Abstract
Castor oil (CAO)/poly (N-vinyl-2-pyrrolidone) (PVP) hybrid was synthesized at different reaction conditions including PVP/CAO weight ratio, temperature, and time. The results indicated that the optimum conditions to synthesis that hybrid is reacting of PVP having a molecular weight of 40,000 Da with CAO at a weight ratio of 30% and temperature of 150°C for 60 min. The synthesized hybrid was characterized via FTIR. Emulsification of such hybrid in water results in a white stable emulsion. The TEM analysis proved that the prepared emulsion of a particle size ranges from 320 to 370 nm. The technical feasibility to apply the produced emulsion for functional finishing of cotton fabric was studied. The results obtained indicated that treating cotton fabric with easy care finishing bath containing the synthesized emulsion results in enhancement in nitrogen content, tensile strength, whiteness index, stiffness, and antibacterial activities along with a decreasing in resiliency, wettability, and surface roughness properties of treated fabric, compared to the control sample. Moreover, incorporation of zinc oxide nano-particles (ZnO-NPs) or dihydroxybenzophenone (DHBP) in the above mentioned finishing bath enhances the functional properties of the finished fabric. Furthermore, dyeing fabric samples with different reactive and direct dyes followed by finishing using the aforementioned finishing bath, in absence of ZnO-NPs and DHBP, gives rise to an enhancement in the color strength of such samples compared to the dyed samples. The fastness to washing and perspiration of only the direct dye dyed/finished sample was improved. Almost all the dyed/finished samples exhibited an improvement in their fastness to the wet rubbing and alkaline perspiration along with a reduction in fastness to light. In addition, the SEM, as well as EDX analysis of treated fabric, was investigated.
Keywords
Introduction
Castor oil is produced from the castor seeds of the castor plant (Ricinus communis). It comprises ricinoleic (about 90%) in addition to other fatty acids such as linoleic, oleic, stearic, palmitic, dihydoxystearic, eicosanoic, and linolenic acids (Figure 1). 1

Fatty acids of castor oil. 1
The presence of a double bond, hydroxyl group, carboxylic group as well as a hydrocarbon long chain in the ricinoleic acid of the castor oil, offers many possibilities for transforming it into versatile materials. Due to the unique chemical structure and renewability of castor oil, it is widely used in versatile industrial applications including cosmetics, textile dyes, paints, coatings, varnishes, plastics, inks, soaps, and lubricants. Besides, castor oil is used traditionally in medicine as a purgative and laxative as well as for treating dry skin and hair.1–4 Castor oil can be emulsified using many emulsifying agents. Emulsions are widely used in the cosmetics, pharmaceutical, and food industries. In the pharmaceutical field, emulsions are able to deliver both hydrophilic and lipophilic drugs. 5 Many gums such as Arabic gum and Terminalia gum can be used to emulsify castor oil.6,7 Zhang et al. 8 reported that an emulsifier formula containing polyoxyethylene sorbitan monostearate and sorbitan monooleate achieved a stable emulsion of castor oil. Castor oil can be sulfated using sulfuric acid to become an anionic textile softener (Turkey red oil). Increasing the castor oil sulfate groups renders it to have a higher ionicity with lower lubrication and ultimately becomes a self-emulsifier. Sulfated castor is easily removed from treated fiber or fabric without an auxiliary surfactant. 9
Poly (N-vinyl-2-pyrrolidone) (PVP) is a water-soluble, nontoxic, and biocompatible synthetic polymer. PVP is a suspending agent, a clarifying agent, protective colloid, stabilizer, viscosity modifier, film former, and complexing agent. It has versatile industrial applications including pharmaceutical, biomedical, and textiles applications. PVP can be crosslinked through heating at 150°C in air, ultraviolet and gamma radiation, or in the presence of persulfates.10,11
Cellulosic fabrics such as cotton, linen, and viscose provide comfort properties such as softness, high absorbency, and breathability for their wearers. Unfortunately, these fabrics are easily wrinkled and soiled, prone to microbial attack, and have low protection against UV radiation. In response to the consumer demands for healthy lifestyles along with developing cellulosic fabrics having durable functional properties such as soft hand, antibacterial, anti-UV radiation, self-cleaning, and easy care properties, extensive research has been performed by R&D institutions and the textile manufacturers to satisfy that consumer needs and marketing.12–18 Various bioactive agents can be used to impart textiles with antimicrobial properties such as metal and metal oxide nanoparticles, for example, Ag, ZnO, and TiO2 nanoparticles (NPs), chitosan, re-generable N-halamine compounds, metal salts, halogenated phenols, for example, triclosan, quaternary ammonium compounds, and immobilized enzymes.19,20 UV absorbers are inorganic or organic colorless compounds having high absorption in the range of 290–360 nm of ultraviolet spectra. These compounds include phenyl esters, cinnamic acid derivatives, benzotriazoles, hydroxyl benzophenone derivatives, oxalic acid dianilide derivatives, benzotriazole derivatives, dyes, optical brighteners, and metal oxides such as TiO2, ZnO, SiO2, CeO2, and Al2O3 nanoparticles.19–23 The ZnO-NPs provide excellent UV protection as well as high antibacterial activity. 24 However, the ability of Zn-NPs as a UV protector is due to the band gap absorption and light scattering mechanism. 25 The band gap between the valance and conduction electrons of the ZnO-NPs confer the generation of reactive oxygen species giving rise to conformational changes/oxidant injury to the microorganism membrane surface. 24 The substituted benzophenones are the most effective organic compounds in protecting against UV radiation. The dihydroxybenzophenone absorbs UV radiations strongly in the UV-A range while benzophenone absorbs in the UV-B range. The strong absorption for UV-A of dihydroxy benzophenone is due to the conjugate chelation between the carbonyl group and the o-hydroxy. 23 Besides, the benzophenone derivatives generate reactive oxygen species under their exposure to sunlight or UV irradiation, providing antibacterial activity to their loaded fabrics. 26
The main goal of that work is to emulsify the castor oil using poly (N-vinyl-2-pyrrolidone) and investigate the technical feasibility to apply the produced emulsion as a textile softener during easy-care finishing of cotton fabric to impart that fabric with multi-functional properties namely softness, anti-crease, antibacterial, and UV protection. In addition, the coloration and fastness properties of dyed/finished cotton fabric samples will be also studied.
