Abstract
Diabetic wounds are associated with prolonged inflammation and impaired healing due to the overproduction of reactive oxygen species (ROS). These reactive molecules damage cellular components and the extracellular matrix, delaying tissue regeneration. Addressing oxidative stress in the wound microenvironment is a promising strategy to improve healing outcomes. To mitigate ROS at the wound site, 3-hydroxyanthranilic acid (3HA), a known antioxidant, was incorporated into polyvinyl alcohol (PVA) electrospun scaffolds at concentrations of 0.5%, 1%, and 1.5% (w/w). The scaffolds were characterized for fiber morphology and diameter. In vitro cell viability assays were performed under both normal and oxidative stress conditions using fibroblasts. The optimal formulation was further evaluated in a streptozotocin-induced diabetic rat wound model. Gene expression analysis of Interleukin-1 beta (IL-1β), glutathione peroxidase, and basic fibroblast growth factor (b-FGF) was conducted using qPCR. The incorporation of 3HA did not significantly alter the nanofiber morphology. The average fiber diameter was 1408.18 ± 31.28 nm for PVA/1%3HA and 1395.61 ± 23.09 nm for pure PVA scaffolds. Among all formulations, the scaffold with 1% 3HA demonstrated the highest cell viability under oxidative stress. In vivo wound healing studies revealed that PVA/1% 3HA-treated wounds closed more rapidly compared to the PVA-only and untreated control groups. Gene expression results showed reduced levels of IL-1β and glutathione peroxidase, while b-FGF expression was significantly upregulated in the treated group. The PVA/1% 3HA scaffold effectively enhanced diabetic wound healing by reducing oxidative stress and inflammation while promoting tissue regeneration. The improved healing outcomes can be attributed to the antioxidant and immunomodulatory effects of 3HA. These results support the potential of 3HA-loaded PVA scaffolds as a bioactive dressing for diabetic wound management.
Introduction
The skin, our body’s largest organ, plays various roles in maintaining overall bodily balance. Wounds are defined as disruptions in the integrity of the skin, which can result from physical, thermal, or electrical injuries.1,2 While the skin possesses a natural ability to heal itself, certain health conditions, like diabetes mellitus, can significantly impede the skin’s regenerative potential. 3 Diabetes-related wounds are characterized by reduced blood flow, nerve damage, diminished growth factor production, heightened inflammation, and an excess of reactive oxygen species (ROS). These factors disrupt the usual stages of wound healing, causing the healing response to become stuck in the inflammation phase and preventing progression to subsequent phases.4,5
In such cases, external interventions are needed to promote wound healing. Among various treatment options, wound dressings have gained significant therapeutic interest, and numerous formulations, such as electrospun fibers, composites, sheets, and membranes, have been explored for diabetic wound healing. Electrospun fibers have garnered special attention for developing wound dressings due to their ability to provide sustained drug release, their extracellular matrix (ECM)-like structure, high drug encapsulation efficiency, adjustable properties, and ease of fabrication.6,7
Choosing the right materials and optimizing them is a crucial initial step in creating effective electrospun wound dressings. In this context, a variety of polymers, whether synthetic or natural, have been tested. Polyvinyl alcohol (PVA) is a versatile synthetic polymer widely utilized in wound healing due to its biocompatibility, biodegradability, and excellent film-forming properties. Various formulations of PVA-based wound dressings have been tested. PVA-based hydrogels maintain a moist environment that promotes cell migration, tissue regeneration, and pain relief. Various formulations of PVA have been developed to enhance wound healing efficacy. Crosslinked PVA hydrogels offer improved mechanical stability and controlled drug release, while electrospun PVA nanofibers mimic the extracellular matrix (ECM), supporting cell adhesion and proliferation. PVA composites blended with natural polymers like chitosan, alginate, or gelatin enhance antibacterial activity, biocompatibility, and bioactivity. 8 Incorporation of nanoparticles (e.g., silver, zinc oxide) or bioactive compounds (e.g., curcumin, antibiotics) further augments antimicrobial and anti-inflammatory properties.9,10 In this regard, Fatahian et al. 11 developed electrospun PVA-based wound dressings loaded with Tranexamic acid (TXA) for blood coagulation and Ceftriaxone (CTX) for antibacterial activity. Nanofibers were uniform and bead-free. Higher drug concentrations increased fiber diameter, coagulation efficiency, and antibacterial effectiveness. El-Attar et al. 12 developed electrospun PVA nanofibers loaded with biosynthesized silver nanoparticles (AgNPs) using snail mucus as a natural reducing agent. The AgNPs were stable and effective against Pseudomonas aeruginosa. The resulting nanofibers were uniform (100–170 nm), showed strong antibacterial activity, and significantly enhanced wound healing in vitro and in vivo after 12 days. Despite its advantages, PVA has limitations in wound healing, including low inherent antibacterial activity, limited biodegradability, and insufficient mechanical strength when used alone. It often requires blending with other polymers or additives to enhance functionality, such as bioactivity, durability, and controlled drug release for more effective wound care applications. The advantage of PVA is its suitability for electrospinning using aqueous solvents under mild conditions, which allows for effective drug loading without compromising the physicochemical and biological properties of the drug. 13 Despite its advantages, PVA has limitations in wound healing, including low inherent antibacterial activity, fast degradation, and insufficient mechanical strength when used alone. It often requires blending with other polymers or additives to enhance functionality, such as bioactivity, durability, and controlled drug release for more effective wound care applications.9,13
The rationale behind using drug-loaded PVA scaffolds for diabetic wound healing is that pure PVA scaffolds lack sufficient bioactivity for successful wound healing. Antioxidant drugs are an ideal choice for this purpose because they can neutralize excessive ROS at the wound site and reduce inflammation. 14 3-hydroxyanthranilic acid (3HA) is an antioxidant intermediate produced in tryptophan metabolism and isolated from the methanol extract of tempeh. Previous studies have well-documented the protective effects of 3HA in various inflammatory disorders. 15 Krause et al. 16 showed that in primary human CNS cell cultures, 3-HA significantly suppressed glial cytokine and chemokine expression and protected neurons from cytokine-induced death. Notably, 3-HAA strongly induced heme oxygenase-1 (HO-1) expression in astrocytes—an antioxidant enzyme with well-established anti-inflammatory and cytoprotective roles. This induction was most effective in the presence of both 3-HAA and proinflammatory cytokines.
