Abstract
This study aimed to graft a commercial amino-terminated hyperbranched polymer (AHP), specifically a low molecular weight branched polyethylenimine, onto cotton fibres through partial oxidation of cellulose with periodate. The primary objective was to assess the feasibility of achieving salt-free or low-salt dyeing of cotton with reactive dyes. The research investigated the washing, rubbing, and light fastness of modified cotton fabrics dyed in the absence or with a low-salt promoter, comparing the results with conventional dyeing methods. The findings indicated that grafting AHP through partial oxidation significantly reduced the required salt levels for dyeing with reactive dyes while maintaining colour intensity and comparable washing fastness properties.
Introduction
Cotton is a cellulosic fibre that has a current market shared of approximately 25% of global fibre production, being the second most important fibre in terms of volume surpassed only by polyester. 1 So, the consumption of dyes for the coloration of textile products made of cellulose is also proportional to that volume, requiring thousands of metric tons of dye worldwide.
Among the different types of dyes that can be used for dyeing cotton, that is, direct, vat, sulphur, reactive, etc., reactive dyes are the ones commonly used because they produce a full range of bright fashion colours with a high degree of wash fastness. 2 However, the affinity of reactive dyes for cotton is generally low due to electrostatic repulsion between the negatively charged reactive dyes and the cellulose fibres, which have a negative zeta potential, 3 and this repulsion is even higher as reactive dyeing is carried out under basic conditions. In other to overcome this issue and to reduce repulsion by a reduction of the zeta potential, inorganic salts, that is, sodium chloride or sodium sulphate, are the most commonly applied dyeing promoters but their use results in severe salt pollution of water streams and associated economic expenses.1,2,4
Traditionally, large amounts of salt (30–120 g/l) are required to overcome the repulsion between the reactive dye and the cotton fibre, increasing significantly the salinity of the wastewater stream that leaves the dyeing process and causing water pollution. Consequently, most research focus on introducing salt-free/low-salt dyeing technology for reducing the environmental impact of the dyeing process by chemical modification of cotton fibre prior to dyeing.5–7 Most of the methods that propose the chemical modification of cotton are based on the introduction of cationic groups like ammonium into cotton for promoting ionic interactions. 8 In this sense, chemical compounds such as glycidyl trimethylammonium chloride known as Glytac, chlorotriazine type quaternary compounds, choline chloride, polyamide epichlorohydrine, N-methylolacrylamide and polyepichlorohydrin dimethylamine have been proposed, among others, for the pretreatment of cotton fibres. 8 Recently, Acharya et al. 9 demonstrated that the cationization of cotton using (3-chloro-2-hydroxylpropyl) trimethyl-ammonium chloride (CHPTAC) enhanced dye uptake properties due to the introduction of cationic sites and resulted in superior dyeing behaviour without the addition of salt.
Among the different progresses made for low salt or salt-free dyeing of cellulose, amino-terminated hyperbranched polymers (AHP) have been developed showing potential application in several fields such as coatings, adhesives, paints, packaging, water treatment10,11 and they have also been postulated as a potential auxiliary for dyeing textiles. 12 Zhang et al. reported that an amino-terminated hyperbranched polymer can be used as auxiliary to pre-treat cotton obtaining a dyeing with enhanced colour strength and good washing and rubbing fastness, when dyeing was carried out in the absence of salt. 13 Nevertheless, a crosslinking agent such as acid citric was used to promote interaction of AHP with the fibre and enhance the dyeability. In a later research, it was observed similar results when applying the AHP by a pad-dry-cure method. 14 Similarly, Wang et al. reported that the pretreatment of ramie fabric with AHP allow to obtain higher dye uptake with a washing and rubbing fastness comparable to that of the untreated fabrics. 15
