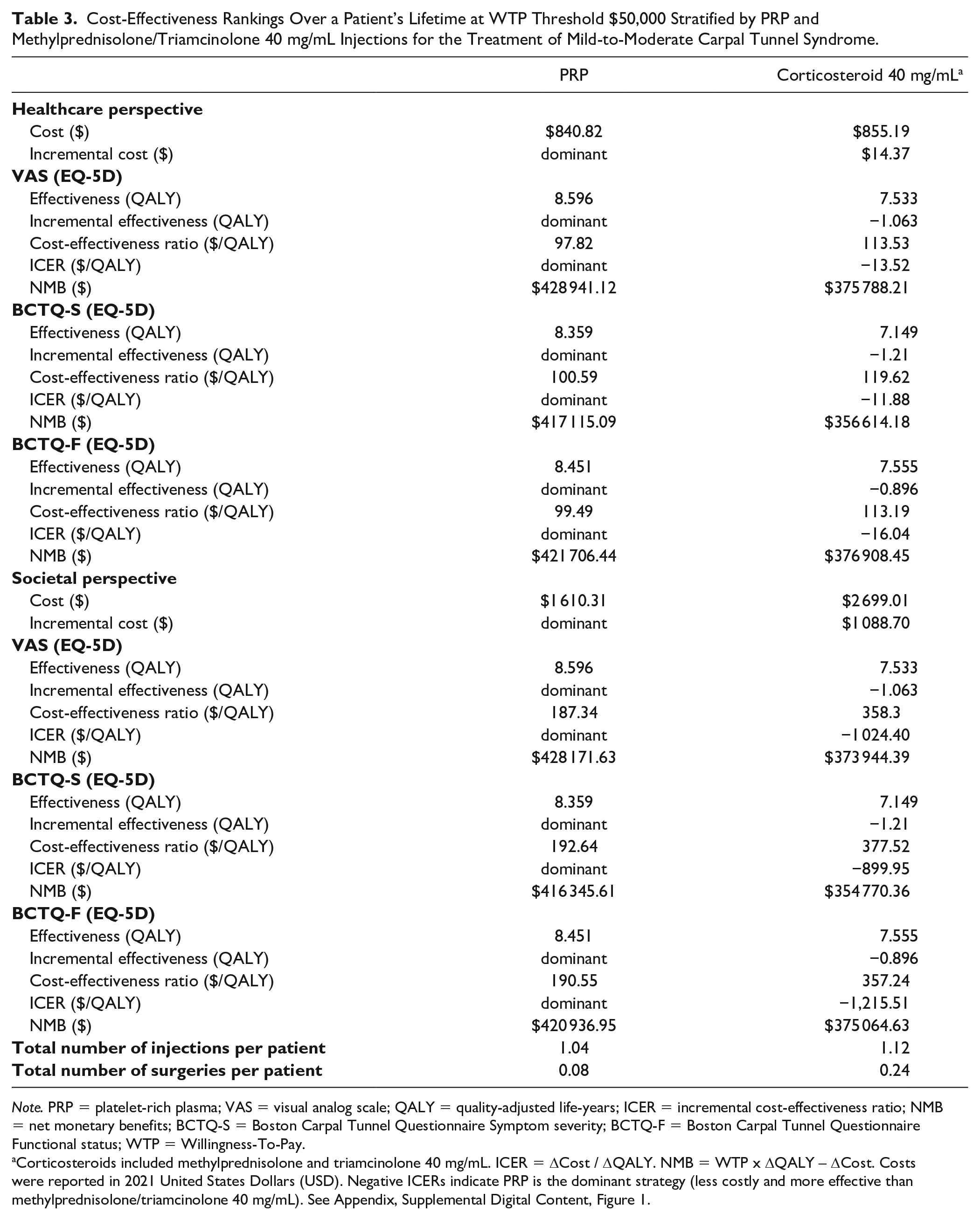
Corticosteroids included methylprednisolone and triamcinolone 40 mg/mL. ICER = ΔCost / ΔQALY. NMB = WTP x ΔQALY – ΔCost. Costs were reported in 2021 United States Dollars (USD). Negative ICERs indicate PRP is the dominant strategy (less costly and more effective than methylprednisolone/triamcinolone 40 mg/mL). See Appendix, Supplemental Digital Content, Figure 1.
