Abstract
This study explores the impact of sodium periodate oxidation time (30 min, 60 min, or 120 min) and alginate coating on the capillarity of jute fabrics. Raw jute fabric exhibits a rapid increase in capillary rise height (h) over time, reaching 72 mm at 1200 s. Fabrics oxidized for 30, 60, and 120 min show significantly lower h values of 19, 18, and 28 mm at 1200 s, respectively, which can be ascribed to the oxidation process itself. Precisely, oxidation-induced conversion of hydroxyl groups of cellulose (the main constituent of jute fiber) to aldehyde groups contributed to the fabric reduced affinity for water, hence negatively affecting its capillarity. Approximately 47% and 55% lower h values of fabrics oxidized for 30 and 60 min compared to that oxidized for 120 min can be explained by their lower weight loss after oxidation as well as less pronounced liberation of elementary fibers. Oxidized fabrics characterized by low capillarity can be used for the manufacturing of packaging materials, outdoor gear, and protective covers for crops. To expand jute fabric applications in biocarpet engineering, that is, to obtain polysaccharide-based supports, raw and oxidized jute fabrics were treated with a 1% sodium alginate solution. At 1200 s, oxidized fabrics coated with alginate exhibited 4.3–5.8 times higher h values compared to solely oxidized counterparts. The diffusion coefficients of oxidized fabrics coated with alginate increase up to 35% with prolonged oxidation, emphasizing the oxidation impact on alginate binding.
Introduction
Oxidation is one of the most studied reactions to which lignocellulosic fibers (like hemp, flax, and jute) have been subjected with the aim of enhancing the existing properties or introducing new functionalities, thereby catering to specific applications. The properties of reaction products strongly depend on several reaction parameters, including the type of oxidation agent, its concentration, pH, reaction duration, and temperature. Depending on the type of oxidation agent, non-selective or selective, and the reaction conditions, different functional groups of cellulose (the main component of jute fiber) take part in the reaction, 1 and the compounds bearing aldehyde, keto, or carboxyl groups can be obtained. The non-selective oxidation agents such as potassium permanganate (KMnO4) and hydrogen peroxide (H2O2) can oxidize primary cellulose hydroxyl groups (to carboxyl groups through intermediate carbonyl groups) and/or secondary cellulose hydroxyl groups (to carbonyl and carboxyl groups, with the ring structure remaining stable, or to aldehyde groups followed by glucopyranose ring-opening). 2 On the other hand, selective oxidation agents like potassium periodate (KIO4) and sodium periodate (NaIO4) oxidize secondary cellulose hydroxyl groups to aldehyde groups followed by glucopyranose ring opening, 3 while (2,2,6,6-tetramethylpiperidin-1-yl)oxyl (TEMPO) converts primary cellulose hydroxyl groups to carboxylic acid via the intermediate aldehyde stage. 4 Oxidation using sodium chlorite (NaClO2) is of particular interest for the functionalization of lignocellulosic fibers since NaClO2 selectively oxidizes only the aldehyde groups in cellulose, leaving its hydroxyl groups and glycosidic bond unaffected. 5 Remarkably, sodium chlorite solution of low concentration (0.7%) also oxidizes lignin (one of the three main jute fiber constituents); it induces benzene ring cleavage and dicarboxylic group formation contributing to lignin selective removal. 6 A comprehensive understanding of the mentioned oxidation mechanisms is essential for tailoring lignocellulosic fibers to meet the evolving demands of sustainable technologies.