Consequently, the incorporation of 3HA into the structure of electrospun wound dressings aims to reduce oxidative stress and alleviate hyper-inflammatory responses. The objective of this study is to investigate the healing effects of 3HA-loaded PVA fibers in a rat model of diabetic wound healing.
This is the first known study to utilize 3HA in a topical scaffold system specifically designed for diabetic skin wound repair. By harnessing 3HA’s proven ability to upregulate heme oxygenase-1 (HO-1) and suppress pro-inflammatory cytokines, this work aims to break the chronic inflammation cycle typical in diabetic wounds and promote progression through the wound healing phases. The combination of a clinically relevant delivery platform (PVA electrospun fibers) with a bioactive metabolite (3HA) offers a novel strategy for diabetic wound management with potential translational impact.
Materials
All materials and solvents were purchased from Sigma Aldrich (Germany), unless otherwise noted.
Methods
Fabrication of 3HA-loaded PVA fibers
To start, polyvinyl alcohol (PVA) was dissolved in distilled water as the solvent, reaching a final concentration of 12%. This solution was thoroughly mixed until it became clear. Subsequently, three different concentrations of 3-hydroxyanthranilic acid (3HA) at 0.5% w/w, 1% w/w, and 1.5% w/w were added to the PVA solution and further mixed for 6 hours at room temperature. The resulting 3HA-loaded polymeric solution was loaded into 10 ml disposable syringes, which were then utilized for the electrospinning process. An 18-gauge metal needle was affixed to the syringe’s tip, connected to a positively charged high voltage source. The impact of various factors, including voltage magnitude, the distance between the needle tip and the collector, polymer concentration, and more, was initially investigated by performing electrospinning on a glass slide, followed by examination under a light microscope. The optimal parameters for the electrospinning process were determined to be a positive high voltage of 16–18 kV, a polymer feeding rate of 0.5–1 mL/h, and a tip-to-collector distance of 15–16 cm. Following the fabrication process, the electrospun fibers were detached from the collector and crosslinked.
Scanning electron microscopy (SEM) analysis
The SEM analysis of both pure PVA and PVA/1% 3HA scaffolds involved examining their microstructures. Prior to analysis, they were coated with gold for a duration of 250 s. The imaging was conducted under 25 kV accelerating high voltage.
Cell viability assay
The viability of L929 fibroblast cells cultured on PVA and 3HA-loaded PVA scaffolds was evaluated using the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. Scaffolds were cut into circular discs and sterilized by exposure to ultraviolet (UV) light for 30 min on each side. Following sterilization, the scaffolds were placed in 24-well culture plates. L929 cells were seeded onto the surface of each scaffold at a density of 1 × 104 cells per well and incubated in DMEM supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 µg/mL streptomycin. The culture plates were maintained at 37°C in a humidified atmosphere with 5% CO2. The MTT assay was performed on days 1, 3, and 7 post-seeding. At each time point, the culture medium was removed and replaced with 500 µL of MTT solution (5 mg/mL in PBS). The plates were incubated for 4 h at 37°C to allow the formation of formazan crystals. After incubation, the MTT-containing medium was carefully removed, and 500 µL of dimethyl sulfoxide (DMSO) was added to each well to dissolve the formazan crystals. The absorbance of the resulting solution was measured at 570 nm using a microplate reader.
Cytoprotection under oxidative stress
The protective effects of 3HA-loaded scaffolds against H2O2-induced oxidative stress were studied according to a method as described previously. 17 Firstly, wound dressings were sterilized and then immersed in 2 mL of Dulbecco’s Modified Eagle Medium (DMEM) culture medium and then kept at 4°C for 4 days. After the incubation time, the remaining culture medium was fileted and kept at 4°C for cell culture assays. The supernatants of different scaffolds were mixed with 1% H2O2, 10% (v/v) fetal bovine serum, 100 unit/mL of penicillin (Sigma-Aldrich, USA) and 100 μg/mL of streptomycin (Sigma-Aldrich, USA) and used for culturing of L929 cells for 24 h. At the end of 24 h post-culturing, cells were assessed for their survival rate using MTT assay kit (Nanjing Duly Biotech Co., Ltd.). Cells cultured with H2O2-containing and H2O2-free scaffolds were control groups.
