Abstract
Introduction
Dupuytren disease (DD) is a fibroproliferative disorder characterized by deposition of collagen in the fascia of the palm and fingers and is estimated to affect approximately 1% of adults in the United States.1,2 The earliest signs of DD include palmar nodules that may, over the course of several years, transform into fibrous cords extending into the fingers (most commonly the ring and little fingers). This can ultimately lead to permanent contracture of the metacarpophalangeal and proximal interphalangeal joints, a condition known as Dupuytren contracture (DC).1,3 Dupuytren contracture can severely impair patients’ ability to perform daily activities, such as dressing or washing, as well as work or leisure activities, resulting in reduced quality of life.2,4,5 The condition is also associated with substantial indirect costs related to high rates of disability and absences from work: in one US study, mean annual indirect costs (disability days or medically related absenteeism) were ~2 times higher for individuals with DC compared with individuals without DC. 6
Treatment options for DC include both surgical procedures, such as fasciectomy or fasciotomy, and nonsurgical procedures, such as percutaneous needle aponeurotomy or collagenase clostridium histolyticum (CCH) injection. 1 Collagenase clostridium histolyticum was approved in the United States in 2010 for the treatment of adults with DC with a palpable cord. 7 Availability of this nonsurgical option has changed the treatment landscape for DC. In 2014, a Medicare database analysis indicated that 11% of patients with DC received treatment with CCH, whereas 21% underwent fasciectomy; in contrast, 33% of patients with DC underwent fasciectomy in 2007, prior to the introduction of CCH (P ≤ .0001). 1 Several studies conducted in the United States 8 and Europe9-12 have reported that, compared with fasciectomy, CCH treatment for DC is associated with lower health care costs and a reduction in the use of health care resources. These studies are supported by a 2019 retrospective analysis of a US adjudicated claims database, using data from 2011 to 2016, which demonstrated that CCH treatment of DC was associated with an 11.5% reduction in total all-cause episode-of-care costs compared with fasciectomy (P < .001). 13 The higher costs associated with fasciectomy were primarily driven by outpatient surgery costs and higher costs for physician office visits and laboratory/pathology tests.
However, currently there are limited real-world data comparing return-to-function after nonsurgical versus surgical therapies for DC. Studies of surgical interventions for carpal tunnel syndrome,14,15 anterior cruciate ligament reconstruction, 16 and total knee or hip arthroplasty17-20 have used physical therapy (PT) and use of pain medication as surrogate prognostic indicators for return-to-work or return-to-function following surgery. Hence, this study was conducted to assess return-to-function with surrogate markers (analgesic use and PT or occupational therapy [OT] visits) in patients with DC treated with CCH versus fasciectomy.
Materials and Methods
The study was a retrospective cohort analysis using the IQVIA Real-World Data Adjudicated Claims—US Database. This database contains fully adjudicated, longitudinal, medical, and pharmaceutical claims data for >150 million patients, and the individuals included are representative of the US national, commercially insured population in terms of age and sex. Data captured include demographics (eg, age, sex, geographic region), diagnoses and comorbidities, prescriptions, and use of medical services (eg, hospitalizations and emergency department visits, physician office visits, procedures, and injections). All data were deidentified and compliant with the 1996 Health Insurance Portability and Accountability Act.
The inclusion and exclusion criteria for the study have been described previously. 13 Patients were included if they were ≥18 years of age and had ≥1 Current Procedural Terminology (CPT) code claim for CCH (CPT code 20527) or fasciectomy (CPT code 26123) between January 1, 2012, and June 30, 2016. The first date from which a claim was documented for either treatment during this time frame (ie, the date of the first treatment) was designated as the index date. Patients were required to have ≥1 medical claim with a DC code (International Classification of Diseases, Ninth Revision, Clinical Modification 728.6 or International Classification of Diseases, Tenth Revision, Clinical Modification M72.0) during the 12-month preindex period and to have continuous health plan enrollment for ≥360 days preindex and ≥90 days postindex (ie, 90-day follow-up period).
Patients were required to be newly treated for DC on the index date and were excluded if they had any claim for DC treatment with CCH, fasciectomy, percutaneous needle aponeurotomy, or open fasciotomy during the 12-month preindex period; any claim for another DC treatment on the index date; or evidence from CPT codes of >1 finger being treated on the index date. Patients were also excluded if they had missing or invalid data, if they had health insurance coverage through Medicare Cost Plans or the State Children’s Health Insurance Program, or if they were aged ≥65 years at the index date and were not covered by a commercial Medicare plan. 13 Ethics committee approval was not required for the study because the data were anonymized and were from a secondary source.
