Abstract
Alignment degradation kinetics of textile implants with tissue healing timelines is critical to mitigate risks from insufficient mechanical retention. This study investigates the structural and mechanical evolution of polyglycolide (PGA) fibers in a knitted interlock fabric during 28-day in vitro degradation, addressing the challenge of rapid PGA degradation that risks premature mechanical failure in biomedical implants. By integrating wide-angle X-ray diffraction (WAXD), small-angle X-ray scattering (SAXS), differential scanning calorimetry (DSC), and mechanical testing, we systematically evaluate how knitted architecture modulates crystallinity, lamellar structure evolution, tensile strength, and mass loss of the PGA fabric. The results indicate that the interlock stitch significantly delays degradation of PGA fibers, retaining 68% of crystallinity and losing only 14.6% of mass after 28 days of in vitro degradation. However, tensile strength sharply declined by 82% within 14 days, driven by hydrolysis-induced flaws in amorphous regions. The degradation process follows three distinct phases: surface erosion (0–7 days), internal amorphous hydrolysis (7–14 days), and gradual crystalline breakdown (14–28 days), governed by heterogeneous solution accessibility and structural constraints of the knitted loops. These findings reveal that textile-based PGA implants have different degradation behavior from PGA fibers, resulting from yarn twisting, intermeshing loops, and localized stress distribution. This work demonstrates that structural design, rather than fiber-level data alone, dictates the balance between mechanical retention and fast degradation in PGA textile implants. The findings provide actionable strategies to optimize surgical implants through tailored textile engineering, ensuring sustained functionality during tissue regeneration while minimizing biocompatibility risks.
Keywords
Introduction
Bioabsorbable polymeric materials are increasingly replacing conventional metal and non-absorbable surgical implants, offering improved long-term safety and efficacy.1,2 Bioabsorbable polymeric materials are engineered to degrade at precisely controlled rates that align with the tissue healing process, thereby minimizing the risk of subsequent complications arising from foreign body reactions. 3
Polyglycolide (PGA) has favorable mechanical properties and excellent biocompatibility. The synthetic and biodegradable sutures made from PGA, known commercially as Dexon®, were developed by American Cyanamide Company in 1962.4,5 Dexon® was approved by the US Food and Drug Administration (FDA) for biomedical applications in 1969. Besides, PGA is also utilized in biodegradable bone grafts,6–10 barrier membranes,11,12 dental materials,13–17 tissue engineering scaffold,18–23 and drug delivery carriers.24–27
Bioabsorbable polymeric materials in medical devices should provide sufficient structural support with adequate mechanical properties throughout their entire service life. PGA has rapid degradation rate, leading to the inevitable loss of mechanical strength and mass during degradation. The acidic degradation products generated can also influence tissue healing. 28 Therefore, understanding the degradation behavior of PGA and its underlying mechanisms is essential for effectively controlling its degradation rate. Fredericks et al.29,30 reported that the PGA hydrolysis process involves two primary mechanisms. Initially, water molecules diffuse into the amorphous regions due to their less organized structure, leading to the breakage of disordered chains. The chain scission enhances the mobility of molecular chains, promoting the crystallization of short-chain molecules. 31 This phenomenon is known as cleavage-induced crystallization.32,33 Subsequently, water molecules gradually attack the crystalline phases, breaking down chain segments into sizes small enough to dissolve in water and be readily metabolized. 34
PGA is typically processed into various morphologies to accommodate different application scenarios, thereby altering its degradation behavior. Melt-spun PGA fibers undergoing in vitro degradation showed that amorphous regions disintegrated within the first 7 days, followed by crystalline phase breakdown within the next 7 days. 35 The fibers lost their tensile strength after 7 days of degradation and experienced an 85% mass loss after 28 days. 35 In the in vitro degradation of Dexon® suture, the amorphous region degraded over 21 days, whereas the crystalline region degraded over 28 days. 36 The PGA suture lost 42% of its mass and all mechanical properties after 4 weeks. 36 Another research found that the PGA suture maintained 80% of its initial strength for 2 weeks, providing adequate structural support for wound healing, which could be fully absorbed within 8–12 weeks. 37 Fibrous PGA scaffolds degraded more slowly in vivo compared to foam PGA scaffolds, and the gradual release of glycolic acid (GA) promoted macrophage polarization toward the M2 pro-healing phenotype. 38 It is evident that, despite the relatively fast degradation rate of PGA, adjusting its morphology and structure enables controlled deceleration of biodegradation in PGA medical devices, thereby enhancing their efficacy.