Water uptake capacity measurement
The water uptake capacity of PVA/1% 3HA scaffolds and pure PVA scaffolds was evaluated to assess their ability to absorb aqueous solutions, following a method previously described with slight modifications. 18 Briefly, dry scaffold samples (n = 3) were weighed (W-dry) and immersed in phosphate-buffered saline (PBS, pH 7.4) at room temperature. Then, the samples were removed, gently blotted to eliminate excess surface moisture, and weighed again (W-wet). The water uptake capacity was calculated using the following formula:
Mechanical properties analysis
The mechanical properties of PVA/1% 3HA scaffolds and PVA-only scaffolds were studied by cutting them into 4.5 × 1.5 cm2 pieces and fixing them into the jaws of a mechanical strength testing device (Instron Universal Testing Machine). A 500 N force at an extension rate of 1-1.5 mm/min was applied on the scaffolds until they failed.
Contact angle measurement
The water contact angle of PVA/1% 3HA scaffolds and PVA-only scaffolds was studied using a contact angle measuring device. A water droplet (0.5 µL) on the surface of the dressings and imaging using a digital camera. The corresponding images were used for the calculation of surface contact angle.
Water vapor permeability test
The water vapor permeability of PVA/1% 3HA scaffolds and PVA-only scaffolds was assessed using an indirect approach. 19 Empty bottles were filled with 10 mL of distilled water, and their openings were sealed with PVA/1% 3HA scaffolds and PVA-only scaffolds. These bottles were then maintained at a temperature of 37°C for a duration of 12 h. The calculation of water vapor permeability for the dressings was carried out using the following formula:
In this equation, “W” represents the weight loss of the water, “A” stands for the surface area of the bottle’s opening, and “T” corresponds to the incubation time.
Release profile
The release profile of 3HA from PVA/1% 3HA scaffolds was studied at 329.52 nm. Briefly, scaffolds with known weight were immersed in 15 mL of phosphate buffer solution (PBS) and kept at 37°C for 24 h. At regular time intervals, 0.5 mL of the release media was removed and the same amount of fresh PBS was added to the release media. The absorbance value of the acquired solution was read using a spectrophotometer.
Microbial penetration assay
The capacity of PVA/1% 3HA scaffolds and PVA-only scaffolds to resist bacterial penetration was examined following a method outlined in a previous reference. 20 Fibrous wound dressings were employed to seal the openings of glass containers containing 5 mL of Brain Heart Infusion (BHI) broth culture medium. These containers, covered with sterile gauze, and uncapped containers were designated as the negative control and positive control groups, respectively. Various groups were maintained at a temperature of 37°C for 7 days. On days 3 and 7, the infiltration of bacteria into the culture medium was assessed using a spectrophotometric technique at 600 nm.
Protein adsorption assay
The protein adsorption on PVA/1% 3HA scaffolds and PVA-only wound dressings was evaluated using batch contact method. 21
Hemolysis assay
The hemolytic activity of PVA/1% 3HA scaffolds and PVA-only wound dressings were studied using a processed human whole blood. Blood samples were obtained from a healthy volunteer and mixed with 3.8% sodium citrate and normal saline solution. Then, scaffolds were cut into 1 × 1 cm2 pieces and incubated with 200 µL of the blood samples for 1 h. Then, scaffolds were removed and the blood samples were centrifuged at 1500 rpm for 10 min. The extent of hemolysis was studied at 545 nm using a spectrophotometer. Normal saline solution and distilled water mixed with the blood samples and used as negative and positive control groups respectively.
Anti-oxidant assay
The capability of PVA/1% 3HA scaffolds and PVA-only scaffolds to neutralize DPPH free radicals was assessed using a 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay. Pure PVA scaffolds and PVA scaffolds loaded with 1% 3HA were cut into pieces measuring 2 × 2 cm2 and submerged in 3 mL of a 0.1 mM DPPH solution in ethanol. These samples were left at room temperature for 9 h. Ascorbic acid was utilized as the standard control, and DPPH-only samples served as the positive control group. The percentage of DPPH radical scavenging was determined using a spectrophotometric method at 517 nm and the following formula:
Animal studies
The animal experiments were approved by Laboratory Animal Welfare Ethics Committee of Yunnan University (Approval No. YNU20240851). To assess the wound healing effects of PVA/1% 3HA wound dressings and PVA-only wound dressings, a rat model of diabetic wounds was employed.
Male Wistar rats received an administration of 55 mg/kg Streptozotocin (STZ) in citrate buffer (pH 4.4, 0.1 M) and were continually monitored for their fasting blood sugar (FBS) levels. Rats with FBS levels exceeding 250 mg/dL were categorized as diabetic and included in the study. The study comprised three groups, each consisting of three rats: (1) PVA/1% 3HA group, (2) PVA group, and (3) a negative control group, where sterile gauze was used for diabetic wound treatment.
The animals were anesthetized via intraperitoneal injection of Ketamine (5%) and Xylazine (2%), administered at a dosage of 70 mg ketamine and 6 mg Xylazine per 1 kg of body weight. Following deep anesthesia, a 1.5 × 1.5 cm2 section of the animals’ skin was excised using a surgical blade. Wound dressings were applied to the wound site in each group and secured in place with adhesive bandages. On the 7th and 14th days, the wounds' macroscopic appearance was assessed by capturing photographs, and the reduction in wound size was calculated using the following formula:
At the conclusion of the 14th day post-surgery, the rats were euthanized, and their wound tissues were harvested for histopathological examinations. Following processing and sectioning, the tissue slides were stained with hematoxylin-eosin (H&E) and Masson’s trichrome for histopathological analysis.