The effect of sodium chlorite oxidation on lignocellulosic fiber sorption and wetting properties (encompassing water retention, moisture sorption, fiber swelling, dyeability, and capillarity) has been extensively investigated.7–10 For example, Pejic et al. 9 studied the capillarity of hemp fiber bundles oxidized with a 0.7% NaClO2 for 5–60 min. The results derived from the capillary rise measurements of 0.1% aqueous solution of eosin pointed out that oxidized fiber bundles are characterized by 2.0–2.7 times higher capillary rise height values (at 2000 s of measurement) compared to the raw hemp fiber bundles. The authors Pejic et al. 9 assert that during the oxidation, selective lignin removal and fiber fibrillation occurred resulting in the formation of new capillary spaces between completely or partially separated fibers within the technical hemp fiber, altogether promoting better wettability of oxidized fiber bundles. Ivanovska et al. 7 used the same oxidation agent to enhance the capillarity of raw jute fabric. About 64%–75% higher h values registered for fabrics oxidized for up to 90 min occurred as a result of the fact that sodium chlorite oxidation induced the changes in jute fiber molecular structure and morphology. Precisely, about 39%–79% of the lignin has been removed from the middle lamellae and secondary wall, which in parallel with the prominent elementary fiber liberation contributed to the formation of new free spaces resulting in improved capillarity of oxidized fabrics compared to the raw one. Bearing in mind that NaClO2 is a powerful oxidation agent, the oxidation of lignocellulosic fibers using it is an elegant approach for examining the effect of selective lignin removal on their sorption and wetting properties. Nevertheless, it is widely acknowledged that this type of oxidation lacks environmental friendliness, can pose human health risks and individuals handling the chemical should be trained in its safe handling and disposal. 11
Considering the aforementioned drawbacks associated with sodium chlorite oxidation and inspired by a recent publication by Ivanovska et al. 12 dealing with the improvement of jute fabric sorption properties upon sodium periodate oxidation, we sought to explore the influence of the same oxidation agent on jute fabric capillarity. Jute was selected as the experimental material since it is the most important lignocellulosic fiber with an annual production of 3.4 million tonnes in 2021 cultivated over 1.4 million hectares mainly in Bangladesh and India. 13 Additionally, it is one of the most versatile natural fibers that has been used for producing packaging materials and other materials for non-textile applications. Exactly in the first part of the manuscript, raw jute fabric was oxidized with 0.2% NaIO4 for 30 min, 60 min, or 120 min with the objective of obtaining fabrics suitable for the production of packaging materials, outdoor gear, and protective covers for crops which was evaluated through the determination of their capillarity. In the second part of the current paper, we attempted to move jute fabric application from conventional to nonconventional, that is, in biocarpet engineering. To achieve this, raw and oxidized jute fabrics were coated with alginate, and the potential application of such fabrics was thoroughly discussed.
Experimental
Materials
Raw jute fabric in the plain weave (bought from Dekoteks Trade, Serbia) with the following chemical composition (64.53% α-cellulose, 16.82% hemicelluloses, 13.68% lignin, 0.68% fats and waxes, 1.82% pectin, and 2.45% water-soluble components) was used as starting material. The chemical composition of raw jute fabric was determined according to the method described in the literature. 14 All chemicals utilized in this investigation were procured from commercial suppliers and were of a purity grade suitable for analytical purposes. The alginic acid sodium salt employed in the study is of low viscosity (Thermo Scientific Chemicals) and was utilized as received.
Sodium periodate oxidation of raw jute and alginate coating of jute fabrics
The raw jute fabric, which had been pre-washed with distilled water at boiling temperature for 30 min and subsequently dried, underwent oxidation with a 0.2% NaIO4 aqueous solution in the dark for 30 min, 60 min, or 120 min, adhering to the protocol outlined by Ivanovska et al. 15 Termination of the oxidation reaction was achieved by immersing the fabric in ice-cold distilled water. Part of the oxidized fabrics was then squeezed and dried at room temperature, while the other part was coated with alginate. The codes assigned to each fabric are presented in Figure 1.

Fabric codes.
After washing or immersing in ice-cold distilled water, raw jute (J) and oxidized jute fabrics (J0.2/30, J0.2/60, and J0.2/120) were squeezed and immediately dipped in 1% sodium alginate solution (with a material-to-liquid ratio of 1:20) for 24 h at room temperature. Subsequently, the fabrics were squeezed and immersed in a 2% CaCl2 aqueous solution (with a material-to-liquid ratio of 1:20) for 24 h at room temperature. Thereafter, the fabrics were thoroughly rinsed with distilled water, and subjected to drying in a laboratory oven for 24 h at 60°C. The abbreviations of alginate-coated fabrics are given in Figure 1.
Fabric capillarity
Fabric capillarity was determined following the method described in the literature Ivanovska et al. 8 Specifically, jute fabrics (20 cm × 2 cm in warp direction) were vertically positioned. One fabric end was secured to a horizontal bar using a clip, while the other end was pre-loaded with a tag and immersed to a depth of approximately 1 cm in a 1% aqueous solution of eosin. The red-colored eosin solution clearly indicated the capillary rise height on the beige jute fabric, which could be easily read with the help of a millimeter ruler placed alongside each sample. During immersion in eosin, photographs were taken at time intervals of 15 s in the first minute, every 30 s up to 5 min, every 1 min up to 10 min, and every 5 min up to 20 min. Utilizing the experimental data obtained through capillarity measurements and following the methodology detailed by Pejić et al., 16 the capillary diffusion coefficient (D, mm2/s) was determined using equation (1).
where h (mm) represents capillary rise height, while t (s) denotes time.