In recent years, textile-structured absorbable surgical implants have attracted much attention owing to their superior flexibility, elasticity, and highly customizable structural characteristics.39,40 Textile technology has advantages in replicating the hierarchical architectures, anisotropic properties, and strain-stiffening behavior of human tissues.41,42 Numerous PGA-based implants have employed knitted structures.43–46 This intermeshed yarn structure enhances porosity and surface area, thereby facilitating cell adhesion, migration, and proliferation, and delivers superior tensile strength and elastic recovery. Given these advantages, it is critical to understand how the knitted structure itself modulates the degradation behavior of PGA, enabling effective performance tailoring from a design perspective. Extensive research has been conducted on the degradation behavior of PGA fibers, sutures, and nonwoven fabrics.35–38 However, a comprehensive investigation into the degradation mechanisms of PGA in textile constructions, particularly in knitted architectures, remains lacking. This gap limits our ability to modulate the degradation rates of PGA textile implants.
In this study, PGA fibers were knitted to fabrics and subjected to 28 days of in vitro degradation. The microstructural evolution of PGA fibers within the knitted architecture was systematically investigated. Mechanical property deterioration, mass loss, and morphological alterations during the degradation process were quantitatively analyzed and interpreted to elucidate the degradation mechanisms of PGA knitted architecture. The findings provide a theoretical basis for enhancing the modulation of the degradation rate of PGA knitted fabrics and improving the efficacy of PGA surgical implants.
Experimental sections
Material
PGA raw material with a melting point (Tm) of 220.8°C was kindly supplied by Shanghai Research Institute of Petrochemical Technology SINOPEC, China. The PGA raw material was added to a single screw extruder and extruded at 250°C for spinning. PGA fibers were obtained after three sequential hot drawing processes at temperatures of 50°C, 90°C, and 95°C, with a drawing ratio of 5.0. The PGA yarn had a linear density of 154.5 dtex with 21 filaments, and a tenacity of 4.5 cN/dtex. The PGA fabrics were knitted with a 1 × 1 interlock stitch on a Stoll CMS 530Ki E10.2 computerized flat knitting machine (Karl Mayer Stoll Textilmaschinenfabrik GmbH, Reutlingen, Germany). The loop length was controlled by setting the NP value (sinking depth) to 10.0. The yarn input tension during knitting was maintained at 2.5 cN through an EFS 920 electronic yarn feeder (Memminger-IRO GmbH, Dornstetten, Germany). As shown in Figure 1(a) and (b), the PGA fabric of 1 × 1 interlock stitch, has dimensions of 50 mm × 25 mm, with an areal density of 265 g/m2, a thickness of 0.99 mm, and a stitch density of 64 courses per 5 cm and 130 wales per 5 cm. The areal density was determined as the average of five samples, each calculated by weighing and dividing by its area, while the thickness was taken as the average of five measurements using a fabric thickness gage (Taizhou Kejia Measuring Tools Co., Ltd., Zhejiang, China). The areal and stitch densities were measured after dry relaxation, where the fabric was conditioned under standard atmosphere (20°C, 65% RH) for 24 h prior to measurement.

(a) Schematic of the 1 × 1 interlock knit stitch and (b) surface morphology of the PGA 1 × 1 interlock knitted fabric.
In vitro degradation
The PGA fabric samples were ultrasonically cleaned with absolute ethanol for 30 min, dried, and subsequently sealed for sterilization using ethylene oxide at 45°C for 15 h. The sterilized PGA fabric specimens were immersed in phosphate-buffered saline (PBS, pH 7.40) contained in glass beakers. The solution volume-to-sample mass ratio was maintained at ⩾30:1 (mL/g). These experimental setups were subsequently incubated at 37°C for 28 days to conduct the in vitro degradation study. The buffer solution remained unchanged throughout the experiment. Systematic pH monitoring was conducted at predetermined intervals (0, 3, 5, 7, 14, 21, and 28 days) following degradation initiation. The PGA fabric samples were extracted from the solution weekly, gently washed three times with distilled water, blotted dry with filter paper, vacuum-dried for 24 h, and then proceeded with subsequent tests. The mass loss (m1%) of PGA fabric during the degradation process was calculated following equation (1)
where m0 is the initial mass of PGA fabrics, md is the dried mass of PGA fabrics remaining at a given degradation time.