Experimental
Materials
Mill-scoured and bleached cotton fabric of weave structure, weight of 128 g/m2 and count (Ne) of 40/1 was supplied by Misr Spinning and Weaving Co., Mahalla El-Kobra, Egypt. Castor oil (CAO) from India, supplied by Iso-Chem. Company was used. Durapret LF (DMDHEU), a low formaldehyde resin of concentration 70%, was used as a crosslinker for easy care finishing of cotton fabric. Poly (N-vinyl-2-pyrrolidone) (PVP) of molecular weight 40,000 Da was supplied by Alpha Chemika. Zinc oxide nanoparticles (ZnO-NPs) and 2,4-dihydroxy benzophenone (DHBP) were supplied by Aldrich. Ammonium sulfate of laboratory-grade chemical was used as a catalyst. The following commercial direct and reactive dyes were used: C.I. Direct Red 84, C.I. Reactive Red 141, C.I. Reactive Blue 19, and C.I. Reactive Black 5.
Methods
Preparation of CAO/PVP hybrid emulsion
The CAO/PVP hybrid emulsion was prepared as follows: 5 g of CAO and different weights of PVP (0–4 g) were mixed together in a 150 mL beaker with stirring at different temperatures (100°C–160°C) for a period of time (0–75 min). After a certain time, the reaction medium was left to cool at room temperature followed by the addition of a specific volume of hot distilled water at 70°C and stirring using a strong homogenizer for 3 min to form a stable CAO/PVP hybrid emulsion. 14
Fabric treatment
Soft finishing of cotton fabric
Cotton fabric samples of 30 × 30 cm2 were padded twice in finishing bathes containing 80 g/L of DMDHEU, ammonium sulfate as a catalyst, and different concentrations of CAO/PVP hybrid emulsion (0–40 g/L), to a wet pick up of 100%. The padded samples were dried at 100°C/3 min in Wenner Mathis AGCH-8155 oven and then cured at 150°C/3 min. The finished fabrics were then washed with distilled water at 50°C for 10 min, thoroughly rinsed, and finally dried for testing.
Dyeing of cotton fabric
Dyeing of cotton fabric with the above-mentioned reactive dyes was performed by dissolving 1% of any of such dyes (owf) in distilled water at 40°C then 30 g/L Na2SO4 was added to the dye bath followed by the addition of the fabric samples using a material to liquor ratio 1:50. After 30 min, the dye bath temperature was raised to 60 °C in case of the C.I. Reactive Blue 19 and C.I. Reactive Black 5 dyes but to 90°C for the C.I. Reactive Red 141, followed that by addition of 15 g/L sodium carbonate dropwise and then the dyeing process was left to proceed for 60 min. At the end, the dyed samples were soaped with 3% nonionic detergent, rinsed with tap water, and finally dried at room temperature. On the other hand, cotton fabric samples were dyed with the nominated direct dye by dissolving 1% of that dye (owf) in distilled water at room temperature and then 30 g/L Na2SO4 was added followed by the addition of the fabric samples to dye bath using a material to liquor ratio 1:50. The dye bath temperature was then raised to 90°C and the dyeing process was preceded for 60 min. At the end, the dyed samples were soaped with 3% nonionic detergent, rinsed with tap, water and finally dried at room temperature.
Analysis and test methods
Fabric weight (W) was assessed according to ATSM (D 3776 – 79).
The nitrogen content was evaluated according to Kjeldahl method. 27
The wrinkle recovery angle of treated fabric samples, WRA, was determined according to ASTM method D-1296-98.
Wettability of the finished fabric sample was assessed according to the AATCC test method 39-1980.
Surface roughness (SMD) was measured using Kawabata evaluation system, Surface tester KES-FB4-A, Kato Tech Co., Ltd, Japan.
Tensile strength (TS) was tested in the warp direction according to ASTM procedure D-2256-98.
The fabric flexural rigidity (stiffness) was carried out according to ASTM (D 1388-1996).
The whiteness index of the finished fabric and color strength of the dyed/finished fabric, expressed as K/S value, were evaluated automatically using Hunter Lab spectrophotometer (Ultra Scan PRO).