Alternatively, a new route based on grafting AHP by previous partial oxidation with periodate of cotton fabrics can also assist in the reduction of the above-mentioned electrostatic repulsion. 16 Grafting will consist in two steps: (i) selective modification of cellulose by reaction with periodate which selectively leads to the breakage of C2–C3 bond in the glucopyranoside ring and the adjacent hydroxyl groups at these positions are converted to aldehydes producing dialdehyde cellulose, 17 (ii) coupling reaction of the aldehyde groups of oxidated cellulose with the amino groups of AHP. This approach using simultaneously the application of AHP onto oxidized cellulose was followed in this work with the aim of obtaining a modified cotton that allow salt-free/low-salt dyeing methodology.
The purpose of this work was to graft a commercial AHP, that is, a low molecular weight branched polyethylenimine, by partial oxidation of cellulose, evaluating the viability to achieve salt-free or low-salt dyeing of cotton fibres with reactive dyes. The washing, rubbing and light fastness of modified cotton fabrics dyed either in absence or with low salt promoter were determined and compared with the ones obtained by conventional dyeing. Taking into account that partial oxidation with periodate it is a crucial step in the grafting process, which affects directly to the mechanical properties of the cotton and to the yield of the grafting reaction, the effect of the time, temperature and concentration of periodate on those parameters was also evaluated and optimized.
Experimental
Materials
Knitted, scoured and bleached cotton fabric weighing 123 g/m2 and with 14 wales/cm and 18 courses/cm was kindly supplied by Tipsa Hilaturas Llandet S.A (Spain). To remove possible impurities, the fabric was washed in a Wascator machine FOM 71 Special (Electrolux, Sweden) using 4 g/l of ECE surfactant at 40°C during 45 min. Subsequently, the samples were rinsed with cold distillated water and air-dried at room temperature.
Commercial amino-terminated polyethyleneimine hyperbranched polymer, Lupasol® PR 8515 (average molar mass of 2000 g/mol), was obtained from BASF (Barcelona, Spain).
Reactive dye C. I. Reactive Blue 52 commercially named Blue Drimaren X-3LR Sandoz was supplied by Sandoz Ltd. (Basel, Switzerland). The chemical class of the reactive dye used is a disazo formazan metal complex having one pyrimidine ring.
Sodium periodate, sodium carbonate, sodium sulphate, sodium hydroxide of technical grade were purchased from Panreac (Barcelona, Spain). Hostapal® DTC from Clariant (Barcelona, Spain) were used as a soap.
Oxidation of cotton with periodate
The partial oxidation of cotton fabrics was analysed using an experimental design: 1 g of cotton fabric was immersed in 30 ml of sodium periodate solution of a certain concentration (1, 2 or 4 g/l), during a certain time (10, 30, and 60 min) at a certain temperature (30, 40, and 50°C). The solution was stirred in a Linitest K1-25 machine (Original Hanau, Germany) at desired temperature and time in absence of light to prevent the photo-induced decomposition of sodium periodate. After completion of the oxidation, the cotton fiber was washed thoroughly with ice-cold distilled water several times to remove the oxidant. Subsequently (without drying the oxidated samples), the oxidized cotton samples were treated using exhaustion bath method with amino-terminated hyperbranched polymer (Lupasol PR 8515®) as described in section “Grafting of AHP onto partially oxidated cotton.” The mechanical resistance of fabrics after treatment was measured as describe in section “Tensile test.” In addition, the aldehyde content was analysed as described in section “Aldehyde content on partially oxidated cotton.” Optimal conditions were selected for subsequent grafting of APH based on the criteria of minimizing fibre weakness while maximizing aldehyde content.
A design of experiments was carried out in order to analyse the effect and interactions between the selected parameters that control the partial oxidation of cotton and to study their influence on the maximum breaking force and aldehyde content. The maximum breaking force (represented by Y1) and the aldehyde content (represented by Y2) may be influenced by the following three parameters: time (represented by X1), temperature (represented by X2) and concentration of sodium periodate (represented by X3). Three different levels for each parameter were studied (Table 1), so 27 experiments were randomly carried out.
Values of each parameter and corresponding codes.