Determination of overall (liquid) moisture management capability (OMMC)
Determination of the overall (liquid) moisture management capability (OMMC) of studied fabrics was performed according to AATCC 195-2020 on a moisture management tester (MMT M290, SDL Atlas). The average of five measurements for each sample was taken into consideration.
Characterization of studied jute fabrics
The weight loss occurred after different oxidations of raw jute fabric was determined using the direct gravimetric method described in Ivanovska et al. 17
The quantification of CHO group content was conducted in accordance with the procedure published by Ivanovska et al. 15
Verification of fabric coating with alginate was established through Field Emission Scanning Electron Microscopy (FESEM, Tescan MIRA 3 XMU). Prior to FESEM analysis, all samples were sputter-coated with a thin layer of gold.
Results and discussion
Capillarity of raw and oxidized jute fabrics
Recent findings 12 have highlighted that sodium periodate oxidation induces significant alteration in the jute fiber molecular (primarily in the content of CHO groups), fine (crystallinity index), and micro (removal of surface impurities and the appearance of micropores) structure. Consequently, certain jute fabric sorption and wettability properties such as moisture sorption and wetting time, are affected by this selective oxidation. It is therefore reasonable to assume that sodium periodate oxidation could also lead to a change in the jute fabric capillarity – an important wettability parameter that represents the fabric capacity to absorb moisture, liquids, lubricants, and steam. 18
In this section, the effect of oxidation time (30 min, 60 min, or 120 min) on fabric capillarity was systematically investigated, wherein the capillarity was presented as a function of a capillary rise height (denoted as “h”) versus time. Figure 2(a) illustrates a rapid increase in h of raw jute fabric (J), which is not the case for oxidized fabrics J0.2/30, J0.2/60, and J0.2/120. Irrespective of the examined time interval, the h values of oxidized fabrics are significantly lower than those of raw jute. Precisely, no h was observed for J0.2/30, J0.2/60, and J0.2/120 up to 420, 240, and 270 s of measurement, respectively, and after that, a slow increase of h was noted. At 600 s, J, J0.2/30, J0.2/60, and J0.2/120 showed h values of 49.00, 10.00, 11.67, and 16.67 mm (Figure 2(a) and (b)), respectively. Prolonging the capillarity measurement to 1200 s resulted in increased h values reaching 72, 19, 18, and 28 mm for J, J0.2/30, J0.2/60, and J0.2/120, respectively. The substantial differences observed between h values of raw and oxidized fabrics J0.2/30, J0.2/60, and J0.2/120, (accounting for 73.6%, 75.0%, and 61.1% at 1200 s), undoubtedly originate from the oxidation process itself and can be attributed to the various factors concurrently influencing the fabric capillarity.