Surface morphological evolution of PGA fabrics was characterized using a Hitachi FLEX 1000 scanning electron microscope (Hitachi High-Technologies Corporation, Tokyo, Japan) with gold sputter coating.
Mechanical property test
Fabric tensile tests were conducted for the samples after different degradation days on a YG028 universal mechanical tester (Ningbo Dahe Instrument Co., Ltd., Zhejiang, China). The fabrics were stretched along the walewise direction to break at a rate of 100 mm/min in the standard environment of 20°C and 65% humidity according to the standard GB/T 3923.1-2013 “Textiles - Tensile properties of fabrics - Part 1: Determination of breaking strength and elongation at break (strip method).” Five tests were conducted for each sample, and the tensile strength and elongation at break were calculated by averaging the five test results. The stress (in MPa) was dividing tensile force (in N) by the original cross-sectional area (in mm2), defined as the product of the sample width (25 mm) and thickness (0.99 mm).
DSC measurements
DSC measurements were performed with Q20 differential scanning calorimeter (TA Instruments, DE, USA) under a nitrogen atmosphere. Samples weighing about 6 mg were placed in small aluminum pans and heated from 30°C to 250°C at a rate of 10°C/min, with three repeated scans conducted per sample and the results averaged. The melting temperature (Tm) was defined as the peak position of the melting endotherm measured via differential scanning calorimetry (DSC). The corresponding melting enthalpy (∆Hm, J/g) was calculated by integrating the peak areas above the baseline within the thermal transition region. The melting enthalpy of 100% crystalline PGA (∆Hm0, J/g) is 139.1 J/g, 47 and the degree of crystallinity (Xc, %) was calculated following equation (2)
For the current PGA knitted fabrics, no distinct cold crystallization peak was observed in the DSC curves. Thus, the value of cold crystallization enthalpy (∆Hc, J/g) was taken as zero.
WAXD and SAXS measurements and data analysis
WAXD measurements were performed on a D8 X-ray diffractometer (Bruker AXS GmbH, Karlsruhe, Germany) with Cu-Kα radiation (λ = 0.1541 nm). Since the PGA fibers became brittle after 14 days of degradation, making it difficult to separate individual intact fibers from the fabric, the samples were ground into powder for testing. The sample-detector distance was 75 mm, and the diffraction angle 2θ ranged from 5° to 60°. The resultant 1D-WAXD curves were analyzed, and the degree of crystallinity and the crystallite sizes (Dhkl) were calculated using peak deconvolution. Due to the inherent subjectivity of manual peak deconvolution, these structural parameters for each sample, although tested once, were determined from five separate calculations, and the average values are reported. The (110) and (020) reflection peaks were used to calculate the Dhkl. This measurement was performed using the Scherrer equation
where λ is the wavelength, θ is the Bragg’s angle, and β is the experimental breadth at the maximum intensity. The shape factor (k) was taken as 0.89, as this value is standard for a spherical crystal model approximation, which appropriately describes the crystal growth habit corresponding to the (110) and (020) diffraction planes used in the calculation.
SAXS measurements were conducted on a Xeuss 3.0 SAXS system (Xenocs, France) equipped with a Cu Kα radiation source (λ = 0.1541 nm) and an Eiger2R 1M hybrid pixel detector (Dectris, Switzerland). The two-dimensional scattering patterns were collected with a sample-to-detector distance of 500 mm. Fabric samples were directly cut into square pieces for the SAXS test as it was hard to extract straight single fibers of sufficient length from the fabrics that have been degraded for 14 days or more. Since SAXS test gives a statistical information of the nanostructures, a single measurement was performed for each sample. 48 The theoretical long period (L) was calculated using the Bragg equation based on the one-dimensional SAXS curve obtained through Lorentz correction
where q is the peak position of the SAXS curve.