Data analysis was performed using a linear model, in which the response (Y1 or Y2) is given by a linear combination of the three parameters:

where bo, bi, bii and bijk are the coefficients of the regression equation; i and j are the integers that represent the reaction variables with i < j ; Y is the response or dependent variable (maximum force (Y1) or aldehyde content (Y2)); and X are the reaction variables (NaIO4 concentration, temperature and time). To estimate the coefficients of the model defined by equation (1), the Student t-test at a confidence level of 95%, was used. The statistical regression analysis was performed with Minitab® 18.1 software.
Grafting of AHP onto partially oxidated cotton
Partial oxidated cotton samples resulted from their treatment under the previously determined optimal conditions were immediately treated with AHP (Lupasol PR 8515®). The oxidized cotton was immersed in AHP aqueous solution of 5 g/l at a temperature of 70°C for 30 minutes with a liquor ratio of 20:1. After treatment, the samples were rinsed with cold tap water and air-dried at room temperature for a least 24 h to produce hyperbranched grafted cotton fibre.
Dyeing cotton
The all-in method was used for dyeing the samples in a Rotawash machine M228B (SDLATLAS, USA). So, dye, salt (sodium sulphate), alkali (sodium carbonate) and cotton fabric was added to the bath at the beginning of the process and the temperature-time manufacturer’s recipe was followed. The dyeing of treated and untreated cotton samples was initiated at 60°C and kept at this temperature for 15 minutes. Then, the temperature was raised to 90°C and left for 30 minutes. Fixation was conducted subsequently for 45 minutes at 90°C. A dye concentration of 2.4% o.w.f., 15 g/l of sodium carbonate, 60 g/l of sodium sulphate at a liquor ratio 20:1 were used for the conventional dyeing. In addition, dyeing with 30, 15, and 0 g/l of sodium sulphate was carried out to study the influence of the salt. After dyeing, the cotton fabric was removed from the dyebath and rinsed thoroughly in hot water (around 60°C) and soaped in a soap solution of 0.5 g/l of Hostapal® DTC for 20 minutes at 95°C. Then, rinsed thoroughly in warm tap water and air-dried.
Characterization
Tensile test
The mechanical resistance from courses-wise direction of cotton fabrics after treatment with periodate was measured using a Uster Tensokid device (Uster, Switzerland) following the ISO 13934-1standard. 18 Specimens had a length of 15 cm and a wide of 5 cm. Before carrying out the test, the samples were conditioned during 24 h at 20°C and 65% of relative humidity. The tensile test was carried out applying a preload of 0.5 kN and subsequently setting the speed at 100 mm/min. Maximum breaking force (in N) and elongation at maximum breaking force (in %) were determined from force-elongation curves. Three replicate specimens were analysed for each sample and the average and standard deviation were calculated.
Aldehyde content on partially oxidated cotton
The aldehyde content was analysed as described elsewhere. 19 Briefly, 100 mg of oxidated fabric was mixed with 25 ml of an aqueous solution 0.25 M of hydroxylamine hydrochloride during 2 h at room temperature. The initial pH of the hydroxylamine hydrochloride solution was measured initially. After the 2 h of reaction, the pH of the solution will decrease due to the delivery of HCl and this solution was titrated with an aqueous solution of 0.01 M of NaOH until the initial measured pH. The aldehyde content (in %) was calculated according to the following equation (2):

where C represent the concentration of NaOH solution (in mol/l), V is the volume of NaOH consumed in the titration, MAGU represents the molecular weight of the oxidated anhidroglucose unit (160 g/mol) and m represent the dry sample weight of cotton.
ATR-FTIR analysis
Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy (ATR-FTIR) was used in order to perform a chemical characterization of the partially oxidated and grafted cotton samples. Analysis were carried out placing the sample on top of the germanium crystal in the attenuated total reflection module of a Bruker Tensor 27 FT-IR spectrometer (ATR-FTIR, Bruker, Ettlingen, Germany), with a resolution of 4 cm−1, measuring the absorption between 600 and 4000 cm−1 and generating 32 scans per spectrum.
Elemental analysis
CHNS elemental analyser Flash EA 2000 CHNS (Thermo Fisher Scientific, USA) was used for the determination of carbon, hydrogen, nitrogen and sulphur of the untreated and treated cotton samples. The sample is weighed inside a capsule of Sn on a microbalance with a resolution of 1 µg. Once carefully sealed, the capsule is placed in the instrument’s sampler, which introduces it into the reaction tube at 1200°C in an O2 atmosphere. Under these conditions, quantitative combustion of the sample occurs producing CO2, H2O, N2, and SO2. Finally, the He stream carries the gas mixture to a gas chromatograph where they are separated, recorded, and quantified. Measurements were made in duplicate and average and relative standard deviation were specified.
Colour measurements
Colour measurements of the dyed samples were performed using a Minolta CM-3600d spectrophotometer (Konica Minolta, Japan) under D65 illuminant at 10° observer. K/S values were calculated from the diffuse reflectance R of the samples at the wavelength of maximum absorption (ʎmax = 630 nm) according to equation (3):