Fastness properties of dyed fabric samples to washing (WF), rubbing (RF), perspiration (PF), and light were determined according to ISO test methods: 105-C06 (2010), 105-X12 (1987), 105-E04 (2008), and 105-B02(2013) respectively.
The antibacterial activity was assessed using the bacterial count method as reported elsewhere
28
against: (a) Gram-positive bacteria: Staphylococcus aureus (SA). (b) Gram-negative bacteria: Escherichia coli (EC).
Durability to washing was assessed by subjecting the finished fabric to 5, 10, and 20 washing cycles according to AATCC test method 61-1994.
UV-Protection Factor (UPF) was determined according to the Australian/New Zealand Standard (AS/NZS 4399-1996), and rated as follows: good (UPF: 15–24), very good (UPF: 25–39), and excellent protection (UPF >40).
Infra-Red (IR) spectroscopy was carried out using FT/IR-4700 FTIR Spectrometer from JASCO.
The morphology and particles size of the hybrid emulsion was obtained by transmission electron microscope (TEM) using a JEOL, JEM 2100 F electron microscope at 200 kV.
Scanning electron microscope (SEM) images of the treated and untreated fabric samples were obtained using SEM Model Quanta 250 FEG (Field Emission Gun) attached to EDX Unit (Energy Dispersive X-ray Analyses), with accelerating voltage 30 kV, magnification 14× up to 1,000,000 and resolution for Gun, FEI company, Netherlands.
Results and discussion
Tentative mechanism
Previous literature reported that PVP can be crosslinked under heating in air at 150°C through a chain scission mechanism.10,11,29,30 Thus, it is expected that heating a CAO/PVP mixture at the conditions employed will give rise to a formation of crosslinked PVP as well as CAO grafted with PVP. Both the hydroxyl groups as well as the double bond present in the castor oil may serve as grafting centers.30 –33 The PVP grafted oil may serve as a self-emulsifiable for the remaining CAO beside the crosslinked PVP that stabilizes the resulting emulsion. 33 For simplicity, the reaction product will be pointed out as “CAO/PVP hybrid.” The following mechanism may investigate the grafting of the CAO with PVP:
(a) Formation of PVP macroradicals

(2) Formation of castor oil radicals

(3) Formation of crosslinked PVP and castor oil grafted with PVP

Factors affecting the CAO/PVP hybrid emulsion formation
Effect of temperature
Table 1 shows the impact of the reaction medium temperature on CAO/PVP hybrid emulsion state. It is clear that raising the reaction temperature from 100°C to 150°C is accompanied by a gradual improvement in the CAO/PVP hybrid emulsion state. The matter that can be associated with the positive impact of temperature, in addition to air oxygen, to form PVP macroradicals that in turn enhance the formation of grafted castor oil as well as crosslinked PVP and hence the upgrading in the hybrid emulsion sate. 30 Increasing the reaction temperature beyond 150°C and up to 160°C has no effect on the emulsion state of the prepared hybrid.
Effect of reaction temperature on CAO/PVP hybrid emulsion state.
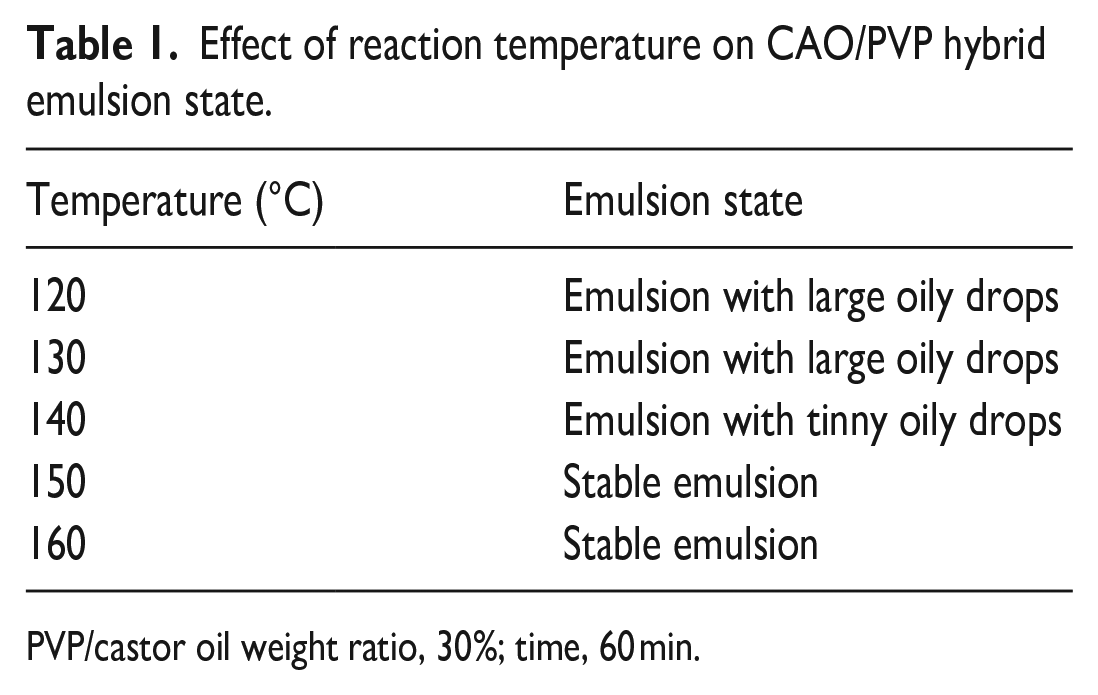
PVP/castor oil weight ratio, 30%; time, 60 min.