From a healthcare perspective, compared to PRP injections, the ICER for corticosteroid injections was -$13.52/QALY measured by VAS, -$11.88/QALY measured by BCTQ-S, and -$16.04/QALY measured by BCTQ-F at a WTP threshold $50 000. PRP injections provided a NMB of $428 941.12 measured by VAS, $417 115.09 measured by BCTQ-S, and $421 706.44 measured by BCTQ-F, compared to a NMB of $375 788.21 measured by VAS, $356 614.18 measured by BCTQ-S, and $376 908.45 measured by BCTQ-F. Differences between NMBs for PRP and corticosteroids were greatest for BCTQ-S utility values ($60 500.91) and least for BCTQ-F utility values ($44 797.99).
Total healthcare perspective direct costs of PRP injections were $840.82, while costs of corticosteroid injections were $855.19. Total societal perspective indirect costs of PRP injections were $769.49, while costs of corticosteroid injections were $1 843.82 (Figure 2). Higher costs of corticosteroid injections were attributed to lost productivity from time off work and unpaid caregiving as a result of suboptimal responses resulting in a greater number of surgeries per patient compared to PRP injections.
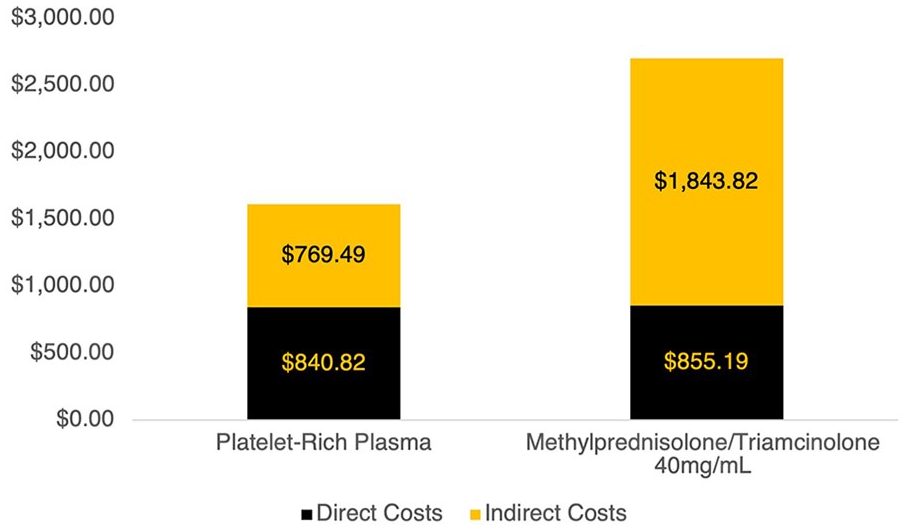
Total direct costs and indirect costs associated with platelet-rich plasma injections and methylprednisolone/triamcinolone 40 mg/mL injections over the patient’s lifetime, until resolution of symptoms, or until death.
From a societal perspective, compared to PRP injections, the ICER for corticosteroid injections was -$1 024.40/QALY measured by VAS, -$899.95/QALY measured by BCTQ-S, and -$1 215.51/QALY measured by BCTQ-F at a WTP threshold $50 000. PRP injections provided a greater NMB compared to corticosteroid injections for VAS, BCTQ-S, and BCTQ-F. PRP injections provided a NMB of $428 171.63 measured by VAS, $416 345.61 measured by BCTQ-S, and $420 936.95 measured by BCTQ-F, compared to a NMB of $373 944.39 measured by VAS, $354 770.36 measured by BCTQ-S, and $375 064.63 measured by BCTQ-F. Differences between NMBs for PRP and corticosteroids were greatest for BCTQ-S utility values ($61 575.25) and least for BCTQ-F utility values ($45 872.32).
Deterministic Sensitivity Analysis
One-way deterministic analysis did not result in major model changes. Overall, PRP injections remained the dominant strategy despite variations across tested model parameters. PRP injections remained more cost-effective compared to corticosteroid injections for patients with mild-to-moderate CTS from healthcare and societal perspectives with VAS, BCTQ-S, and BCTQ-F utility values.