Data on analgesic prescriptions (ie, opioid analgesics and nonsteroidal anti-inflammatory drugs [NSAIDs]) and PT and OT visits during the 90-day follow-up period were collected as surrogate markers for return-to-function after DC treatment. Analgesic utilization was assessed overall and for new initiators (those who had not used opioids or NSAIDs in the 12-month preindex period). Dupuytren contracture–specific PT and OT visits were assessed based on an associated diagnosis code for DC on the claim during the 90-day follow-up period, by weekly interval. Comparisons between cohorts were performed using parametric t tests (for means) or nonparametric Wilcoxon rank sum tests (for medians) for continuous variables and χ2 tests for categorical variables. All analyses were performed on observed data, without imputation of missing values. Analyses were performed using SAS version 9.2 software (SAS Institute Inc., Cary, North Carolina), and P < .05 was considered significant.
Results
A total of 15 491 patients filed ≥1 claim for CCH or fasciectomy (CCH, n = 5558; fasciectomy, n = 9933) during the assessment period. Of these, 4399 patients (CCH, n = 1654; fasciectomy, n = 2745) met all analysis criteria and were included in the study (Table 1). 13 In general, the demographics and baseline characteristics between the CCH and fasciectomy cohorts were similar, although there were significant differences observed, such as sex, geographic location, and payer type. In addition, the percentage of patients receiving opioid analgesics during the 12 months prior to the index date was significantly lower in the CCH cohort compared with the fasciectomy cohort (28.9% vs 38.5%; P < .001), but there was no significant difference between the 2 groups in the percentage of patients using NSAIDs. 13
Baseline Demographic and Clinical Characteristics. 13
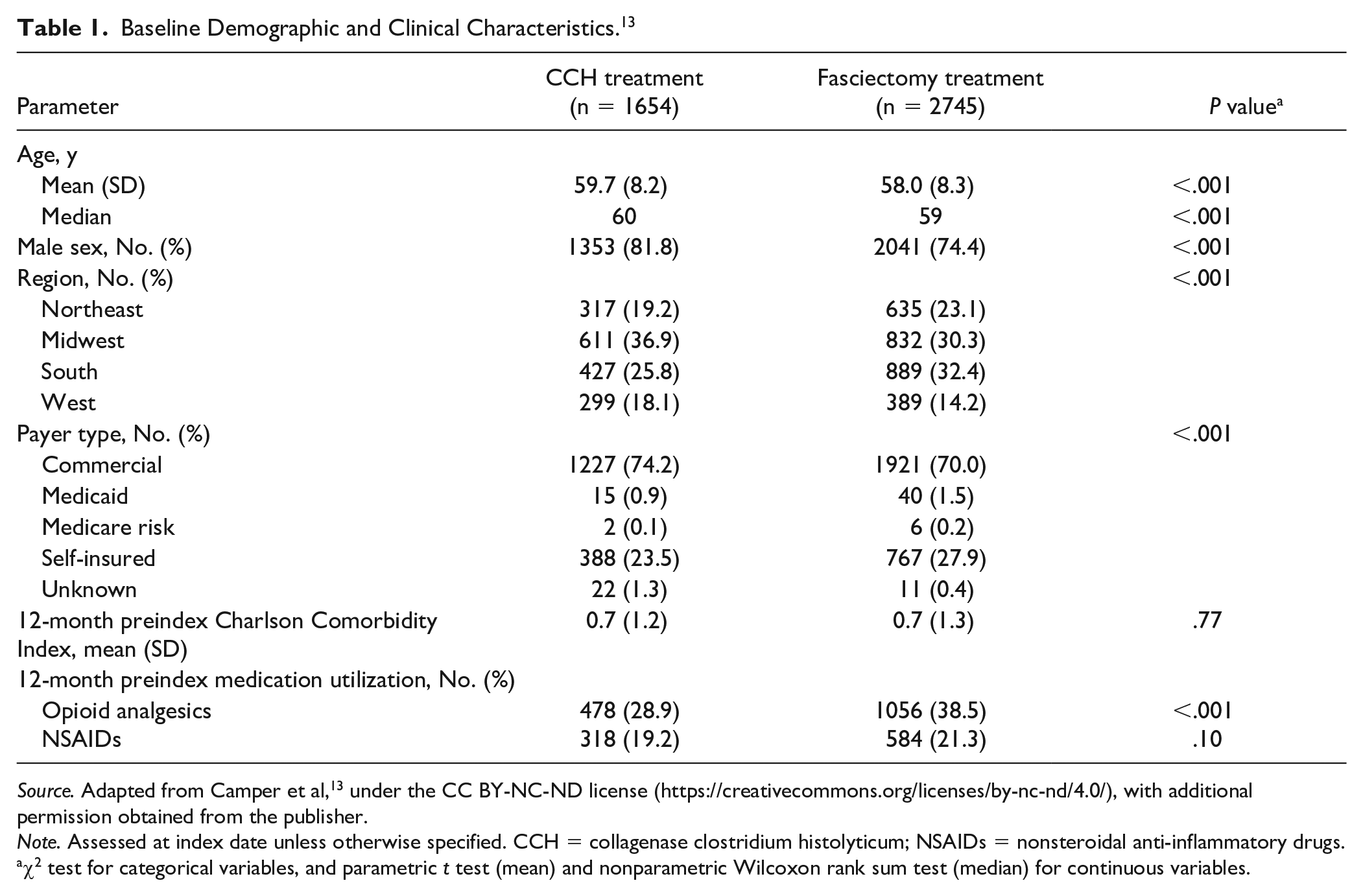