In addition, colour difference CIE1976 was determined as follows:

where L*, a* and b* are the CIELab colour coordinates calculated from triestimulus values X, Y and Z according to ISO 105-J01 standard.
Also, colour difference CMC (2:1) according to ISO 105-J03, 20 was calculated from L*, a* and b* colour coordinates to assess colour matching acceptability.
Measurements were made in triplicate.
Colour fastness
Washing fastness tests were performed according to ISO 105-C06 21 selecting the C1S method (T = 60°C, and 30 min) and using a Rotawash machine M228B (SDLATLAS, USA). Rubbing fastness tests were performed according to ISO 105-X12 22 using Crockmeter. Artificial light fastness tests were performed according to ISO 105 B02 23 using a Xenotest 150 machine (Original Hanau, Germany).
Results
Cotton fabrics were partially oxidated with sodium periodate achieving a chemical modification of cellulose. Particularly, sodium periodate oxidation is highly selective resulting in the cleavage of C2–C3 groups of anhidroglucose rings and the adjacent hydroxyls groups at this positions are converted to aldehyde groups. As a consequence, the oxidation causes a degradation of the cellulose that manifest itself in a loss of the mechanical properties. The decrease of mechanical properties and the change in the aldehyde content will depend of the conditions of the treatment, that is, time, temperature and concentration of periodate. Consequently, the effect of the conditions of periodate treatment on both the aldehyde content and mechanical properties were optimized by multifactorial analysis according to the proposed design of experiments.
On the one hand, regression analysis of the data shows that maximum breaking force (Y1 in N) is influenced by time (X1 in min), temperature (X2 in °C), and concentration of sodium periodate (X3 in g/l). Interaction between the aforementioned variables were not found significant so the regression equation with a R 2 = 74,5% is as follow:

By other hand, regression analysis for the oxidation degree or aldehyde content (Y2 in %) shows that this variable is influenced by time, temperature and concentration of sodium periodate and also by the interaction of these parameters. The model resulted in a R 2 = 98% and the equation is as follow:

The general trend obtained when plotting maximum breaking force versus aldehyde content of all the treatments for different concentration of sodium periodate, temperature and time is shown in Figure 1. From the results, it was shown that treatments producing a high amount of aldehyde groups (~1%–1.5%) affect significantly the strength of the cotton fabric decreasing the maximum force from 180 N to values of approximately 100 N (44% loss). In addition, it was observed that treatments in mild conditions can introduce a minimum quantity of aldehyde groups (~0.5% of aldehyde groups) resulting in a loss of maximum force varying from 180 N–125 N (30% loss). In this case, accepting a loss of strength of 30%, optimal conditions were selected to minimize fibre weakness while maximizing aldehyde content resulting in a treatment with a 2 g/l of sodium periodate at 40°C for 60 min which are close to the optimal conditions proposed by Zhang et al. who pointed out that short time (30–60 min) yields adequate aldehyde content for the oxidized cotton fiber to ensure sufficient amount of the AHP grafted to the oxidized cotton fiber for achieving high K/S value when dyed with reactive dyes in salt-free dyeing. 14 Minimum quantity of aldehyde groups is necessary for the subsequent step of grafting AHP onto oxidised cotton despite significantly reducing the mechanical properties of cotton.

Relationship between the warp-wise maximum breaking force (N) and aldehyde content (%) for all the treatments carried out at different concentration sodium periodate (1, 2 or 4 g/l), temperature (30, 40, and 50°C) and time (10, 30, and 60 min).
Amino terminated hyperbranched polymer (AHP) was grafted onto partially oxidised cotton fabrics. The presence of grafted AHP in treated cotton sample was followed by ATR-FTIR and elemental analysis (Table 2).
CHNS elemental analysis of the untreated and treated cotton samples.