Results and discussion
Crystallinity evolution and lamellar dynamics in degraded knitted architecture
The microstructural evolution of PGA during degradation dictates its macroscopic behavior, thereby governing the load-bearing capacity and structural integrity retention of knitted implants. It is well-established that crystalline PGA adopts an orthorhombic unit cell. The reported lattice parameters are a = 0.522 nm, b = 0.619 nm, and c = 0.702 nm, with molecular chains in a planar zigzag conformation yielding a crystalline density of 1.69 g/cm3. 49 One-dimensional WAXD patterns enable quantitative assessment of crystallinity and crystallite dimensions through diffraction peak broadening analysis and intensity integration. The WAXD results of PGA in the knitted fabrics at different degradation days are presented in Figure 2. As shown in Figure 2(a), the characteristic diffraction peaks were the (110) and (020) crystal planes, with the corresponding 2θ values being 22.0° and 28.7°, respectively. The degree of crystallinity calculated from the WAXD results is shown in Figure 2(b). The degree of crystallinity of PGA gradually increased during the initial 21 days of degradation and subsequently decreased. This trend is consistent with the degree of crystallinity obtained from DSC results, as shown in Figure 3. The DSC curves in Figure 3 exhibit no clear glass transition or cold crystallization events, consistent with the behavior of highly crystalline PGA. This is because the substantial crystallinity (approximately 60%) confines chain mobility within the amorphous regions, and the absence of cold crystallization is attributed to rapid chain scission induced by hydrolysis during degradation which inhibits molecular reorganization upon subsequent heating. 47 This observation is in agreement with previous reports on PGA fibers, where the Tg is frequently obscured in highly crystalline samples.47,50

Crystalline structural evolution during degradation: (a) 1D-WAXD profiles, (b) crystallinity, and (c) (110) and (020) crystallite sizes.

Thermal analysis and crystallinity changes: (a) DSC curves and (b) calculated crystallinity.
The increase in crystallinity during the initial stage of degradation is attributed to the preferential hydrolytic degradation of the amorphous regions. 35 The cleavage-induced crystallization could further increase the degree of crystallinity. 50 It is worth noting that, due to the high crystallinity of PGA fibers used in this study, the cleavage-induced crystallization phenomenon was likely to be minor. The degradation of lamellar crystals manifested through progressive crystallinity reduction between days 21 and 28. The PGA fibers in the knitted fabric retained a crystallinity of 68% after the 28 days of degradation, indicating that a substantial portion of the crystals had not degraded yet.
PGA knitted fabric degraded slower than PGA fibers. This is supported by a comparison with previous work on fibers of comparable initial crystallinity (approximately 60%), which exhibited a mass loss of approximately 40% within 21 days, suggesting significant degradation of the crystalline regions. 51 This is also confirmed by another study which reported mass losses exceeding 80% for PGA fibers alongside a crystallinity reduction of approximately 10% over a comparable period. 35
The delayed overall degradation of the PGA fibers in knitted construction stems from heterogeneous solution accessibility among fibers, which will be systematically analyzed through morphological changes and mass loss below.
SAXS measurement was implemented to investigate the lamellar structure evolution of PGA fibers in knitted fabrics during degradation. The 2D-SAXS patterns in Figure 4(a) exhibit anisotropic scattering rings which indicate the slight orientation of lamellar crystals. During the initial 14 days of degradation, the intensity of the scattering ring progressively increases, indicating a growing difference in electron density between the crystalline and amorphous phases, which is corresponded to pronounced degradation of the amorphous region. Subsequently, the scattering signal intensity from the lamellar crystals gradually decreases, reflecting the degradation of the crystalline phases. Figure 4(b) presents the Lorentz-corrected SAXS profiles extracted from the 2D-SAXS pattern, and the theoretical long period (L) calculated using the Bragg equation is shown in Figure 4(c). The L of PGA fibers in knitted fabrics exhibits a slight decrease during the first 14 days and remains nearly constant during the subsequent period. The L value is the sum of the lamellar thickness (Lc) and the amorphous layer thickness (La). PGA fiber amorphous regions typically contracted during initial degradation, accompanied by a slight decrease in La. Moreover, the minor cleavage-induced crystallization formed thinner crystals due to spatial constraints in amorphous regions and lower molecular mass species, potentially reducing Lc.49,52

Lamellar dynamics during degradation: (a) 2D-SAXS patterns, (b) Lorentz-corrected SAXS profiles, and (c) long period calculation.
The WAXD and SAXS results demonstrate that knitting PGA fibers into fabrics substantially reduced the degradation rate. The microstructural characteristics of the knitted fabrics composed of PGA fibers after 28 days of in vitro degradation were comparable to those of directly degraded PGA fibers after 7 days. 35 This delayed degradation in microstructure could affect the mechanical properties, mass loss, and morphological changes of PGA knitted fabrics.