Real-time PCR
After a 14-day period following the surgery, the expression levels of Glutathione peroxidase (GPx), b-FGF, and IL-1β genes in the tissue were analyzed. The wound samples were extracted from −80°C, and their RNA content was isolated using an RNA extraction kit from Shaanxi Pharmaceutical Holdings Group’s Paiang Medical Devices Co., Ltd. Subsequently, cDNA templates were generated in accordance with the manufacturer’s instructions using a Reverse Transcription Kit from Beijing Solarbio Science & Technology Co., Ltd. For gene amplification, 3 µL of the resulting cDNA templates were combined with 17 µL of a reaction mix containing Power SYBR Green PCR master mix from Beijing Solarbio Science & Technology Co., Ltd. The 2−ΔΔct relative quantification method was applied to determine the relative gene expression, with GAPDH serving as the reference gene.
Statistical analysis
The collected data were subjected to analysis using Graphpad Prism version 5, employing the one-way ANOVA test and Student’s t-test. Each experiment was conducted a minimum of three times, and statistical significance was considered when the p-value was less than 0.05.
Results
SEM imaging
Results of SEM imaging (Figure 1) showed that both 3HA-loaded and 3HA-free wound dressings were composed of randomly oriented fibers with smooth morphology. Fiber size measurement showed that PVA/1% 3HA scaffolds had 1408.18 ± 31.28 nm and PVA-only scaffolds demonstrated 1395.61 ± 23.09 nm of fiber diameter. Statistically no significant difference was observed between two scaffolds, p-value > 0.05.

SEM images of (a) PVA/1%3HA scaffolds and (b) PVA scaffolds, showing randomly oriented, smooth nanofibers with no significant difference in diameter.
Results of MTT assay
The outcomes of the cell viability assessment indicated that none of the scaffolds exhibited notable toxicity towards the L929 cells at any of the examined time intervals. Although on day 1, the discrepancies in optical density (OD) values did not exhibit statistical significance, by day 3 and 7, cells cultured on PVA/1% 3HA scaffolds displayed considerably higher absorbance values in comparison to the other groups, with a p-value below 0.05. As the MTT assay results indicated that the PVA/1% 3HA formulation provided the highest cell viability, we selected this formulation for the subsequent in vitro and in vivo assays.
Figure 2 displays a bar chart illustrating the cell viability of L929 cells grown on various types of scaffolds, including PVA scaffolds, PVA/0.5% 3HA scaffolds, PVA/1% 3HA scaffolds, and PVA/1.5% 3HA scaffolds. In the chart, “a” denotes a p-value less than 0.05 compared to PVA, “b” signifies a p-value less than 0.05 compared to PVA/0.5% 3HA, “c” indicates a p-value less than 0.05 compared to PVA/1.5% 3HA, and “d” represents a p-value less than 0.05 compared to the control group.

Bar chart illustrating the cell viability of L929 cells grown on various types of scaffolds, including PVA scaffolds, PVA/0.5% 3HA scaffolds, PVA/1% 3HA scaffolds, and PVA/1.5% 3HA scaffolds. In the chart, “a” denotes a p-value less than 0.05 compared to PVA, “b” signifies a p-value less than 0.05 compared to PVA/0.5% 3HA, “c” indicates a p-value less than 0.05 compared to PVA/1.5% 3HA, and “d” represents a p-value less than 0.05 compared to the control group.
Results of cell viability assay under oxidative stress
The findings from the MTS assay revealed that, when subjected to the same conditions, scaffolds containing 1% 3HA demonstrated notably superior protection against oxidative stress in comparison to the other formulations, with a p-value less than 0.05. Moreover, dressings loaded with 1.5% and 0.5% 3HA exhibited significantly higher relative cell viability when contrasted with the PVA group, also with a p-value below 0.05.
Figure 3 displays a bar chart comparing the relative cell viability of L929 cells following their exposure to extracts from PVA scaffolds, PVA/0.5% 3HA scaffolds, PVA/1% 3HA scaffolds, and PVA/1.5% 3HA scaffolds. In the chart, “a” indicates a p-value less than 0.05 in relation to PVA, “b” represents a p-value less than 0.05 compared to PVA/0.5% 3HA, “c” signifies a p-value less than 0.05 relative to PVA/1.5% 3HA, and “d” denotes a p-value less than 0.05 in comparison to the control group.

Bar chart comparing the relative cell viability of L929 cells following their exposure to extracts from PVA scaffolds, PVA/0.5% 3HA scaffolds, PVA/1% 3HA scaffolds, and PVA/1.5% 3HA scaffolds. In the chart, “a” indicates a p-value less than 0.05 in relation to PVA, “b” represents a p-value less than 0.05 compared to PVA/0.5% 3HA, “c” signifies a p-value less than 0.05 relative to PVA/1.5% 3HA, and “d” denotes a p-value less than 0.05 in comparison to the control group.