Effect of time
Table 2 illustrates the effect of the reaction time on the CAO/PVP hybrid emulsion state. It is obvious that prolonging the reaction time from 15 to 60 min gives rise to a gradual enhancement in the emulsion state of the CAO/PVP hybrid to be a stable emulsion. This can be attributed to increasing the extent of formation of the PVP grafted castor as well as crosslinked PVP over time. The longer reaction time, up to 75 min, does not affect the hybrid emulsion state. 30
Effect of reaction time on CAO/PVP hybrid emulsion state.

PVP/castor oil weight ratio, 30%; temperature, 150°C.
PVP/castor oil weight ratio
Table 3 shows the effect of PVP/castor oil weight ratio on the emulsion state of the CAO/PVP hybrid. It is well seen that increasing that ratio up to 30% brings about a progressive upgrading in the hybrid emulsion state which can be ascribed to increasing of the extent of formation of both the grafted castor oil and the crosslinked PVP species. 30 Beyond the ratio of 30%, within the range studied, plenty of the aforementioned species is present in the reaction medium that keeps the stability of the hybrid emulsion. It seems that the PVP/CAO weight ratio of 30% is the appropriate ratio to achieve a stable CAO/PVP hybrid emulsion. Figure 2 represents a visual appearance of the CAO/PVP hybrid emulsion at the optimum conditions of synthesis.
Effect of PVP/castor oil weight ratio on CAO/PVP hybrid emulsion state.

Time, 60 min; temperature, 150°C.

A visual appearance of the CAO/PVP hybrid emulsion.
Characterization of CAO/PVP hybrid and its emulsion
FTIR of the CAO/PVP hybrid
The FTIR spectra of CAO/PVP hybrid, PVP, and castor oil are represented by Figure 3(a) to (c) respectively. It is clear that Figure 3(a) includes:
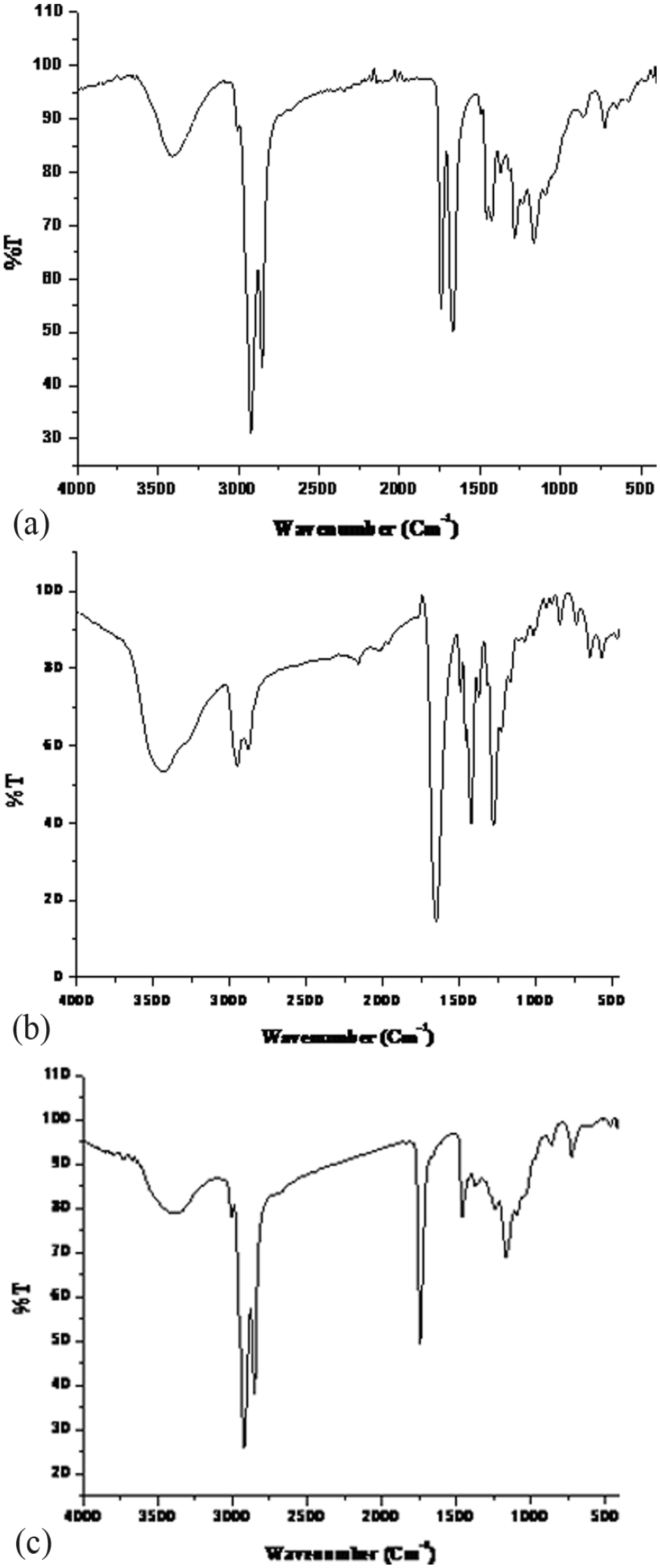
FTIR spectra of: (a) CAO/PVP hybrid, (b) PVP, and (c) castor oil.