Source. Adapted from Camper et al, 13 under the CC BY-NC-ND license (https://creativecommons.org/licenses/by-nc-nd/4.0/), with additional permission obtained from the publisher.
Note. Assessed at index date unless otherwise specified. CCH = collagenase clostridium histolyticum; NSAIDs = nonsteroidal anti-inflammatory drugs.
χ2 test for categorical variables, and parametric t test (mean) and nonparametric Wilcoxon rank sum test (median) for continuous variables.
Fewer patients in the CCH cohort used opioid analgesics or NSAIDs during follow-up (P < .0001 for both), and patients in the CCH cohort were significantly less likely to be new users of these medications (P < .0001 for both; Figure 1). In addition, during follow-up, 806 (48.7%) of the 1654 patients in the CCH cohort had ≥1 all-cause PT visit compared with 1717 (62.6%) of the 2745 patients in the fasciectomy cohort (P < .0001). Among patients with ≥1 all-cause PT visit, the mean (SD) number of PT visits per patient was 3.3 (3.8) in the CCH cohort compared with 7.0 (5.9) in the fasciectomy cohort (P < .0001). The percentage of patients with ≥1 all-cause OT visit was also significantly lower in the CCH cohort than in the fasciectomy cohort (38.5% vs 51.7%; P < .0001). Among patients with ≥1 all-cause OT visit, the mean (SD) number of OT visits per patient was significantly lower in the CCH cohort than in the fasciectomy cohort: 1.4 (1.3) vs 2.2 (3.1); P < .0001. Assessment of all-cause PT or OT visits by weekly interval during follow-up (Figure 2a and 2b) showed that the percentage of patients with ≥1 visit during the first week of the postindex period was significantly higher in the CCH cohort than in the fasciectomy cohort. After the first week postindex, the percentage of patients with an all-cause PT or OT visit during each week in the CCH cohort was significantly lower than in the fasciectomy cohort, except for OT visits during the last week of follow-up (week 13).