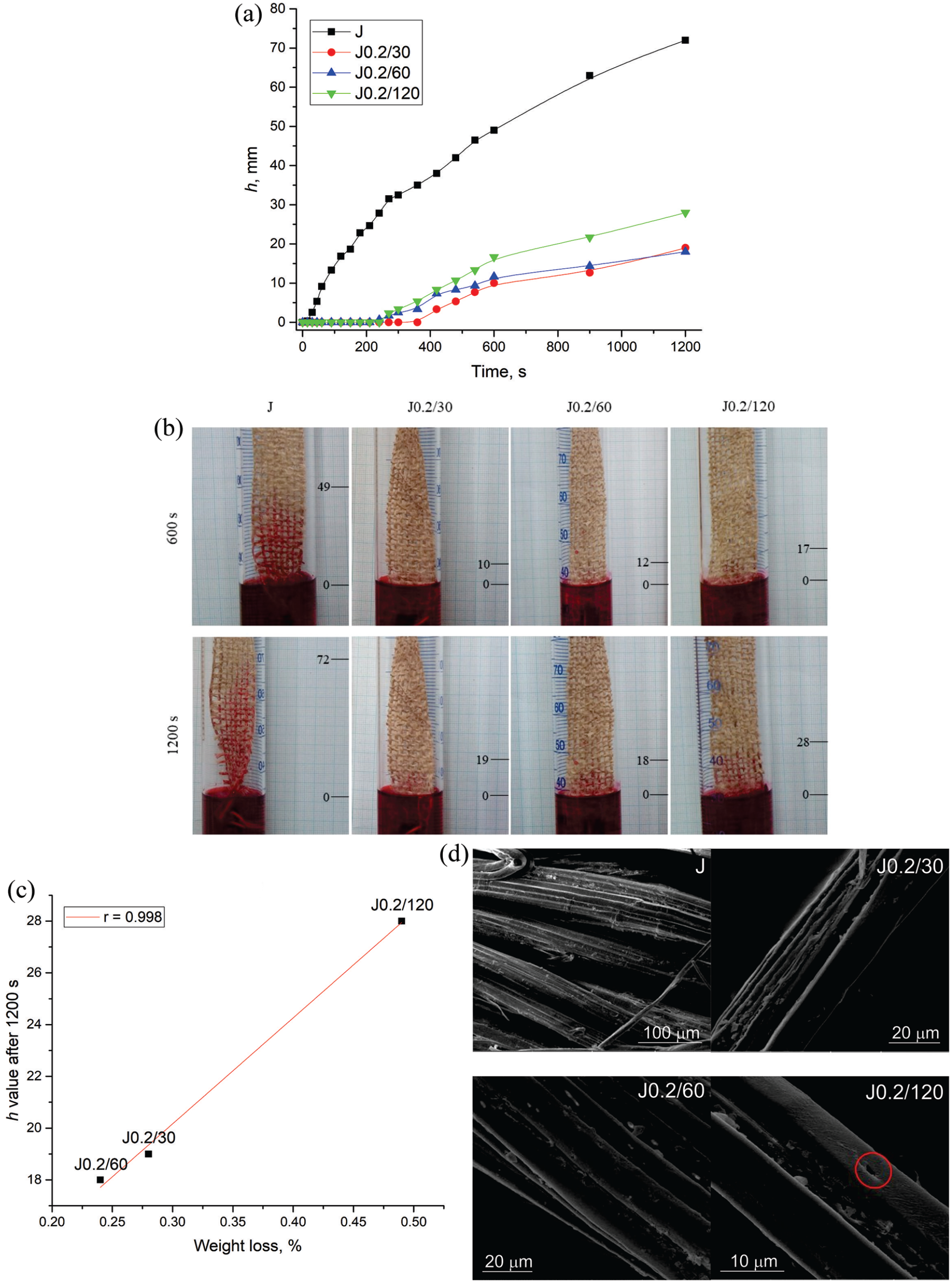
(a) Capillary rise high (h) of studied fabrics, (b) capillarity measurement at 600 and 1200 s, (c) linear correlation between weight loss and h values, and (d) SEM photographs of examined fabrics.
On one hand, the observed behavior of oxidized fabrics aligns with the expectations since sodium periodate oxidation induces the oxidation of cellulose secondary hydroxyl to aldehyde groups, which in parallel results in ring cleavage, and the formation of 2,3-dialdehyde cellulose. 19 The newly introduced CHO groups (0.239, 0.272, and 0.327 mmol/g in J0.2/30, J0.2/60, and J0.2/120, respectively vs 0.185 mmol/g in J) exhibit reduced affinity for water, and hence, negatively affecting fabric capillarity. The absence of a clear correlation between the CHO group content and h values of oxidized fabrics suggests the existence of additional contributors to approximately 47% and 55% lower h values of J0.2/30 and J0.2/60 than h of J0.2/120 at 1200 s, Figure 2(a). In addition to negligible weight loss occurred after oxidations (Figure 2(c)), these data are crucial as they imply removal of certain jute fiber components. Considering the layered structure of jute fibers, it becomes apparent that the observed weight loss is attributed to the removal of non-cellulosic hydrophobic impurities like fats, waxes, and pectin from the fiber surface. As a result, the surfaces of oxidized fabrics become more hydrophilic contributing to elevated h values, as affirmed by the linear correlation presented in Figure 2(c). Furthermore, the impact of sodium periodate oxidation on fiber microstructure should not be disregarded. The SEM images depicting fabric surface morphology revealed not only the removal of fiber surface impurities but also the liberation of elementary fibers (Figure 2(d)). The prominence of this phenomenon intensifies with prolonged oxidation duration and the new capillary spaces between completely or partially separated elementary fibers within technical jute fiber were created. According to Ivanovska et al., 8 during wetting, these spaces demonstrate the capability to transfer the fluid and act as a void system thereby promoting capillarity. The eye-catching difference on fabric J0.2/120 surface occurred due to the long oxidation is the existence of a small micropore, which according to Pejić et al. 16 contributes to the greater liquid advancement during the capillarity rise.