(1) Peaks resemble that of PVP spectrum Figure 3(b) that are16,30,34,35: ● A peak at 2931 cm−1 characteristic to the stretching vibration of –CH of PVP, ● A peak at 1285 cm−1 characteristic to C–N bond, and ● A peak at 1656 cm−1 corresponding to stretching vibration the C=O group in the PVP and crosslinked PVP.
(2) Peaks resemble that of the castor oil spectrum Figure 3(c) that are36–39: ● A broad stretching vibration band at 3356 cm−1 was assigned to the OH group, ● A stretching vibration band at 3007 cm−1 assigned to C=C but with lower intensity than that of the castor oil which indicates opening some of the double bonds of castor oil and grafting of castor oil with PVP segments, ● Symmetric and asymmetric stretching vibration bands at 2924 and 2848 cm−1 respectively corresponding to the of CH2, ● A stretching band at 1740 cm−1 assigned to C=O of castor oil, ● Asymmetric and symmetric vibrations of –CH3 groups at 1459 and 1372 cm−1 respectively, ● Stretching vibration bands at 1230 and 1162 cm−1 assigned to C–O of the ester group, and, ● A peak at 729 cm−1 characteristic to –CH.
TEM image of the CAO/PVP hybrid emulsion
The CAO/PVP hybrid emulsion was characterized using the TEM analysis. The TEM image (Figure 4) shows that such emulsion particles are in a size ranges between 320 and 370 nm.

TEM image of the CAO/PVP hybrid emulsion.
Inclusion of the CAO/PVP hybrid emulsion in easy-care finishing of cotton fabric
The performance properties of the finished fabric
The performance properties of cotton fabric samples treated with easy-care finishing formulations containing different concentrations of the prepared CAO/PVP hybrid emulsion is illustrated in Table 4. It is well seen that: (i) finishing of fabric samples in absence of the nominated emulsion, results in an enhancement in nitrogen content, resiliency, stiffness, and surface roughness along with a decrease in tensile strength, wettability, and whiteness index properties of the treated fabric, compared to the untreated sample. The matter that can be attributed to crosslinking of hydroxyl groups of cotton cellulose with DMDHEU is represented by the following equation1,12,17:
Effect of the CAO/PVP hybrid emulsion concentration on performance properties of treated fabric.

%N: nitrogen content; WRA: wrinkle recovery angle (warp + weft); TS: tensile strength; W: wettability in seconds; WI: whiteness index; S: stiffness; SMD: surface roughness.
Finishing conditions: [DMDHEU], 60 g/L; [(NH4)2SO4], 6 g/L; wet pick up, 100%; drying, 100°C/3 min; curing, 160°C/3 min. Control is a fabric sample finished in absence of the hybrid emulsion.
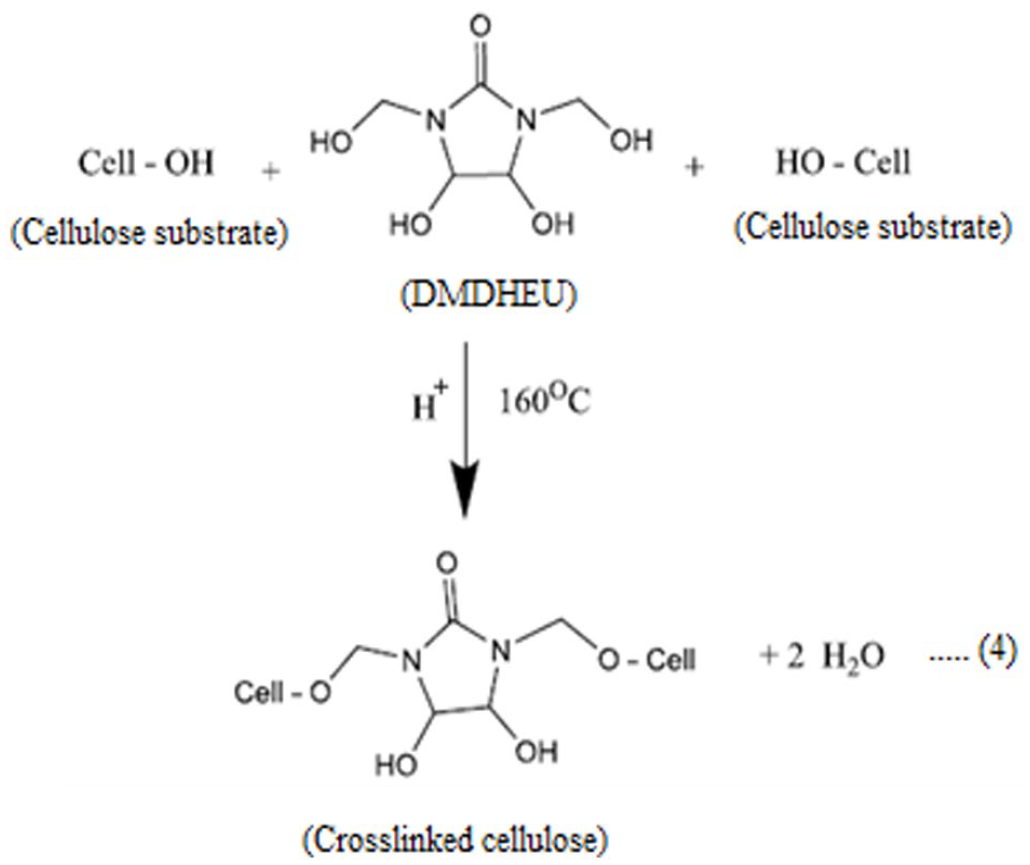
(ii) Incorporation of the prepared hybrid emulsion in the above-mentioned finishing bath gives rise to an enhancement in nitrogen content, tensile strength, whiteness index, and stiffness accompanied by a decrease in resiliency, wettability, and surface roughness properties of the treated fabric, compared to the control sample. The matter that can be associated with the fixation and deposition of such hybrid ingredients onto/within the fabric structure is shown by the following equations1,12,17:
(a) Fixation of the hybrid emulsion ingredients onto/within cotton cellulose