Mechanical failure driven by amorphous hydrolysis and structural disintegration
Artificial implants must provide necessary mechanical strength to support tissue function until complete healing occurs. Figure 5 shows the changes in mechanical properties of PGA knitted fabric throughout the degradation process. The tensile strength of PGA fabric has an initial decline during the first 7 days of degradation, followed by a dramatic reduction of 56% between days 7 and 14. The PGA knitted fabric exhibits 82% tensile strength loss after 14-day degradation, with complete mechanical failure occurring upon 21-day degradation.

Mechanical deterioration over degradation time: (a) tensile behavior and (b) ultimate tensile strength.
The deterioration of the mechanical properties of PGA knitted fabrics can be attributed to the synergistic effects of structural disintegration and intrinsic material degradation. Weft knitted fabrics are formed by yarns being laterally bent into loops and subsequently intermeshing with the loops from the preceding course. The load-bearing mechanism of weft knitted structures primarily involves the dispersion of loads through the elastic deformation of loops, complemented by stress dissipation via frictional energy absorption between yarns. Figure 6 schematically illustrates the mechanical failure process of the PGA fabric. Stress concentration was likely to occur at the contact points between loops when the fabric was stretched. Moreover, the force distribution on fibers within the fabric was not entirely along the axial direction of the fibers, but local forces at different points along the fibers vary. This distributed loading of fibers in fabric is markedly different from the uniform loading of uniaxially stretching fibers. Flaws originating from the intrinsic material degradation of fiber preferentially initiated fracture under the distributed loading configuration. The cumulative fiber fracture led to yarn failure, subsequently triggering loop disentanglement. The loop disentanglement progressed along the walewise direction under the tensile loading, resulting in stress concentration that initiated further yarn fracture and laddering. Ultimately, the knitted construction collapsed and lost load-bearing capacity.

Tensile-induced structural failure modes in degraded knitted fabrics: failure yarns (red) with critical fracture points (A and B), disentangled loops (orange), structurally intact yarns (yellow).
The above microstructural analysis demonstrates that amorphous phase degradation induced progressive internal flaw formation in PGA fibers within the initial 14 days, resulting in 82% strength loss in PGA knitted fabrics. After 21 days of degradation, PGA fabrics exhibited complete mechanical strength loss, attributable to lamellar crystal disintegration. Remarkably, most crystalline regions of PGA fibers had not degraded by day 28 of the in vitro degradation. These observations indicate that the deterioration rate of PGA knitted architecture’s mechanical properties is predominated by amorphous region degradation.
Morphological heterogeneity and progressive mass loss in knitted architecture
Aliphatic polyesters undergo hydrolytic cleavage of their ester backbone in aqueous environments. The degradation products of PGA include glycolic acid (GA) and its oligomers. GA is water-soluble and can be metabolized through the tricarboxylic acid cycle or excreted by the kidneys. The morphology of PGA knitted fabrics and fibers evolved during degradation are shown in Figure 7. The yarns composed of multiple PGA fibers were not twisted before being fed into the knitting machine. However, SEM micrographs of the PGA knitted fabric reveal that the yarns were slightly twisted during knitting, leading to fiber migration between the interior and exterior of the yarns. This observation directly verifies the formation of a coherent yarn structure through mechanical entanglement during the knitting process, which critically influences the heterogeneous degradation behavior by creating solution-accessibility gradients. Fiber segments exposed on the yarn’s exterior contacted the external solution earlier and more extensively, leading to faster degradation than those inside the yarn. Additionally, the degradation extent varied among different segments of the same yarn due to mutual shielding and overlapping between adjacent yarns.

Morphological evolution of PGA knitted fabrics and fibers during degradation.