rsd: relative standard deviation (%).
The ATR-FTIR spectra (not shown) did not reveal significant differences in the absorption bands between untreated and treated cotton samples. The spectra of the untreated and treated cotton samples indicated the primary absorption bands of cotton, with no evidence of aldehyde or amino groups in the oxidized and AHP-grafted samples, respectively. A broad peak at 3300 cm−1 corresponding to O–H stretching and a broad peak at 3000–2800 cm−1 region for C–H stretching were observed. Also, peaks around 1420, 1310, 1100 and 1030 cm−1 characteristic of cotton were observed. 24 Therefore, owing to the predominant presence of cotton, the ATR-FTIR technique was not sufficiently sensitive to detect the characteristic chemical groups (i.e., amino groups) corresponding to the AHP in treated samples.
Given the limited sensitivity of the ATR-FTIR technique to detect characteristic chemical groups in treated samples due to the predominant presence of cotton, additional analysis was pursued. To ascertain the presence of amino-terminated hyperbranched polymer in the sample, elemental analysis of the cotton samples was employed and the results are shown in Table 2. This complementary approach provided a more thorough examination of the chemical composition, particularly focusing on the verification of amino groups that may not have been effectively captured by ATR-FTIR.
The results presented in Table 2 illustrate the presence of amino-terminated hyperbranched polymer (AHP) in the cotton sample treated with AHP. When compared to untreated and partially oxidized cotton samples, the percentage of nitrogen is significant, reaching a value of 0.59%.
Data regarding colour strength (K/S) values and CIELab colour coordinates for untreated and treated cotton fabrics dyed with different amounts of salt are plotted in Figure 2 and shown in Table 3, respectively.

Colour strength (K/S) values at λmax = 630 nm for untreated, oxidated and AHP treated cotton fabrics dyed with different amounts of salt.
CIELab colour coordinates for untreated, oxidated and AHP treated cotton fabrics dyed with different amounts of salt.

Firstly, colour strength (K/S) achieved when using AHP on oxidized cotton was comparable to the untreated cotton dyed by standard procedure using 60 g/l of salt. Using AHP with 15 or 30 g/l provided similar K/S value that for untreated cotton dyed using 60 g/l of salt. The sample treated with AHP and dyed in absence of salt doubled the colour intensity obtained for untreated cotton when dyeing with zero salt. However, oxidized samples without AHP behaved similarly to untreated cotton regardless the amount of salt used. Thus, it can be concluded that the most significant effect on colour strength was attributed solely to the presence of AHP, allowing for comparable colour strength to conventional dyeing while reducing salt usage by up to 75%.
Differences in colour coordinates (ΔL*, Δa*, Δb*) referred to untreated cotton fabric dyed using 60 g/l of salt has been calculated and plotted in Figures 3 to 5.

ΔL* for untreated, oxidated and AHP treated cotton fabrics dyed with different amounts of salt (60, 30, 15, and 0 g/l). Sample used as reference is the untreated dyed with 60 g/l of salt.

Δa* for untreated, oxidated and AHP treated cotton fabrics dyed with different amounts of salt (60, 30, 15, and 0 g/l). Sample used as reference is the untreated dyed with 60 g/l of salt.
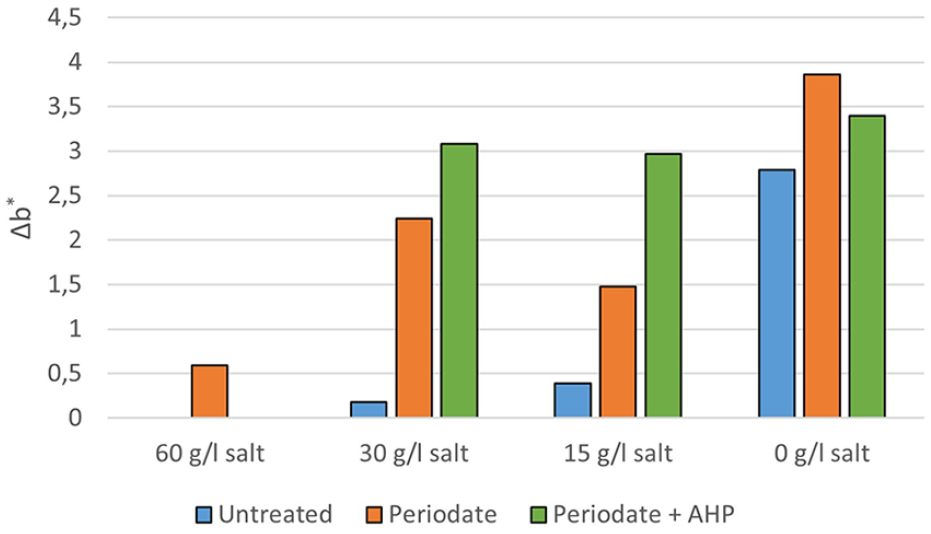
Δb* for untreated, oxidated and AHP treated cotton fabrics dyed with different amounts of salt (60, 30, 15, and 0 g/l). Sample used as reference is the untreated dyed with 60 g/l of salt.
Dyeing AHP treated cotton using 0 g/l of salt resulted in a colour with a lightness comparable to the reference sample since ΔL* is close to zero. In addition, ΔL* is lower for all cotton samples grafted with AHP regardless the amount of salt used. However, when decreasing the amount of salt in the bath the Δa* negatively increased for cotton samples treated with AHP indicating that the colour changed to a greener hue, obtaining the worst results when dyeing was carried out in absence of salt. In addition, colour difference Δb* was more stable for AHP treated cotton samples (Δb* ~ 3) regardless the amount of salt used, showing a more yellowish hue for samples treated with AHP.
Taking into account the aforementioned colour differences, the overall colour difference ΔE* CMC (2:1) between the samples was measured in order to evaluate the colour matching (Figure 6). As it is shown in Figure 6, the colour difference was lower for cotton samples grafted with AHP compared to untreated samples dyed at salt concentration below 15 g/l. However, differences were significant when comparing the resulted values with the colour obtained for the untreated sample dyed with 60 g/l of salt. In this regard, colour matching acceptability, assessed through colour difference ΔE* CMC (2:1), indicated that the colour differences were significant and can not be acceptable since ΔE* CMC (2:1) is higher than 1 for all the samples treated with AHP. Nevertheless, the colour difference ΔE* CMC (2:1) obtained for AHP treated samples for zero salt dyeing was approximately around two indicating that although the colour match is not acceptable, colour matching is reasonably better than that obtained for untreated and oxidated samples especially when the content of salt was reduced to zero.