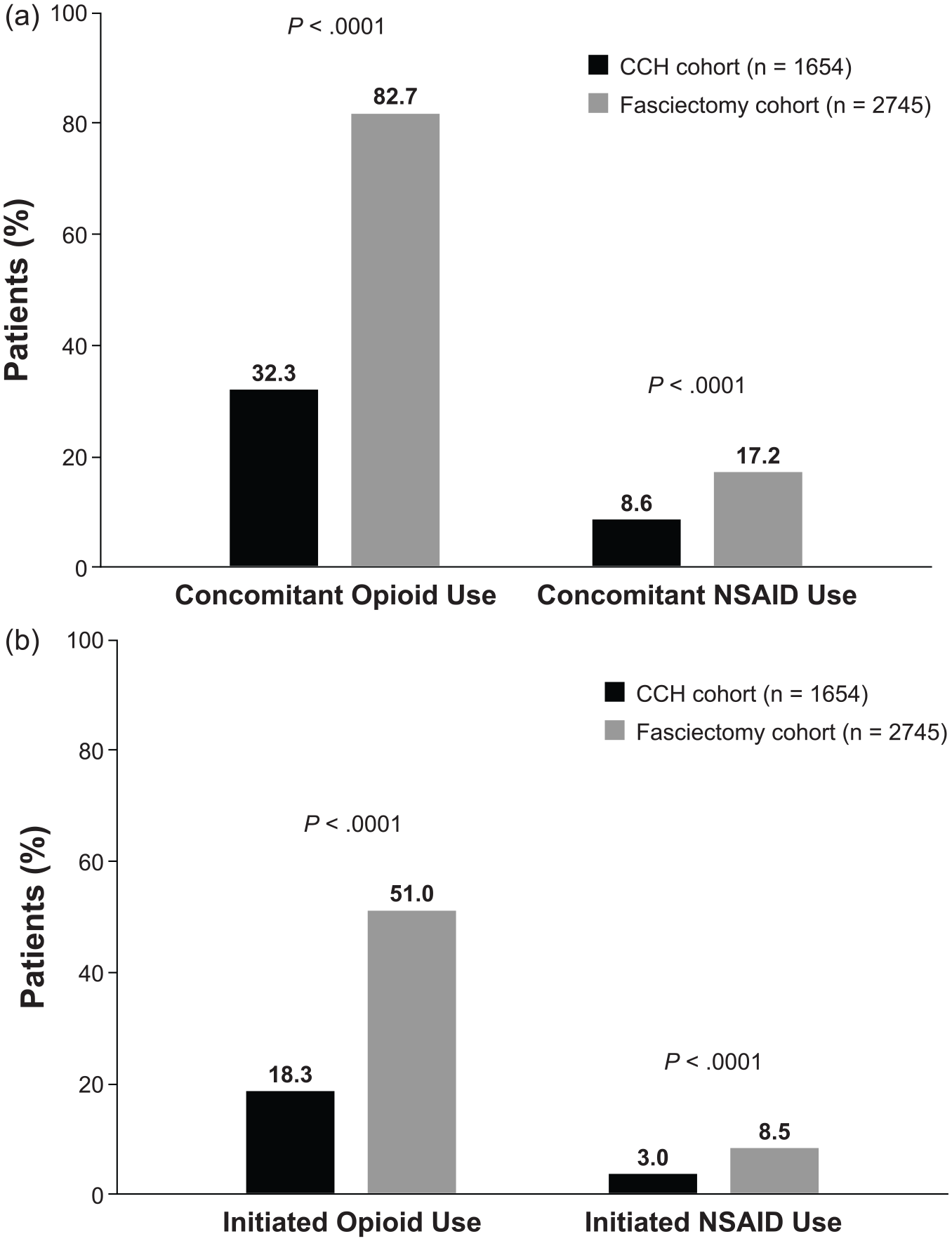
Percentage of patients with (a) any utilization or (b) initiation of opioid analgesics or NSAIDs during the 90-day follow-up period.