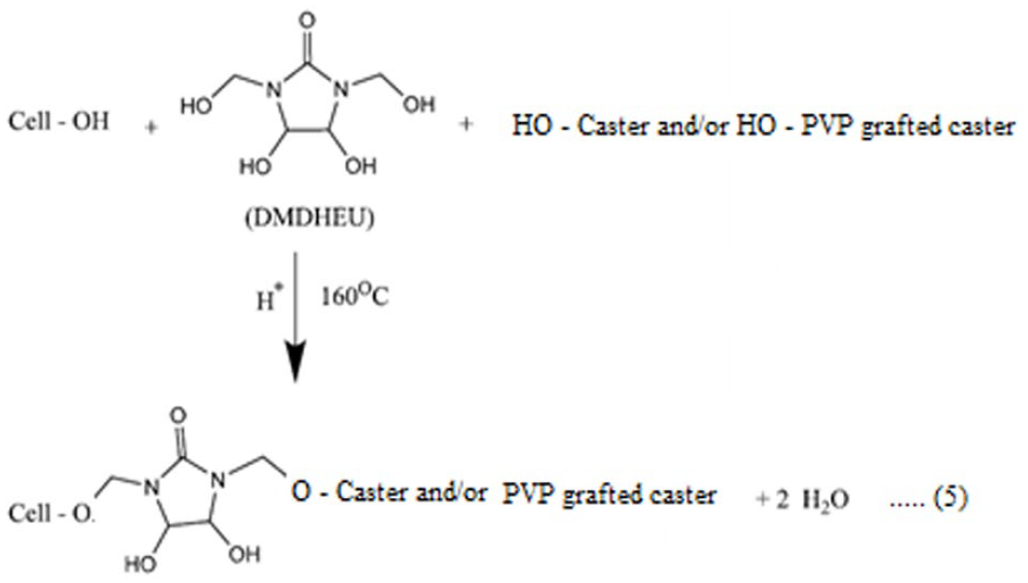
(b) Deposition of the hybrid emulsion ingredients onto/within cotton cellulose

And, (iii) increasing such emulsion concentration in the finishing bath from 20 to 40 g/L is accompanied by an increase in the extent of nitrogen content, tensile strength, and stiffness along with a reduction in resiliency, whiteness index, wettability, and surface roughness properties of treated fabric reflecting of increasing of the extent of fixation of such hybrid ingredients onto/within treated fabric structure.1,12,17
The functional properties of the finished fabric
Table 5 signifies the functional properties of cotton fabric samples treated with easy care finishing formulations containing the CAO/PVP hybrid emulsion individually or in combinations with ZnO-NPs or DHBP. It is obvious that treating fabric sample with the hybrid emulsion acquires the treated fabric with antibacterial activities against the pathogenic Gram-negative (E. coli) and Gram-positive (S. aureus) bacteria, reflecting the antibacterial properties of castor oil. 40 Combining the hybrid emulsion with the Zn-NPs or DHBP, regardless of their concentration, in different easy-care finishing formulations gives rise to an enhancement in the antibacterial properties as well as UV-blocking ability. The extent of improvement in such functional properties reflects the differences between such bio-additives in their chemical structure, functional groups, molecular size, the extent of fixation, mode of interaction, anti-bacterial activity, and ability to reflect or absorb harmful UV radiation.41,42 The upgrading in the antibacterial activity of ZnO-NPs treated fabric is attributed to a generation of reactive oxygen species such as HO·, O2·, and H2O2 and their subsequent damaging effect on the bacterial cells.41,43 The promotion in the antibacterial activities of DHBP finished fabric can be associated with the impact of exiting the benzophenone chromophoric groups to active radicals under UV light on destroying of the pathogenic bacteria.44,45 The improvement in UPF values of DHBP-loaded fabric is a direct consequence for the change in the benzophenone structure from keto to enol form, the matter that enhances the absorption of the harmful UVB radiation and prevents it to penetrate the fabric structure. 46 The higher the prepared hybrid, ZnO-NPs, or DHBP concentration in the finishing bath, the higher is the resistance against such bacterial strains. On the other hand, increasing the laundering of the treated fabric from 5 to 20 cycles gives rise to a reasonable decrease in extents of the functional properties of the treated fabric, irrespective of the bio-active type and concentration. However, the functional properties of the DHBP-loaded fabric are more durable to washing compared to the other treated fabric samples, the matter which can be attributed to the binding of DHBP molecules chemically, in addition to castor oil, to the cellulose structure through their hydroxyl groups during easy-care finishing of the treated fabric as represented by equation (7) 47 :
The functional properties of the CAO/PVP hybrid emulsion treated fabric.