Water uptake capacity measurement
The findings from the assessment of water absorption capacity revealed that PVA/1% 3HA scaffolds and PVA-only scaffolds exhibited water uptake capacities of 24.37 ± 2.15% and 26.43 ± 3.08%, respectively. Importantly, there was no statistically significant difference observed between the two groups, with a p-value exceeding 0.05.
Tensile strength studies
Results of tensile strength measurement showed that incorporation of 1% 3HA into PVA’s matrix significantly improved its tensile strength from 1.63 ± 0.30 MPa to 2.68 ± 0.27 MPa, p-value < 0.05.
Contact angle measurement
The outcomes of the contact angle measurement indicated that PVA/1% 3HA scaffolds and PVA-only scaffolds displayed contact angles of 57.92 ± 8.81° and 62.43 ± 3.19°, respectively. Importantly, there was no significant difference observed between the two groups, with a p-value greater than 0.05.
Water vapor permeability studies
Results of water vapor permeability measurement showed that PVA/1% 3HA scaffolds and PVA scaffolds demonstrated 146.96 ± 12.54 mg/cm2/h and 138.42 ± 9.46 mg/cm2/h of water vapor permeation respectively. Statistically no significant difference was observed between two groups, p-value > 0.05.
Release profile assay
Cumulative drug release assay (Figure 4) showed that PVA-based wound dressings could release almost all their drug cargo within 96 h of scaffolds’ immersion in PBS solution. This time stem coincided with completed disintegration of the electrospun scaffolds. At 24 h, PVA/1% 3HA scaffolds could release almost 50.87 ± 4.14 of their drug cargo.

Cumulative release profile of 3HA from PVA/1% 3HA scaffolds.
Microbial penetration test
In the bacterial penetration test (Figure 5), it was evident that the positive control group exhibited notably higher absorbance values in comparison to the other groups at both 3 and 7 days, with a p-value below 0.05. However, the drug-loaded and drug-free wound dressings did not demonstrate a significant difference in preventing bacterial infiltration when compared to the negative control group, as the p-value exceeded 0.05.
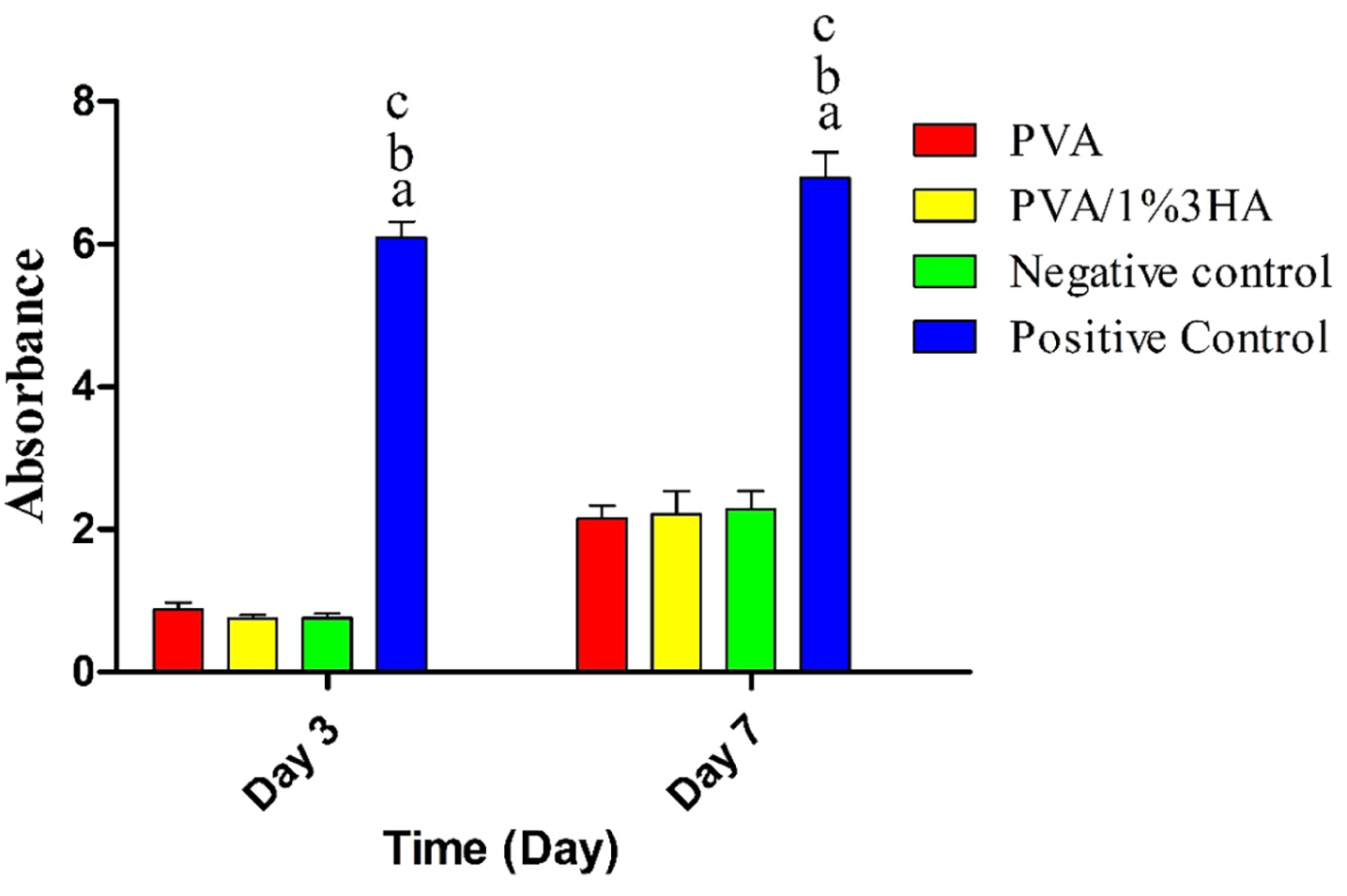
Bacterial resistance characteristics of PVA/1% 3HA scaffolds and PVA scaffolds in comparison to sterile gauze (negative control) and open bottles (positive control) on days 3 and 7. In this figure, “a” indicates a p-value less than 0.05 in relation to PVA, “b” represents a p-value less than 0.05 in comparison to PVA/1% 3HA, and “c” denotes a p-value less than 0.05 relative to the negative control group.