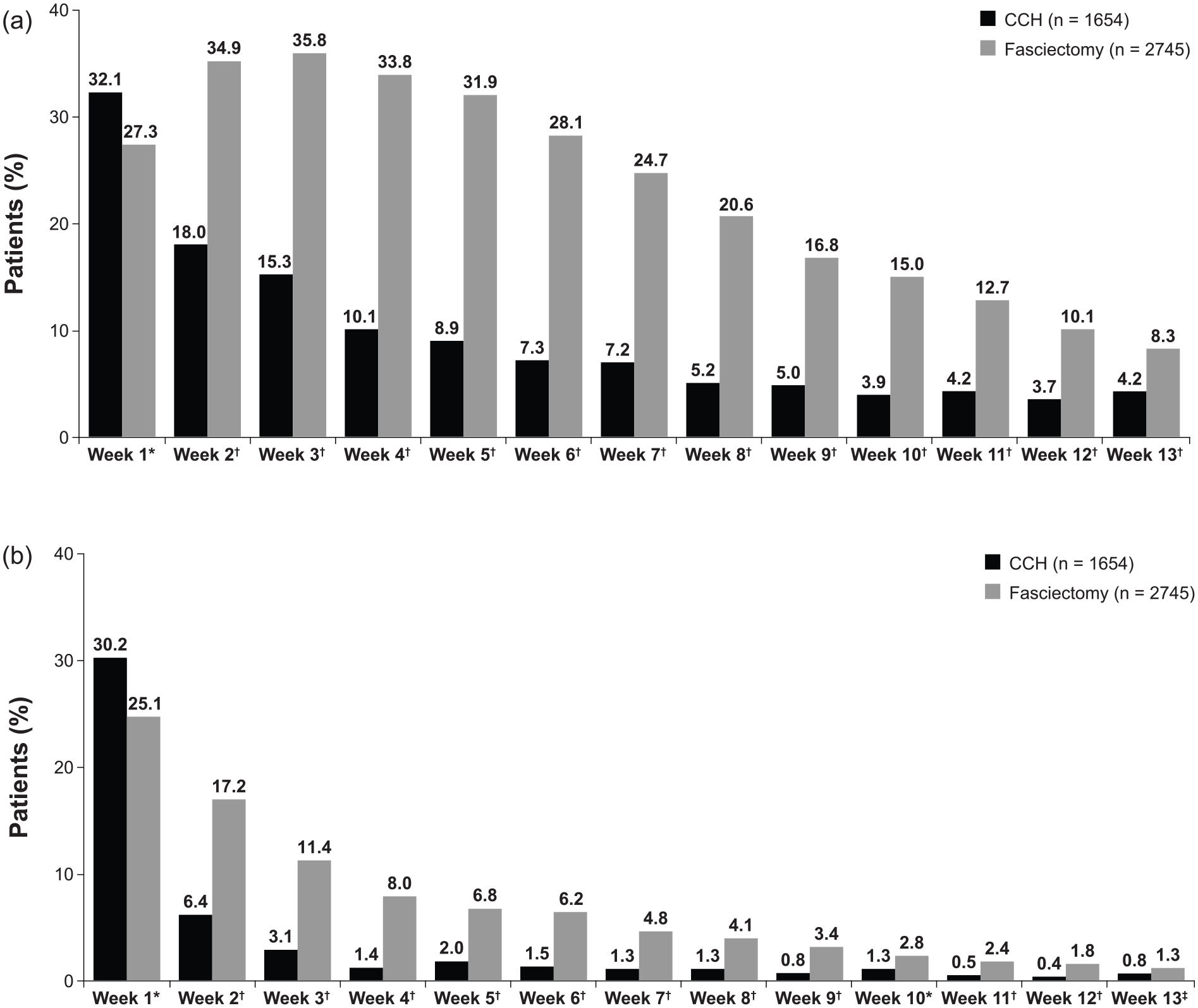
Percentage of patients with ≥1 all-cause (a) physical therapy or (b) occupational therapy visit during the 90-day follow-up period.
Similar trends were observed for DC-specific PT and OT visits. In the CCH cohort, 643 (38.9%) of 1654 patients had ≥1 DC-specific PT visit compared with 1243 (45.3%) of 2745 patients in the fasciectomy cohort (P < .0001). For patients with ≥1 DC-specific PT visit, the mean (SD) number of DC-specific PT visits per patient was 2.5 (2.9) in the CCH cohort versus 6.4 (5.6) in the fasciectomy cohort (P < .0001). In the CCH cohort, 543 patients (32.8%) had ≥1 DC-specific OT visit compared with 1042 patients (38.0%) in the fasciectomy cohort (P = .0006). For patients with ≥1 DC-specific OT visit, the mean (SD) number of DC-specific OT visits per patient was 1.4 (1.4) in the CCH cohort versus 1.9 (2.6) in the fasciectomy cohort (P < .0001). Assessment of DC-specific PT or OT visits by weekly interval during follow-up indicated a significantly higher percentage of patients with ≥1 visit during the first week postindex in the CCH cohort (Figure 3a and 3b). After the first week postindex, the percentage of patients with a DC-specific PT or OT visit during each week in the CCH cohort was significantly lower than in the fasciectomy cohort, with the exception of OT visits during weeks 10 and 13.