Oxidized fabrics characterized by low h values could be used for the manufacturing of packaging materials like hessian and sackings that can be employed for the storage of different commodities. For such application, fabric overall (liquid) moisture management capability (OMMC) should also be taken into consideration since fabrics with high OMMC can efficiently wick moisture away from the surface, promoting evaporation and keeping stored items dry. AATCC TM 195-2020 defines OMMC as an index that reflects the overall capability of the fabric to manage the transport of liquid moisture, encompassing three key performance aspects: the liquid moisture absorption rate on the bottom surface, the one-way liquid transport capability, and the maximum liquid moisture spreading speed on the bottom surface. The determined OMMC indexes of J, J0.2/30, J0.2/60, and J0.2/120 are 0.073, 0, 0, and 0.061, respectively. These fabrics show poor moisture management capability 20 (grade 1 according to AATCC TM 195-2020), which in combination with fabric h values (Figure 2) indicate that J0.2/120 is the most appropriate fabric for storing commodities. Additionally, J0.2/30, J0.2/60, and J0.2/120, have low h values as well as poor to fair adsorption rate on the bottom surface, poor one-way transport capability with no spreading of liquid moisture on the bottom surface (evaluation based on MMT measurements), also present viable options for the production of outdoor gear, including backpacks and tents. Furthermore, the utility of oxidized fabrics extends to serving as protective covers for crops, presenting a natural and biodegradable solution to shield plants from rain and water.
The effect of alginate coating on the capillarity of jute fabrics
Expanding the application of jute fabric from conventional to nonconventional areas involves the exploration of novel innovative approaches. The rapid evolution of textile technology opens up new possibilities for integrating jute fabrics into sustainable and environmentally friendly practices, for instance in the context of biocarpet engineering. The idea for such an unconventional jute fabric application is connected to the fact that land degradation, whether resulting from natural or anthropogenic processes, affects the functionality of terrestrial ecosystems and exerts indirect repercussions on aquatic ecosystems. Over 40% of global terrestrial ecosystems are confronting significant anthropogenic degradation resulting in the loss of ecosystem integrity and services, and the deterioration of associated socioeconomic values. 21 In the quest for an effective approach to land restoration, Chamizo et al. 22 have proposed the utilization of induced biological soil crusts (biocrusts). These biocrusts host complex communities of microorganisms, including cyanobacteria, inhabiting and permeating the soil, and undertaking several indispensable ecosystem functions. 22 While certain research endeavors have been conducted in the realm of biocrust-assisted restoration,23–26 finding optimal methodologies remains a big challenge. For instance, a field investigation conducted by Wu et al. 26 showed that intricate biocrusts, following artificial cyanobacterial inoculation on a sandy substrate, could develop within a span of 7 years. However, this process can often take much longer. 27 Consequently, expediting the development and succession of biocrusts by enhanced inoculum viability through improved water availability emerges as a critical determinant of success. The survival of the biocrust inoculum and the efficacy of biocrust restoration can be promoted through a careful selection of cyanobacterial strains specific to targeted substrates. Additionally, the design of geo-prebiotic polysaccharide-based supports, possessing a favorable wettability, can foster a sustainable interplay between microbiota and abiotic constituents on degraded lands, sediments, and substrates, while also contributing to air and water pollution control.