Probabilistic Sensitivity Analysis
Figure 3 illustrates cost-effectiveness scatterplot results of 10 000 iterations of the analysis when considering uncertainty for all probabilities, utilities, and costs of PRP injections, corticosteroid injections, and surgery, for patients with CTS from healthcare and societal perspectives. Figure 4 illustrates acceptability curves displaying how many of the 10 000 iterations of PRP injections and corticosteroid injections provided cost-effectiveness across WTP thresholds from healthcare and societal perspectives (Appendix, Supplemental Digital Content, Figure 1). From both perspectives, PRP injections were accepted as optimal treatment ranging from 92.8% to 95.5% across most WTP thresholds with VAS, BCTQ-S, and BCTQ-F utility values.
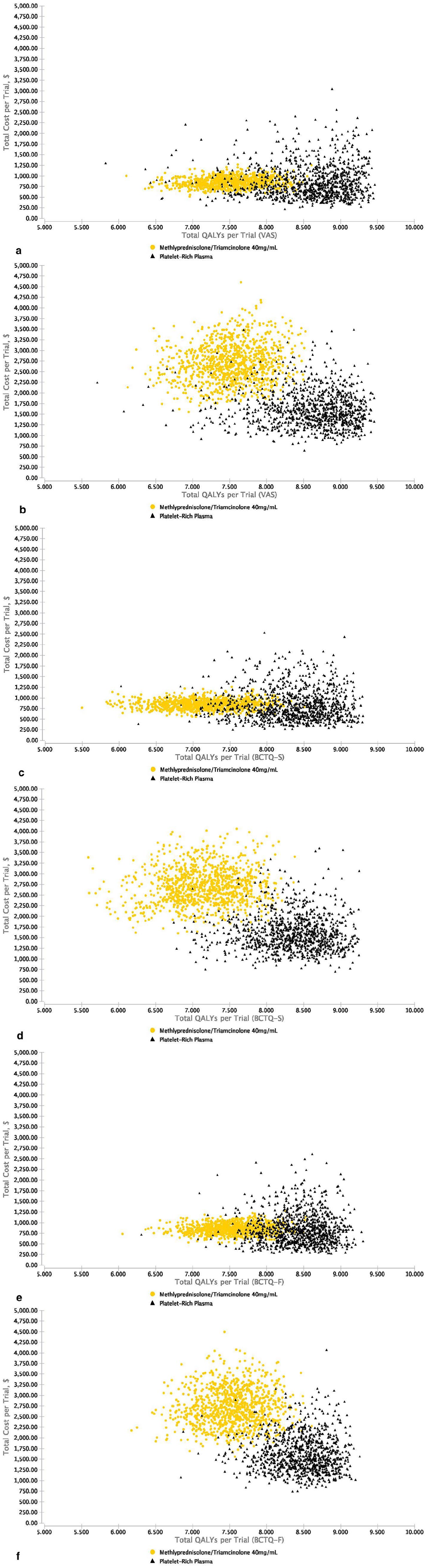
Probabilistic sensitivity analyses scatterplots illustrating cost-effectiveness results of 10 000 iterations for platelet-rich plasma (PRP) injections and methylprednisolone/triamcinolone 40 mg/mL injections for patients with mild-to-moderate carpal tunnel syndrome (CTS).