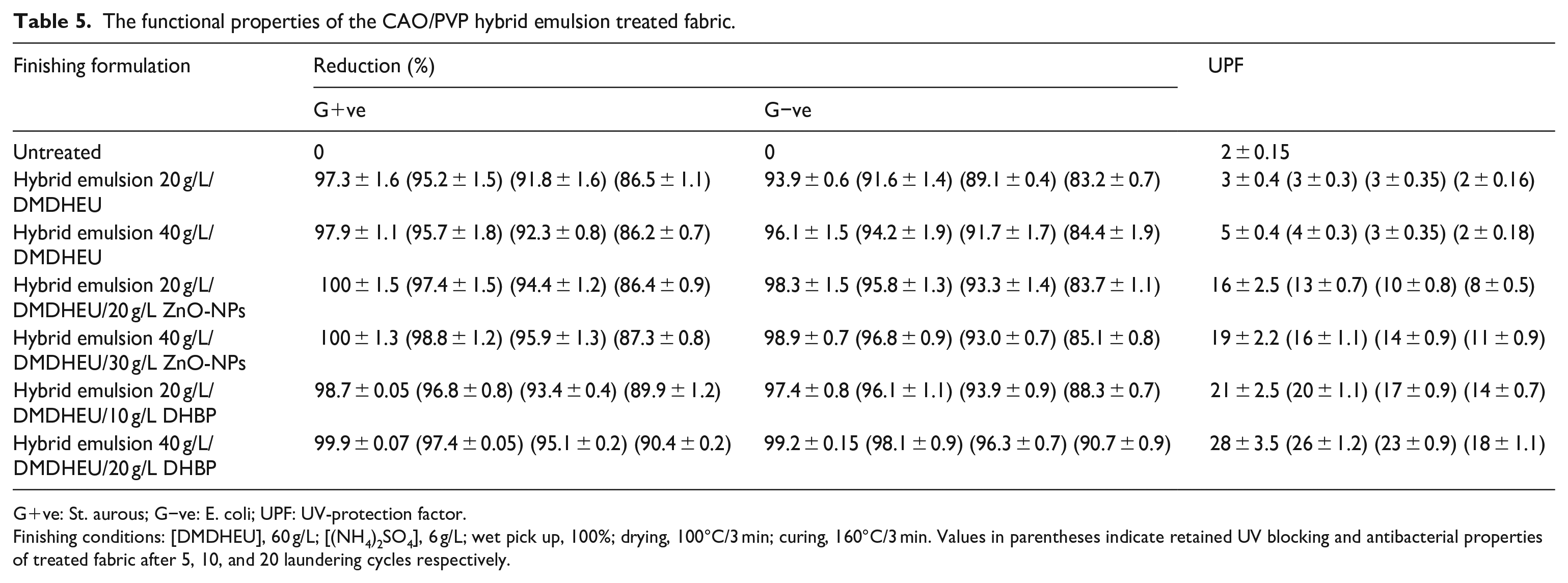
G+ve: St. aurous; G−ve: E. coli; UPF: UV-protection factor.
Finishing conditions: [DMDHEU], 60 g/L; [(NH4)2SO4], 6 g/L; wet pick up, 100%; drying, 100°C/3 min; curing, 160°C/3 min. Values in parentheses indicate retained UV blocking and antibacterial properties of treated fabric after 5, 10, and 20 laundering cycles respectively.

In contrast, Zn-NPs are not firmly attached to the cellulose structure as they lack reactive groups capable to bind to the cellulose structure, the matter which indeed lowers the durability of the finished to laundering.
Coloration properties and fastness properties of the dyed and dyed/finished cotton fabric
Wavelength and coloration properties of the dyed and dyed/finished cotton fabric
Table 6 shows the wavelength (λmax), color strength (K/S), and colorimetric data (L*, a*, and b*) values of the dyed samples before and after treatment with easy care finishing bath containing the prepared CAO/PVP hybrid emulsion. It is clear that the variation in dye type results in dyed samples having different wavelengths, color strength as well as colorimetric data, which can be associated with the alteration in affinity, molecular size, functionality, as well as the mode of interaction of that dyes.18,20,48,49 In addition, treating the dyed samples with the aforementioned emulsion, irrespective of dye type, brings about an enhancement in K/S of the treated samples, which may be attributed to the color-deepening effect of the CAO/PVP hybrid emulsion as a textile softener. 50
Effect of the dye type on wavelength and coloration properties of the dyed and dyed/finished cotton fabric.