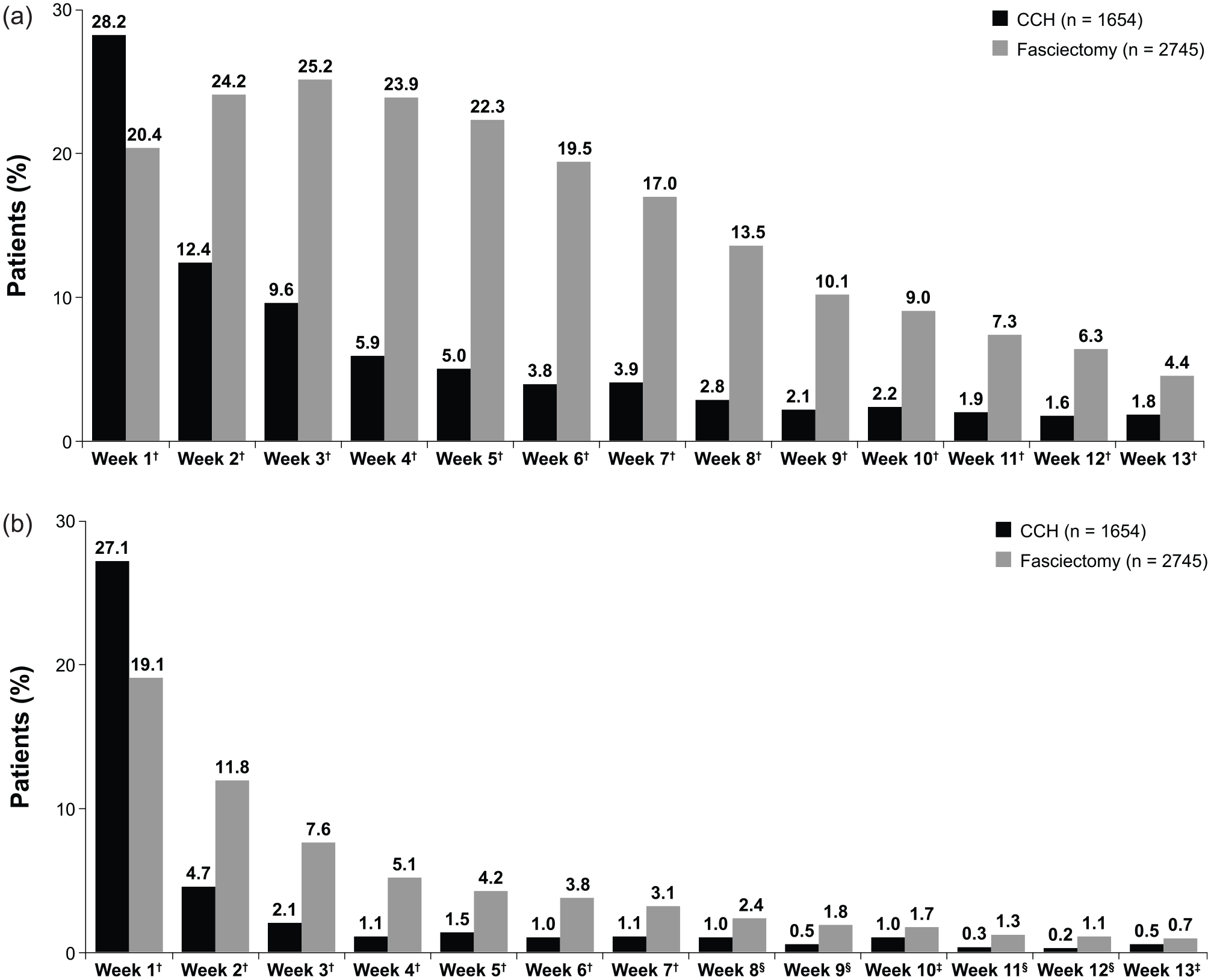
Percentage of patients with ≥1 Dupuytren contracture–specific (a) Dupuytren contracture–specific physical therapy or (b) occupational therapy visit during the 90-day follow-up.
Discussion
Dupuytren disease, including DC, can markedly affect a patient’s ability to perform daily occupational and leisure activities and negatively impacts quality of life.2,4,5 The resulting functional impairment is a key motivation for patients to seek treatment for the condition.21,22 The type of intervention for DC treatment may impact return-to-function during the first few months after treatment and could impact a patient’s ability to perform everyday activities, and thus his or her quality of life. In this study, utilization of prescription pain medication and the number of PT and/or OT visits were less frequent in the CCH cohort compared with the fasciectomy cohort. When these measurements were considered as surrogates for patient return-to-function, the data indicated that patients with DC treated with CCH were able to return to function more quickly than those who underwent fasciectomy.