Figure 5 illustrates the bacterial resistance characteristics of PVA/1% 3HA scaffolds and PVA scaffolds in comparison to sterile gauze (negative control) and open bottles (positive control) on days 3 and 7. In this figure, “a” indicates a p-value less than 0.05 in relation to PVA, “b” represents a p-value less than 0.05 in comparison to PVA/1% 3HA, and “c” denotes a p-value less than 0.05 relative to the negative control group.
Protein adsorption assay
The findings from the protein adsorption test indicated that PVA/1% 3HA scaffolds and PVA wound dressings were able to adsorb approximately 4.23 ± 0.64 mg and 4.16 ± 0.28 mg of BSA proteins onto their surfaces, respectively. Importantly, the observed differences did not achieve statistical significance, as the p-value exceeded 0.05.
Hemostatic activity measurement
The outcomes of the hemostatic activity assessment, as depicted in Figure 6, revealed that the positive control group displayed notably higher absorbance values when compared to the other groups, with a p-value less than 0.05. However, there was no statistically significant difference observed between the electrospun scaffolds and the negative control group, as the p-value exceeded 0.05.

Bar chart comparing the hemostatic activity of PVA/1% 3HA scaffolds and PVA scaffolds in contrast to normal saline (negative control) and distilled water (positive control). The values provided are the means with their corresponding standard deviations. In this figure, “a” signifies a p-value less than 0.05 when compared to PVA, “b” represents a p-value less than 0.05 relative to PVA/1% 3HA, and “c” denotes a p-value less than 0.05 concerning the negative control group.
Figure 6 presents a bar chart comparing the hemostatic activity of PVA/1% 3HA scaffolds and PVA scaffolds in contrast to normal saline (negative control) and distilled water (positive control). The values provided are the means with their corresponding standard deviations. In this figure, “a” signifies a p-value less than 0.05 when compared to PVA, “b” represents a p-value less than 0.05 relative to PVA/1% 3HA, and “c” denotes a p-value less than 0.05 concerning the negative control group.
Results of antioxidant assay
The assessment of radical scavenging activity, as depicted in Figure 7, demonstrated that ascorbic acid exhibited the highest potential compared to the other groups across various concentrations. The IC50 (The term “IC50” represents the lowest concentration needed to neutralize 50% of DPPH free radicals) value for this group was approximately 77.06 µg/mL. The free 3HA group displayed an IC50 value of approximately 189.37 µg/mL. Notably, pure PVA scaffolds exhibited minimal radical scavenging activity, but the inclusion of 3HA into the scaffold's structure substantially enhanced its antioxidant properties.

Radical scavenging activity of free 3HA, PVA/1% 3HA scaffolds, and PVA scaffolds in comparison to the standard ascorbic acid control.
Figure 7 illustrates the radical scavenging activity of free 3HA, PVA/1% 3HA scaffolds, and PVA scaffolds in comparison to the standard ascorbic acid control.
Wound healing assay
The results regarding wound size reduction indicated that wounds treated with PVA/1% 3HA wound dressings exhibited a significantly higher rate of wound closure compared to PVA wound dressings and the negative control group on both the 7th and 14th days after the initial wounding, as presented in Figure 8. The statistical analysis showed a p-value less than 0.05.

Bar chart comparing the percentage of wound contraction in wounds treated with PVA/1% 3HA scaffolds, PVA scaffolds, and sterile gauze. In this figure, “a” indicates a p-value less than 0.05 compared to the negative control group, and “b” signifies a p-value less than 0.05 relative to the PVA group.
Specifically, wounds treated with PVA/1% 3HA wound dressings displayed a wound size reduction of 67.59 ± 7.68% on the 7th day and 92.93 ± 6.32% on the 14th day post-surgery. In contrast, the PVA-only group showed a wound closure of 50.26 ± 3.85% and 75.94 ± 6.19% at the end of the first- and second-week following surgery, respectively.
Histopathological examinations presented in Figure 9 revealed that the negative control group exhibited the formation of immature epithelial tissue with numerous inflammatory cells. In this group, collagen fibers were also immature and disintegrated. Conversely, rats treated with PVA/1% 3HA scaffolds displayed a robust formation of epithelial tissue with a lower number of inflammatory cells. Skin appendages were formed, and the tissue was partially able to restore its normal structure. Moreover, more aligned collagen fibers were deposited in this group. The PVA-only group exhibited some degree of collagen deposition and epithelial tissue formation, though the extent of tissue repair was significantly greater in the PVA/1% 3HA group.