The surface of PGA fibers is initially smooth, but distinct blocky damages are evident on the fiber surface after 7 days of degradation, indicating that hydrolysis predominantly occurred in the surface during this period. It could be confirmed by monitoring the changes in the pH value of the external solution and the mass loss of PGA knitted fabric, as shown in Figure 8(a) and (b). During the initial 7 days of degradation, both the pH value of the solution and the fabric mass decreased slightly. SEM micrographs also reveal deep internal cracks propagating within PGA fibers during days 7–14 of degradation. The internal cracks accelerated both water molecule penetration and acidic degradation product release. This dissolution process, occurring specifically in the 7–14 day period, drove concurrent sharp pH decline and fabric mass loss through mass transfer mechanisms. After 21 days of degradation, numerous fiber breaks were observed on the PGA knitted fabric surface. Most broken fiber cross-sections showed ductile fracture morphology, due to extensive degradation of amorphous regions and limited degradation of lamellar crystals. From 14 to 21 days of degradation, the amounts of acidic degradation products continued to be generated and dissolved in the external solution, driving a more rapid pH decline and a sustained increase in fabric mass loss. After 28 days of degradation, the lamellar crystals were further degraded. Macroscopically, more broken fibers appeared on the fabric surface, and fracture surfaces resembled brittle fractures. The mass loss of PGA knitted fabrics progressed during this degradation phase. The degradation solution was not replaced throughout the process, and the solution’s pH had reached a low level by day 21, which is likely contributed to the slowed rate of pH decrease thereafter.
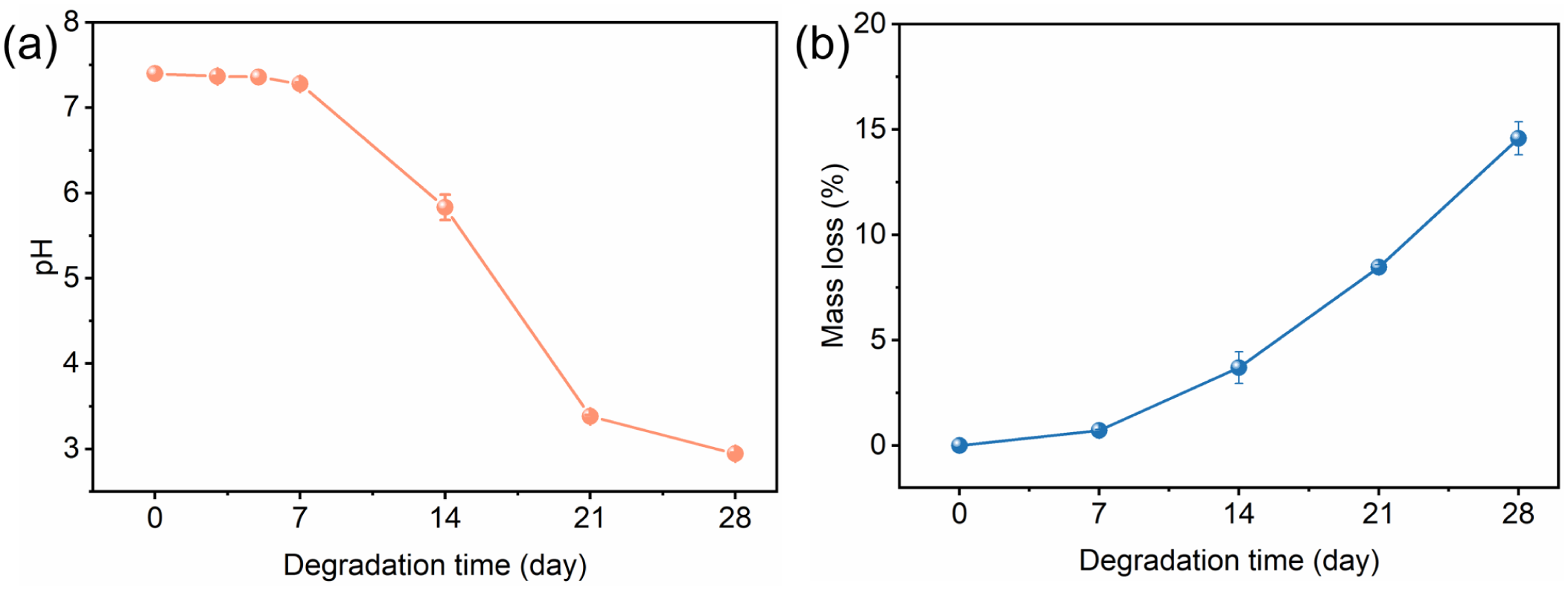
Environmental and mass loss monitoring: (a) pH of degradation medium and (b) cumulative mass loss of fabrics.
After 28 days of in vitro degradation, the PGA knitted construction remained largely intact, with a mass loss of only 14.6%. The extensive degradation of the amorphous region was primarily responsible for the increased acidity of the external solution. It can be expected that, even after losing all mechanical strength, the PGA textile-based implants could continue to promote tissue thickening and nutrient delivery at the injury site.