Most surgeons recommend PT following surgery for the treatment of DC to preserve the gain in finger extension and to restore normal hand functioning.3,22 This likely contributes to higher medical costs reported with fasciectomy compared with CCH treatment. For example, in a 2015 study, the mean total DC-specific cost of PT or OT over a 1-year follow-up period was $442.87 in patients with DC who underwent fasciectomy compared with $182.84 in those who received CCH treatment during 2010-2011 in the United States (2012 US dollars). 8 Similarly, in a 2014 UK single-center, cost-comparison study, all 20 patients with DC who underwent fasciectomy in 2013 had postoperative PT. 11 The mean number of visits per patient was 4 (range, 1-9), at an average cost of £152 (range, £38.82-£342), equivalent to approximately $231 (2013 US dollars), per visit. In contrast, only 3 of 20 CCH-treated patients required PT, each of whom had just 1 visit at a cost of £38.82 per visit ($59 [2013 US dollars]).
A number of factors may affect functional recovery following treatment of DC. For example, a Swedish study of 81 patients undergoing fasciectomy for DC reported that functional recovery was largely related to safety and social issues (eg, the need to take special precautions due to hand function, avoidance of using the hand in social situations) and health-related quality of life. 22 There is also evidence that functional recovery is reflected in patient satisfaction with treatment. In a UK study of 213 patients treated with CCH for DD, the highest satisfaction rates were observed in the patients with the greatest improvements in hand function (measured using the Southampton Dupuytren Scoring Scheme and Quick Disabilities of the Arm, Shoulder, and Hand [QuickDASH]). 23 A faster return-to-function following CCH treatment, compared with fasciectomy, might partly explain the finding in the same study that, among patients who had previously undergone surgery (n = 78), 70% would “definitely” or “probably” prefer CCH injection versus surgery.
A strength of this study is the large data set evaluated, which can be considered representative of the US commercially insured population. Limitations include those inherent in any retrospective analysis of claims data, particularly the lack of clinical data on DC severity at the time of treatment and the extent to which covered benefits may have influenced treatment-related decision making. Although only patients with a single treated digit were included in the analysis, there may be a bias toward patients with DC and severe multijoint, multidigit involvement receiving fasciectomy because there is a limit to the number of areas that should be treated during a CCH treatment session. However, data have shown that functional recovery is not related solely to the degree of improvement of contracture; as noted above, emotional factors and health-related quality of life are important contributors to functional recovery. 22 A further limitation of the study is the use of surrogate markers to assess return-to-function in patients with DC. Given the use of a large claims database, the retrospective nature of the study limited the analyses to these types of markers. However, the utilization of analgesic prescriptions and the number of PT and OT visits as surrogates for return-to-function is are supported by the experience in clinical trials of surgical interventions for conditions such as carpal tunnel syndrome,14,15 anterior cruciate ligament reconstruction, 16 and total knee or hip arthroplasty.17-20 These clinical trials have evaluated PT and pain medication utilization as surrogate prognostic indicators for return-to-work or return-to-function following surgery. Nevertheless, it would be valuable to include other outcome measures, such as the QuickDASH questionnaire, in future prospective studies. 24 In addition, the financial implications of disease recurrence following treatment are another aspect that was not evaluated in the current analysis.
In conclusion, this real-world database analysis using surrogate markers (ie, pain medication utilization and PT and OT visits) suggests that CCH treatment may allow for earlier return-to-function, compared with fasciectomy, in adults treated for DC. Given the high indirect costs associated with the condition, which include costs associated with the loss of employment related to disability and medically related absenteeism from work, 6 this finding may have important implications from a health economic perspective, in addition to that of patient-related quality of life.
Footnotes
Acknowledgements
Technical editorial and medical writing assistance were provided, under the direction of the authors, by Mary Beth Moncrief, PhD, and Michael Shaw, PhD, Synchrony Medical Communications, LLC, West Chester, PA.
Ethical Approval
Ethical approval was not required because this study was a retrospective analysis of anonymized data.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Informed consent was not required because this study was a retrospective analysis of anonymized data.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: V.D. and M.D. are employees of IQVIA, which conducted this analysis with funding from Endo Pharmaceuticals Inc. D.H. is an employee of Endo Pharmaceuticals Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Endo Pharmaceuticals Inc., Malvern, Pennsylvania.