H&E and Masson’s Trichrome staining images of wounds treated with different wound dressings. Arrows show infiltration of inflammatory cells and asterisks show epithelium tissue.
Histomorphometric analysis in Figure 10(a) and (b) indicated that PVA/1% 3HA scaffolds demonstrated significantly greater epithelial thickness and a higher percentage of collagen deposition compared to PVA-only wound dressings and the negative control group, with a p-value less than 0.05. The drug-loaded wound dressings showed an epithelial thickness of 54.16 ± 6.47 µm and a percentage of wound closure of 57.91 ± 8.20%. In contrast, drug-free dressings displayed an epithelial thickness of 29.25 ± 7.73 µm and a percentage of wound closure of 38.80 ± 8.25%.

Histomorphometric analysis in H&E and Masson’s trichrome staining images of wound tissues on day 14th, a and b show p-value < 0.05 relative to PVA and negative control groups, respectively.
Figure 8 illustrates a bar chart comparing the percentage of wound contraction in wounds treated with PVA/1% 3HA scaffolds, PVA scaffolds, and sterile gauze. In this figure, “a” indicates a p-value less than 0.05 compared to the negative control group, and “b” signifies a p-value less than 0.05 relative to the PVA group.
Real time PCR assay
The results obtained from the gene expression analysis, as depicted in Figure 11, revealed that wounds treated with PVA/1% 3HA wound dressings were capable of significantly decreasing the tissue expression levels of GPx and IL-1β genes in comparison to PVA-only scaffolds and the negative control group, with a p-value less than 0.05. Additionally, the analysis of b-FGF gene expression showed that the tissue expression level of this gene in the PVA/1% 3HA group was significantly higher when compared to the other groups, also with a p-value less than 0.05.

Bar charts comparing the tissue expression levels of (a) GPx, (b) IL-1β, and (c) b-FGF genes at the 14th day post-wounding. In this figure, “a” denotes a p-value less than 0.05 in comparison to the PVA group, and “b” signifies a p-value less than 0.05 relative to the control group.
Figure 10 presents bar charts that compare the tissue expression levels of (a) GPx, (b) IL-1β, and (c) b-FGF genes at the 14th day post-wounding. In this figure, “a” denotes a p-value less than 0.05 in comparison to the PVA group, and “b” signifies a p-value less than 0.05 relative to the control group.
Discussion
Electrospun wound dressings hold great potential for the development of next generation of smart wound dressing materials. Due to ECM-like structure and versatile properties, they can be easily modified with various functionalization methods.22,23 In the current study, an exogenous antioxidant was incorporated into the matrix of PVA scaffolds in order to develop a drug-delivering wound dressing. The produced scaffolds were shown to be highly similar to ECM architecture. The beneficial roles of electrospun wound dressings in wound healing applications have been shown in previous studies. These scaffolds are known to trigger various cellular functions and promote the proliferation phase of wound healing. 24 Results of cell viability assays under normal and high oxidative stress conditions showed that the scaffolds loaded with 1% 3HA had higher viable cells. The dose dependent effect of 3HA on cell viability could be due to the fact that under a critical concentration, 3HA does not possess protective effects on L929 cells; while, above this concentration 3HA may have demonstrated toxic effect on skin cells. Based on these assays, we were convinced that PVA/1% 3HA scaffolds were optimal for in vivo studies and therefore was chosen for the treatment of diabetic wounds. Water uptake potential indicates the ability of the dressings to absorb wound exudates. Diabetic wounds are often associated with high wound exudates which are abundant with various pro-inflammatory cytokines. These factors exacerbate the inflammatory condition and damage the skin tissues. The water absorption capacity findings of our manufactured dressings were in line with the outcomes of prior research. 25 Therefore; in this study, we could produce scaffolds with ideal mechanical strength. The surface hydrophilicity of the wound dressings has a profound effect on their healing effects. Generally, cells do not favor hydrophobic surfaces and hydrophilic surface possess higher potential to absorb wound exudates. 26 Results of contact angle measurement showed that both drug-loaded and drug-free dressings was within the range of hydrophilic materials (contact angle below 90°). 25 Water vapor permeability of wound dressings shows their ability to facilitate gas exchange. This property is essential for diabetic wound healing since it prevents establishment of anaerobic conditions for bacterial growth and also reduce the risk of wound maceration. 27 However, we expect that upon absorption of wound exudate, PVA-based wound dressings would undergo significant swelling due to the hydrophilic nature of polyvinyl alcohol. This swelling can lead to a reduction in pore size and an increase in the density of the hydrogel network, thereby limiting gaseous exchange, particularly the diffusion of oxygen and carbon dioxide essential for effective wound healing. Impaired gas exchange can result in a hypoxic wound environment, potentially delaying tissue regeneration and promoting bacterial growth. To mitigate this limitation, strategies such as incorporating porogens or sacrificial particles to create interconnected micropores, blending PVA with more hydrophobic polymers, or crosslinking with agents that reduce excessive swelling while maintaining permeability could be employed.28 –30 These approaches can help preserve sufficient porosity and gas permeability even after fluid uptake. Drug release assay showed that the PVA/1% 3HA wound dressings release their drug cargo within 96 h of incubation in PBS solution which coincided with complete disintegration of the electrospun dressings. The mechanism of drug release from the PVA’s matrix may be the combination of diffusion, PVA’s swelling, and gradual degradation of PVA. However, in clinical applications, it seems that swelling and diffusion are the key role players, because wound dressings are replaced each day and in short time period the wound dressing would not undergo a significant degradation. 