Colour difference CMC (2:1) for untreated, oxidated and AHP treated cotton fabrics dyed with different amounts of salt (60, 30, 15, and 0 g/l). Sample used as reference is the untreated dyed with 60 g/l of salt.
Concerning to fastness properties (Table 4), treated fabrics only showed acceptable wash fastness since in this case AHP treated cotton samples behaved similarly to untreated fabric. The AHP treated cotton sample has found to have good to very good wash fastness ratings of 4–5 on grey scale (Colour difference CMC (2:1) has been also included between parenthesis). However, the pretreated dyed fabrics performed worse against rubbing, particularly in wet state, and light fastness with ratings of 2–3. The worsening of rubbing and light fastness could be explained considering that a high amount of dye introduced in the treated fabrics was located on the surface, being less stable to the mechanical action and light radiation.
Washing, rubbing and light fastness of untreated, oxidated and AHP treated cotton fabrics.

Washing and rubbing fastness: 5: Excellent, 4: Good, 3: Fair, 2: Poor, 1: Very poor.
Light fastness: 8: Excellent, 7: Very good, 6: Good, 5: Fair, 4: Average, 3: Low, 2: Poor, 1: Very poor.
Colour difference CMC (2:1) has been also included between parentheses besides fastness ratings on grey scale.
Conclusions
Grafting amino terminated hyperbranched polymer (AHP) by partial oxidation of cotton can dramatically reduce the amount of salts needed for dyeing with reactive dyes while obtaining similar colour intensities and comparable washing fastness properties. However, although the approach is acceptable from the point of view of washing fastness, some limitations were observed in AHP treated fabrics regarding the rubbing and light fastness that could limit the use of this low-salt method to applications for which rubbing and/or light fastness are a crucial issue. In addition, the grafting process using periodate to generate aldehyde groups produce a decrease of the strength of the cotton fabrics that should be controlled by selecting the appropriate process conditions of time, temperature and concentration of periodate. In this regard, it was observed that to minimize fibre weakness while maximizing aldehyde content it is recommended to use a 2 g/l of sodium periodate at 40°C for 60 min.
Footnotes
Acknowledgements
The authors would like to express their gratitude to Nur Syafiqah for her assistance in the experimental work and extend appreciation to BASF for supplying the hyperbranched polymer used in this study.
Sample credit author statement
A.D.S. and R.P.F.: Investigation; F.C.N.: Original draft preparation, Writing-Reviewing and Editing; F.C.N, A.D.S., R.P.F.: Methodology, Validation; F.C.N.: Supervision, Funding acquisition.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