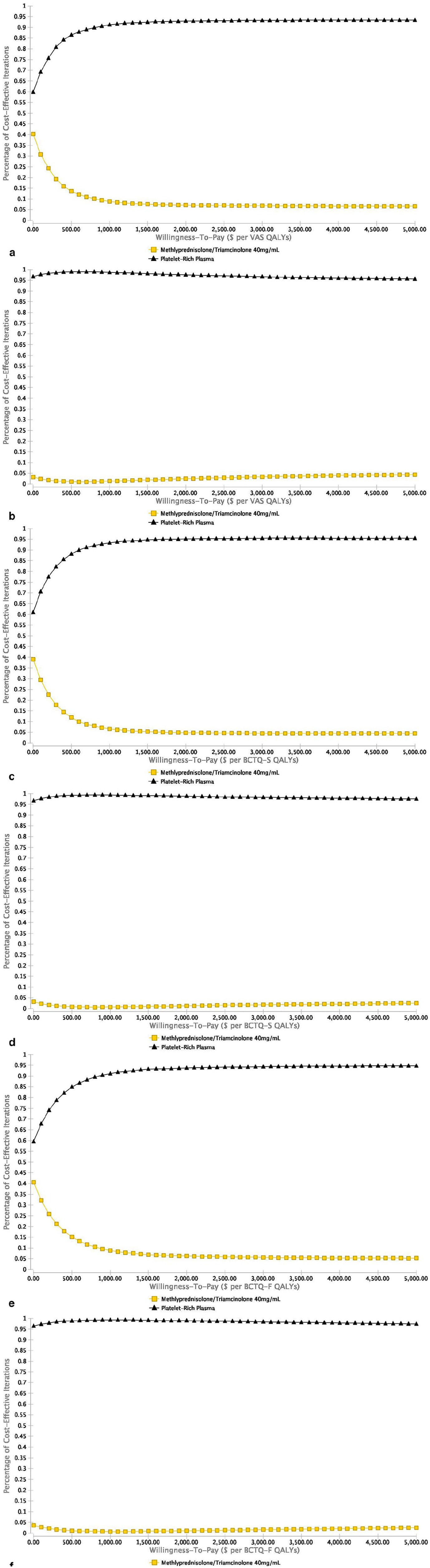
Cost-effectiveness acceptability curves illustrating the percentage of the 10 000 probabilistic sensitivity analyses iterations that demonstrated cost-effectiveness across various willingness-to-pay (WTP) thresholds for platelet-rich plasma (PRP) injections and methylprednisolone/triamcinolone 40 mg/mL injections for patients with mild-to-moderate carpal tunnel syndrome (CTS).
Discussion
The Markov model and Monte Carlo simulation analysis over a patient’s lifetime demonstrated PRP injections were more cost-effective treatments compared to methylprednisolone/triamcinolone 40 mg/mL injections from healthcare and societal perspectives, for mild-to-moderate CTS at a WTP threshold of $50 000. Although we hypothesized PRP injections provided greater effectiveness, we were surprised to find they also provided lower overall costs, dominating corticosteroid injections. Base-case patient scenarios were presented from data extracted from Level-I/II studies, that included a population where PRP or corticosteroid injections may be appropriate, necessitating the need for cost-effectiveness. Utility values crosswalked from VAS, BCTQ-S, and BCTQ-F to EQ-5D values demonstrated similar trends from baseline to 12-weeks following treatment. Assessing the cost-effectiveness of each intervention independently using VAS, BCTQ-S, and BCTQ-F utility values allowed for additional comparisons between healthcare and societal perspectives. The BCTQ-S demonstrated the greatest fluctuations in utility values, followed by VAS, and BCTQ-F. Deterministic and probabilistic sensitivity analyses demonstrated the robustness and generalizability of the model following variations in input parameters.
Dose variations, splinting, and the impact of injections at the time of surgery were unable to be compared between PRP and corticosteroids for CTS. Studies comparing splinting alone to corticosteroid injections alone were performed using 20 mg/mL or 48.3 mg/mL equivalents of methylprednisolone or triamcinolone.31,32 Studies comparing splinting alone to splinting and corticosteroid injections were performed using 16 mg/mL equivalents of methylprednisolone or triamcinolone.33,34 We were unable to find any studies comparing PRP injections alone to surgery alone. Studies comparing surgery alone to surgery and perioperative injections did not have similar follow-up durations. PRP injections were compared at 6-weeks, while corticosteroids were compared at 1, 4, 8, and 12-weeks.35,36 Similarly, no level-I/II studies have been performed directly comparing perioperative PRP to perioperative corticosteroids for the treatment of CTS. For additional model comparisons, studies need to compare 40 mg/mL equivalent corticosteroid doses, similar follow-up durations, and similar outcome measures.