31 The bacterial penetration assay showed that there was not a significant difference between electrospun scaffolds and negative control group. Indeed, not PVA nor 3HA possess antibacterial properties and cannot efficiently prevent bacterial invasion. This drawback may be compensated by blending of PVA with an antibacterial polymer such as chitosan or by incorporation of an antibiotic into its matrix. After applying the wound dressing on the skin wound, its surface gets hydrated and then wound fluid’s proteins gradually replace this water layer. Our observations showed that there was no significant disparity in protein adsorption between PVA and PVA/1% 3HA scaffolds. This outcome can be attributed to their comparable contact angles. It appears that the interaction of proteins with the scaffolds is primarily influenced by their surface chemistry, with hydrophobic surfaces being more conducive to protein adsorption, as the energy required for replacing the water layer with proteins is lower. 32 Hemostasis is the first phase of wound healing which is initiated via the interaction of circulating platelets with ECM components. Lack of hemolytic activity of PVA and PVA/1% 3HA scaffolds may be beneficial for wound healing since their removal would not harm wound tissue. The radical scavenging activity of a substance is governed by the presence of carboxyl, phenolic, and hydroxyl groups in its structure.33,34 In this study, we could show that the incorporation of 3HA into PVA’s matrix significantly enhanced its antioxidant properties. This result is accordance with the results of cell viability under oxidative stress. In fact, 3HA possesses an antioxidant property and can quench free radicals. Leipnitz et al. 35 showed that 3HA reduced lipid peroxidation and significantly reduced the release of peroxyl radicals. The wound healing investigation demonstrated that PVA/1% 3HA scaffolds exhibited the highest rate of wound healing. This heightened healing activity can be explained by several factors. As indicated by the results of the cytoprotection assay and DPPH test, the findings from the gene expression analysis indicated that wound dressings delivering 3HA led to a notable reduction in the tissue expression level of the GPx gene, suggesting that 3HA significantly mitigated oxidative stress in diabetic wounds. Elevated levels of reactive oxygen species (ROS) in diabetic wounds can inflict damage on the tissues and hinder the progression of wound healing beyond a stagnant inflammation phase.36,37 3HA’s ability to quench ROS plays a crucial role in alleviating oxidative stress. Additionally, the immunomodulatory properties of 3HA may have also contributed to the process of wound healing. Krause et al. 16 demonstrated the anti-inflammatory activity of 3HA in a brain inflammation model by upregulating the expression of heme oxygenase-1, indicating its potential role in reducing inflammation. Pérez-González et al. 38 examined the dual antioxidant/pro-oxidant behavior of 3-hydroxyanthranilic acid (3-OHAA) using density functional theory. In metal-free environments, 3-OHAA acted as a potent antioxidant, efficiently scavenging peroxyl radicals—outperforming Trolox in both lipid and aqueous phases. However, in the presence of metal ions at physiological pH, 3-OHAA showed pro-oxidant behavior by reducing Cu(II) to Cu(I), facilitating hydroxyl radical (•OH) formation via the Fenton reaction. Metal ion presence and pH were key factors influencing this dual activity. We could also show that PVA/1% 3HA dressings decreased IL-1β expression, suggesting that 3HA-delivering wound dressings have alleviated inflammation responses. IL-1β is a pro-inflammatory cytokine and is involved in various inflammation-induced tissue damages. Our study for b-FGF gene expression showed that the tissue expression profile of this gene was significantly increased via 3HA-delivering wound dressings. Various studies have shown beneficial role of b-FGF-delivering wound dressings on skin wound healing. This growth factor can accelerate wound healing via promoting different phase of wound healing such as proliferation phase and tissue remodeling. 39
Conclusion
The primary objective of this study was to develop an electrospun wound dressing for the localized delivery of 3HA to diabetic wounds. Different formulations of PVA and 3HA were prepared and evaluated. The selection of the optimal formulation was based on cell viability assays, with the results indicating that PVA/1% 3HA scaffolds exhibited significantly higher cell viability and cytoprotection. Subsequently, the wound healing potential of this particular formulation was assessed using a rat model of diabetic wounds. The results of the wound healing study revealed that PVA/1% 3HA wound dressings exhibited superior wound healing activity in comparison to both the PVA-only group and the sterile gauze group. Furthermore, gene expression analysis demonstrated that PVA/1%3HA wound dressings reduced the tissue expression levels of GPx and IL-1β genes while simultaneously enhancing the expression profile of the b-FGF gene. Future studies should focus on scaling up the production process, conducting long-term in vivo evaluations, and exploring the molecular mechanisms underlying the dual antioxidant/pro-oxidant role of 3HA in different wound microenvironments.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by Yunnan Province Endocrinology Clinical Key Specialty Yunnan University Affiliated Hospital Internal Open Project: ZKF2024084 and Kunming Medical University Basic Research Joint Special Project (202501AY070001-213)