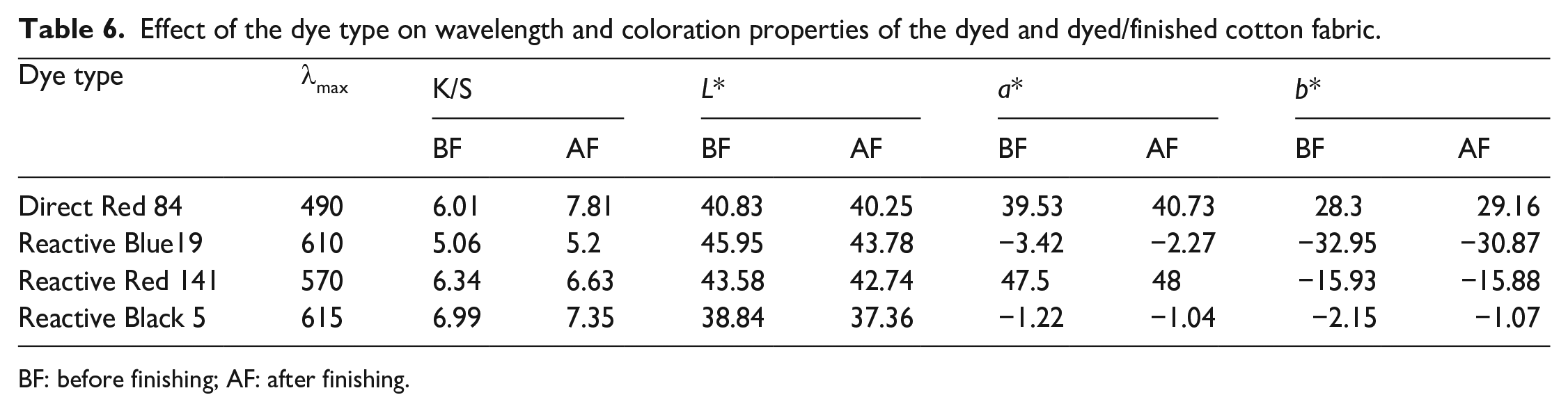
BF: before finishing; AF: after finishing.
The fastness properties dyed/finished fabric
Table 7 shows the fastness properties of the dyed and dyed/finished fabric samples. It is clear that finishing of the dyed samples results in: (i) an enhancement in the washing and perspiration fastness of only the sample dyed with the direct dye, (ii) an improvement in the wet rubbing fastness as well as the alkaline perspiration fastness of almost all the dyed/finished samples, and (iii) a lowering in the lightfastness of all the dyed/finished fabric samples. The variation in such fastness properties reflects the impact of the prepared CAO/PVP hybrid ingredients deposition as a film onto the dyed cotton fabric structure.
The effect of finishing of the dyed fabric with the CAO/PVP hybrid on the fastness properties of that fabric.

Alt: alteration in color; St: staining on cotton; St: staining on wool; BF: before finishing; AF: after finishing.
SEM and EDX images of the treated fabric
Figure 5 shows SEM images of (a) untreated cotton fabric and (b) cotton fabric treated with a finishing bath containing 60 g/L of DMDHEU, 6 g/L of ammonium sulfate, 30 g/L of the prepared emulsion, and 2 g/L of ZnO-NPs. It is clearly seen that the SEM image of fabric sample (b) shows a homogenous deposited film of the hybrid ingredients onto the surface of that sample, compared to that of the untreated sample. On the other hand, Figure 6 shows EDX analysis of the above-mentioned fabric samples. It is clear that, EDX image of the untreated sample contains only the elements of C and O whereas the EDX image of the sample (b) contains the elements of C, O, N, and Zn.

SEM images of: (a) untreated cotton fabric and (b) finished cotton fabric.

EDX images of: (a) untreated cotton fabric and (b) finished cotton fabric.
Storage stability of the CAO/PVP hybrid emulsion
Upon storage of the CAO/PVP hybrid emulsion for one month, tinny oil drops appeared on the emulsion surface after 1 week that continued for a month without almost any change, and then disappeared upon shaking and a stable emulsion was formed again.
Conclusion
New textile softener CAO/PVP hybrid emulsion was synthesized at different reaction conditions.
The hybrid was synthesized at optimum conditions of PVP molecular weight, 40,000 Da; PVP/CAO weight ratio, 30%; temperature, 150°C; and time, 50 min.
Emulsification of such hybrid in water results in a white stable emulsion.
The TEM image confirmed that the particle size of such emulsion ranges from 320 to 370 nm.
Treating cotton fabric sample with easy care finishing bath containing the synthesized emulsion brings about an enhancement of nitrogen content, tensile strength, whiteness index, and stiffness accompanied by a reduction in resiliency, wettability, and surface roughness properties of the treated fabric.
Incorporation of the produced emulsion in easy care finishing formulations containing ZnO-NPs or dihydroxybenzophenone imparted the cotton fabric with remarkable antibacterial properties as well as protection against UV radiation.
All the dyed/finished fabric samples showed higher color strength, compared to the dyed samples.
The washing and perspiration fastness of only the direct dye dyed sample was improved upon finishing of sample with the aforementioned hybrid emulsion.
Almost all the dyed/finished samples exhibited an improvement in their fastness to the wet rubbing and alkaline perspiration along with a reduction in their fastness to light.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper is based upon work supported by Science, Technology & Innovation Funding Authority (STDF) under grant number 43342.