The literature defines PRP as a sample of autologous blood with platelet concentrations above normal physiologic levels, produced by centrifugation of whole blood. 37 Its efficacy relates directly to regenerative properties promoted by locally released growth factors, estimated from over 30 biologically active proteins. 38 Reported costs of PRP range anywhere from $4.88 to $2 301.39,40 Low costs of $4.88 reflect prices of materials required to extract and administer PRP in a setting with an available centrifuge. In the United States, PRP is billed using the temporary CPT code 0232 T. This CPT code is a level III categorization with Status Code C that bundles the harvesting, preparation, and image guidance for service.
Limitations were related to inconsistency of PRP formulations and required assumptions for decision analysis methodologies. Even among the same PRP preparation kits, the product is different. Depending on the physiologic concentrations of cells or fragments within the blood, different concentrations may be observed at different time periods within the same patient. This limitation is characteristic of all PRP studies. We attempted to evaluate Disabilities of the Arm, Shoulder and Hand (DASH) scores and grip strength scores (GSS); however, scores were unavailable for prospective studies comparing PRP injections to methylprednisolone/triamcinolone 40 mg/mL injections. This prevented crosswalked estimations at 12-weeks for utility values proportional to changes in DASH scores and GSS. Follow-up evaluations at 12-weeks were the longest available. Longer, prospective, comparative assessments are required to determine the long-term benefits of each intervention. We assumed events were mutually exclusive, characteristic of decision analysis methodologies. Once a patient entered an optimal state, we assumed they remained in that state over the course of their lifetime. In reality, patients may relapse following injections or surgery. Assumptions were made using the longest available level-I/II comparative data from the literature to maintain a structured model. We assumed two-thirds of patients with suboptimal states following injections would receive median nerve decompression surgery, while the remaining third would receive a reinjection. This assumption was determined by authors as reasonable to standardize the model. Additional publications comparing PRP injections to corticosteroid injections with longer follow-up durations are required to properly assess long-term outcomes for recommendations. Following deterministic and probabilistic sensitivity analyses over the course of a patient’s lifetime, model assumptions were reasonable. Costs began at the first evaluation by a hand surgeon, directly attributed to PRP injections or corticosteroid injections. This limited including past costs of conservative treatments, previous laboratory and diagnostic tests, and lost productivity. Societal costs do not entirely consider all costs to the patient. It remains difficult to estimate costs specific to individual patients in cost-effectiveness studies. In contrast to corticosteroid injections, payers rarely cover costs of PRP injections. As more evidence becomes available, PRP injections may become covered by more payers.
Model findings should not be applied to all patients without proper clinical context and judgment. Variations in base-case parameters may potentially impact outcomes (demographics, comorbidities, occupations, underlying etiologies, previous interventions, medications, and outcome measures). Nevertheless, our base-case demonstrated a single PRP injection was more cost-effective than a single methylprednisolone/triamcinolone 40 mg/mL injection from healthcare and societal perspectives for the treatment of mild-to-moderate CTS.
Supplemental Material
sj-docx-1-han-10.1177_15589447221092056 – Supplemental material for Platelet-Rich Plasma Versus Corticosteroid Injections for the Treatment of Mild-to-Moderate Carpal Tunnel Syndrome: A Markov Cost-Effectiveness Decision Analysis
Supplemental material, sj-docx-1-han-10.1177_15589447221092056 for Platelet-Rich Plasma Versus Corticosteroid Injections for the Treatment of Mild-to-Moderate Carpal Tunnel Syndrome: A Markov Cost-Effectiveness Decision Analysis by Kevin M. Klifto, Christopher S. Klifto, Tyler S. Pidgeon, Marc J. Richard, David S. Ruch and Stephen H. Colbert in HAND
Footnotes
Acknowledgements
We thank Andrew Munzer, Director of Product Management & Training at TreeAge Software for assistance and confirmation of accuracy of our model design.
Supplemental material is available in the online version of the article.
Ethical Approval
All data from studies included within this larger study were approved by that respective institution’s institutional review board.
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from all patients for being included in the study.
Statement of Informed Consent
Informed consent was obtained from all patients for being included in the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors K. M. K., S. H. C., T. S. P., and C. S. K. declare that they have no conflict of interest. M. J. R. receives royalties or licenses from Acumed, Bioventus, DJO, Depuy Synthes, Medartis within the past 36 months. D. S. R. receives royalties or licenses from Acumed within the past 36 months.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