To meet the aforementioned requirements, we have decided to prepare polysaccharide-based supports which involved the treatment of raw and oxidized jute fabrics (Figure 1) with a 1% aqueous solution of sodium alginate. An inquiry that may arise pertains to the rationale behind selecting this linear copolymer consisting of (1 → 4) linked β-
The alterations in the surface chemistry and morphology of fabric J0.2/120 upon coating with alginate (J0.2/120 + AL) were documented using ATR-FTIR spectroscopy and SEM, respectively, Figure 3. To elucidate the variations in intensity and position of the bands observed in the spectra of J0.2/120 and J0.2/120 + AL, an FTIR spectrum of sodium alginate was recorded, Figure 3(a). This spectrum reveals characteristic bands at 3215 cm−1 (O–H stretching vibrations), and at 1595−1 and 1411 cm−1 (COO– asymmetric and symmetric stretching vibrations, respectively 30 ). The alginate coating onto J0.2/120 led to discernible alterations in the FTIR spectrum of J0.2/120 + AL. Notably, a band at 3338 cm−1 (hydrogen-bonded O–H stretching in J0.2/120) intensified and shifted to a lower wavenumber (3292 cm−1) in the spectra of J0.2/120 + AL. Furthermore, the spectrum of fabric J0.2/120 + AL exhibited a dominant band at 1595 cm−1 associated with the vibrations of the alginate COO– groups. Additionally, bands attributed to symmetric COO– vibrations shifted from 1411 cm−1 (observed in the spectrum of sodium alginate) to 1418 cm−1 (observed in the spectrum of J0.2/120 + AL) due to the replacement of Na+ ions from sodium alginate with Ca2+ ions from calcium chloride, which was introduced for cross-linking purposes. 31 Moreover, the band observed at 1030 cm−1, corresponding to the vibration of the C–O–C pyranose ring, exhibits increased intensity and sharper characteristics after coating, Figure 3(a).

(a) ATR-FTIR spectra and (b) SEM images of selected fabrics.
The successful application of alginate coating onto jute fabrics was further confirmed through SEM photographs. As depicted in Figure 3(b), the J0.2/120 exhibited a relatively smooth fiber surface without the existence of surface impurities. In the SEM image of J0.2/120 + AL, it is notable that alginate appeared as a thicker biopolymer layer.
The enhancement of fabric capillarity after coating with alginate is obvious especially during the initial 420, 240, and 270 s since at the mentioned times, J0.2/30 + AL, J0.2/60 + AL, and J0.2/120 + AL exhibited h values of 79.7, 68.0, and 80.0mm (Figure 4(a)), respectively. To recall, their uncoated counterparts J, J0.2/30, J0.2/60, and J0.2/120 showed h values of 0 at targeted time intervals (Figure 2(a)). Observing in parallel the fabric h values at 1200 s, before and after coating (Figures 2(a) and 4(a)), it is apparent that alginate-coated fabrics J + AL, J0.2/30 + AL, J0.2/60 + AL, and J0.2/120 + AL are characterized by 15.3%, 5.3, 5.8, and 4.3 times higher h values compared to those before coating. It is interesting to emphasize that by prolonging the oxidation time, h values of alginate-coated fabrics increase too. This phenomenon is the most obvious at 1200 s wherein their h values account for 85, 100, 104, and 120 mm, respectively. To better describe such behavior, the capillary measurement data were employed to calculate the diffusion coefficient (D) of alginate-coated fabrics, Figure 4(b). Notably, in comparison to J + AL, J0.2/120 + AL demonstrated a 35% higher rate (Figure 4(c)) at which a liquid moves through the fabric due to the capillary action signifying the substantial impact of oxidation in the binding of alginate.

(a) Capillary rise high (h) of alginate-coated jute fabrics, (b) diffusion coefficient (D) of alginate-coated jute fabrics, and (c) capillarity measurement at 1200 s.
As indicated in our previous publication, 12 the sodium periodate oxidation of the jute fabrics not only increased the number of aldehyde groups but also increased their negative zeta potential. The oxidation step, thus, further promotes the coating of jute fabric with alginate. The reason behind this behavior lies in the significant electrostatic repulsion between the jute surface and alginate carboxylate groups dictating the orientation of these groups in a way that is more favorable for effective coating (Figure 5). Namely, to minimize the energy of repulsion, alginate carboxylate groups are oriented away from the jute surface, while alginate –OH groups are enabled to establish effective hydrogen bonds with hydrogen bond donor groups of jute fabrics (mainly –OH groups originating from cellulose, as depicted in Figure 5). Upon treatment with CaCl2, carboxylate groups on the fabric surface attract Ca2+ cations and the gel network is formed by lodging of Ca2+ ions into the cavities formed by two adjacent alginate chains bearing GG blocks in helical conformation. 28 The cross-linking process produces the “egg-box” structure as shown in Figure 5. Higher capillarity observed for oxidized fabrics coated with alginate could be explained by the possible mechanism of coating: the more negative jute fabric surface, the stronger repulsion between the fabric surface and alginate carboxylate groups, thus, more effective coating with alginate results in improved capillarity.