Three-phase degradation mechanism: Structural constraints and solution accessibility
The PGA yarns inevitably twisted and intermeshed during loop formation, causing fiber migration between the interior and exterior of the yarns. Attributed to the differences in exposure to the solution, fibers within the fabric degraded more slowly compared to those exposed on the surface of fabrics. Hence, knitting PGA fibers into fabrics can significantly delay the degradation of PGA. The structural and morphological evolution of PGA knitted architecture during 28 days of degradation can be roughly divided into three stages.
In stage I (0–7 days), the PGA fibers exposed on the fabric surface begin to degrade. Water molecules initially degraded the surface and limited amorphous regions of PGA fibers, thereby increasing crystallinity and decreasing the mechanical strength of the PGA knitted fabrics. The oligomers generated from the degradation of internal amorphous regions were retained within the fibers, leading to minimal mass loss and a slight decrease in pH of solution.
In stage II (7–14 days), the fibers in the interior of the fabric progressively initiate degradation, while water molecules penetrate deeper into the fibers degraded previously. Extensive degradation of amorphous regions drove a continuous rise in crystallinity and generated multiple flaws within fibers. The structural flaws fractured preferentially at stress concentration areas during loading, accelerating mechanical deterioration of the knitted fabric through loop disentanglement. Fiber cracks served as channels for acidic degradation product release, causing sharp pH decline and substantial fabric mass loss. Amorphous region contraction and slight cleavage-induced crystallization reduced L.
In stage III (14–28 days), lamellar crystals within fibers initiate degradation, evidenced by diminishing scattering intensity patterns. Preferential degradation of amorphous chains drove an increasing PGA crystallinity during days 14–21, while extensive lamellar crystal degradation between days 21 and 28 reversed this trend. The PGA knitted fabric exhibited 82% strength loss by degradation day 14. Remarkably, while the mechanical strength of the PGA fabric was entirely lost on day 28, the degree of crystallinity maintained 68% with only 14.6% mass loss, revealing substantial crystalline survival in PGA fibers. The delayed microstructural degradation was governed by constrained solution accessibility to PGA fibers in the knitted architecture.
To sum up, knitting PGA fibers into knitted architecture significantly decelerates the microstructural degradation in vitro, yet the mechanical strength deterioration rate of PGA knitted architecture exceeded that of pristine fibers. The faster strength loss stems from the combined effects of the load-bearing mechanism of the weft knitted architecture and the growth of internal flaws initiated by amorphous region degradation.
Conclusions
In this study, WAXD and SAXS were utilized to investigate the microstructural changes of PGA fibers in knitted fabrics during in vitro degradation. The evolution of microstructure was correlated with alterations in mechanical properties, mass loss, and morphology of PGA knitted fabrics. The comprehensive analysis elucidated the in vitro degradation mechanism of PGA knitted fabric of 1 × 1 interlock stitch. The following conclusions can be drawn:
Fabricating PGA fibers into knitted architecture significantly decelerates microstructural degradation, retaining 68% crystallinity and exhibiting only 14.6% mass loss after 28 days of in vitro degradation.
The degradation of PGA knitted fabric is divided into three stages. In stage I (0–7 days), the PGA fibers exposed on the fabric surface start to degrade. In stage II (7–14 days), internal fibers progressively degrade, while the amorphous regions of surface fibers are significantly hydrolyzed. In stage III (14–28 days), the lamellar crystals of PGA fibers within knitted fabrics gradually degrade.
PGA knitted fabrics exhibit 82% tensile strength loss after 14-day degradation, with substantial hydrolysis of amorphous regions predominantly governing this mechanical deterioration.
The findings of this work demonstrate that the PGA textile-based implants could provide robust mechanical support to damaged tissues during the initial implantation phase. Even after the loss of mechanical strength, they may continue to facilitate tendon or tissue thickening while providing a bioactive scaffold for nutrient diffusion and spatial accommodation. The findings of this work also confirm that solely extrapolating biodegradable fiber degradation cannot reliably predict the behavior of the bioabsorbable textile-based implants. Processing PGA fibers into knitted fabrics and conducting degradation studies on them provide a more comprehensive understanding of the degradation performance of PGA implants, which helps to improve the efficacy of PGA surgical implants.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research project of China Petroleum & Chemical Corporation (grant number 224259) and the Fundamental Research Funds for the Central Universities (grant number CUSF-DH-T-2024046).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.