Putative mechanism of coating jute fabrics with alginate.
Despite the targeted potential application of alginate-coated oxidized fabrics in biocarpet engineering, jute fabrics with enhanced capillarity, that is, wettability properties as well as several times increased OMMC (0.101, 0.148, 0.206, and 0.225 for J + AL, J0.2/30 + AL, J0.2/60 + AL, and J0.2/120 + AL, Supplemental Material Figure 1S) can be beneficial in filtration application. Fabrics J0.2/60 + AL and J0.2/120 + AL showed fast absorption and slow drying,20,32 and can be characterized by fair moisture management capability (grade 2 according to AATCC TM 195-2020). Therefore, they enable efficient transport of fluids through them, allowing for water purification or other liquid filtration processes.
Conclusion
This study systematically investigates the influence of sodium periodate oxidation time (30 min, 60 min, or 120 min) and coating with alginate on the capillarity of jute fabrics. The obtained results reveal that raw jute fabric exhibits a rapid increase in capillary rise height (h) with the measuring time, reaching 72 mm at 1200 s. In contrast, h values of sodium periodate oxidized fabrics are significantly lower than those of the raw fabric, regardless of the observed time interval. For example, at 1200 s, h values of fabrics oxidized for 30, 60, and 120 min are 19, 18, and 28 mm, respectively. The substantial difference observed between the capillarity of raw and oxidized fabrics is attributed to the oxidation process itself. The low h values observed for oxidized jute fabrics make them suitable for manufacturing packaging materials, outdoor gear (backpacks, tents, and sleeping bags), and protective covers for crops.
To further broaden the application of jute fabrics in biocarpet engineering, that is, water-binding geo-prebiotic polysaccharide-based supports, raw and oxidized jute fabrics were treated with a 1% aqueous solution of sodium alginate. Upon examination of fabric h values at 1200 s before and after coating, it was observed that alginate-coated fabrics showed between 15% and 5.8 times higher h values compared to their uncoated counterparts. Furthermore, the h values of oxidized alginate-coated fabrics are about 15%–29% higher than that of alginate-coated raw jute at 1200 s. The calculated diffusion coefficients, as h values of alginate-coated fabrics demonstrated an increasing trend with prolonged oxidation duration, highlighting the significant impact of oxidation on the binding of alginate. Beyond their potential application in biocarpet engineering, alginate-coated oxidized jute fabrics can find application in water purification and various liquid filtration processes.
Supplemental Material
sj-docx-1-jef-10.1177_15589250241246071 – Supplemental material for The impact of sodium periodate oxidation and alginate coating on the capillarity of jute fabric
Supplemental material, sj-docx-1-jef-10.1177_15589250241246071 for The impact of sodium periodate oxidation and alginate coating on the capillarity of jute fabric by Aleksandra Ivanovska, Marija Milošević, Jelena Lađarević, Anita Tarbuk, Zorica Svirčev and Mirjana Kostić in Journal of Engineered Fibers and Fabrics
Footnotes
Acknowledgements
The authors thank Tamara Matić (University of Belgrade, Faculty of Technology and Metallurgy) for SEM photographs. The authors express their gratitude to the Laboratory for Controlled Crosslinking Processes (University of Zagreb, Faculty of Textile Technology) for providing access to the Moisture Management Tester. A.I., M.M., Z.S., and M.K. would like to acknowledge financial support from the Science Fund of the Republic of Serbia, #7726976, Integrated Strategy for Rehabilitation of Disturbed Land Surfaces and Control of Air Pollution -RECAP.
Author statement
The manuscript entitled: “The impact of sodium periodate oxidation and alginate coating on the capillarity of jute fabric” has not been published elsewhere and it has not been submitted simultaneously for publication elsewhere.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Science Fund of the Republic of Serbia, #7726976, Integrated Strategy for Rehabilitation of Disturbed Land Surfaces and Control of Air Pollution – RECAP and Ministry of Science, Technological Development and Innovation of the Republic of Serbia (Contract No. 451-03-47/2023-01/200287 and 451-03-47/2023-01/200135).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

